Polyphenols are an important family of molecules of vegetal origin present in many medicinal and edible plants, which represent important alimentary sources in the human diet. Polyphenols are known for their beneficial health effects and have been investigated for their potential protective role against various pathologies, including cancer, brain dysfunctions, cardiovascular diseases and stroke.
- polyphenols
- flavonoids
- stroke
- polyphenols,flavonoids, stroke
1. Polyphenols: Definition and Classification
Polyphenols are molecules chemically characterized by the presence of at least one aromatic ring with one or more hydroxyl groups attached [1][2]. Polyphenols are plant secondary metabolites that are thought to help plants to survive and proliferate, protecting them against microbial infections or herbivorous animals, or luring pollinators [3]. Polyphenols are found in many medicinal and edible plants which represent important alimentary sources, including fruits, vegetables, beverages (such as tea and red wine) and extra virgin oil [4].
This group of natural products includes a broad number of different compounds, ranging from simple molecules with low molecular weight to complex and large derived polyphenols [1][2]. According to their chemical structure, polyphenols can be classified into various classes including flavonoids, phenolic acids, stilbenes, curcuminoids, lignans, ellagitannins and ellagic acid and coumarins [1][2]. Flavonoids are structurally based on a skeleton of fifteen carbons, with two aromatic rings connected by a three-carbon bridge. They are the most numerous of polyphenols and are widely distributed through the plant kingdom [1][2]. The main subclasses of dietary flavonoids include flavonols, flavan-3-ols, flavanones, flavones, isoflavones, anthocyanins, dihydrochalcones and proanthocyanidins [1][2]. Among the non-flavonoid polyphenols, phenolic acids can be further divided into hydroxycinnamic acids and hydroxybenzoic acids [1][2]. Figure 1 depicts the classification of polyphenols and describes some known food sources for each molecular class.
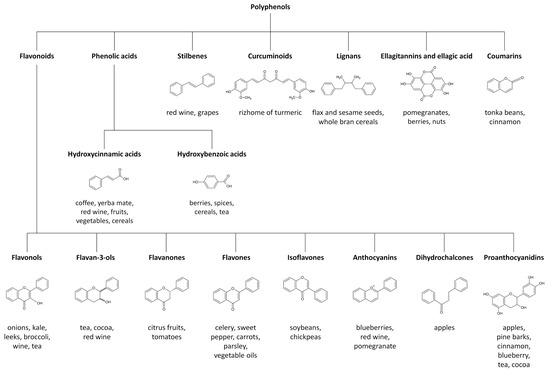
Figure 1.
Major polyphenol classes and their most important food sources.
Among the many micronutrients present in plants, polyphenols are the most numerous and particularly endowed with beneficial properties [5]. For these reasons, polyphenols have been widely investigated for the prevention and treatment of several pathological conditions, including cancer, neurodegenerative disorders, metabolic and cardiovascular diseases and stroke [6][7][8].
2. Polyphenol Metabolism: Role of Gut Microbiota
Polyphenol bioavailability is generally poor, and only 1–10% of total polyphenol intake is detectable in blood and urine samples [6]. Bioavailability is particularly low for flavones, stilbenes and curcumin and is slightly higher for tea flavan-3-ols, flavanones in citrus fruits, soy isoflavones and red wine anthocyanidins [9][10][11]. However, maximum polyphenol concentration in plasma remains extremely low and rarely exceeds 1 μM, even in individuals consuming a polyphenol-rich diet [9]. How polyphenols exert their beneficial actions despite their poor bioavailability is not clear yet. A possible explanation may rely on the fact that many polyphenol metabolites exhibit a biological activity [12].
Polyphenols are generally consumed with the diet or as supplements. A proportion of ingested polyphenolic compounds can be absorbed in the small intestine and metabolized by phase II enzymes. However, the major part of polyphenols reach the large intestine where they are degraded by intestinal microbiota. A large body of evidence indicates a fundamental role of colonic microorganisms in determining the bioavailability and activity of polyphenols by transforming them into readily absorbable molecules or biologically active metabolites [5][13][14]. The relationship between polyphenols and microbiota is bidirectional and, if the intestinal bacteria modulate polyphenol metabolism, polyphenolic compounds can in their turn influence the composition of the microbial population [5][15].
Different findings suggest the gut microbiota could modulate the activity of polyphenols potentially active against stroke. For example, flavan-3-ols, phenolic compounds characterized by a generally low bioavailability, are extensively metabolized by host and gut microbiota enzymes. Phenyl-γ-valerolactones and phenylvaleric acids, the main microbial metabolites of flavan-3-ols, might be responsible for the beneficial effects attributed to their parent compounds, including neuroprotection [16]. Daidzein, an isoflavone endowed with beneficial properties enriched in soy food, is metabolized by gut microbiota to equol, which possesses higher antioxidant activity and affinity for estrogen receptors than the parent compound. The neuroprotective flavone glycoside baicalin (baicalein 7-O-glucuronide) can only be absorbed after hydrolysis by gut microbiota β-glucuronidase to the aglycone form baicalein [17]. Similarly, the neuroprotective anthocyanin cyanidin-3-O-glucoside displays a poor bioavailability, while its microbiota degradation products are more easily absorbable [18]. Ellagic acid and ellagitannins are degraded by intestinal microorganisms to form urolithins, molecules characterized by higher bioavailability and better anti-inflammatory and antioxidant properties than their compounds of origin [19].
3. Polyphenols and Stroke: Mechanisms of Action
The major mechanism of natural polyphenols in preventing stroke relies on their protective action on the cardiovascular system [7][20][21].
Many polyphenols are endowed with anticoagulant and antiplatelet activities, potentially contributing to the prevention of thrombus formation, the main cause of ischemic stroke [22][23]. For example, several coumarin derivatives exert anticoagulant properties by inhibiting the vitamin K epoxide reductase complex and are widely used as clinical anticoagulant agents [24]. Among the polyphenols endowed with antiplatelet activity, the isoflavones genistein and daidzein possess a marked and physiologically relevant cyclooxygenase-1 (COX-1) inhibitory activity [25]. Other flavonoids with antiaggregant effects, including the isoflavone tectorigenin, have been reported to act as antagonists on thromboxane receptors [25][26].
Hypertension, a long-term medical condition affecting millions of individuals worldwide [27], is an important risk factor in particular for intracerebral hemorrhage (ICH) and subarachnoid hemorrhage (SAH) subtypes of stroke [28][29]. Dietary intake of flavonoids belonging to anthocyanin, flavone and flavan-3-ol subclasses may contribute to the prevention of hypertension [30][31]. The underlying biological mechanisms by which polyphenols regulate blood pressure include vasodilation through the regulation of nitric oxide (NO) and endothelium-derived hyperpolarizing factor (EDHF) [20][30][31].
Besides the well-documented beneficial effects of polyphenols on cardio- and cerebrovascular systems, a growing number of studies in cellular and animal stroke models indicates a direct protective effect of many polyphenols on the brain. Notably, several polyphenols exert neuroprotective actions in preclinical models even when administered after stroke induction, indicating that these molecules may be useful not only in increasing resilience to brain damage, but also for the recovery of patients suffering from stroke. Moreover, the fact that different polyphenolic compounds act on the same molecular pathways raises the possibility that they may promote synergistic effects at very low doses. Therefore, the possible synergistic effect between polyphenols with each other or with other compounds may provide the rationale to overcome the limitations caused by the poor bioavailability of these molecules.
At the mechanistic level, polyphenols exert their neuroprotective benefits by acting on several targets simultaneously. These compounds are generally strong antioxidants, working as reactive oxygen species (ROS) scavengers and metal chelators due to the presence of hydroxyl groups and neutrophilic centers [6]. Furthermore, many polyphenols are able to activate transcription factors involved in antioxidant-responsive element pathways, such as erythroid 2-related factor 2 (Nrf2), thus promoting the expression of antioxidant enzymes including superoxide dismutase (SOD), heme oxygenase-1 (HO-1), catalase, glutathione reductase and glutathione-S-transferase [32].
Apoptosis is a process that can play a primary role in various pathologies, including cardiovascular diseases and stroke [33]. Many polyphenols are able to interact with proteins regulating apoptosis, including proapoptotic (Bax, Bad) and antiapoptotic (Bcl-2, Bcl-XL) members of Bcl-2 family, p53, mitogen-activated protein kinases (MAPKs) and protein kinase B (AKT) [33]. These compounds can act as pro- or antiapoptotic agents, depending on their concentrations, cellular system and stage of pathological process [33].
The polyphenol-mediated neuroprotection not only involves a direct effect on neurons, but also modulatory effects on different inflammation players in the brain, including microglia and mast cells (MCs) [34][35]. The anti-inflammatory properties of polyphenols are based on their capability to interfere with immune cell regulation, inflammatory gene expression and the synthesis of inflammatory mediators [36]. For example, a number of polyphenols have been shown to modulate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB), toll-like receptor (TLR) and arachidonic acid pathways, suppressing the production of tumor necrosis factor α (TNF-α), interleukin (IL)-1β, IL-6, IL-1 and IL-8, as well as cyclooxygenase-2 (COX-2), inducible nitric oxide synthase (iNOS) and nitric oxide (NO) [36].
Epigenetic modifications, including DNA methylation, histone modifications and RNA-based mechanisms, modify gene expression without altering the DNA sequence. Epigenetic modifications regulate important physiological processes in living organisms, but they have also been associated with the pathogenesis of various diseases, including stroke [37]. Various polyphenols can influence epigenetic mechanisms underlying stroke pathogenesis and progression by modulating DNA methylation and histone modifications through the interaction with histone deacetylases (HDACs) and DNA methyltransferases (DNMTs) [38].
References
- Alan Crozier; Indu B. Jaganath; Michael N. Clifford; Dietary phenolics: chemistry, bioavailability and effects on health. Natural Product Reports 2009, 26, 1001-1043, 10.1039/b802662a.
- Rong Tsao; Chemistry and Biochemistry of Dietary Polyphenols. Nutrients 2010, 2, 1231-1246, 10.3390/nu2121231.
- Sandro Rogerio De Almeida; Marco G. Alves; Maria De Sousa; Pedro F. Oliveira; Branca M. Silva; Are Polyphenols Strong Dietary Agents Against Neurotoxicity and Neurodegeneration?. Neurotoxicity Research 2016, 30, 345-366, 10.1007/s12640-015-9590-4.
- Jara Perezjimenez; Vanessa Neveu; F M Vos; Augustin Scalbert; Identification of the 100 richest dietary sources of polyphenols: an application of the Phenol-Explorer database. European Journal of Clinical Nutrition 2010, 64, S112-S120, 10.1038/ejcn.2010.221.
- Cesar G. Fraga; Kevin D Croft; David O. Kennedy; Francisco A. Tomás-Barberán; The effects of polyphenols and other bioactives on human health. Food & Function 2019, 10, 514-528, 10.1039/c8fo01997e.
- Anna Tresserra-Rimbau; Rosa M. Lamuela-Raventos; J. J. Moreno; Polyphenols, food and pharma. Current knowledge and directions for future research. Biochemical Pharmacology 2018, 156, 186-195, 10.1016/j.bcp.2018.07.050.
- Anna Tresserra-Rimbau; Sara Arranz; Matthias Eder; Anna Vallverdú-Queralt; Dietary Polyphenols in the Prevention of Stroke. Oxidative Medicine and Cellular Longevity 2017, 2017, 1-10, 10.1155/2017/7467962.
- Lea Pogačnik; Ajda Ota; Jian Bo Xiao; An Overview of Crucial Dietary Substances and Their Modes of Action for Prevention of Neurodegenerative Diseases. Cells 2020, 9, 576, 10.3390/cells9030576.
- Massimo D'archivio; Carmela Filesi; Roberta Di Benedetto; Raffaella Gargiulo; Claudio Giovannini; R. Masella; Polyphenols, dietary sources and bioavailability.. Annali dell'Istituto Superiore di Sanità 2007, 43, 348–361.
- Adele Chimento; Francesca De Amicis; Rosa Sirianni; Maria Stefania Sinicropi; Francesco Puoci; Ivan Casaburi; Carmela Saturnino; Vincenzo Pezzi; Progress to Improve Oral Bioavailability and Beneficial Effects of Resveratrol. International Journal of Molecular Sciences 2019, 20, 1381, 10.3390/ijms20061381.
- Adrian L. Lopresti; The Problem of Curcumin and Its Bioavailability: Could Its Gastrointestinal Influence Contribute to Its Overall Health-Enhancing Effects?. Advances in Nutrition 2018, 9, 41-50, 10.1093/advances/nmx011.
- Joshua D. Lambert; Shengmin Sang; Chung S. Yang; Biotransformation of Green Tea Polyphenols and the Biological Activities of Those Metabolites. Molecular Pharmaceutics 2007, 4, 819-825, 10.1021/mp700075m.
- Priyankar Dey; Gut microbiota in phytopharmacology: A comprehensive overview of concepts, reciprocal interactions, biotransformations and mode of actions. Pharmacological Research 2019, 147, 104367, 10.1016/j.phrs.2019.104367.
- Wuwen Feng; Hui Ao; Cheng Peng; Dan Yan; Gut microbiota, a new frontier to understand traditional Chinese medicines. Pharmacological Research 2019, 142, 176-191, 10.1016/j.phrs.2019.02.024.
- Michał Wiciński; Jakub Gębalski; Ewelina Mazurek; Marta Podhorecka; Maciej Śniegocki; Paweł Szychta; Ewelina Sawicka; Bartosz Malinowski; The Influence of Polyphenol Compounds on Human Gastrointestinal Tract Microbiota. Nutrients 2020, 12, 350, 10.3390/nu12020350.
- Estefanía Márquez Campos; Peter Stehle; Marie-Christine Simon; Microbial Metabolites of Flavan-3-Ols and Their Biological Activity. Nutrients 2019, 11, 2260, 10.3390/nu11102260.
- Keumhan Noh; Youra Kang; Mahesh R. Nepal; Ki Sun Jeong; Do Gyeong Oh; Mi Jeong Kang; Sangkyu Lee; Wonku Kang; Hye Gwang Jeong; Tae Cheon Jeong; et al. Role of Intestinal Microbiota in Baicalin-Induced Drug Interaction and Its Pharmacokinetics. Molecules 2016, 21, 337, 10.3390/molecules21030337.
- Paola Vitaglione; Giovanna Donnarumma; Aurora Napolitano; Fabio Galvano; Assunta Gallo; Luca Scalfi; Vincenzo Fogliano; Protocatechuic Acid Is the Major Human Metabolite of Cyanidin-Glucosides. The Journal of Nutrition 2007, 137, 2043-2048, 10.1093/jn/137.9.2043.
- J. M. Landete; Ellagitannins, ellagic acid and their derived metabolites: A review about source, metabolism, functions and health. Food Research International 2011, 44, 1150-1160, 10.1016/j.foodres.2011.04.027.
- Monica Galleano; Olga Pechanova; Cesar G. Fraga; Hypertension, Nitric Oxide, Oxidants, and Dietary Plant Polyphenols. Current Pharmaceutical Biotechnology 2010, 11, 837-848, 10.2174/138920110793262114.
- Charlotte Grootaert; Senem Kamiloglu; Esra Capanoglu; John Van Camp; Cell Systems to Investigate the Impact of Polyphenols on Cardiovascular Health. Nutrients 2015, 7, 9229-9255, 10.3390/nu7115462.
- Mirza Bojić; Željan Maleš; Andrea Antolić; Ivana Babić; Maja Tomičić; Antithrombotic activity of flavonoids and polyphenols rich plant species. Acta Pharmaceutica 2019, 69, 483-495, 10.2478/acph-2019-0050.
- Michał Bijak; Agnieszka Sut; Anna Kosiorek; Joanna Saluk-Bijak; Jacek Golanski; Dual Anticoagulant/Antiplatelet Activity of Polyphenolic Grape Seeds Extract. Nutrients 2019, 11, 93, 10.3390/nu11010093.
- Kinga Kasperkiewicz; Michał B. Ponczek; Jacek Owczarek; Piotr Guga; E. Budzisz; Antagonists of Vitamin K—Popular Coumarin Drugs and New Synthetic and Natural Coumarin Derivatives. Molecules 2020, 25, 1465, 10.3390/molecules25061465.
- Jana Karlíčková; Michal Říha; Tomáš Filipský; Kateřina Macáková; Radomír Hrdina; Přemysl Mladěnka; Antiplatelet Effects of Flavonoids Mediated by Inhibition of Arachidonic Acid Based Pathway. Planta Medica 2015, 82, 76-83, 10.1055/s-0035-1557902.
- Applová, Lenka; Karlíčková, Jana; Říha, Michal; Filipský, Tomáš; Macáková, Kateřina; Spilková, Jiřina; Mladěnka, Přemysl; The isoflavonoid tectorigenin has better antiplatelet potential than acetylsalicylic acid. Phytomedicine 2017, 35, 11-17.
- Mohammad H. Forouzanfar; Patrick Liu; Gregory A. Roth; Marie Ng; Stan Biryukov; Laurie Marczak; Lily Alexander; Kara Estep; Kalkidan Hassen Abate; Tomi F. Akinyemiju; et al.Raghib AliNelson Alvis-GuzmanPeter AzzopardiAmitava BanerjeeTill BärnighausenArindam BasuTolesa BekeleDerrick A. BennettSibhatu BiadgilignFerrán Catalá-LópezValery L. FeiginJoao C. FernandesFlorian FischerAlemseged Aregay GebruPhilimon GonaRajeev GuptaGraeme J. HankeyJost B. JonasSuzanne E. JuddYoung-Ho KhangArdeshir KhosraviYun Jin KimRuth W. KimokotiYoshihiro KokuboDhaval KolteAlan LopezPaulo Andrade LotufoReza MalekzadehYohannes Adama MelakuGeorge A. MensahAwoke MisganawAli H. MokdadAndrew E. MoranHaseeb NawazBruce NealFrida Namnyak NgalesoniTakayoshi OhkuboFarshad PourmalekAnwar RafayRajesh Kumar RaiDavid Rojas-RuedaUchechukwu K. SampsonItamar S. SantosMonika SawhneyAletta E. SchutteSadaf G. SepanlouGirma Temam ShifaIvy ShiueBemnet Amare TedlaAmanda G. ThriftMarcello TonelliThomas TruelsenNikolaos TsilimparisKingsley Nnanna UkwajaOlalekan A. UthmanTommi VasankariNarayanaswamy VenketasubramanianVasiliy Victorovich VlassovTheo VosRonny WestermanLijing L. YanYuichiro YanoNaohiro YonemotoMaysaa El Sayed ZakiChristopher J. L. Murray Global Burden of Hypertension and Systolic Blood Pressure of at Least 110 to 115 mm Hg, 1990-2015. JAMA 2017, 317, 165-182, 10.1001/jama.2016.19043.
- Daniel Woo; Mary Haverbusch; Padmini Sekar; Brett Kissela; Jane Khoury; Alexander Schneider; Dawn Kleindorfer; Jerzy Szaflarski; Arthur Pancioli; Edward Jauch; et al.Charles J. MoomawLaura SauerbeckJames GebelJoseph P. Broderick Effect of Untreated Hypertension on Hemorrhagic Stroke. Stroke 2004, 35, 1703-1708, 10.1161/01.str.0000130855.70683.c8.
- Francesca Pistoia; Simona Sacco; Diana Degan; Cindy Tiseo; Raffaele Ornello; Antonio Carolei; Hypertension and Stroke: Epidemiological Aspects and Clinical Evaluation. High Blood Pressure & Cardiovascular Prevention 2015, 23, 9-18, 10.1007/s40292-015-0115-2.
- Aedín Cassidy; Éilis J O’Reilly; Colin Kay; Laura Sampson; Mary Franz; J P Forman; Gary Curhan; Eric B. Rimm; Habitual intake of flavonoid subclasses and incident hypertension in adults. The American Journal of Clinical Nutrition 2010, 93, 338-347, 10.3945/ajcn.110.006783.
- Alexander Medina Remon; Ramón Estruch; Anna Tresserra-Rimbau; Anna Vallverdú-Queralt; Rosa M. Lamuela-Raventós; The effect of polyphenol consumption on blood pressure.. Mini-Reviews in Medicinal Chemistry 2013, 13, 1137-1149, 10.2174/1389557511313080002.
- Giovanni Scapagnini; Vasto Sonya; Abraham G. Nader; Caruso Calogero; Davide Zella; Galvano Fabio; Modulation of Nrf2/ARE Pathway by Food Polyphenols: A Nutritional Neuroprotective Strategy for Cognitive and Neurodegenerative Disorders. Molecular Neurobiology 2011, 44, 192-201, 10.1007/s12035-011-8181-5.
- Claudio Giovannini; Roberta Masella; Role of polyphenols in cell death control. Nutritional Neuroscience 2012, 15, 134-149, 10.1179/1476830512y.0000000006.
- Rui F. M. Silva; Lea Pogačnik; Polyphenols from Food and Natural Products: Neuroprotection and Safety. Antioxidants 2020, 9, 61, 10.3390/antiox9010061.
- Edoardo Parrella; Vanessa Porrini; Marina Benarese; Marina Pizzi; The Role of Mast Cells in Stroke.. Cells 2019, 8, 437, 10.3390/cells8050437.
- Nour Yahfoufi; Nawal Alsadi; Majed Jambi; Chantal Matar; The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618, 10.3390/nu10111618.
- Rosita Stanzione; Maria Cotugno; Franca Bianchi; Simona Marchitti; Maurizio Forte; Massimo Volpe; Speranza Rubattu; Pathogenesis of Ischemic Stroke: Role of Epigenetic Mechanisms. Genes 2020, 11, 89, 10.3390/genes11010089.
- Min-Hsiung Pan; Ching-Shu Lai; Jia-Ching Wu; Chi-Tang Ho; Epigenetic and disease targets by polyphenols.. Current Pharmaceutical Design 2013, 19, 6156-6185, 10.2174/1381612811319340010.
