Root-lesion nematodes of the genus Pratylenchus are migratory endoparasites belonging to the family Pratylenchidae (Nematoda, Tylenchina), with around 100 species recognized today.
- Pratylenchus,root lesion nematode
- Pratylenchus
- root lesion nematode
1. Introduction
Pratylenchus species can cause yield losses of up to 85% of expected production [4], and even higher losses when nematodes interact synergistically with certain soilborne plant pathogens [5]. Hence, Pratylenchus species are highly relevant to agriculture.
Israel is a small semiarid country located in western Asia, only 22,000 km2 in size. Despite the fact that the geography of the country is not naturally conducive to agriculture, advanced irrigation, cultivation mastery, use of elite varieties, and the introduction of state-of-the-art agricultural technologies contribute, in practice, to intensive and efficient farming. On the other hand, this success in agricultural productivity along with a diversity of climatic conditions have led to the proliferation of devastating plant-parasitic nematodes. Among them, Pratylenchus species are widely distributed in vegetable and crop fields in Israel and are associated with a major reduction in quality and yield. The genus Pratylenchus was first reported from Israel in 1957 [6]. Since then, several studies related to this nematode have been published [7,8,9,10,11,12,13,14][7][8][9][10][11][12][13][14]. However, these studies are largely scattered. Some of them are published in less accessible local journals, such as master’s or PhD theses, or in scientific reports written in Hebrew.
2. Biology and Pathogenicity of Pratylenchus Species
Pratylenchus species are polyphagous, migratory root endoparasites, developing and reproducing in the soil or roots. Their life cycle is simple and direct. The female lays its eggs singly or in small groups in the host root or in the soil near the root surface. Although little information is available about the true length of the Pratylenchus life cycle, on the basis of laboratory observations, research has estimated it to last from 45 to 65 days [45]. Symptoms caused by Pratylenchus are variable and depend on the host; they can include stunted and inefficient plant growth with reduced numbers of tillers and yellowed leaves.
Pathogenicity studies indicate that Pratylenchus species are very well adapted to parasitism, as extremely high populations in the soil do not kill their host plants. Nevertheless, damage thresholds range from 0.05 to 30 nematodes/cm3 of soil. Apart from direct damage to the roots, Pratylenchus species may also predispose plants to other pathogens (e.g., Verticillium and Fusarium), thereby increasing the damage extent [46,47][46][47]. Consequently, elimination of the nematode or reduction of its population causes a marked reduction in the incidence of fungi and an increase in crop yield. In Israel, the synergistic relationship between P. thornei and the fungus Verticillium dahliae caused a significant increase in the populations of both pathogens and in their damage to potato crops in the northern Negev [48].
Among the nine species recorded in Israel, P. mediterraneus, P. thornei, and P. capsici have been relatively more studied, and their biology and pathogenicity are discussed below.
Pratylenchus mediterraneus parasitism occurs mainly in the winter, but the nematode can survive for 7–8 months in a state of anhydrobiosis during the hot and dry season [8,49][8][49]. It is reactivated by the subsequent winter rains. In a field observation conducted by Orion et al. [10] [10] from 1974 to 1983, the highest population of P. mediterraneus (as P. thornei in their paper) was recorded in the drought of 1978 and partial drought of 1982, and the lowest population in the unusually wet years of 1980 and 1983. Moreover, nematode populations with auxiliary irrigation treatments were extremely low. These data suggest that low moisture level—the natural condition in the northern Negev region—is a major ecological factor required for P. mediterraneus to build up its population, supporting the notion that P. mediterraneus is native to the semiarid zones of the Middle East [8][19] [8,19] or, more specifically, the eastern Mediterranean region [50]. During the long hot season (April–November), the nematode population level remains stable due to anhydrobiosis [8]. In this state, the nematode can withstand conditions of 0% relative humidity, and desiccated nematodes can withstand temperatures of up to 40 °C. This characteristic enables their survival and facilitates their field or regional transmission in the northern Negev, where soil temperatures typically reach 40 °C in the hot season. This species is also likely to require the higher temperatures found in the Mediterranean region for its development, but this needs to be further studied.
In contrast to P. mediterraneus, the optimal temperature for P. thornei reproduction seems to be lower, ranging between 20 and 25° C [51,52][51][52], suggesting that the northern Negev may not be a suitable area for its survival. However, our molecular- and morphological-based analyses suggested that P. thornei is present not only in the mild northern Israel (Mesilot, Avuka, Shif’a), but also in the hot and dry region of the northern Negev [41]. In comparison, P. mediterraneus was only recovered from the northern Negev, suggesting that P. thornei may be able to adapt to a wider range of environmental conditions than P. mediterraneus, with the latter being more specialized for the hot and dry northern Negev.
The pathogenic effect of P. mediterraneus is limited to the early plant stages, resulting in reduced foliage and root growth of cereals and legumes, and thus influencing final plant density at harvest [12,14][12][14]. Pratylenchus mediterraneus was shown to be most concentrated in the root-tip region of hosts Vicia sativa and Trifolium alexandrinum. A histopathological study using scanning electron microscopy (SEM) showed nematodes penetrating the root epidermis and the cortical parenchyma through a clean-cut hole, probably a result of enzymatic activity and mechanical force [53]. When passing through parenchyma cells, P. mediterraneus can consume the cell contents, and these cells are thus void of cytoplasmic structures compared to the prominent nuclei and cytoplasmic structures in adjacent intact cells [12]. Typical symptoms caused by P. mediterraneus on common vetch were lesions produced along roots. These lesions lacked root hairs, with necrotic epidermal cells consisting of many holes, leading to severely deformed roots. Similar to P. penetrans [54], Orion and co-workers [12,37][12][37] speculated that P. mediterraneus can infect root tips as ectoparasites as well. Further SEM analysis showed the collapse of the parenchyma cells in the root lesion as the result of nematode feeding activity. The observed destruction was limited to the root cortex with an intact central cylinder, while nematode egg deposition was observed in cavities of the root cortex. These findings were similar to observations of P. vulnus in sour orange [55], P. penetrans in broad beans [56], and Pratylenchus zeae in maize [57].
Pratylenchus penetrans and P. crenatus Loof, 1960 have been reported worldwide as the major causal agent of carrots and Kuroda-type carrots [58,59,60,61][58][59][60][61]. In an investigation of carrot nematodes in Shoval, located in the northern Negev, we failed to detect these species. Instead, the field was infested with P. thornei, resulting in significant quality loss due to forking of carrot taproots [41]. However, whether P. thornei is the causal agent of these symptoms still needs to be confirmed, as continuous sampling from carrots demonstrated that the forking symptoms were not necessarily related to nematode occurrence [41].
Pratylenchus capsici is an endemic Israeli species that is widely distributed in the Arava Rift Valley, causing significant yield reduction of pepper (Figure 2).

Figure 2. Symptoms caused by Pratylenchus capsici. (A) Pepper plant decline in the Arava Rift Valley characterized by stunted growth and wilting. (B) Heavily infected roots, with pronounced lesions along primary and secondary roots. (C) Photograph of developed root lesion taken under a dissecting microscope.
The emergence of this species was surprising, as this remote region is isolated from the country’s other agricultural areas. Moreover, until 1995, the entire region was free of reported nematodes, mainly due to intensive soil fumigation with methyl bromide [62]. Since the phase-out of this fumigant, certain species of Meloidogyne and Pratylenchus have become established in the soil, causing substantial damage to vegetable crops. Further biogeographical analysis suggested that a P. capsici population in weeds (Chenopodium album and Sonchus oleraceus) was an important source for P. capsici dispersal, either as the original nematode source or in maintaining the population between growing seasons (Figure 3).

Figure 3. Weed distribution and function as a reservoir for Pratylenchus capsici during and in between growing seasons. (A) Weeds emerging early after pepper seedling planting, and (B) throughout the pepper-growing season. (C) Lesions caused by P. capsici on Chenopodium album growing alongside the pepper plants.
Similar findings were observed for P. penetrans [63], P. brachyurus [64[64][65],65], Pratylenchus coffeae [66], P. zeae [67], P. scribneri and P. vulnus [68], and P. thornei and P. neglectus [69].
Pratylenchus capsici has been shown to survive through the seasons with no host from April to July. During this period, nematode activation is prevented by the high temperature and low moisture in the soil. Extensive nematode extraction from roots and soils yielded a high number of nematodes in the former and low numbers in the latter, supporting its exclusive endoparasitic life strategy. Therefore, these observations raise the question of whether P. capsici is ever anhydrobiotic, and if so, whether it goes through anhydrobiosis in the roots or in the soil. Similarly, P. capsici’s capacity to migrate to lower soil levels during the off seasons is not known. Further study is needed to clarify this question.
3. Phylogeny and Evolution of Pratylenchus Species Occurring in Israel
To date, nine species of Pratylenchus have been reported from Israel, with molecular data available for only three of them (P. thornei, P. mediterraneus, and P. capsici) (Figure 4). The concatenated phylogeny based on 18S and 28S rDNA and internal transcribed spacer (ITS) suggests that P. thornei and P. mediterraneus form a well-supported (posterior probability (PP) = 1, bootstrap (BS) = 100) monophyletic group, concurring with previous studies [18,24][18][24].
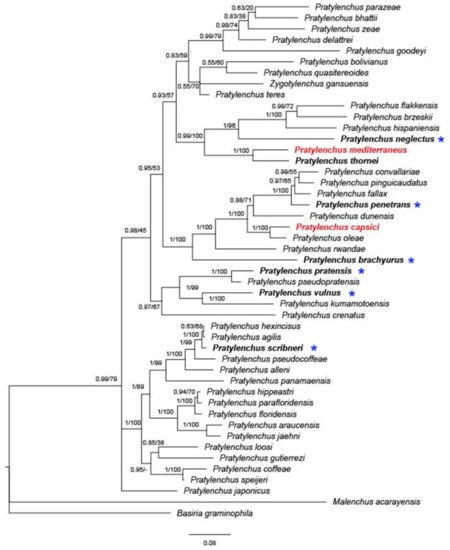
Figure 4. Bayesian 50% majority rule consensus tree inferred on concatenated sequences of 28S; asterisks indicate species that were only identified by morphology. The dataset was aligned by MAFFT v. 7.205 [70] [70] using the G-INS-i algorithm. The phylogeny was reconstructed by maximum likelihood (ML) and Bayesian inference (BI) using RAxML v.8.1.11 [71] and MrBayes 3.2.3. [72]. Branch support is indicated in the following order: posterior probability (PP) value from BI analysis/bootstrap (BS) value from ML analysis. Red marked species indicate local Israeli isolates.
Orion [50] [50] suspected that P. mediterraneus is a native or at least old inhabitant of the semiarid region of the Eastern Mediterranean. Given the similarities in morphology and morphometric features, the overlapping geographical area (Mediterranean region), the same hosts (mostly cereal and legumes), and the anhydrobiotic survival properties, P. thornei and P. mediterraneus could be derived from recent speciation events, with insufficient time to attain complete morphological differentiation.
Similarly, P. capsici is sister to P. oleae in concatenated phylogeny (Figure 4, PP = 1, BS = 100), as well as in a previous study [18]. Pratylenchus oleae was found in the Mediterranean region, parasitizing both wild and cultivated olive trees in Spain and Tunisia, with the presence of the nematode in wild olive not showing any clear symptoms in the aboveground plant or roots [3]. Interestingly, P. capsici was found in both pepper and weeds, markedly damaging the pepper but causing very mild symptoms on the weeds. Later, population genetic analysis revealed that P. capsici is likely to have been native to wild grass and transmitted to pepper by a recent expansion [18]. The adjacent distribution, similar morphology and presumably similar transmission background give rise to the idea that the two closely related species, P. capsici and P. oleae, may be native to the Mediterranean region.
References
- Geraert, E. The Pratylenchidae of the World: Identification of the Family Pratylenchidae (Nematoda: Tylenchida); Academia Press: Ghent, Belgium, 2013. [Google Scholar]
- Hodda, M.; Collins, S.J.; Vanstone, V.A.; Hartley, D.; Wanjura, W.; Kehoe, M. Pratylenchus quasitereoides n. sp. from cereals in Western Australia. Zootaxa 2014, 3866, 277–288. [Google Scholar] [CrossRef]
- Palomares-Rius, J.E.; Guesmi, I.; Horrigue-Raouani, N.; Cantalapiedra-Navarrete, C.; Liébanas, G.; Castillo, P. Morphological and molecular characterisation of Pratylenchus oleae n. sp.(Nematoda: Pratylenchidae) parasitizing wild and cultivated olives in Spain and Tunisia. Eur. J. Plant Pathol. 2014, 140, 53–67. [Google Scholar] [CrossRef]
- Nicol, J.; Turner, S.; Coyne, D.; Den Nijs, L.; Hockland, S.; Maafi, Z.T. Current nematode threats to world agriculture. In Genomics and Molecular Genetics of Plant-Nematode Interactions; Springer: Dordrecht, The Netherlands, 2011; pp. 21–43. [Google Scholar]
- Jones, M.; Fosu-Nyarko, J. Molecular biology of root lesion nematodes (Pratylenchus spp.) and their interaction with host plants. Ann. Appl. Biol. 2014, 164, 163–181. [Google Scholar] [CrossRef]
- Minz, G. Free-living plant-parasitic and possible plant-parasitic nematodes in Israel. Plant Dis. Report. 1957, 41, 92–94. [Google Scholar]
- Cohn, E.; Sher, S.; Bell, A.; Minz, G. Soil Nematodes Occurring in Israel; The Volcani Center: Rishon Letzion, Israel, 1973. [Google Scholar]
- Glazer, I.; Orion, D. Studies on anhydrobiosis of Pratylenchus thornei. J. Nematol. 1983, 15, 333. [Google Scholar]
- Krikun, J.; Orion, D. Verticillium wilt of potato: Importance and control. Phytoparasitica 1979, 7, 107. [Google Scholar] [CrossRef]
- Orion, D.; Amir, J.; Krikun, J. Field observations on Pratylenchus thornei and its effects on wheat under arid conditions. Rev. Nématol. 1984, 7, 341–345. [Google Scholar]
- Orion, D.; Glazer, I. Nematicide seed dressing for Pratylenchus mediterraneus control in wheat. Phytoparasitica 1987, 15, 225–228. [Google Scholar] [CrossRef]
- Orion, D.; Lapid, D. Scanning Electron-Microscope Study on the Interaction of Pratylenchus-Mediterraneus and Vicia-Sativa Roots. Nematologica 1993, 39, 322–327. [Google Scholar] [CrossRef]
- Orion, D.; Nachmias, A.; Lapid, D.; Orenstein, J. Observations on the parasitic behavior of Pratylenchus mediterraneus on excised potato roots. Nematropica 1995, 25, 71–74. [Google Scholar]
- Orion, D.; Shlevin, E. Nematicide seed dressing for cyst and lesion nematode control in wheat. J. Nematol. 1989, 21, 629. [Google Scholar] [PubMed]
- Goldreich, Y.; Karni, O. Climate and precipitation regime in the Arava Valley, Israel. Isr. J. Earth Sci. 2001, 50, 53–59. [Google Scholar] [CrossRef]
- Yair, A. Runoff generation in a sandy area—The Nizzana sands, Western Negev, Israel. Earth Surf. Process. Landf. 1990, 15, 597–609. [Google Scholar] [CrossRef]
- Pasternak, D.; De Malach, Y.; Borovic, I.; Shram, M.; Aviram, C. Irrigation with brackish water under desert conditions IV. Salt tolerance studies with lettuce (Lactuca sativa L.). Agric. Water Manag. 1986, 11, 303–311. [Google Scholar] [CrossRef]
- Qing, X.; Bert, W.; Gamliel, A.; Bucki, P.; Duvrinin, S.; Alon, T.; Braun Miyara, S. Phylogeography and Molecular Species Delimitation of Pratylenchus capsici n. sp., a New Root-Lesion Nematode in Israel on Pepper (Capsicum annuum). Phytopathology 2019, 109, 847–858. [Google Scholar] [CrossRef]
- Orion, D.; Krikun, J.; Sullami, M. The distribution, pathogenicity and ecology ofPratylenchus Thornei in the northern negev. Phytoparasitica 1979, 7, 3–9. [Google Scholar] [CrossRef]
- Cobertt, D.; Clark, A. Surface features in the taxonomy of Pratylenchus species. Rev. Néinatol 1983, 6, 85–98. [Google Scholar]
- Ryss, A.Y. Root Parasitic Nematodes of the Family Pratylenchidae (Tylenchida) of the World Fauna; Nauka: Leningrad, Russia, 1988. [Google Scholar]
- Café Filho, A.C.; Huang, C. Description of Pratylenchus pseudofallax n. sp. with a key to species of the genus Pratylenchus Filipjev, 1936 (Nematoda: Pratylenchidae). Rev. Nématol. 1989, 12, 7–15. [Google Scholar]
- Castillo, P.; Vovlas, N. Pratylenchus (Nematoda: Pratylenchidae): Diagnosis, biology, pathogenicity and management. In Nematology Monographs and Perspectives; Brill: Leiden, The Netherlands, 2007; Volume 6. [Google Scholar]
- De Luca, F.; Reyes, A.; Troccoli, A.; Castillo, P. Molecular variability and phylogenetic relationships among different species and populations of Pratylenchus (Nematoda: Pratylenchidae) as inferred from the analysis of the ITS rDNA. Eur. J. Plant Pathol. 2011, 130, 415–426. [Google Scholar] [CrossRef]
- Troccoli, A.; De Luca, F.; Handoo, Z.; Di Vito, M. Morphological and molecular characterization of Pratylenchus lentis n. sp.(Nematoda: Pratylenchidae) from Sicily. J. Nematol. 2008, 40, 190. [Google Scholar]
- Waeyenberge, L.; Ryss, A.; Moens, M.; Pinochet, J.; Vrain, T. Molecular characterisation of 18 Pratylenchus species using rDNA restriction fragment length polymorphism. Nematology 2000, 2, 135–142. [Google Scholar] [CrossRef]
- de Luca, F.; Fanelli, E.; Di vito, M.; Reyes, A.; De giorg, C. Comparison of the sequences of the D3 expansion of the 26S ribosomal genes reveals different degrees of heterogeneity in different populations and species of Pratylenchus from the Mediterranean region. Eur. J. Plant Pathol. 2004, 110, 949–957. [Google Scholar] [CrossRef]
- Corbett, D. Three new species of Pratylenchus with a redescription of P. andinus Lordello, Zamith & Boock, 1961 (Nematoda: Pratylenchidae). Nematologica 1983, 29, 390–403. [Google Scholar]
- Greco, N.; Di Vito, M.; Saxena, M.; Reddy, M. Investigation on the root lesion nematode, Pratylenchus thornei, in Syria. Nematol. Mediterr. 1988, 16, 101–105. [Google Scholar]
- Saxena, M.C.; Singh, K. The Chickpea [Cicer Arietinum]; Commonwealth Agricultural Bureaux International: Wallingford, UK, 1987. [Google Scholar]
- Di Vito, M.; Greco, N.; Ores, G.; Saxena, M.; Singh, K.; Kusmenoglu, I. Plant parasitic nematodes of legumes in Turkey. Nematol. Mediterr. 1994, 22, 245–251. [Google Scholar]
- Kepenekcİ, I. Plant parasitic nematode species of Tylenchida (Nematoda) associated with sesame (Sesamum indicum L.) growing in the Mediterranean region of Turkey. Turk. J. Agric. For. 2002, 26, 323–330. [Google Scholar]
- Greco, N.; Di Vito, M. Nematodes of food legumes in the Mediterranean Basin 1. Eppo Bull. 1994, 24, 393–398. [Google Scholar] [CrossRef]
- Greco, N.; Di Vito, M.; Saxena, M. Plant parasitic nematodes of cool season food legumes in Syria. Nematol. Mediterr. 1992, 20, 37–46. [Google Scholar]
- Troccoli, A.; Di Vito, M. Root lesion and stem nematodes associated with faba bean in North Africa. Nematol. Mediterr. 2002, 30, 79–81. [Google Scholar]
- Choi, D.-R.; Lee, J.-K.; Parte, B.-Y.; Han, H.-R.; Choi, Y.-E. A new and one unrecorded species of Pratylenchus from Korea (Nematoda: Pratylenchidae). J. Asia-Pac. Entomol. 2006, 9, 5–9. [Google Scholar] [CrossRef]
- Orion, D.; Shlevin, E.; Yaniv, A. Controlling the migratory nematode Pratylenchus mediterraneus improves carrot yield quality. Hassadeh 1988, 69, 72–74. [Google Scholar]
- Development MoAaR. Database of Plant Pests in Israel: Ministry of Agriculture and Rural Development; Development MoAaR: Rishon Letzion, Israel, 2020. [Google Scholar]
- Nicol, J. The Distribution, Pathogenicity and Population Dynamics of Pratylenchus Thornei on Wheat in South Australia; Department of Crop Protection, University of Adelaide: Adelaide, Australia, 1996. [Google Scholar]
- Nicol, J.; Rivoal, R.; Taylor, S.; Zaharieva, M. (Eds.) Global importance of cyst (Heterodera spp) and lesion nematode (Pratylenchus spp.) on cereals: Distribution, yield loss, use of host resistance and integration of molecular tools. In Proceedings of the Fourth International Congress of Nematology, Tenerife, Spain, 8–13 June 2002. [Google Scholar]
- Braun Miyara, S.; Nematology Unit, Plant Protection Institute, ARO, The Volcani Center, Rishon Letzion, Israel. Personal communication, 2020.
- Kozodoi, E.; Plant Protection and Inspection Services, Rishon Letzion, Israel. Personal Communication, 2020.
- Pivonia, S.; Central-and Northern-Arava Research and Development, Arava Sapir, Israel. Personal Communication, 2020.
- Oka, Y. Nematicidal activity of fluensulfone against some migratory nematodes under laboratory conditions. Pest Manag. Sci. 2014, 70, 1850–1858. [Google Scholar] [CrossRef]
- Agrios, G.N. Introduction to Plant Pathology; Academic Press: New York, NY, USA, 1988; pp. 3–39. [Google Scholar]
- Rotenberg, D.; MacGuidwin, A.; Saeed, I.; Rouse, D. Interaction of spatially separated Pratylenchus penetrans and Verticillium dahliae on potato measured by impaired photosynthesis. Plant Pathol. 2004, 53, 294–302. [Google Scholar] [CrossRef]
- Rowe, R.C.; Powelson, M.L. Potato early dying: Management challenges in a changing production environment. Plant Dis. 2002, 86, 1184–1193. [Google Scholar] [CrossRef]
- Siti, E. The interrelationships between Pratylenchus tornei and Verticilium daliae, and their effect on potatoes. In Jerusalem HUo; Faculty of Agriculture—Hebrew University of Jerusalem: Rehovot, Israel, 1978. [Google Scholar]
- Talavera, M.; Valor, H.; Tobar, A. Post-anhydrobiotic viability of Pratylenchus thornei and Merlinius brevidens. Phytoparasitica 1998, 26, 293. [Google Scholar] [CrossRef]
- Orion, D. Nematodes of agricultural importance in Israel. Nematology 2000, 2, 735–736. [Google Scholar] [CrossRef]
- Castillo, P.; Trapero-Casas, J.; Jiménez-Díaz, R. Effect of time, temperature, and inoculum density on reproduction of Pratylenchus thornei in carrot disk cultures. J. Nematol. 1995, 27, 120. [Google Scholar]
- Thompson, J.; Clewett, T.; O’Reilly, M. Temperature response of root-lesion nematode (Pratylenchus thornei) reproduction on wheat cultivars has implications for resistance screening and wheat production. Ann. Appl. Biol. 2015, 167, 1–10. [Google Scholar] [CrossRef]
- Kurppa, S.; Vrain, T.C. Penetration and feeding behavior of Pratylenchus penetrans in strawberry roots. Rev. Nématol. 1985, 8, 273–276. [Google Scholar]
- Zunke, U. Observations on the invasion and endoparasitic behavior of the root lesion nematode Pratylenchus penetrans. J. Nematol. 1990, 22, 309. [Google Scholar] [PubMed]
- Inserra, R.; Vovlas, N. Effects of Pratylenchus vulnus on the growth of sour orange. J. Nematol. 1977, 9, 154. [Google Scholar]
- Vovlas, N.; Troccoli, A. Histopathology of broad bean roots infected by the lesion nematode Pratylenchus penetrans. Nematol. Mediterr. 1990, 18, 239–242. [Google Scholar]
- De Waele, D.; Jordaan, E. Plant-parasitic nematodes on field crops in South Africa. 1. Maize. Rev. Nématol. 1988, 11, 65–74. [Google Scholar]
- Coosemans, J. The influence of Pratylenchus penetrans on growth of Impatiens balsamina L. Daucus carota L., Linum usitatissimum L. and Crysanthemum indicum L. Symp. Int. Phytopharm Gent Belg. 1975, 27, 465–471. [Google Scholar]
- Hay, F.; Pethybridge, J. Nematodes associated with carrot production in Tasmania, Australia, and the effect of Pratylenchus crenatus on yield and quality of Kuroda-type carrot. Plant Dis. 2005, 89, 1175–1180. [Google Scholar] [CrossRef]
- Potter, J.; Olthof, T.H. Nematode pests of vegetable crops. In Plant Parasitic Nematodes in Temperate Agriculture; CABI: Wallinford, UK, 1993; pp. 171–207. [Google Scholar]
- Vrain, T.; Belair, G. Symptoms induced by the lesion nematode, Pratylenchus penetrans on carrot taproots in organic soil. Phytoprotection 1981, 62, 79–81. [Google Scholar]
- Gamliel, A.; ARO-Volcani Center, Rishon LeTsiyon, Israel. Personal communication, 2020.
- Townshend, J.; Davidson, T. Some weed hosts of Pratylenchus penetrans in premier strawberry plantations. Can. J. Bot. 1960, 38, 267–273. [Google Scholar] [CrossRef]
- Hogger, C.; Bird, G. Weed and indicator hosts of plant-parasitis nematodes in georgia cotton and soybean fields. Plant Dis. Rep. 1976, 60, 223–226. [Google Scholar]
- Koen, H. Notes on the host range, ecology and population dynamics of Pratylenchus brachyurus. Nematologica 1967, 13, 118–124. [Google Scholar] [CrossRef]
- Edwards, D.; Wehunt, E. Hosts of Pratylenchus coffeae with additions from central american banana-producing areas. Plant Dis. Rep. 1973, 57, 47. [Google Scholar]
- Ayoub, S. Pratylenchus zeae found on corn, milo, and three suspected new hosts in California. Plant Dis. Rep. 1961, 45, 940. [Google Scholar]
- Manuel, J.S.; Reynolds, D.; Bendixen, L.; Riedel, R. Weeds as Hosts of Pratylenchus; Ohio Agricultural Research and Development Center: Wooster, OH, USA, 1980. [Google Scholar]
- Vanstone, V.A.; Russ, M.H. Ability of weeds to host the root lesion nematodes Pratylenchus neglectus and P. thornei I. Grass weeds. Australas. Plant Pathol. 2001, 30, 245–250. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A.; Hoover, P.; Rougemont, J. A rapid bootstrap algorithm for the RAxML web servers. Syst. Biol. 2008, 57, 758–771. [Google Scholar] [CrossRef]
- Ronquist, F.; Teslenko, M.; Van Der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542.
