- marine organism
- anticancer medicine
- small peptide
- liner peptide
- cyclic peptide
1. Introduction
Oceans cover about 70% of the earth’s surface and 95% of the biosphere. Water was the cradle of the earliest living organisms, containing approximately 75% of all living organisms. The marine environment offers a rich source of natural products with potential therapeutic applications. More than 1 million marine invertebrates and more than 25,000 species of fish have been discovered, and some of these have been shown to contain natural products with potential biological activity [1][2]. In recent years, marine microorganisms, have also been regarded as a valuable source of bioactive compounds, with the advantages of easy cultivation and good compound extraction repeatability [3]. More than 10,000 bioactive molecules that have been isolated from marine organisms, and several have been found to possess anticancer activity [4]. Most of these natural products with anticancer activity originate from microorganisms (bacteria, fungi, protozoa, viruses, and chromista), plantae (flowering plants like mangroves and macroalgae), and animalia (invertebrates such as sponges, tunicates, and vertebrates such as fish and whale), etc.
Oceans cover about 70% of the earth’s surface and 95% of the biosphere. Water was the cradle of the earliest living organisms, containing approximately 75% of all living organisms. The marine environment offers a rich source of natural products with potential therapeutic applications. More than 1 million marine invertebrates and more than 25,000 species of fish have been discovered, and some of these have been shown to contain natural products with potential biological activity [1,2]. In recent years, marine microorganisms, have also been regarded as a valuable source of bioactive compounds, with the advantages of easy cultivation and good compound extraction repeatability [3]. More than 10,000 bioactive molecules that have been isolated from marine organisms, and several have been found to possess anticancer activity [4]. Most of these natural products with anticancer activity originate from microorganisms (bacteria, fungi, protozoa, viruses, and chromista), plantae (flowering plants like mangroves and macroalgae), and animalia (invertebrates such as sponges, tunicates, and vertebrates such as fish and whale), etc.
Cancer is one of the leading causes of death in the world. An estimated 9.6 million people died of cancer in 2018 [5]. Almost 1 in 6 people die of cancer globally. With the application of new theories, modern technologies, and new drugs in basic tumor research and clinical treatment, the rising trend in tumor death in many countries has been effectively controlled [5]. Chemotherapy is part of the major categories of medical oncology. Despite these successes, chemotherapy’s lingering toxic side-effects are still a primary cause of morbidity and mortality in cancer survivors [6]. As for traditional chemotherapy drugs, most of these inhibit tumor cell proliferation by acting on the DNA synthesis and the replication of tumor cells, which have been shown to be effective but at the price of high toxicity due to a lack of selectivity. Nowadays, there are novel molecular methods for treatment using cancer drugs, including target therapy by cell surface receptors, immune-directed therapy, therapeutic vaccines, and antibody–drug conjugates (ADCs) [7][8]. According to the 2020 ASCO’s Annual Report, a large number of innovative drugs, which can be categorized into targeted drugs and immune drugs, have entered trials and clinical trials [9]. Targeted medicine can restrain tumor cell growth by blocking the signal transduction, but the recurrence rate is extremely high [10]. Antibody–drug conjugates have the potential for increased tumor penetration and drug resistance. It has been demonstrated that a knottin peptide–drug conjugate (KDC) can selectively deliver gemcitabine to malignant cells expressing tumor-associated integrins [11]. In recent years, major pharmaceutical companies and research centers have focused on monoclonal antibody drugs and bi-specific antibody drugs in targeted therapy, as well as CAR-T and immunoassay point inhibitors in immunotherapy [12][13][14]. Immunotherapy achieves anti-tumor therapy by stimulating the body’s immune system. In immunotherapy, mainly immune cell therapy, immune checkpoint inhibitors, tumor vaccines, and immune system regulators, the immune system is used to recognize and regulate the body’s attack on abnormal cell functions [14]. Small peptides, such as the thymic peptide, with their unique advantages in immunotherapy, are also very prominent. The thymic peptide used has been a non-specific adjuvant therapy for various tumors, as it can induce T cell differentiation and development, promoting its proliferation and improving T cell response to the antigen at the same time [15]. This kind of drug enhances the patient’s immunity, with fewer side effects.
Cancer is one of the leading causes of death in the world. An estimated 9.6 million people died of cancer in 2018 [5]. Almost 1 in 6 people die of cancer globally. With the application of new theories, modern technologies, and new drugs in basic tumor research and clinical treatment, the rising trend in tumor death in many countries has been effectively controlled [5]. Chemotherapy is part of the major categories of medical oncology. Despite these successes, chemotherapy’s lingering toxic side-effects are still a primary cause of morbidity and mortality in cancer survivors [6]. As for traditional chemotherapy drugs, most of these inhibit tumor cell proliferation by acting on the DNA synthesis and the replication of tumor cells, which have been shown to be effective but at the price of high toxicity due to a lack of selectivity. Nowadays, there are novel molecular methods for treatment using cancer drugs, including target therapy by cell surface receptors, immune-directed therapy, therapeutic vaccines, and antibody–drug conjugates (ADCs) [7,8]. According to the 2020 ASCO’s Annual Report, a large number of innovative drugs, which can be categorized into targeted drugs and immune drugs, have entered trials and clinical trials [9]. Targeted medicine can restrain tumor cell growth by blocking the signal transduction, but the recurrence rate is extremely high [10]. Antibody–drug conjugates have the potential for increased tumor penetration and drug resistance. It has been demonstrated that a knottin peptide–drug conjugate (KDC) can selectively deliver gemcitabine to malignant cells expressing tumor-associated integrins [11]. In recent years, major pharmaceutical companies and research centers have focused on monoclonal antibody drugs and bi-specific antibody drugs in targeted therapy, as well as CAR-T and immunoassay point inhibitors in immunotherapy [12,13,14]. Immunotherapy achieves anti-tumor therapy by stimulating the body’s immune system. In immunotherapy, mainly immune cell therapy, immune checkpoint inhibitors, tumor vaccines, and immune system regulators, the immune system is used to recognize and regulate the body’s attack on abnormal cell functions [14]. Small peptides, such as the thymic peptide, with their unique advantages in immunotherapy, are also very prominent. The thymic peptide used has been a non-specific adjuvant therapy for various tumors, as it can induce T cell differentiation and development, promoting its proliferation and improving T cell response to the antigen at the same time [15]. This kind of drug enhances the patient’s immunity, with fewer side effects.
The resistance adaptation ability of small peptides in many drug treatments and their fewer toxic side effects indicate their potential application in further developing novel drugs. Due to these medicines’ unique metabolic processes, many new study areas on the pharmaceutical aspects of protein and peptide drugs have recently emerged [16][17]. Many natural and synthetic peptides were characterized in recent decades, and public databases were established, such as APD3 (database of antimicrobial peptides), the Defensins Knowledgebase, the antiviral AVPdb (database of antiviral peptides), the antiparasitic ParaPep, and the CancerPPD (database of anticancer peptides and proteins) [18][19][20][21][22]. Bioactive small peptides are composed of 2–10 amino acids linked by peptide bonds. Studies have found that amino acids such as Trp, Tyr, Met, Gly, Cys, His, and Pro in the peptide chain can significantly improve the bioactivity [23][24]. In other words, compared with traditional chemotherapy drugs, small peptides have several advantages, such as high absorption, small size, well-defined signaling targets, and minimal toxicity. As such, they offer a new promising area of research. Besides, small peptides are just connected by a few peptide linkages, and their small molecular weight makes them easy to be modified and synthesized. Additionally, small peptides can be used as vectors to deliver drugs specifically to every cell and tissue [25]. However, small peptides have some disadvantages, including a short half-life, easy degradation in vivo, poor stability, etc. Many researchers have tried to modify or develop corresponding pseudo-peptide drugs or have combined small peptides with traditional therapy for tumor combination therapy to achieve better results than with a single treatment. The application of several cyclic peptides as noncovalent nuclear targeting molecular transporters of Dox has been reported [26]. Additionally, another work designed new peptides based on the molecular dynamics simulation (MDs) of the matuzumab-EGFR complex in a water environment. These peptides had a higher affinity to the EGFR relative to that previously reported [27]. The peptide modification of anticancer drugs could enhance their activity and selectivity, perhaps even circumventing multi-drug resistance [28].
The resistance adaptation ability of small peptides in many drug treatments and their fewer toxic side effects indicate their potential application in further developing novel drugs. Due to these medicines’ unique metabolic processes, many new study areas on the pharmaceutical aspects of protein and peptide drugs have recently emerged [16,17]. Many natural and synthetic peptides were characterized in recent decades, and public databases were established, such as APD3 (database of antimicrobial peptides), the Defensins Knowledgebase, the antiviral AVPdb (database of antiviral peptides), the antiparasitic ParaPep, and the CancerPPD (database of anticancer peptides and proteins) [18,19,20,21,22]. Bioactive small peptides are composed of 2–10 amino acids linked by peptide bonds. Studies have found that amino acids such as Trp, Tyr, Met, Gly, Cys, His, and Pro in the peptide chain can significantly improve the bioactivity [23,24]. In other words, compared with traditional chemotherapy drugs, small peptides have several advantages, such as high absorption, small size, well-defined signaling targets, and minimal toxicity. As such, they offer a new promising area of research. Besides, small peptides are just connected by a few peptide linkages, and their small molecular weight makes them easy to be modified and synthesized. Additionally, small peptides can be used as vectors to deliver drugs specifically to every cell and tissue [25]. However, small peptides have some disadvantages, including a short half-life, easy degradation in vivo, poor stability, etc. Many researchers have tried to modify or develop corresponding pseudo-peptide drugs or have combined small peptides with traditional therapy for tumor combination therapy to achieve better results than with a single treatment. The application of several cyclic peptides as noncovalent nuclear targeting molecular transporters of Dox has been reported [26]. Additionally, another work designed new peptides based on the molecular dynamics simulation (MDs) of the matuzumab-EGFR complex in a water environment. These peptides had a higher affinity to the EGFR relative to that previously reported [27]. The peptide modification of anticancer drugs could enhance their activity and selectivity, perhaps even circumventing multi-drug resistance [28].
The marine environment is a crucial biological kingdom with the richest source of novel functional proteins and peptides. Additionally, it is gradually becoming a vital field of drug development [29]. As marine organisms live in a special environment that is hypersaline, high-pressure, hypoxia, and hypothermic, and lacks sunlight, these proteins and peptides have strong bioactivities and a specific structure. There are many effects associated with marine peptides, such as antioxidant, antimicrobial, antitumor, antiviral, cardioprotective, immunomodulatory, and tissue regeneration properties [16][30][31][32]. Approximately 49 marine-derived active substances or their derivatives have been approved for the market or entered clinical trials globally [33]. Most of these bioactive molecules are extracted from marine sponges, mollusca, and algae. There are 11 kinds of marine drugs approved by European and American drug authorities, of which, four are listed as anticancer drugs: Cytosar-U, Yondelis, Halaven, and Adcetris. In recent years, more and more studies on marine bioactive peptides have appeared. Many bioactive peptides with anticancer potential have been extracted from various marine organisms. Small peptides from marine sources are gradually gaining attention because of their uniqueness. Compared with high-molecular-weight peptides, low-molecular-weight peptides show greater molecular mobility and diffusivity, contributing to their enhanced interaction with cancer cell components and increasing their anticancer activity [34]. Nowadays, several newly discovered anticancer small peptides and their derivatives thereof from marine organisms have been widely applied to clinical research [35][36][37] (
The marine environment is a crucial biological kingdom with the richest source of novel functional proteins and peptides. Additionally, it is gradually becoming a vital field of drug development [29]. As marine organisms live in a special environment that is hypersaline, high-pressure, hypoxia, and hypothermic, and lacks sunlight, these proteins and peptides have strong bioactivities and a specific structure. There are many effects associated with marine peptides, such as antioxidant, antimicrobial, antitumor, antiviral, cardioprotective, immunomodulatory, and tissue regeneration properties [16,30,31,32]. Approximately 49 marine-derived active substances or their derivatives have been approved for the market or entered clinical trials globally [33]. Most of these bioactive molecules are extracted from marine sponges, mollusca, and algae. There are 11 kinds of marine drugs approved by European and American drug authorities, of which, four are listed as anticancer drugs: Cytosar-U, Yondelis, Halaven, and Adcetris. In recent years, more and more studies on marine bioactive peptides have appeared. Many bioactive peptides with anticancer potential have been extracted from various marine organisms. Small peptides from marine sources are gradually gaining attention because of their uniqueness. Compared with high-molecular-weight peptides, low-molecular-weight peptides show greater molecular mobility and diffusivity, contributing to their enhanced interaction with cancer cell components and increasing their anticancer activity [34]. Nowadays, several newly discovered anticancer small peptides and their derivatives thereof from marine organisms have been widely applied to clinical research [35,36,37] (
). Marine small peptides are molecules that participates in all processes of life activities. They can bear anticancer roles in diversiform aspects in different ways, such as preventing cell migration, induction of apoptosis, disorganization of tubulin structure and inducing cell cycle arrest, and more (
) [38].

Figure 1.
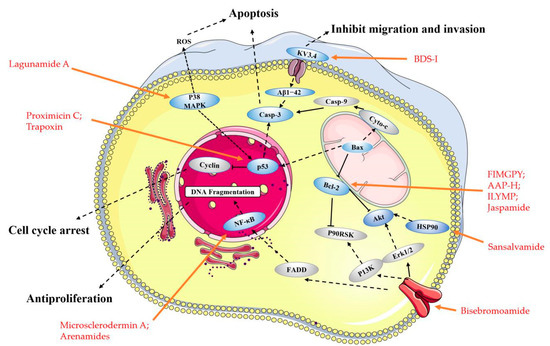
Figure 2.
2. Linear Peptides and Derivatives
2.1. Animals
In recent years, some anticancer peptides have been discovered from proteolytic products and secondary metabolites of marine animals, and these purified anticancer peptides have cytotoxic, anti-proliferative and protease inhibition effects [39]. The early discovery of the anti-tumor effect of small marine peptides was mostly based on cytotoxicity assessment, and the subsequent mechanism is not clear. Ding Guo-Fang et al. discovered a tripeptide QPK (50
Sarcophyton glaucum
50
Table 1.
| Compound | Source | Mechanism | Cell Lines | IC50/(GI50) b | Reference | |||
|---|---|---|---|---|---|---|---|---|
| QPK | Sepia ink | Cytotoxicity a | DU-145 | 9.50 mg/mL (24 h); | [40] | |||
| 1.00 mg/mL (48 h) | ||||||||
| 1.6 µM; | 1.6 µM; |
AGAPGG; | Sarcophyton glaucum | Cytotoxicity a | HeLa | 8.6 mmol/L; | [41] | |
| AERQ; | 4.9 mmol/L; |
Streptomyces
50 value of 30.9 µM [60]. Tasiamide (
Symploca
50 values of 0.48 and 3.47 μg/mL, respectively [61]. Cathepsin D (Cath D) has been considered a potential target to treat cancer [72]. Tasiamide’s C-terminal modified derivatives have inhibitory activity against Cath D/Cath E/BACE1, potentially making them highly potent and selective Cath D inhibitors [62]. Belamide A (
50: 0.74 μM). At a concentration of 20 μM, it destroyed the micro-tubule network in rat aortic smooth muscle A-10 cells and showed the classic tubulin destabilizing mitotic characteristics [63]. Symplostatin
A
10
Symploca hydnoides, can cause a loss of interphase micro-tubules, G2/M arrest, and active caspase 3 and initiate the phosphorylation of Bcl-2 [64]. Proximicins A, B, and C (
Verrucosispora
50 values of 12.7 and 11.4 μg/mL, respectively [65]. Bisebromoamide (
Lyngbya
50) value across all of the cell lines (a panel of 39 human cancer cell lines (termed JFCR39)) [66]. Additionally, it can also inhibit the Raf/MEK/ERK and PI3K/Akt/mTOR pathways, showing a potent protein kinase inhibitor effect [67].
2.3. Other Small Peptides
Currently, the number of small linear anti-cancer peptides derived from marine plants is relatively small. The peptide, HVLSRAPR (
2.3. Other Small Peptides
Currently, the number of small linear anti-cancer peptides derived from marine plants is relatively small. The peptide, HVLSRAPR (
), exhibited strong inhibitory activity on HT-29 cells, with an IC
50 value of 99.88 μg/mL, while it had little inhibitory activity on LO2 cells (normal liver cells) (5.37% at 500 μg/mL). It was also shown to be selectively active on cancer cells, including Hep G2 (human liver cancer cells), MCF-7 (human breast cancer cells), SGC-7901 (human gastric cancer cells), and A549 cells [68].Most of the small peptides are still in the stage of structural optimization and in vitro activity verification. The lack of progress in subsequent research on their mechanisms prevents them from entering clinical development. Additionally, these small peptides merit further investigation as a potential therapeutic agent.
3. Cyclic Peptides and Derivatives
Cyclic peptides combine several favorable properties, such as a good binding affinity, target selectivity, and low toxicity, which make them attractive in anticancer drug researches [73]. Besides, most of the small peptides are concentrated in marine animals, and they are found to be secondary metabolites from sponges, sea squirts, cnidaria, and mollusks.
3.1. Animals
value of 99.88 μg/mL, while it had little inhibitory activity on LO2 cells (normal liver cells) (5.37% at 500 μg/mL). It was also shown to be selectively active on cancer cells, including Hep G2 (human liver cancer cells), MCF-7 (human breast cancer cells), SGC-7901 (human gastric cancer cells), and A549 cells [72].
Most of the small peptides are still in the stage of structural optimization and in vitro activity verification. The lack of progress in subsequent research on their mechanisms prevents them from entering clinical development. Additionally, these small peptides merit further investigation as a potential therapeutic agent.
3. Cyclic Peptides and Derivatives
Cyclic peptides combine several favorable properties, such as a good binding affinity, target selectivity, and low toxicity, which make them attractive in anticancer drug researches [73]. Besides, most of the small peptides are concentrated in marine animals, and they are found to be secondary metabolites from sponges, sea squirts, cnidaria, and mollusks.
3.1. Animals
3.1.1. Metabolites of Ascidians
Didemnum mole
50
50
Lissoclinum
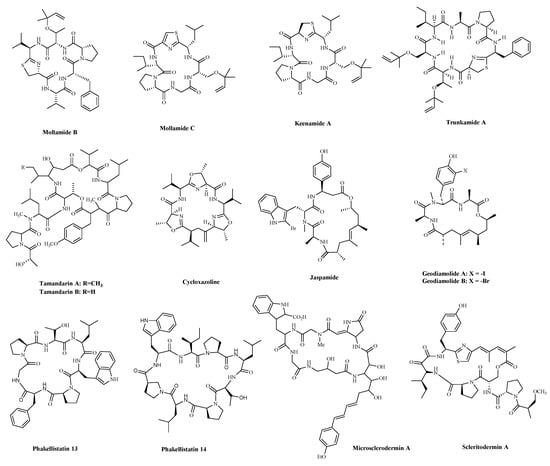
Figure 4.
Table 2.
| Compound | Source | Mechanism | Cell Lines | IC50/(GI50) b | Reference | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mollamide B | Tunicate Didemnum | Cytotoxicity a | H460; | (>100 µM) | [74] | |||||
| MCF-7; | ||||||||||
| SF-268 | ||||||||||
| Mixirin C
Mixirin C |
HCT-116
HCT-116 |
1.26 µM;
1.26 µM; |
Keenamide A | Tunicate Didemnum. | ||||||
| Mechercharmycin A
Mechercharmycin A |
Thermoactinomyces sp. YM3-251
Thermoactinomyces sp. YM3-251 | Cytotoxicity a | P-388; | Cytotoxicity a
Cytotoxicity a | 2.5 µg/mL; | A549;
A549; | [75] | RDTQ | 5.6 mmol/L | |
| A-549; | 2.5 µg/mL; | Virenamides A; | The Didemnid ascidian Diplosoma virens | Inhibiting the Topoisomerase II | P388; | 2.5 µg/mL | [42] | |||
| MEL-20; | 2.5 µg/mL;A549; | 10 µg/mL | ||||||||
| HT-29 | 5.0 µg/mL | HT-29; | 10 µg/mL | |||||||
| Trunkamide A | Didemnid ascidians | Cytotoxicity a | DU-145; | 7.08 nM; | [76] | CV1 | 10 µg/mL | |||
| IGROV; | 7.31 nM; | Virenamides B | The Didemnid ascidian Diplosoma virens | Inhibiting the Topoisomerase II | P388; | |||||
| SK-BR-3; | 5 µg/mL | |||||||||
| 5.44 nM; | A549; | 5 µg/mL | ||||||||
| HT-29; | 5 µg/mL | |||||||||
| CV1 | 5 µg/mL | |||||||||
| Hela | 3.90 nM | |||||||||
| Tamandarin A | Didemnid ascidians | Cytotoxicity a | NCI-60 | 1.4 µM (2.3 µM) |
[77] | Virenamides C | The Didemnid ascidian Diplosoma virens | Inhibiting the Topoisomerase II | ||
| Tamandarin B | P388; | 5 µg/mL | ||||||||
| A549; | 5 µg/mL | |||||||||
| HT-29; | 5 µg/mL | |||||||||
| CV1 | 5 µg/mL | |||||||||
| SCAP1; (Leu-Ala-Asn-Ala-Lys) |
Oyster (Saccostrea cucullata) | Enhancing oxidative DNA damage; Inducing apoptosis |
HT-29 | 90.31 mg/mL (24 h); | [43][44][43,44] | |||||
| 70.87 mg/mL (48 h); | ||||||||||
| 60.21 mg/mL (72 h) | ||||||||||
| YALPAH | Half-fin anchovy (Setipinna taty) | Inducing apoptosis | PC‑3 | 8.1 mg/mL | [45][46][45,46] | |||||
| BCP-A (Trp-Pro-Pro) |
Blood clam (Tegillarca granosa) muscle | Inducing apoptosis and inhibiting lipid peroxidation | PC-3; | 1.99 mg/mL; | [47] | |||||
| DU-145; | 2.80 mg/mL; | |||||||||
| H-1299; | 3.3 mg/mL; |
90][91]. In a way similar to other depsipeptides (Jaspamide and Dolastatins), it keeps the normal microtubule organization and regulates actin cytoskeleton, migration, and invasion of breast cancer cells [81][92]. Phakellistatins, a class of cycloheptapeptides, isolated from
Phalkellia
50
50) against BEL-7404 (human liver cancer cells) cells [82][83]. Due to its good antitumor activity in vitro and in vivo, many researchers have modified this structure in the hope of synthesizing potential Marine drugs with a better anti-cancer effect [93]. The Microsclerodermins are cyclic hexapeptides isolated from a deep-water sponge of the genus
Microscleroderma
50 values were 2.3, 0.8, 4.3, and 4.0 μM against the four cells, respectively [84]. Additionally, recent research has discovered the congeners of the Microsclerodermins, Microsclerodermins N and O. They exhibit cytotoxic activity against HeLa cells with IC
50
Scleritoderma nodosum in 2004. Scleritodermin A has significant in vitro cytotoxicity against a panel of human tumor cell lines and acts through tubulin polymerization inhibition and the resulting disruption of microtubules [85]. Subsequent studies have led to discovering several novel cyclopeptides, but their antitumor activity is relatively weak, and the mechanism of action is unclear [95]. These peptides have unique structures as compared with those from other sources. This attribute makes sponge- and tunicate-derived peptides highly attractive as potential drug and molecular probes.
3.2. Fungi
Zygosporamide (
3.2. Fungi
Zygosporamide (Zygosporium masonii.
50
50
50
Acremonium persicinum
50

Figure 5.
Table 3.
| Compound | Source | Mechanism | Cell Lines | IC50/(GI50) b | Reference | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Zygosporamide | Zygosporium masonii | Cytotoxicity a | SF-268; | (6.5 nM) | [96] | ||||||
| RXF 393 | (5.0 nM) | ||||||||||
| Cordyheptapeptide C | Acremonium persicinum | Cytotoxicity a | SF-268; | 3.7 μM; | [97] | ||||||
| MCF-7; | 3.0 μM; | ||||||||||
| 40 nM; | 40 nM; | [109]
[109] |
NCI-H460 | 11.6 μM | |||||||
| Jurkat
Jurkat |
46 nM;
46 nM; |
Cordyheptapeptide D | Acremonium persicinum | ||||||||
| Urukthapelstatin A
Urukthapelstatin A |
Thermoactinomycetaceae bacterium Mechercharimyces asporophorigenens YM11-542 | Cytotoxicity a | SF-268; | Thermoactinomycetaceae bacterium 45.6 μM; |
|||||||
Mechercharimyces asporophorigenens | YM11-542 |
Cytotoxicity a
Cytotoxicity a |
A549
A549 |
12 nM;
12 nM; |
[110]
[110] |
MCF-7; | 82.7 μM; | ||||
| Arenamide A
Arenamide A |
Salinispora Arenicola.
Salinispora Arenicola. |
Inhibiting NF kappa B
Inhibiting NF kappa B |
293/NF-κB-Luc
293/NF-κB-Luc |
3.7 μM;
3.7 μM; |
[111] | NCI-H460 | >100 μM | ||||
[ | ] |
Cordyheptapeptide E | Acremonium persicinum | Cytotoxicity a | SF-268; | 3.2 μM; | |||||
| MCF-7; | 2.7 μM; | ||||||||||
| NCI-H460 | 4.5 μM | ||||||||||
| Asperterrestide A | Aspergillus terreus | Cytotoxicity a | U937; | 6.4 μM; | [98] | NCI-60 | 1.4 µM (2.3 µM) |
||||
| MOLT4 | 6.2 μM | Cycloxazoline | Didemnid ascidians | Cell cycle G2/M arrest, Induction of apoptosis | |||||||
| Arenamide B
Arenamide B |
293/NF-κB-Luc
293/NF-κB-Luc |
1.7 μM
1.7 μM |
MRC5CV1; T24 | ||||||||
| Sansalvamide A | 0.5 μg/mL | Microsporum cf. gypseum | [ | Inhibiting cell growth, and proliferation, and inducing cell apoptosis by regulating the expression of HSP90 | 77] | ||||||
| HCT-116; | 1.5 µM; | [ | 99 | ] | [100][99,100] | Jaspamide (Jasplakinolide, NSC-613009) |
Sponge Jaspis johnstoni | Induced apoptosis is associated with caspase-3 activation, increased Bax level, and decreased Bcl-2 protein expression | T24; MCF-7; 15NCI/ADR; A-10 |
60 to 150 µg /mL | [79][80][85,86] |
| Geodiamolide A | Sponge Geodia corticostylifera | Induction of apoptosis; Tubulin polymerization inhibition |
T47D; | 18.82 nM; | [81][89] | ||||||
| MCF7 | |||||||||||
| HeLa | |||||||||||
| 2.54 mg/mL | |||||||||||
| 17.83 nM; | |||||||||||
| Geodiamolides B | T47D; | 113.90 nM; | |||||||||
| MCF7 | 9.82 nM; | ||||||||||
| Phakellistatin 13 | Sponge Phakellia sp. | Induction of both intrinsic and extrinsic apoptosis | BEL-7404 | (10 ng/mL) | [82][83][91,92] | BDS-I; (Ala-Ala-Pro-Ala-Phe-Ala-Ser-Gly) |
The sea anemone toxin | Blocking KV3.4 currents prevented (the neurotoxic β-amyloid peptide1-42) Aβ1−42-induced caspase-3 activation and apoptotic processes | PC-12 | 75 nM | [48][49][49,50] |
| FIMGPY | The skate (R. porosa) cartilage protein hydrolysate | Inducing apoptosis by upregulating the Bax/Bcl-2 ratio and caspase-3 activation | HeLa | 4.81 mg/mL | [50][51] | ||||||
| AAP-H; (Tyr-Val-Pro-Gly-Pro) |
The sea anemone Anthopleura anjunae | Inducing apoptosis, decreasing the mitochondrial membrane potential, and increasing Bax/Bcl-2 ratio, cytochrome-C, caspase-3, and caspase-9 | DU-145 | 9.605 mM (24 h); | [51][52] | ||||||
| 7.910 mM (48 h); | |||||||||||
| 2.298 mM (72 h) | |||||||||||
| Phakellistatin 14 | P388 | (5 µg/ mL) | |||||||||
| Microsclerodermin A | Sponge of the genus Amphibleptula | Inhibit NFκB, Induction of apoptosis; | AsPC-1; | 2.3 μM; | [84][82] | ||||||
| BxPC-3; | 0.8 μM; | ||||||||||
| MIA PaCa-2; | 4.3 μM; | ||||||||||
| PANC-1; | 4.0 μM | ||||||||||
| Scleritodermin A | Sponge Scleritoderma nodosum | Tubulin polymerization inhibition |
HCT-116; | 1.9 µM; | [85][83] | ILYMP | |||||
| A2780; | 0.940 µM; | Cyclina sinensis | Enhancing expression of Bax, cleaved caspase-3/9 as well as suppression of Bcl-2 expression | ||||||||
| SKBR3 | DU-145 | 11.25 mM | [ | 52 | ][53] | ||||||
| SCH-P9 (Leu-Pro-Gly-Pro) |
Sinonovacula constricta hydrolysates | Inducing apoptosis and sub-G1 phase cell cycle arrest | DU‑145; | 1.21 mg/mL (24 h); |
[53][54] | ||||||
| 0.670 µM | PC‑3 | 1.09 mg/mL (24 h) | |||||||||
| SCH-P10 (Asp-Tyr-Val-Pro) |
DU‑145; | 1.41 mg/mL (24 h); |
|||||||||
| PC‑3 | 0.91 mg/mL (24 h) |
||||||||||
| SIO | Sepia ink | Inducing apoptosis, and S and G2/M phase cell cycle arrest | DU-145; | <5 mg/mL | [54][55][55,56] | ||||||
| PC-3; | <5 mg/mL | ||||||||||
| LNCaP | <10 mg/mL | ||||||||||
| Psammaplin A (PsA) | The two Sponges, Jaspis sp.and Poecillastra wondoensis. | Inducing S or S-G2/M phase cell cycle arrest; Inhibting HDAC | P388; HCT-116; A549 |
(40 nM) | [56][57][58,59] | ||||||
| NVP-LAQ824 | Psammaplysilla sp. | Inducing S or S-G2/M phase cell cycle arrest; Inhibting HDAC | H-1299 | 150 nM | [58][60] | ||||||
| HCT-116 | 10 nM | ||||||||||
| Lucentamycins A; | The fermentation broth of a marine-derived actinomycete | Cytotoxicity a | HCT-116 | 0.20 µM; | [59][62] | ||||||
| Lucentamycins B | 11 µM | ||||||||||
| Padanamides A and B | Sediment in the culture of Streptomyces sp. | Cytotoxicity a | Jurkat | 30.9 µM | [60][63] | ||||||
| Tasiamide | Cyanobacterial compound derived from Symploca sp. | Inhibiting the expression of Cath D | KB; | 0.48 μg/mL; | [61][62][64,66] | ||||||
| LoVo | 3.47 μg/mL | ||||||||||
| Belamide A | Cyanobacterium | Tubulin polymerization inhibition |
HCT-116; | 0.74 μM; | [ |
Notes: a Mechanism is yet to be investigated; b If there are parentheses around the value, it means the GI50 value is displayed.
3.1.2. Metabolites of Sponges
Sponges are an excellent source of bioactive metabolites with novel chemical architectures. They produce a diverse array of highly modified peptides, especially cyclic peptides with nonproteinogenic amino acids and polyketide-derived moieties. The depsipeptides isolated from sponges or associated organisms are usually described as cytotoxic substances, such as Jaspamide (Jasplakinolide) [86], Geodiamolides [87], Phakellistatin [88], Microsclerodermin A [84] and Scleritodermin A [85]. However, some of those researched have an anticancer mechanism for a specific cancer cell. Jaspamide (jasplakinolide, NSC-613009,
Jaspis Johnstoni in 1986, has been considered a classical actin stabilizer [89]. Jaspamide-induced apoptosis is associated with caspase-3 activation, and increased Bax level, and decreased Bcl-2 protein expression. It exhibits antitumor activity in multiple in vitro tumor models for prostate and breast carcinomas and acute myeloid leukemia [79][80]. Geodiamolides A, B (
Geodia sp. in 1987 [
| HCT-15 | |||||
| 1 µM | |||||
| Trapoxin | |||||
| Fungal product the culture broth of | |||||
| Helicoma ambiens | |||||
| RF-1023 | |||||
| Inhibiting HDAC | |||||
| NIH3T3 | |||||
| 200 ng/mL | [ | 101 | ] | [ | 102][104,105] |
| Microsporin A | Microsporum cf. gypseum | Inhibiting HDAC | HCT-116 | 0.6 mg/mL; | [103][106] |
| Microsporin B | HCT-116 | 8.5 mg/mL | |||
| 63 | |||||
| ] | |||||
| [ | |||||
| 67 | |||||
| ] | |||||
| A-10 | |||||
| 20 μM | |||||
| Symplostatin A | |||||
| Cyanobacterium | |||||
| Microtubule assembly Inhibiting cell cycle arrest | |||||
| MDA-MB-435 | |||||
| 0.15 μM | |||||
| [ | |||||
| 64 | |||||
| ] | |||||
| [ | |||||
| 68 | |||||
| ] | |||||
| SK-OV-3; | |||||
| 0.09 μM | |||||
| NCI/ADR; | 2.90 μM | ||||
| NCI/ADR with Verapamil; | 0.09 μM | ||||
| A-10; | 1.8 μM | ||||
| HUVEC | 0.16 μM | ||||
| Proximicins C | Actinomycetes of the genus Verrucosispora, | Inducing Cell cycle G1 to S phase arrest and inducing apoptotic cell death | U-87 MG; | 12.7 μg/mL | [65][69] |
| MDA-MD-231 | 11.4 μg/mL | ||||
| Bisebromoamide | Cyanobacterium of the genus Lyngbya sp. | Inhibiting both the Raf/MEK/ERK and PI3K/Akt/mTOR pathways | JFCR39 | (40 nM) | [66][67][70,71] |
| HVLSRAPR | Spirulina platensis | Cytotoxicity a | HT-29; | 99.88 µg/mL | [68][72] |
Notes: a Mechanism is yet to be investigated; b If there are parentheses around the value, it means the GI50 value is displayed.
Diplosoma virens
50
50
50 values of 90.31 to 60.21 μg/mL [43][44].

Figure 3. The structures of bioactive marine linear peptides and derivatives with anticancer potential.
With the deepening of understanding, researchers have discovered that a variety of marine small peptides can induce tumor cell apoptosis to exert anti-tumor effects. The peptide sequence was identified as YALPAH (
Setipinna taty
50
Tegillarca granosa
50
Table 1), had been successfully identified as a new inhibitor of the KV3.4 channel subunits. In particular, it had been reported that KV3.4 channels play a crucial role in cancer cell migration [69]. BDS-I blocking KV3.4 currents prevented (the neurotoxic β-amyloid peptide1–42) Aβ1−42-induced caspase-3 activation and apoptotic processes [48][49].
Moreover, several small peptides are closely associated with the mitochondrial-mediated apoptosis pathway. The hexapeptide FIMGPY (
Raja porosa
50 of 4.81 mg/mL. It also could induce apoptosis by upregulating the Bax/Bcl-2 ratio and caspase-3 activation [50]. The anticancer peptide AAP-H (
Anthopleura anjunae with an amino acid sequence Tyr-Val-Pro-Gly-Pro. It has been shown that AAP-H induces apoptosis by decreasing the mitochondrial membrane potential and increasing Bax/Bcl-2 ratio, cytochrome-C, caspase-3, and caspase-9 [51]. An antiproliferative pentapeptide ILYMP (
Cyclina sinensis
ILYMP enhances Bax and cleaved caspase-3/9 expression and the suppression of Bcl-2 expression in DU-145 cells [52].
Apoptosis is closely related to cell cycle arrest. At present, some small peptides discovered cannot only induce cancer cells apoptosis, but also cause cell cycle arrest and ultimately lead to cell death. The sequences of SCH-P9 and SCH-P10 (
Sinonovacula constricta hydrolysates. The researches illustrated that SCH-P9 and SCH-P10 inhibited the growth of DU-145 cells and PC-3 cells by reducing the number of cells in the G0/G1 phase, thus increasing the number in the sub G1 phase and inducing apoptosis [53]. SIO (
sepia ink. The research found that it significantly inhibited the proliferation of DU-145, PC-3, LNCaP (human prostate cancer cells), A549 and H-1299 cells, in a time and dose-dependent manner by inducing apoptosis and arresting cell at S or G2/M phase [54][55]. The anticancer mechanism is similar to another decapeptide SHP, which is accompanied by the activation of cellular tumor antigen p53 and caspase-3, the upregulation of pro-apoptosis regulator Bax, and the downregulation of anti-apoptosis regulator Bcl-2 [70]. Psammaplin A (PsA) (
Figure 3) is a natural product that has been isolated from sponges and has been suggested to be a promising novel HDAC (histone deacetylase) inhibitor. Some researchers found that PsA exhibited antiproliferative effects on cancer cells by the induction of cell cycle arrest and apoptosis. However, the psammaplin class has the disadvantage of physiologic instability [56][57]. Latest research reports that the indole derivatives of Psammaplin are more potent modulators of epigenetic enzymes than the original natural product. Additionally, positional isomers at the bromoindole ring also showed cell cycle block and apoptosis induction [57]. NVP-LAQ824 (
Table 1) is a more stable indolic cinnamyl hydroxamate analogue of Psammaplin A, has entered phase I clinical trials in patients with solid tumors or leukemia [58]. A toxicity evaluation in rats identified the hematopoietic and lymphatic systems as the primary target organs, with a reversible dose-dependent reduction in RBC (red blood cell) and WBC (white blood cell) counts and lymphoid atrophy [58].
2.2. Fungi and Bacteria
Hundreds of secondary metabolites obtained from marine fungal strains revealed potent pharmacological and biological activities [71]. Lucentamycins A–D (
2.2. Fungi and Bacteria
Hundreds of secondary metabolites obtained from marine fungal strains revealed potent pharmacological and biological activities [61]. Lucentamycins A–D (Nocardiopsis lucentensis
50 values of 0.20 and 11 µM [59]. Two highly modified linear tetrapeptides, Padanamides A and B (
Notes: a Mechanism is yet to be investigated; b If there are parentheses around the value, it means the GI50 value is displayed.
Microsporum
gypseum. Since ring-opening enzymes easily inactivate natural products, many analogues have been synthesized and modified to provide better stability, and 86 analogues have been reported synthesized [99]. Recently researchers have shown that Sansalvamide A and Sansalvamide analogues inhibit cell growth and proliferation, and induce cell apoptosis by regulating the expression of HSP90 [99][100]. New research has shown that HSP90A strengthens AKT activation through TCLlA-stabilization, promoting multi-aggressive properties in tumor cells [104].
Trapoxin (
l-Aoe) [105][106]. Some finding displayed that the primary target molecule of the agent in vivo is the histone deacetylase itself, and it also induced growth inhibition in several cell lines regardless of their p53 status [101][102]. Other cyclic peptides, Microsporins A and B (
Microsporum
gypseum
50
S
50 values of 0.14 and 0.55 µM, respectively [103].
3.3. BacteriaBacterial proteins and peptides are a class of promising bioactive compounds and potential anticancer drugs [107]. Mixirins are cyclic acyl-peptides derived from the bacterium
Bacillus
species. Mixirins A, B and C (
and
) blocked the growth of the HCT-116 with an IC
50
value at the level of 0.65, 1.6, and 1.26 µM, respectively [108]. A new cytotoxic substance named Mechercharmycin A (
and
) was isolated from
Thermoactinomyces
sp. YM3-251 showed relatively strong antitumor activity against A549 and Jurkat cells with an IC
50
value of 40 nM and 46 nM, respectively [109]. Urukthapelstatin A (
and
), a novel cyclic peptide, was isolated from the cultured mycelia of Thermoactinomycetaceae bacterium
Mechercharimyces asporophorigenens
YM11-542. Research showed that Urukthapelstatin A inhibited human growth lung cancer A549 cells with an IC
50
value of 12 nM. Recently analogues of Urukthapelstatin A were synthesized. Cytotoxicity data showed the phenyl ring attached to the eastern oxazole and the rigid, lipophilic tripeptide section are critical structural features of the bio-activity [110]. Three new cyclohexadepsipeptides, Arenamides A–C (
and
), were isolated from the fermentation broth of a bacterial strain, identified as
Salinispora arenicola
. Arenamide A and B’s effect on NF-κB activity was studied with stably transfected 293/NF-κB-Luc human embryonic kidney cells, induced by treatment with tumor necrosis factor (TNF). Arenamides A and B blocked TNF-induced activation in a dose- and time-dependent manner with IC
50
values of 3.7 and 1.7 μM, respectively [111].
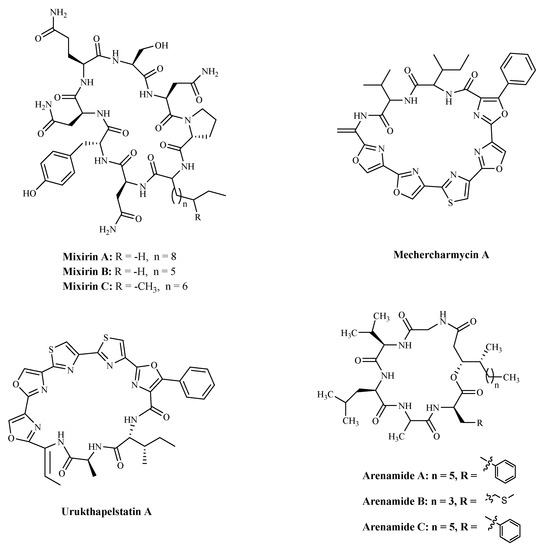
Figure 6. The structures of marine bioactive cyclic peptides from Bacteria.
Table 4.
Marine bioactive cyclic peptides from Bacteria.
| Compound
Compound |
Source
Source |
Mechanism
Mechanism |
Cell Lines
Cell Lines |
IC50
IC50 |
Reference
Reference |
|---|---|---|---|---|---|
| Mixirin A
Mixirin A |
Bacillus species.
Bacillus species. |
Cytotoxicity a
Cytotoxicity a |
HCT-116
HCT-116 |
0.65 µM;
0.65 µM; |
[108]
[108] |
| Mixirin B
Mixirin B |
HCT-116
HCT-116 | ||||
Notes: a Mechanism is yet to be investigated.
