Coupling of CO
2
with epoxides is a green emerging alternative for the synthesis of cyclic organic carbonates (COC) and aliphatic polycarbonates (APC). The scope of this work is to provide a comprehensive overview of metal complexes having sulfur-containing ligands as homogeneous catalytic systems able to efficiently promote this transformation with a concise discussion of the most significant results. The crucial role of sulfur as the hemilabile ligand and its influence on the catalytic activity are highlighted as well.
- homogeneous catalysis
- sulfur
- carbon dioxide
- epoxides
- cyclic carbonates
- polycarbonates
1. Introduction
Homogeneous catalysis is one of the most fast-developing areas of chemistry because of the possibility to finely tune the course of a given reaction by rational design of the catalyst architecture. Indeed, compared to heterogeneous catalysis, mechanisms governing the activity and selectivity are easier to be understood and therefore the overall performances can be consequentially greatly improved. In particular, the use of soluble transition-metal complexes in catalysis opened unprecedented possibilities to control the product features by a judicious modification of the electronic and steric properties of the ancillary ligand. Inspired by this simple concept, chemists have unfolded countless stereoselective reactions encompassing all families of chemical transformations (oxidations, cross-coupling reactions, polymerizations, etc.) [1][2][3].
Homogeneous catalysis is one of the most fast-developing areas of chemistry because of the possibility to finely tune the course of a given reaction by rational design of the catalyst architecture. Indeed, compared to heterogeneous catalysis, mechanisms governing the activity and selectivity are easier to be understood and therefore the overall performances can be consequentially greatly improved. In particular, the use of soluble transition-metal complexes in catalysis opened unprecedented possibilities to control the product features by a judicious modification of the electronic and steric properties of the ancillary ligand. Inspired by this simple concept, chemists have unfolded countless stereoselective reactions encompassing all families of chemical transformations (oxidations, cross-coupling reactions, polymerizations, etc.) [1,2,3].
Parallel to this trend, in the last two decades, the use of renewable feedstocks has gained momentum because of the growing demand for more sustainable chemical processes. In particular, carbon dioxide utilization (CDU) has emerged as an important tool for the transition to a carbon-neutral society because of the possibility to close, through the use of renewable energies, an anthropogenic carbon cycle avoiding net emission of greenhouse gases. Besides, CO
2
displays evident advantages in terms of toxicity, flammability, and the possibility of storage with respect to other C
1 feedstocks (such as phosgene and carbon monoxide) that render the implementation of industrial processes based on this molecule quite promising [4][5][6][7][8][9].
feedstocks (such as phosgene and carbon monoxide) that render the implementation of industrial processes based on this molecule quite promising [4,5,6,7,8,9].
Among the possible products obtainable from CO
2, cyclic organic carbonates (COCs) and aliphatic polycarbonates (APCs) have received considerable attention because of their wide range of application and straightforward synthetic pathway starting from the corresponding epoxides [10][11][12][13][14][15][16][17][18][19]. The coupling between CO
, cyclic organic carbonates (COCs) and aliphatic polycarbonates (APCs) have received considerable attention because of their wide range of application and straightforward synthetic pathway starting from the corresponding epoxides [10,11,12,13,14,15,16,17,18,19]. The coupling between CO
2
and an epoxide can result in the formation of the cyclic product or of the polymeric one depending on many factors such as the nature of the substrate, the reaction conditions, and the catalytic system. Notwithstanding the efforts to develop efficient metal-free catalysts, most of the active systems are based on the combination of a metal complex activated by a suitable nucleophile, often added as ammonium or phosphonium salt. The basic reaction mechanism can be exemplified following the steps depicted in
: (i) The coordination of the epoxide to the metal center; (ii) the ring-opening of the activated epoxide by the nucleophilic attack and the formation of the metal-alkoxo bond; (iii) the insertion of CO
2
. At this stage, the intermediate can give an intramolecular ring-closing with the formation of the COC (path A) or the alternating insertion of epoxide and CO
2 with the formation of the APC (path B). Since two consecutive insertions of the epoxide are possible as well (path C), often the resulting polymer also contains some polyether linkages [20].
with the formation of the APC (path B). Since two consecutive insertions of the epoxide are possible as well (path C), often the resulting polymer also contains some polyether linkages [20].
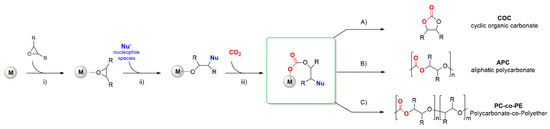
Scheme 1.
2 with epoxides [20].
The metal complexes successfully employed as catalysts in this reaction can be based both on main group metals such as Al, Mg, Ca [21][22][23] and on transition metals such as Ti, Cr, Fe, Co, Nb, La, and Zn [17][18][20][24][25][26][27][28]. Some examples of highly active catalysts for the formation of cyclic carbonates are given in
The metal complexes successfully employed as catalysts in this reaction can be based both on main group metals such as Al, Mg, Ca [21,22,23] and on transition metals such as Ti, Cr, Fe, Co, Nb, La, and Zn [17,18,20,24,25,26,27,28]. Some examples of highly active catalysts for the formation of cyclic carbonates are given in
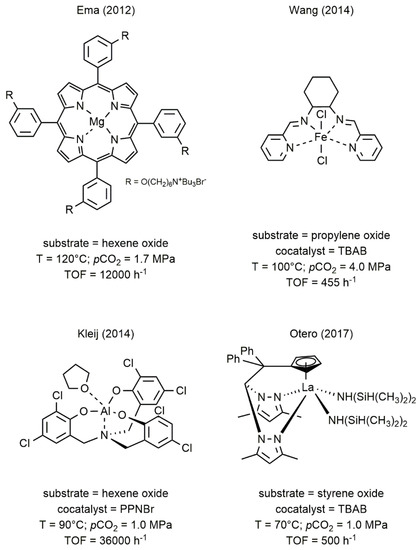
Figure 1.
2 and epoxides [22][25][29][30].
2. Metal Complexes Bearing Sulfur-Containing Ligands Active in the Coupling of CO2 and Epoxides
2.1. Chromium-Based Systems
Chromium Salen complexes are among the most studied coordination compounds in catalysis. Indeed, in the last years, their chemistry had been deeply investigated because of their unique properties in the oxidation, even in the enantioselective version, of various substrates [31]. The easy synthesis and the possibility to design chiral metal species sparked the interest in using these complexes also in the CO
Chromium Salen complexes are among the most studied coordination compounds in catalysis. Indeed, in the last years, their chemistry had been deeply investigated because of their unique properties in the oxidation, even in the enantioselective version, of various substrates [38]. The easy synthesis and the possibility to design chiral metal species sparked the interest in using these complexes also in the CO2
/epoxide coupling. In this context, Darensbourg and Nguyen separately reported the use of chromium Salen complexes for the coupling of cyclohexene oxide (CHO) or propylene oxide (PO) with CO2
for the formation of polycyclohexene carbonate (PCHC) and propylene carbonate (PC), respectively (5) [54,55].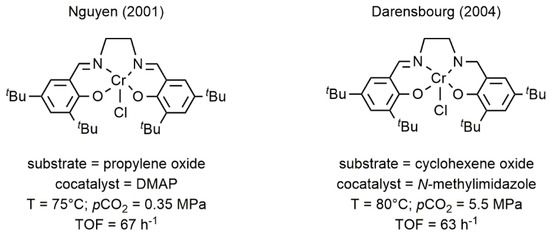
Figure 25.
2
allowing the synthesis, with high activity, of new copolymer microstructures (6) [56,57].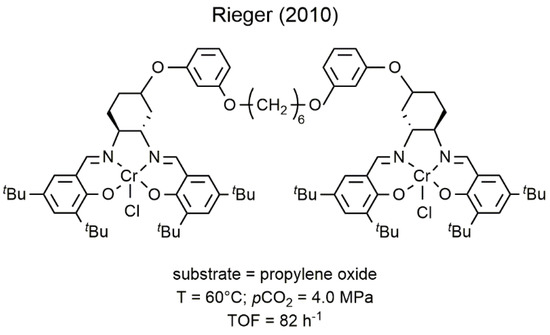
Figure 36.
2 with PO [34].
2
/epoxide chemistry, the first example of chromium catalysts bearing a soft donor atom in the ligand skeleton was reported by Duchateau and coworkers only in 2014 and regarded the utilization of two chromium(III) complexes having an aminophosphine bidentate ligand (Figure 4) [36]. These catalysts showed moderate catalytic activity in the copolymerization of CO
7) [58]. These catalysts showed moderate catalytic activity in the copolymerization of CO2
with CHO affording low molecular weight PCHC (Mn
up to 1930 g mol−1
).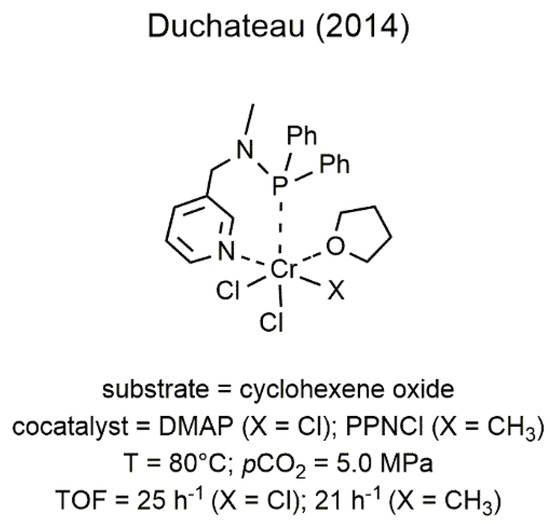
Figure 47.
2 with CHO [36].
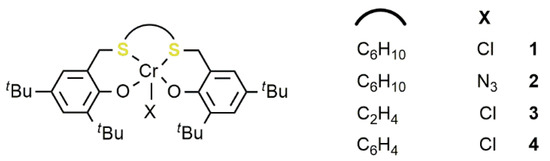
2
with vinylcyclohexeneoxide (VCHO) (8) [59]. Complexes1–4
, activated by bis(triphenylphosphoranylidene) ammonium chloride (PPNCl) are all active catalysts in the CO2
/VCHO coupling by using a [VCHO]/[[OSSO]CrX]/[PPNCl] ratio of 1000/1/2 at 90 °C and high pressure (p
CO2
= 3.0 MPa). Activity and chemoselectivity toward the APC vis a vis COC depended on the catalyst structure. Indeed, the complexes1
–3
with a cyclohexyl or an ethyl carbon bridge showed better performances compared to the phenylene analogue4
. In addition,4
also produced a low molecular weight polymer, in comparison to the other complexes, albeit with a narrow dispersity (Ð
= 1.24–1.49). For the most active catalyst1
, the authors also explored the use of different co-catalysts, such as bis(triphenylphosphoranylidene)ammonium azide, (PPNN3
), tetrabutylammonium chloride (TBAC), and 4-dimethylaminopyridine (DMAP) obtaining the best compromise between activity and selectivity toward the polymeric product by using 2 equivalents, with respect to the metal, of PPNCl. Notably, by adjusting the [VCHO]/[1
]/[PPNCl] ratio to 500/1/2, the activity reached the value of 134 h−1
with 76% of selectivity for the polymeric product at 90 °C and with a CO2
pressure of 3.0 MPa.13
C NMR analysis showed that the resulting polymer is atactic in all cases. The reaction of terminal epoxides such as PO or epichlorohydrine (EPC) invariably resulted in the formation of the corresponding COC.2.2. Iron-Based Systems
Despite the abundance of this metal on the Earth-crust, the use of iron in homogeneous catalysis has been longtime neglected and only in the last two decades emerged as a viable alternative to catalysts based on rare/precious metals. In this trend, the development of iron catalysts for the reaction of CO2
with epoxides was not an exception, with only few examples of complexes that showed significant activity in the cycloaddition of CO2 to epoxide or in the alternating copolymerization [18][38][39][40][41]. As for other metals, the structural motifs of choice were based on macrocyclic or polydentate ligands having oxygen and nitrogen as donor atoms. In particular, the complexes developed by Williams and Nozaki [38][39], depicted in
to epoxide or in the alternating copolymerization [18,62,63,64,65]. As for other metals, the structural motifs of choice were based on macrocyclic or polydentate ligands having oxygen and nitrogen as donor atoms. In particular, the complexes developed by Williams and Nozaki [62,63], depicted inFigure 6, were the first examples of iron catalysts able to promote the copolymerization of CO
11, were the first examples of iron catalysts able to promote the copolymerization of CO2 with CHO and PO, respectively. The iron complexes developed by Kleij [40][41] (
with CHO and PO, respectively. The iron complexes developed by Kleij [64,65] (Figure 6), efficiently produced cyclic carbonates from a variety of terminal and internal epoxides.
11), efficiently produced cyclic carbonates from a variety of terminal and internal epoxides.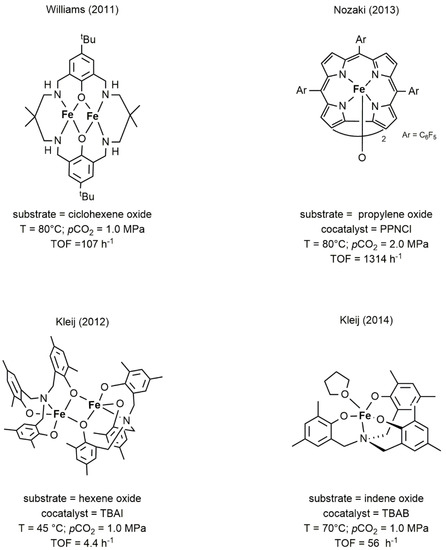
Figure 611.
2
with epoxides was reported by Abu-Surrah and coworkers in 2013 (10
,Figure 7) [42]. This thiophenaldimine Fe(II) complex, activated by TBAB, at 130 °C and 0.5 MPa of CO
12) [66]. This thiophenaldimine Fe(II) complex, activated by TBAB, at 130 °C and 0.5 MPa of CO2
pressure, promoted the formation of styrene carbonate (SC) with low activity (TON = 799; TOF = 33.3 h−1
).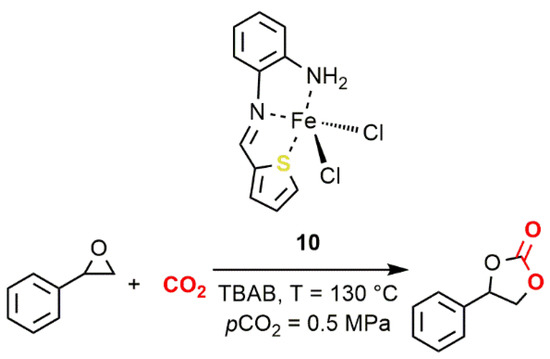
Figure 712.
2
10 [42].
11
,Figure 8) bearing a bis-thioether-triphenolate ligand [43][44][45]. These complexes were stable in solution, and when activated by TBAB (
13) bearing a bis-thioether-triphenolate ligand [67,68,69]. These complexes were stable in solution, and when activated by TBAB (11
/TBAB = 1/2), were able to promote the transformation of various terminal or internal epoxides to the corresponding COCs with good activity and selectivity. In particular, PO was converted to PC reaching a TOF of 580 h−1
at 100 °C with a CO2
pressure of 2.0 MPa, and with a [PO]/[11
]/[TBAB] ratio of 4000/1/5. Furthermore, this catalyst showed to be also highly stereoselective affording exclusively thecis
-COC by using CHO as the substrate regardless the TBAB/[11
] ratio. Moreover, by using the enantiopure SO as the epoxide (ee
= 94%) the corresponding SC was gained with a good degree of stereoretention (ee
= 74%).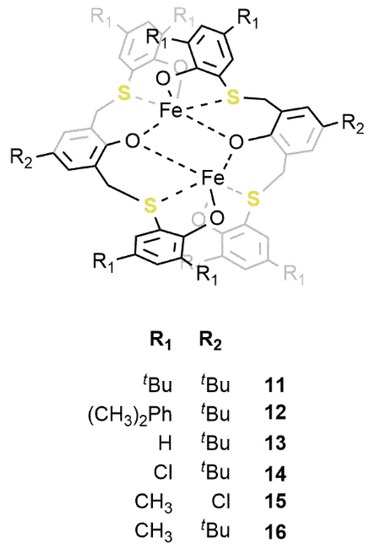
Figure 813.
11
16
, bearing methyl substituents on the phenol ring, displayed the highest activity so far reported for an iron catalyst with a TOF of 5200 and 7000 h−1
in the conversion of PO and EPC, respectively, in the corresponding COCs at 120 °C with a CO2
pressure of 2.0 MPa and a [PO]/[16
]/[TBAB] ratio of 10000/1/10. Conversely, the introduction of bulkier substituents in catalyst12
and electron-withdrawing groups in catalyst14
resulted in a lowering of the catalyst activity. In order to shed more light on the reaction mechanism, DFT calculations were performed and highlighted three important features regarding the formation of COCs promoted by these catalysts: (i) The coordination of the epoxide to the iron(III) center requires the detachment of a sulfur atom from the coordination sphere; (ii) the insertion of CO2
is not mediated by the second iron center; (iii) the ring-closure of the COCs is the rate-limiting step (Figure 9). As a matter of fact, the hemilability of the sulfur atoms plays a fundamental role in determining the catalytic activity. This was also confirmed by the worse performances of catalyst
14). As a matter of fact, the hemilability of the sulfur atoms plays a fundamental role in determining the catalytic activity. This was also confirmed by the worse performances of catalyst14
, which were reasonably caused by a stronger sulfur-iron interaction which, in turn, was caused by the presence of electron-withdrawing substituents on the phenoxo rings.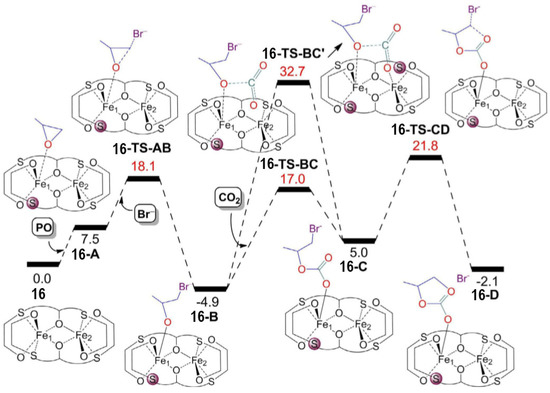
Figure 914.
2
16 [44]. Reprinted with permission from ref. 68. Copyright (2020) John Wiley and Sons.
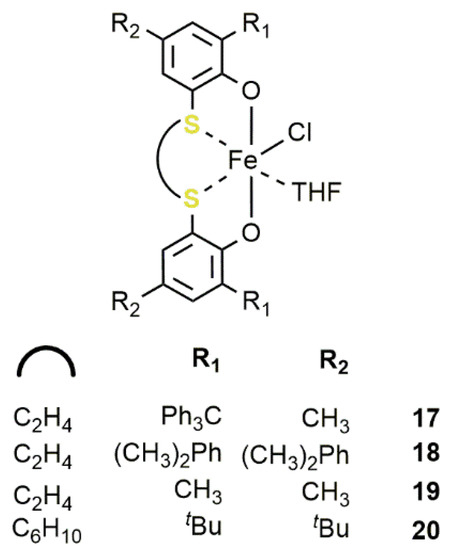
17
–20
(5) [52]. Notably, when activated by TBAB, these catalysts efficiently performed in the cycloaddition of CO2
to terminal and internal epoxides giving the corresponding COC under mild reaction conditions (35 °C;p
CO2
= 0.1 MPa) and, by using catalyst20
, reaching a TOF of 290 h−1
in the case of PO with a [PO]/[20
]/[TBAB] ratio of 1000/1/5. Substituents on the ligand framework had a negligible influence on the catalytic activity. In analogy with the bis-thiother-triphenolate complexes11
–16
, the reaction occurred with a high degree of stereoretention (98%) in the case of internal epoxides such as 2-buteneoxide. More importantly, when the substrate was CHO, only the alternating PCHC was observed. In this case, the best cocatalyst was TBAC and the activity order was18
>20
≈17
>19
.2.3. Bismuth-Based Systems
Among the possible Lewis acidic metal centers in catalysis, the last two decades have witnessed a rising interest in the use of bismuth. Indeed, bismuth is cheap and low-toxic and therefore the implementation of catalytic processes based on this metal are an active task of research in green chemistry [47][48][49][50].
Among the possible Lewis acidic metal centers in catalysis, the last two decades have witnessed a rising interest in the use of bismuth. Indeed, bismuth is cheap and low-toxic and therefore the implementation of catalytic processes based on this metal are an active task of research in green chemistry [73,74,75,76].
Actually, the use in CO
2/epoxide reactions was barely explored and only recently received more attention [51][52]. In this field, one of the most efficient catalytic systems was developed by Shimada e Yin in 2009 [53]. In this study, the synthesis of Bi(III) compounds
/epoxide reactions was barely explored and only recently received more attention [77,78]. In this field, one of the most efficient catalytic systems was developed by Shimada e Yin in 2009 [79]. In this study, the synthesis of Bi(III) compounds
35
and
36
, bearing a sulfur bridged bis(phenolato) ligand, was reported (
20).
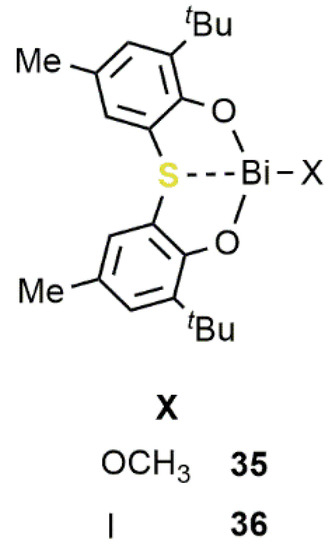
Figure 1120.
35
36 containing an [OSO]-bisphenolate ligand [53].
In the presence of iodide as the nucleophile, by using sources such as Ph
3
PI, tetrabutylammonium iodide (TBAI), NaI or LiI, these compounds were found to be active catalysts for the conversion of PO to PC under mild reaction conditions (RT;
p
CO
2
= 0.1 MPa; [PO]/[Bi]/[I
−
] = 820/1/4) reaching a TOF of 34 h
−1
. More recently, Wong, Au, and coworkers reported that the dinuclear sulfide-bridged complexes
37
–
40
(
Figure 212) performed as active catalysts for the conversion of EPC to the corresponding carbonate at 140 °C and a CO
) performed as active catalysts for the conversion of EPC to the corresponding carbonate at 140 °C and a CO
2
pressure of 3.0 MPa ([EPC]/[Bi]/[TBAI] = 200/1/0.2) reaching TOFs up to 25 h
−1 [54]. Complexes
[80]. Complexes
37
–
39
are also among the few air-stable organobismuth compounds reported so far.
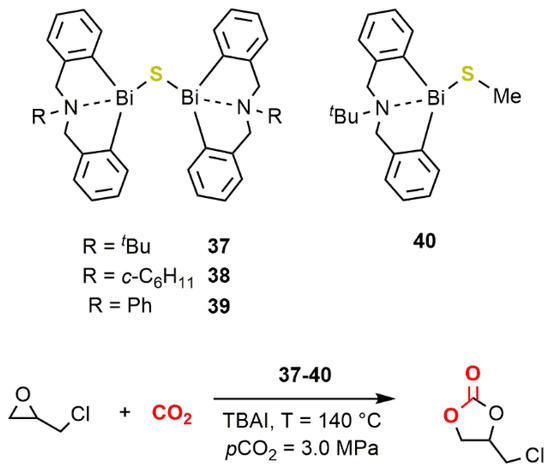
Figure 212.
Dinuclear sulfide-bridged complexes
37
–
40 [54].
[80].
3. Conclusions
The use of sulfur-containing ligands to support metal ions in the reaction between CO
2
and epoxides has attracted an increasing interest in the past years. In many cases, the presence of sulfur in the ligand backbone imparts unique properties in terms of activity, selectivity, and stability of the catalyst precursor. Furthermore, both cyclic carbonates and polycarbonates were obtained with good activity and selectivity for a wide range of substrates showing a high potential for further development. In particular, the use of these ligands can be extended to a major variety of metal centers, allowing an increase in the catalytic performances and expanding the portfolio of possible products. Finally, the design and the synthesis of new structural motifs containing sulfur atoms in the ligand backbone is another important objective for a possible expansion of the catalytic applications.