Bloodstream infections still constitute an outstanding cause of in-hospital morbidity and mortality, especially among critically ill patients. Follow up blood cultures (FUBCs) are widely recommended for proper management of Staphylococcus aureus and Candida spp infections. On the other hand, their role is still a matter of controversy as far as Gram negative bacteremias are concerned. We revised, analyzed, and commented on the literature addressing this issue, to define the clinical settings in which the application of FUBCs could better reveal its value.
- follow-up blood cultures
- Gram negative bacteremia
- Gram negative
- Bloodstream infections
1. Introduction
Bloodstream infections (BSIs) represent a leading cause of death in industrialized countries, with an estimate of two million episodes and 250,000 deaths annually in North America and Europe, despite the availability of new potent antimicrobial therapies and advances in supportive care. In particular, hospital-acquired BSIs are a major cause of morbidity and mortality in intensive care units (ICU), and septic shock still represents the first cause of ICU total mortality [1]. This burden is likely to grow over the next few decades due to the increase in life-expectancy and in median number of patient comorbidity [2]. Unlike Gram positive (GP) BSIs, whose incidence rate has declined over the last few decades, Gram negative (GN) BSIs have markedly increased overtime and nowadays account for up to half of BSIs, with a mortality rate of 20–40% [3–5][3][4][5].
When considering GP-BSIs, international guidelines and consolidated evidence-based procedure bundles are available for the management of the leading pathogen species, Staphylococcus aureus. In this setting, follow-up blood cultures (FUBCs) are regarded as essential to document clearance of bacteremia after treatment initiation and exclude seeding [6–8][6][7][8]. On the other hand, FUBCs are mandatory in the case of Candida spp. BSIs in order to determine the end of candidemia and optimize treatment duration [9].
As for GN-BSIs, relevant advances in management strategies have been made in the last few years, such as the non-inferiority demonstration of 7 vs 14 day antibiotic courses [10] and of oral step-down vs continued parental therapy [11] in uncomplicated GN-BSIs. Recently, a combined approach of rapid diagnostic testing with a bundle of antimicrobial stewardship found a decrease in readmission rate and in cost per case [12]. Anyway, the management of GN-BSIs remains poorly codified and thus prone to personal clinician judgement, as compared to Gram positive settings. In a recently published scoping review, Fabre et al. proposed an algorithm for bacterial blood culture (BC) recommendations. They found that in bacteremias due to Enterobacterales, FUBCs are unlikely to grow unless the source of infection is endovascular or there is inadequate source control. Although with small numbers, similar results were found in Pseudomonas infections. The authors suggest clinical judgment to evaluate the need of FUBCs for GN. Of note, this review considered studies published from 1 January 2004 to 1 June 2019 [13], but the majority of studies addressing the topic of FUBCs in GN infections are actually subsequent to this time frame. At the time of writing, the role of FUBCs in Gram negative bacteremia (GNB) still represents an important matter of debate, with controversial results [14].
The early studies conducted have focused on the disadvantages of FUBCs, mainly represented by the risk of false positive results, prolonged hospitalization, inappropriate antibiotic use and increased cost [15,16][15][16].
2. Discussion
GNB still represents an extremely relevant cause of morbidity and mortality in hospitalized patients, especially in ICU settings. Therefore, it is crucial to achieve an optimization of patient care, which, at the same time, could reduce mortality rates and meet the growing demands of antimicrobial stewardship and cost control. In this regard, the use of FUBCs in patients with GNB has represented a contentious topic in the past few years and especially in the very last period.
Indeed, unnecessary FUBCs may cause patient discomfort and carry the risk of false positive results. According to previous reports, as many as 90% of all BCs grow no organisms and of the approximate 10% that do grow organisms, almost half are considered contaminants (false positives) [27]. Thus, given a constant rate of contamination, performing more FUBCs may result in a higher chance of encountering contaminant organisms, and consequently, in increased costs and patients discomfort, longer hospital stays, unnecessary consultations, and inappropriate antimicrobial therapy [15,16]. Of note, this reasoning includes an inherent fallacy. Although theoretically acceptable in cases of GP yield, the growth of GN bacteria in BCs should always be regarded as relevant and never, or just anecdotally, be considered as a contamination [28].
On the other side, performing FUBCs may have a relevant impact on patient management and outcome, reducing mortality rates. When FUBCs are performed in more severe patients with comorbidities and without adequate infection source control, in cases of bacteremia due to multidrug resistant (MDR) GNs and without an appropriate empiric therapy, the positive or negative results may guide the clinician to the correct decision about type and duration of antibiotic therapy [22,23][22][23].
Not surprisingly, the evidence concerning the usefulness of FUBCs underwent a progressive shift in the last few years, going from a restrictive to a selective approach. Taking a look at Table 1, it appears that papers with a higher rate of FUBCs performed, more frequently found no evidence of benefit in contrast to those that applied FUBCs more selectively. In fact, Kang et al. [18] and Jung et al. [24] performed FUBCs in GNB in 81% and 86% of the cases, respectively, as compared to Maskarinec and Giannella that used this tool only in 68% and 17% of cases, respectively. Likely, Spaziante et al. [26] found a high rate of PB by only performing FUBCs in high-risk ICU patients with GNB. Furthermore, the inclusion in the analysis of a mixed GP and GN population was also associated with pessimistic results. Therefore, it seems that the more refined the selection of patients in which to draw FUBCs, the more evident the benefits they bring. Indeed, the results of our literature review show that, while in some subgroups of patients the use of FUBCs may not translate into a clear benefit, it is possible to identify several situations where the application of this tool may steer the clinical decision making in the correct way. Thus, selection is a sticking point in this topic.
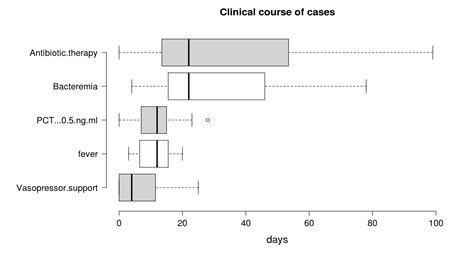
For the purposes of performing rational FUBCs, physicians well trained in infectious diseases should be available in all settings where risk factors for positive FUBCs are present. For this reason, ICU patients might be evaluated from dedicated infectious disease consultants in order to avoid unnecessary BCs [22,26][26]. The study written by Ceccarelli et al. shows an example of this management [29]. In fact, they reported some cases of GN-related septic thrombosis (ST) with indolent clinical course and long-term positive BCs despite adequate antibiotic treatment as one possible exception to the restricted use of FUBCs. In these cases, FUBCs allowed the determination of the correct treatment duration, representing a tool of critical importance for patient management [29]. The same group of authors further stressed this concept in a case series of 13 critical care patients with ST caused by GN bacilli (Figure 1): this disease was characterized by PB despite prompt source control and appropriate antibiotic treatment, an indolent clinical course and, even more important, a rapid defervescence with normalization of procalcitonin (PCT) values preceding bacteremia clearance. This phenomenon was interpreted as a mechanism of immune tolerance [30].
Figure 1. Duration of bacteremia and clinical course features of Gram negative septic thrombosis in critically ill patients, modified from Spaziante et al. [30]. This figure shows that bacteremia may persist despite clinical improvement (fever disappearance, negative procalcitonin (PCT) values and no vasopressor support); under these circumstances, FUBCs may remain the only driver of antibiotic therapy.
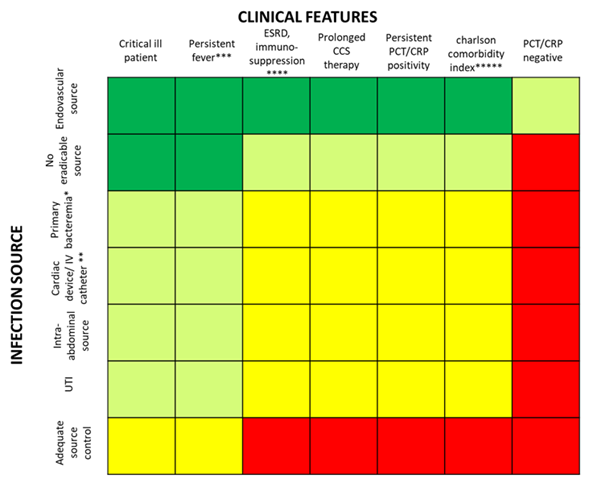
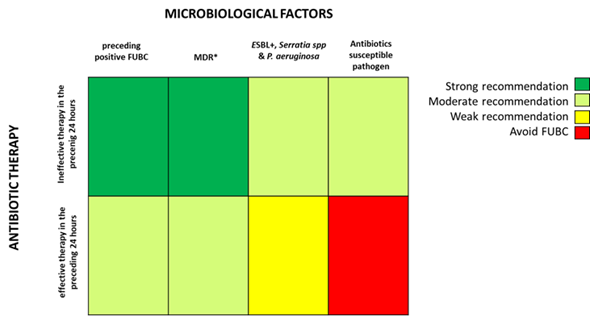
Based on the fascinating hypothesis that FUBCs in BSIs due to GNs could be part of clinical practice, we tried to investigate which conditions make FUBCs either necessary or unfounded. Figures 2 and 3 show clinical and microbiological risk factors for PB, respectively. Rows and columns of each figure intersect in a colored square and every color means a risk threshold of PB, from green (FUBCs highly recommended), light green (moderate recommendation), yellow (weak recommendation) to red (avoid FUBCs), of various combinations of all risk factors recognized in this review.
In order to point out the right setting where FUBCs should be prescribed (otherwise when they are not warranted), we created Figures 2 and 3 analyzing the risk factors for persistent bacteremia found in the revised articles. On the other hand, we identified settings where FUBCs are moderate or even weakly recommended based on risk factors cited by less authors/articles and our judgment. For these reasons, a clinical decision on a case-by-case basis is needed to judge when to perform FUBCs.
In general, FUBCs should be always considered in critically ill patients because they frequently present multiple risk factors that may account for resistant or persistent GNB: intravascular catheter, antibiotic resistant pathogen or an occult source of infection that requires control. As an example, FUBCs are warranted in patients with an endovascular source of GNB that represents the single most important indication to this procedure, even in some instances where a biomarker of active infection, such as PCT, is decreased to negative values. In fact, Spaziante et al. stressed this point by analyzing Gram negative ST where positive FUBCs played a crucial role in patient outcomes (Figure 1). Along the same line, the presence of a non-eradicable infection source appears to be a condition in which FUBCs should be performed, especially in patients that require ICU, with persistent fever or as a moderate recommendation (light green) in individuals afflicted with ESRD on hemodialysis [23–25][23][24][25]. FUBCs might also be indicated in persistently febrile patients with primary bacteremia, long term intravascular catheter or urinary tract infection, to a lower degree of evidence. Additionally, we suggest that even in some instances of a lower positivity rate of FUBCs, a clinical decision might be reached in cases of light green and yellow squares both in Figures 2 and 3. For this reason, in the case of patients without a clear clinical indication to perform FUBCs as shown in Figure 2, they should be checked for microbiological risk factors, as shown in Figure 3. To this end, in our opinion, FUBCs might be performed even in instances of microbiological risk factors alone. In fact, taking clinically stable patients with UTI as an example (yellow to red squares in Figure 2), instances where the column of MDR microorganisms’ etiology and the row of ineffective therapy intersect in a green square might represent a recommendation to perform FUBCs [20,21,24,25][21][24][25].
Getting to the point, if Figure 2 recommends FUBCs, it deals with a “green light” and they should be performed in any case. Additionally, when FUBCs are not indicated by clinical risk factors, clinicians should check the presence of microbiological risk factors, as shown in Figure 3. In the case of neither clinical nor microbiological risk factors, we are in front of a “red light” and FUBCs are not warranted.
A possible limitation of Figures 2 and 3 is that the definitions of risk factors are often different between studies or even not provided at all. For instance, the dosage and duration of corticosteroid treatment with a significant immunosuppressive effect are not clearly defined in the literature as they are considered to depend on the characteristics of the patient and underlying disease [31].
Finally, progressive acquisition of resistance during antimicrobial therapy through the selection of a hidden resistant subpopulation (named hetero-resistance) is a growing concern in the case of persistent bacteremia [32]. Future studies should elucidate the possible role of FUBCs in early detection and management of this increasingly appreciated mechanism of resistance.
Figure 2. Analysis of recommendations for FUBC sampling in the setting of Gram negative bacteremia on the basis of clinical features and source of infection. Note: infection source: * when apparently there is not an infection source; ** if another infection source is presented; *** fever for more than 72–96 h; **** malignancy, solid organ transplantation; ***** Charlson comorbidity index ≥ 6. References: critical ill patient [20,22,23,25,26]; persistent fever [15,20,26]; ESRD, immuno-suppression [15,18,20,23–25]; prolonged corticosteroid (CCS) therapy [23]; persistent PCT/C-reactive protein (CRP) positivity [20]; Charlson comorbidity index [18][17]; PCT/CRP negative [20]; endovascular source [19,23,24,26][19]; no eradicable source [22,24,25][24][25]; primary bacteremia [26]; cardiac device/intravenous (IV) catheter [15,21,23–25][23][24][25]; intra-abdominal source [18]; UTI [20,23,25][23][25]; adequate source control [24]. Abbreviations: UTI, urinary tract infections; ICU, intensive care unit; ESRD, end stage renal disease; CCS, corticosteroids; PCT, procalcitonin; CRP, C-reactive protein.
Figure 3. Analysis of recommendations for FUBC sampling in the setting of Gram negative bacteremia on the basis of microbiological factors and efficacy of antibiotic therapy. Note: * MDR definition by Magiorakos AP et al., 2012 [33]; ESBL, extended-spectrum beta-lactamases. References: ineffective therapy in the preceding 24 h [18,21–23][21][22][23]; effective therapy in the preceding 24 h [18,21–24][21][22][23][24]; preceding positive FUBC [26]; MDR [22,23,25,26][25][26]; ESBL+, Serratia spp and P. aeruginosa [23–25][23][24][25]; antibiotic susceptible pathogen [22,23,25,26][25][26].
References
- Siddiqui S. Mortality profile across our Intensive Care Units: A 5-year database report from a Singapore restructured hospital. Indian J Crit Care Med. 2015;19(12):726-727. doi:10.4103/0972-5229.171401
- Goto M, Al-Hasan MN. Overall burden of bloodstream infection and nosocomial bloodstream infection in North America and Europe. Clin Microbiol Infect. 2013;19(6):501-509. doi:10.1111/1469-0691.12195
- Kang CI, Song JH, Chung DR, Peck KR, Ko KS, Yeom JS, Ki H K, Son J S, Lee S S, Kim Y S, et al. Risk factors and pathogenic significance of severe sepsis and septic shock in 2286 patients with gram negative bacteremia. J Infect. 2011;62(1):26-33. doi:10.1016/j.jinf.2010.10.010
- Sligl WI, Dragan T, Smith SW. Nosocomial Gram-negative bacteremia in intensive care: epidemiology, antimicrobial susceptibilities, and outcomes. Int J Infect Dis. 2015;37:129-134. doi:10.1016/j.ijid.2015.06.024
- Islas-Muñoz B, Volkow-Fernández P, Ibanes-Gutiérrez C, Villamar-Ramírez A, Vilar-Compte D, Cornejo-Juárez P. Bloodstream infections in cancer patients. Risk factors associated with mortality. Int J Infect Dis. 2018; 71:59-64. doi:10.1016/j.ijid.2018.03.022
- Liu C, Bayer A, Cosgrove SE, Daum R S, Fridkin S K, Gorwitz R J, Kaplan S L, Karchmer A W, Levine D P, Murray B E, et al. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children [published correction appears in Clin Infect Dis. 2011 Aug 1;53(3):319]. Clin Infect Dis. 2011; 52(3):e18-e55. doi:10.1093/cid/ciq146
- López-Cortés LE, Del Toro MD, Gálvez-Acebal J, Bereciartua-Bastarrica E, Fariñas M C, Sanz-Franco M, Natera C, Corzo J E, Lomas J M, Pasquau J, et al. Impact of an evidence-based bundle intervention in the quality-of-care management and outcome of Staphylococcus aureus bacteremia. Clin Infect Dis. 2013 Nov;57(9):1225-33. doi: 10.1093/cid/cit499.
- Holland TL, Arnold C, Fowler VG Jr. Clinical management of Staphylococcus aureus bacteremia: a review. JAMA. 2014;312(13):1330-1341. doi:10.1001/jama.2014.9743
- Cornely OA, Bassetti M, Calandra T, Garbino J, Kullberg BJ, Lortholary O, Meersseman W, Akova M, Arendrup M C, Arikan-Akdagli S, et al. ESCMID guideline for the diagnosis and management of Candida diseases 2012: non-neutropenic adult patients, Clin Microbiol Infect 2012; Dec;18 Suppl 7:19-37. doi: 10.1111/1469-0691.12039
- Yahav D, Franceschini E, Koppel F, Turjeman A, Babich T, Bitterman R, Neuberger A, Ghanem-Zoubi N, Santoro A, Eliakim-Raz N, et al. Seven Versus 14 Days of Antibiotic Therapy for Uncomplicated Gram-negative Bacteremia: A Noninferiority Randomized Controlled Trial. Clin Infect Dis. 2019 Sep 13;69(7):1091-1098. doi: 10.1093/cid/ciy1054
- Tamma PD, Conley AT, Cosgrove SE, Harris A D, Lautenbach E, Amoah J, Avdic E, Tolomeo P, Wise J, Subudhi S, et al. Association of 30-Day Mortality With Oral Step-Down vs Continued Intravenous Therapy in Patients Hospitalized With Enterobacteriaceae Bacteremia [published correction appears in JAMA Intern Med. 2019 Nov 1;179(11):1607]. JAMA Intern Med. 2019;179(3):316-323. doi: 10.1001/jamainternmed.2018.6226
- Erickson RM, Tritle BJ, Spivak ES, Timbrook TT. Impact of an Antimicrobial Stewardship Bundle for Uncomplicated Gram-Negative Bacteremia. Open Forum Infect Dis. 2019; 6(12):ofz490. doi:10.1093/ofid/ofz490
- Fabre V, Sharara SL, Salinas AB, Carroll KC, Desai S, Cosgrove SE. Does This Patient Need Blood Cultures? A Scoping Review of Indications for Blood Cultures in Adult Nonneutropenic Inpatients. Clin Infect Dis. 2020 Aug 22;71(5):1339-1347. doi: 10.1093/cid/ciaa039.
- Chan JD, Bryson-Cahn C, Kassamali-Escobar Z, Lynch JB, Schleyer AM. The Changing Landscape of Uncomplicated Gram-Negative Bacteremia: A Narrative Review to Guide Inpatient Management. J Hosp Med. 2020 Aug 19. doi: 10.12788/jhm.3414. Epub ahead of print.
- Canzoneri CN, Akhavan BJ, Tosur Z, Andrade PEA, Aisenberg GM. Follow-up Blood Cultures in Gram-Negative Bacteremia: Are They Needed? Clin Infect Dis. 2017;13;65(11):1776-1779. doi: 10.1093/cid/cix648.
- Dempsey C, Skoglund E, Muldrew KL, Garey KW. Economic health care costs of blood culture contamination: A systematic review. Am J Infect Control. 2019;47(8):963-967. doi:10.1016/j.ajic.2018.12.020.
- Kang CK, Kim ES, Song KH, Kim H B, Kim T S, Kim N H, Kim C J, Choe P G, Bang J H, et al. Can a routine follow-up blood culture be justified in Klebsiella pneumoniae bacteremia? A retrospective case-control study. BMC Infect Dis. 2013; Aug 2;13:365. doi: 10.1186/1471-2334-13-365
- Wiggers JB, Xiong W, Daneman N. Sending repeat cultures: is there a role in the management of bacteremic episodes? (SCRIBE study). BMC Infect Dis. 2016; Jun 13;16:286 doi:10.1186/s12879-016-1622-z
- Shi H, Kang CI, Cho SY, Huh K, Chung DR, Peck KR. Follow-up blood cultures add little value in the management of bacteremic urinary tract infections. Eur J Clin Microbiol Infect Dis. 2019; 38(4):695-702. doi:10.1007/s10096-019-03484-4
- Uehara E, Shoji K, Mikami M, Ishiguro A, Miyairi I. Utility of follow-up blood cultures for Gram-negative rod bacteremia in children. J Infect Chemother. 2019; 25(9):738-741. doi:10.1016/j.jiac.2019.04.014
- Giannella M, Pascale R, Pancaldi L, Monari C, Ianniruberto S, Malosso P, Bussini L, Bartoletti M, Tedeschi S, Ambretti S, et al. Follow-up blood cultures are associated with improved outcome of patients with gram-negative bloodstream infections: retrospective observational cohort study. Clin Microbiol Infect. 2020; 26(7):897-903. doi:10.1016/j.cmi.2020.01.023
- Maskarinec SA, Park LP, Ruffin F, Turner N A, Patel N, Eichenberger E M, van Duin D, Lodise T, Fowler V G Jr, Thaden J T. Positive follow-up blood cultures identify high mortality risk among patients with Gram-negative bacteraemia. Clin Microbiol Infect. 2020;26(7):904-910. doi:10.1016/j.cmi.2020.01.025
- Jung J, Song KH, Jun KI, Kang CK, Kim NH, Choe PG, Park WB, Bang JH, Kim ES, Park SW, et al. Predictive scoring models for persistent gram-negative bacteremia that reduce the need for follow-up blood cultures: a retrospective observational cohort study. BMC Infect Dis. 2020 Sep 17;20(1):680. doi: 10.1186/s12879-020-05395-8
- Mitaka H, Gomez T, Lee YI, Perlman DC. Risk Factors for Positive Follow-up Blood Cultures in Gram-negative Bacilli Bacteremia: Implications for Selecting Who Needs Follow-up Blood Cultures. Open forum Infect Dis. 2020; Mar 28;7(4):ofaa110. doi: 10.1093/ofid/ofaa110
- Spaziante M, Oliva A, Ceccarelli G, Alessandri F, Pugliese F, Venditti M. Follow-up blood cultures in Gram-negative bacilli bacteremia: are they needed for critically ill patients? Minerva Anestesiol. 2020 May;86(5):498-506. doi: 10.23736/S0375-9393.20.14040-9
- Weinstein MP, Towns ML, Quartey SM, Mirrett S, Reimer L G, Parmigiani G, Reller L B. The clinical significance of positive blood cultures in the 1990s: a prospective comprehensive evaluation of the microbiology, epidemiology, and outcome of bacteremia and fungemia in adults. Clin Infect Dis. 1997; Apr 24(4):584–602. doi: 10.1093/clind/24.4.584
- Dargère S, Cormier H, Verdon R. Contaminants in blood cultures: importance, implications, interpretation and prevention. Clin Microbiol Infect. 2018 Sep;24(9):964-969. doi: 10.1016/j.cmi.2018.03.030.
- Ceccarelli G, Giuliano S, Falcone M, Venditti M. Follow-up Blood Cultures: A 2.0 Diagnostic Tool in Patients With Gram-Negative Bacteremia and Septic Thrombophlebitis. Clin Infect Dis. 2018; 66(7):1154-1155. doi:10.1093/cid/cix949
- Spaziante M, Giuliano S, Ceccarelli G, Alessandri F, Borrazzo C, Russo A, Venditti M. Gram-negative septic thrombosis in critically ill patients: A retrospective case-control study. Int J Infect Dis. 2020;94:110-115. doi: 10.1016/j.ijid.2020.02.054
- Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, Harbarth S, Hindler JF, Kahlmeter G, Olsson-Liljequist B et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268-81. doi: 10.1111/j.1469-0691.2011.03570.
- Youssef J, Novosad SA, Winthrop KL. Infection Risk and Safety of Corticosteroid Use. Rheum Dis Clin North Am. 2016 Feb;42(1):157-76, ix-x. doi: 10.1016/j.rdc.2015.08.004.
- Brukner I, Oughton M. A Fundamental Change in Antibiotic Susceptibility Testing Would Better Prevent Therapeutic Failure: From Individual to Population-Based Analysis. Front Microbiol. 2020 Aug 18;11:1820. doi: 10.3389/fmicb.2020.01820.
References
- Siddiqui S. Mortality profile across our Intensive Care Units: A 5-year database report from a Singapore restructured hospital. Indian J Crit Care Med. 2015;19(12):726-727. doi:10.4103/0972-5229.171401
- Goto M, Al-Hasan MN. Overall burden of bloodstream infection and nosocomial bloodstream infection in North America and Europe. Clin Microbiol Infect. 2013;19(6):501-509. doi:10.1111/1469-0691.12195
- Kang CI, Song JH, Chung DR, Peck KR, Ko KS, Yeom JS, Ki H K, Son J S, Lee S S, Kim Y S, et al. Risk factors and pathogenic significance of severe sepsis and septic shock in 2286 patients with gram negative bacteremia. J Infect. 2011;62(1):26-33. doi:10.1016/j.jinf.2010.10.010
- Sligl WI, Dragan T, Smith SW. Nosocomial Gram-negative bacteremia in intensive care: epidemiology, antimicrobial susceptibilities, and outcomes. Int J Infect Dis. 2015;37:129-134. doi:10.1016/j.ijid.2015.06.024
- Islas-Muñoz B, Volkow-Fernández P, Ibanes-Gutiérrez C, Villamar-Ramírez A, Vilar-Compte D, Cornejo-Juárez P. Bloodstream infections in cancer patients. Risk factors associated with mortality. Int J Infect Dis. 2018; 71:59-64. doi:10.1016/j.ijid.2018.03.022
- Liu C, Bayer A, Cosgrove SE, Daum R S, Fridkin S K, Gorwitz R J, Kaplan S L, Karchmer A W, Levine D P, Murray B E, et al. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children [published correction appears in Clin Infect Dis. 2011 Aug 1;53(3):319]. Clin Infect Dis. 2011; 52(3):e18-e55. doi:10.1093/cid/ciq146
- López-Cortés LE, Del Toro MD, Gálvez-Acebal J, Bereciartua-Bastarrica E, Fariñas M C, Sanz-Franco M, Natera C, Corzo J E, Lomas J M, Pasquau J, et al. Impact of an evidence-based bundle intervention in the quality-of-care management and outcome of Staphylococcus aureus bacteremia. Clin Infect Dis. 2013 Nov;57(9):1225-33. doi: 10.1093/cid/cit499.
- Holland TL, Arnold C, Fowler VG Jr. Clinical management of Staphylococcus aureus bacteremia: a review. JAMA. 2014;312(13):1330-1341. doi:10.1001/jama.2014.9743
- Cornely OA, Bassetti M, Calandra T, Garbino J, Kullberg BJ, Lortholary O, Meersseman W, Akova M, Arendrup M C, Arikan-Akdagli S, et al. ESCMID guideline for the diagnosis and management of Candida diseases 2012: non-neutropenic adult patients, Clin Microbiol Infect 2012; Dec;18 Suppl 7:19-37. doi: 10.1111/1469-0691.12039
- Yahav D, Franceschini E, Koppel F, Turjeman A, Babich T, Bitterman R, Neuberger A, Ghanem-Zoubi N, Santoro A, Eliakim-Raz N, et al. Seven Versus 14 Days of Antibiotic Therapy for Uncomplicated Gram-negative Bacteremia: A Noninferiority Randomized Controlled Trial. Clin Infect Dis. 2019 Sep 13;69(7):1091-1098. doi: 10.1093/cid/ciy1054
- Tamma PD, Conley AT, Cosgrove SE, Harris A D, Lautenbach E, Amoah J, Avdic E, Tolomeo P, Wise J, Subudhi S, et al. Association of 30-Day Mortality With Oral Step-Down vs Continued Intravenous Therapy in Patients Hospitalized With Enterobacteriaceae Bacteremia [published correction appears in JAMA Intern Med. 2019 Nov 1;179(11):1607]. JAMA Intern Med. 2019;179(3):316-323. doi: 10.1001/jamainternmed.2018.6226
- Erickson RM, Tritle BJ, Spivak ES, Timbrook TT. Impact of an Antimicrobial Stewardship Bundle for Uncomplicated Gram-Negative Bacteremia. Open Forum Infect Dis. 2019; 6(12):ofz490. doi:10.1093/ofid/ofz490
- Fabre V, Sharara SL, Salinas AB, Carroll KC, Desai S, Cosgrove SE. Does This Patient Need Blood Cultures? A Scoping Review of Indications for Blood Cultures in Adult Nonneutropenic Inpatients. Clin Infect Dis. 2020 Aug 22;71(5):1339-1347. doi: 10.1093/cid/ciaa039.
- Chan JD, Bryson-Cahn C, Kassamali-Escobar Z, Lynch JB, Schleyer AM. The Changing Landscape of Uncomplicated Gram-Negative Bacteremia: A Narrative Review to Guide Inpatient Management. J Hosp Med. 2020 Aug 19. doi: 10.12788/jhm.3414. Epub ahead of print.
- Canzoneri CN, Akhavan BJ, Tosur Z, Andrade PEA, Aisenberg GM. Follow-up Blood Cultures in Gram-Negative Bacteremia: Are They Needed? Clin Infect Dis. 2017;13;65(11):1776-1779. doi: 10.1093/cid/cix648.
- Dempsey C, Skoglund E, Muldrew KL, Garey KW. Economic health care costs of blood culture contamination: A systematic review. Am J Infect Control. 2019;47(8):963-967. doi:10.1016/j.ajic.2018.12.020.
- Kang CK, Kim ES, Song KH, Kim H B, Kim T S, Kim N H, Kim C J, Choe P G, Bang J H, et al. Can a routine follow-up blood culture be justified in Klebsiella pneumoniae bacteremia? A retrospective case-control study. BMC Infect Dis. 2013; Aug 2;13:365. doi: 10.1186/1471-2334-13-365
- Wiggers JB, Xiong W, Daneman N. Sending repeat cultures: is there a role in the management of bacteremic episodes? (SCRIBE study). BMC Infect Dis. 2016; Jun 13;16:286 doi:10.1186/s12879-016-1622-z
- Shi H, Kang CI, Cho SY, Huh K, Chung DR, Peck KR. Follow-up blood cultures add little value in the management of bacteremic urinary tract infections. Eur J Clin Microbiol Infect Dis. 2019; 38(4):695-702. doi:10.1007/s10096-019-03484-4
- Uehara E, Shoji K, Mikami M, Ishiguro A, Miyairi I. Utility of follow-up blood cultures for Gram-negative rod bacteremia in children. J Infect Chemother. 2019; 25(9):738-741. doi:10.1016/j.jiac.2019.04.014
- Giannella M, Pascale R, Pancaldi L, Monari C, Ianniruberto S, Malosso P, Bussini L, Bartoletti M, Tedeschi S, Ambretti S, et al. Follow-up blood cultures are associated with improved outcome of patients with gram-negative bloodstream infections: retrospective observational cohort study. Clin Microbiol Infect. 2020; 26(7):897-903. doi:10.1016/j.cmi.2020.01.023
- Maskarinec SA, Park LP, Ruffin F, Turner N A, Patel N, Eichenberger E M, van Duin D, Lodise T, Fowler V G Jr, Thaden J T. Positive follow-up blood cultures identify high mortality risk among patients with Gram-negative bacteraemia. Clin Microbiol Infect. 2020;26(7):904-910. doi:10.1016/j.cmi.2020.01.025
- Jung J, Song KH, Jun KI, Kang CK, Kim NH, Choe PG, Park WB, Bang JH, Kim ES, Park SW, et al. Predictive scoring models for persistent gram-negative bacteremia that reduce the need for follow-up blood cultures: a retrospective observational cohort study. BMC Infect Dis. 2020 Sep 17;20(1):680. doi: 10.1186/s12879-020-05395-8
- Mitaka H, Gomez T, Lee YI, Perlman DC. Risk Factors for Positive Follow-up Blood Cultures in Gram-negative Bacilli Bacteremia: Implications for Selecting Who Needs Follow-up Blood Cultures. Open forum Infect Dis. 2020; Mar 28;7(4):ofaa110. doi: 10.1093/ofid/ofaa110
- Spaziante M, Oliva A, Ceccarelli G, Alessandri F, Pugliese F, Venditti M. Follow-up blood cultures in Gram-negative bacilli bacteremia: are they needed for critically ill patients? Minerva Anestesiol. 2020 May;86(5):498-506. doi: 10.23736/S0375-9393.20.14040-9
- Weinstein MP, Towns ML, Quartey SM, Mirrett S, Reimer L G, Parmigiani G, Reller L B. The clinical significance of positive blood cultures in the 1990s: a prospective comprehensive evaluation of the microbiology, epidemiology, and outcome of bacteremia and fungemia in adults. Clin Infect Dis. 1997; Apr 24(4):584–602. doi: 10.1093/clind/24.4.584
- Dargère S, Cormier H, Verdon R. Contaminants in blood cultures: importance, implications, interpretation and prevention. Clin Microbiol Infect. 2018 Sep;24(9):964-969. doi: 10.1016/j.cmi.2018.03.030.
- Ceccarelli G, Giuliano S, Falcone M, Venditti M. Follow-up Blood Cultures: A 2.0 Diagnostic Tool in Patients With Gram-Negative Bacteremia and Septic Thrombophlebitis. Clin Infect Dis. 2018; 66(7):1154-1155. doi:10.1093/cid/cix949
- Spaziante M, Giuliano S, Ceccarelli G, Alessandri F, Borrazzo C, Russo A, Venditti M. Gram-negative septic thrombosis in critically ill patients: A retrospective case-control study. Int J Infect Dis. 2020;94:110-115. doi: 10.1016/j.ijid.2020.02.054
- Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, Harbarth S, Hindler JF, Kahlmeter G, Olsson-Liljequist B et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268-81. doi: 10.1111/j.1469-0691.2011.03570.
- Youssef J, Novosad SA, Winthrop KL. Infection Risk and Safety of Corticosteroid Use. Rheum Dis Clin North Am. 2016 Feb;42(1):157-76, ix-x. doi: 10.1016/j.rdc.2015.08.004.
- Brukner I, Oughton M. A Fundamental Change in Antibiotic Susceptibility Testing Would Better Prevent Therapeutic Failure: From Individual to Population-Based Analysis. Front Microbiol. 2020 Aug 18;11:1820. doi: 10.3389/fmicb.2020.01820.