Clinical reports suggest a potential link between excess retinoids and development of depression. Although it has been shown that all-trans retinoic acid (ATRA) administration induces behavioral changes, further insight into how ATRA is involved is lacking. The hippocampus seems to be a major target of retinoids, and abnormal synaptic plasticity of the hippocampus is involved in depression. We examined two genes associated with synaptic function, discs large homolog 2 (DLG2), and synapse differentiation-inducing gene protein 1 (SynDIG1) in terms of hippocampal expression and correlation with behavior. Three different doses of ATRA were injected into young mice and 10 mg/kg ATRA was found to induce depression-like behavior. In the hippocampus, DLG2 mRNA was significantly decreased by ATRA. mRNA levels were positively correlated with central area duration and distance in the open-field test. Increased SynDIG1 mRNA levels were observed. There was a negative correlation between SynDIG1 mRNA levels and mobility time in the forced swimming test. Retinoic acid receptor γ mRNA was significantly positively correlated with DLG2 and negatively correlated with SynDIG1. To summarize, ATRA administration induced anxiety- and depression-like behavior accompanied by a decreased expression of DLG2 and an increased expression of SynDIG1. Moreover, DLG2 was correlated with anxiety-like behavior and SynDIG1 was correlated with depression-like behavior. These results might constitute a novel target underlying ATRA-induced anxiety- and depression-like behavior.
- all-trans retinoic acid
- discs large homolog 2
- synapse differentiation-inducing gene protein 1
- hippocampus
- anxiety-like behavior
- depression-like behavior
1. Introduction
Retinoic signaling is reportedly linked with the development of the central nervous system (CNS) and the pathogenesis of depression in adults [1][2][3][4]. Excessive consumption of vitamin A (hypervitaminosis A) has long been known to cause adverse psychiatric events [5]. A synthetic retinoid used to treat acne, 13-cis-retinoic acid (13-cis-RA; isotretinoin), has been linked to depression and suicide, since its approval in 1982 [6]. All-trans retinoic acid (ATRA), the endogenous active derivative of vitamin A, plays a role in cell growth, neural differentiation, and synaptic plasticity during development and operates exclusively by regulating gene transcription [7][8][9]. Recently, our group found that chronic ATRA administration could induce depression-like behavioral changes in adult rats [10][11][12].
The hippocampus is involved in mood disorders, such as anxiety and depression. Brain imaging and post-mortem studies provide evidence of changes in the cellular architecture and/or morphology within this brain region, including a reduction in hippocampal volume, and atrophy of hippocampal pyramidal neurons [13][14][15][16]. Recent studies have indicated that abnormal synaptic plasticity in specific areas of the CNS, particularly the hippocampus, may be a core factor in the pathophysiology of depression [17]. Studies in rodents have provided evidence in support of a reduced synapse number and decreased levels of synaptic signaling proteins in the hippocampus in a depression model [18][19]. Furthermore, antidepressants, such as the highly prescribed selective serotonin reuptake inhibitors, could enhance synaptic plasticity in the hippocampus, as demonstrated in electrophysiological studies [20][21]. All of the above studies confirm a link between altered synaptic plasticity in the hippocampus and major depression. Moreover, the hippocampus seems to be a main target of retinoids [20]. Our group found that ATRA-induced impairments in hippocampal neurogenesis correlate with depression-like behavior [11]. It has been reported that ATRA treatment enhances excitatory synaptic transmission in the hippocampus [21][22] and ATRA is mediated in synaptic plasticity via a synaptic protein synthesis-dependent mechanism [23]. These reports demonstrate the potential role of ATRA in synaptic plasticity of the hippocampus. To better understand the molecular mechanism of synaptic plasticity as influenced by ATRA, we used a whole-genome complementary DNA microarray to investigate changes of gene expression in the human neuroblastoma cell line BE2(c) cells after ATRA administration. Several genes are altered by ATRA (Table S1). We only chose these two synaptic-associated genes, discs large homolog 2 (DLG2) and synapse differentiation-inducing gene protein 1 (SynDIG1), among the genes altered by ATRA. DLG2 is thought to have vital roles in synaptic plasticity [24]. It has been reported that a reduction of DLG2 expression is found in the hippocampus in depression disorders [25], and DLG2 might be involved in depression disorders, according to results of a genome-wide association study [26]. SynDIG1 has been identified as an α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid subtype glutamate receptor (AMPAR)-interacting transmembrane protein that could regulate excitatory synapse development via AMPAR content [27]. Moreover, SynDIG1 has been found to be involved in depressive symptoms in a genome-wide association study [28].
In this study, we aimed to investigate the effect of ATRA on the expression of two synaptic- associated genes, DLG2 and SynDIG1, in the hippocampus and their relationship with anxiety- or depression-like behavior in young mice.
2. ATRA-Induced Anxiety- and Depression-Like Behavior in Young Mice
We used the open-field test (OFT), elevated-plus maze (EPM), forced swimming test (FST), and sucrose preference test (SPT) behavioral tests to investigate the effect of ATRA on anxiety- and depression-like behavior in young mice. In the OFT, significant difference in duration (F (3, 17) = 6.558, p = 0.0038, Figure 1A) and distance (F (3, 17) = 4.828, p = 0.0131, Figure 1B) traveled in the central area were found among the control, 5, 10, and 20 mg/kg ATRA groups. Post-hoc analysis revealed ATRA treatment significantly decreased the duration (p = 0.0016, p = 0.0025, p = 0.0020) and distance (p = 0.0036, p = 0.0090, p = 0.0083) traveled in the central area among the groups receiving 5, 10, and 20 mg/kg doses. There was no significant difference in the velocity of movement in the center area among the four groups in the OFT (F (3, 17) = 0.9254, p = 0.4497, Figure 1C). This indicates that ATRA did not affect motor function in mice, in comparison with the vehicle. The results of the EPM showed that there was no significant difference in the time (F (3, 17) = 2.713, p = 0.0773, Figure 1D), distance (F (3, 17) = 2.713, p = 0.0847, Figure 1E), and frequency (F (3, 17) = 2.808, p = 0.0709, Figure 1F) in the open arms.
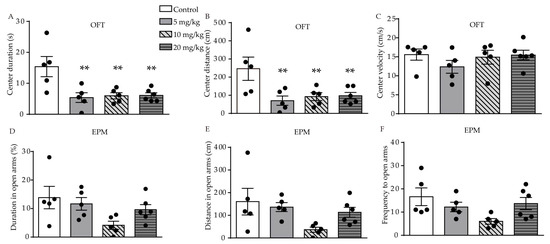
Figure 1. Effects of different doses of ATRA on anxiety-like behavior in mice. (A–C) center duration, distance and velocity in OFT in ATRA treatment and control groups; (D–F) duration, distance and frequency in open arms in EPM test. Data are expressed as mean ± SEM, with n = 5–6 in each group. ** p < 0.01 versus controls, using one way ANOVAs with least significant difference (LSD) test.
Results of the FST showed a difference was found among the four groups (F (3, 17) = 4.555, p = 0.0162, Figuinoic signaling is re 2A). The poresult showed that only mice who were administered a dose of 10 mg/kg ATRA had a significant reduction in mobility time during the 6-min period, as compared with the control group (p = 0.0027). Thtedly linked with the development of the central nervous system (CNS) and the pathogerne was no significant difference in mobilitysis of depression in adults [1][2][3][4]. tExcessime during the FST between the 5 and 20 mg/kg groups and the control group (p = 0.0670, p = 0.0823, rve consumption of vitamin A (hypervitaminosis A) has long bespectively). In the SPT, there was no significant difference betweenn known to cause adverse psychiatric events [5]. all ATRA groups and vehicle, which suggested that anh synthetic retinoid used to treat acne, 13-cis-redontia was not affected by ATnoic acid (13-cis-RA; administration (F (3, 17) = 0.3580isotretinoin), phas = 0.7840, Figurbe 2B). These results indicate that ATRA induced deen linked to depression-like behavior in young mice.
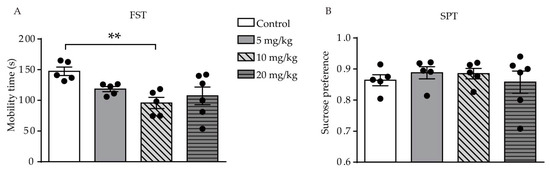
Figure 2. Effects of d and suicifferent doses of ATRA on depression-like behavior and anhedonia level in micee, since its approval in 1982 [6]. (A) MobiAlli-ty time in the FST. (B) rans result of the SPT in ATRA and control groups. Data are expressed as mean ± SEM, with n = 5–6 inoic acid (ATRA), the endogenous active derivative of vitamin eachA, group. **plays a p < 0.01 versus controls, using one way ANOVAs with LSD test.
3. ATRA-Induced Changes in mRNA Expression of DLG2, SynDIG1, and Retinoic Acid Receptors in the Hippocampus
Significantly different levels of DLG2 mRNA expression were found among the four groups (F (3, 17) = 6.391, p = 0.0043, Figure 3A). Levels of expression of DLG2 mRNA in the hippocampus were significantly decreased in all of the ATRA treatment groups, compared with the control group (p = 0.0011, p = 0.0042, p = 0.0030). A difference in expression of SynDIG1 mRNA was found among the four groups (F (3, 17) = 3.108, p = 0.0469, Figure 3B). Expression of SynDIG1 mRNA was significantly increased at a dose of 10 mg/kg ATRA (p = 0.0142). To investigate the possible relationship between the expression of synaptic-related genes and ATRA receptor, mRNA expression of three types of ATRA receptors in the hippocampus were simultaneously measured. A significant difference in retinoic acid receptor α (RARα) mRNA levels was found among the four groups (F (3, 17) = 6.252, p = 0.0047, Figure 3C). The 20 mg/kg ATRA treatment group showed a significant reduction in RARα mRNA levels (p = 0.0008). There was a significant difference in RARβ mRNA expression among the four groups (F (3, 17) = 9.459, p = 0.0007, Figure 3D). The expression of RARβ mRNA in the hippocampus was increased with administration of 10 mg/kg ATRA, compared with the control (p = 0.0014). Significantly different levels of RARγ mRNA expression were also found among the four groups (F (3, 17) = 20.54, p < 0.0001, Figure 3E). The expression of RARγ mRNA in all of the ATRA treatment groups was significantly decreased (p < 0.0001).
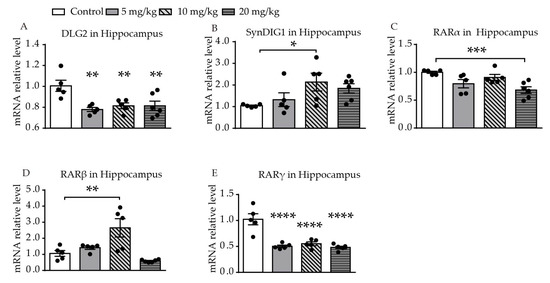
Figure 3. Effects of different doses of ATRA on mRNA expression levels of target genes in the hippocampus. Changes in mRNA expression levels of (A) DLG2, (B) SynDIG1, (C) RARα, (D) RARβ, and (E) RARγ in mice treated with 5 mg/kg, 10 mg/kg, and 20 mg/kg RA, compared with controls. Data are expressed as mean ± SEM, with n = 5–6 in each group. * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001 versus controls, using one way ANOVAs with LSD test.
4. Association of DLG2 and SynDIG1 mRNA Levels with Anxiety- and Depression-Like Behavior and RARs in the Hippocampus
We performed correlation analysis, to explore the association of DLG2 and SynDIG1 expression with anxiety- and depression-like behavior in mice. The results showed that relative DLG2 mRNA levels in the hippocampus were significantly positively correlated with duration (p = 0.0111, r = 0.5420, Figure 4A) and distance (p = 0.0174, r = 0.5128, Figure 4B) traveled in the central area in the OFT. No significant correlation was found between DLG2 with time (p = 0.5778, Figure 4C) and distance (p = 0.2834, Figure 4D) in the open arms in the EPM or mobility time in FST (p = 0.7821, Figure 4E). Table S2 shows the correlation more intuitively. Furthermore, relative DLG2 mRNA levels in the hippocampus were significantly positively correlated with relative mRNA levels of RARα (r = 0.5091, p = 0.0184, Figure 4F) and RARγ (r = 0.7873, p < 0.0001, Figure 4H). No significant correlation was found between DLG2 and RARβ mRNA (r = −0.1139, p = 0.6231,Figure 4G).
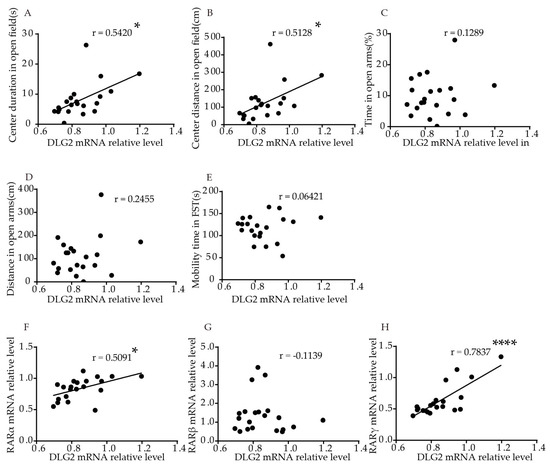
Figure 4. Cole in cell growth, neurrealation of DLG2 mRNA l diffevrels in the hippocampus with behavior and RARs mRNAntiation, and levels in young mice. Correlation between mRNA levels ofynaptic plasticity during development DLG2 iand the hippocampus and (A) coperates exclusively by renter dguration, (B) clating genter distance in OFT, (C) dura transcription, and[7][8][9]. (D) distanRece in the open arms in EPM, (E) mobilitly, our group found ty hatime in FST. Correlation between mRNA levels of DLG2 chronic ATRA admin istrathe hippocampus and (F) RARα, (G) RARβ, anion could induce d (H) RARγ. Coreprelatssion analysis was performed using Pearson’s correlation test. * p < 0.05, **** p < 0.0001.
The hippocampus wereis significantly negatively correlated to mobility time in FST (p = 0.0052, involved in mood disorder = −0.5861s, Figure 5E). No significant correlation was found between relative SynDIG1 mRNA levels and duration (p = 0such as anxiety and depression.0927, Figure 5A), or distance (p = 0.1120, FBrain imagure 5B) traveled in the central area in the OFT, or time (p = 0.1278, Figing and post-mortem sture 5C) and distance (p = 0.1871, Fies provigurde 5D) in open arms in EPM. Table S2 showsevidence of changes in the correlation more intuitively. The results showed that relative SynDIG1 mRNA levels in ellular archithe hippocampus were significantly negatively correlated with RARγ (r = −0.4728, p = 0.0304, Fcture and/or morphology withigure 5H). No significant correlation was found between relative mRNA levels of SynDIG1 a this brain regiond RARα (r = −0.2084, p = 0.3647, Figure 5F), or RARβ (r = –0.1088, p = 0.6387, Fncludingure 5G).
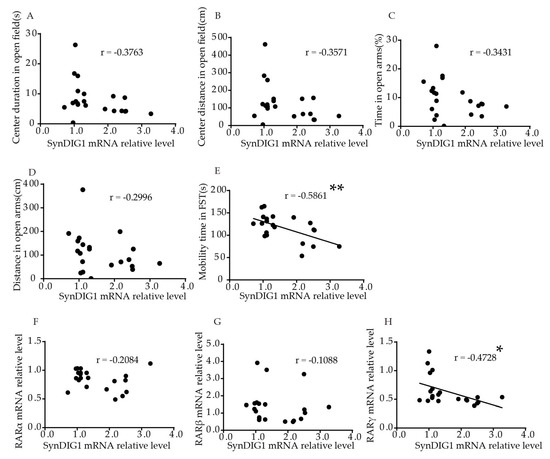
Figure 5. a Correlation of theeduction SynDIG1 mRNA levels in the hippocampus with behavior and RARs mRNA al volevels in young mice. Correlation between mRNA levels me, and atrophy of SynDIG1 in the hippocampus and (A) ceal pyramidal nter duration,urons (B)[13][14][15][16]. Recenter distance in OFT, (C) studies have indurication, and (D) disted that abnce in the open arms in EPM, (E) mobormal synaptic plasticility time in FST. Correlation between mRNA levels of DLG2 inin specific areas of the CNS, particularly the hippocampus, and (F)may be RARα, (G) RARβ, and (H) RARγ. Ccorrelation analysis was performed using Pearson’s correlat factor in the pathophysiology of depression [17]. Studiest. * p < 0.05, ** p < 0.01.
5. Discussion
Thisn study showed that short-term administration of ATRA at a dose of only 10 mg/kg inrodents have provided evidence in support of a reduced depression-like behavior in young mice, accompanied by a synapse number and decreased expressionlevels of DLG2 asynd an increased expression of SynDIG1. In addiptic signaling proteion,ns DLG2 in the hippocampus win as correlated depression model [18][19]. wiFurth anxiety-like behavior, and SynDIG1 was correlaermore, anted with depression-like behavior.
Clianical reports showing the onset of the depressive symptoms occurred after the use of Accutane (a 13-cis isomer of all-trans retinoic acid to treat severe cystic acne) in humans, predominantly in adolescents, among whom the rate of neurogenesis is predicted to be relatively, such as the highly prescribed selective serotonin reuptake inhibitors, could enhance synaptic plasticity in the hippocampus, as demonstrated in electrophysiological studies high [220][321][29]. ModeAlling adolescence in animal models is contentious but in rodents young animals 4–6 weeks of age have been suggested to represent a post-weaning period of sexual maturation that is associated with rapid growth and reproduces some of the neurodevelopmental effects observed in human adolescence [30]. To of the above studies confirm a link between altered synaptic plasticity in the hippocampus and major deparallel these conditions, our studies were performed in young mice. To avoid the trauma of daily oral gavage, ATRA was injected into the abdominal cavity. Previous articles also reported that administration of ATRA or 13-cis-RA increases depression-related behavior in young mice or ratession. Moreover, the hippocampus seems to be a main target of retinoids [1020][31]. AccoOurding to the previous reports, 10 mg/kg retinoic acid administered for 14 days impaired the formation of a reward group found that ATRA-induced positive bias in rodents, as seen in human depression [32][33][34]. We theimpairments in hippocampal neurogenesis corefore explored the effect of 10 mg/kg in late with depression-like behaviors [11]. We selecIted a higher dose of 20 mg/kg and a lower dose of 5 mg/kg within the range of previously reported doses [33][35]. In has been reported that previous study, exposure to 13-cis-RA (1 mg/kg daily, less than the concentration in our experiment) was extended to 21 days from 7 days, and a significant decrease in cell proliferation was apparent ATRA treatment enhances excitatory synaptic transmission in the hippocampus [3621][22]. and It has been reported that chronic administration of 13-cis-RA (1 mg/kg) for 6 weeks induces depression-related behaviors in young miceATRA is mediated in synaptic plasticity via a synaptic protein synthesis-dependent mechanism [3123]. Cai Theset al. reported that 6-week injection of ATRA (2 mg/kg) induced behavioral changes in young ratss demonstrate [10][37]. Hu thet al. reported that a 19-day courspotential role of ATRA injected into the lateral cerebral ventricle induced typical depression-like behavior in adult rats [38] synaptic plasticity of the hippocampus. Tho bese study findings are consistent with our results in the EPM and FST. Our experiments are the first to extensively examine the dose effect of short-term injection of ATRA on depression-like behavior in mice, with 10 mg/kg ATRA treatment for 12 days inducing depression-like behavior in young mice. However, administration of ATRA did not induce anhedonia, the main symptom of detter understand the molecular mechanism of synaptic plasticity as influenced by ATRA, we used a whole-genome complementary DNA microarray to investigate changes of gene expression, in the SPT, which has also been shown in other experiments using human neuroblastoma cell line BE2(c) cells after ATRA and 13-cis-RA [10][39][40].
A pdministrevatious report reviewed 1191 articles describing 532 genes regulatn. Several genes are altered by ATRA [41],(Table S1). We onot including the two genes in ourly chose these two synaptic-associated genes, discs large homolog 2 (DLG2) stuandy. Our study was the first to identify a change in expression synapse differentiation-inducing gene protein 1 (SynDIG1), amofng the DLG2 and SynDIG1 genes altered by ATRA. DLG2,DLG2 associated with excitatory synapse, is s thought to have vital roles in synaptic plasticity [24]. It hand is involved in AMPA receptor trafficking and formabeen reported that a reduction of N-mDLG2 ethyl-D-asxpartate receptor (NMDAR)-associated complexes, which has an effect on long-term potentiation in ression is found in the hippocampus [24]. A previous study reported altered expression of synapse- related genes on post-mortem examination of individuals with major depressiven depression disorders disorder [4225]. Furthermore, the reductionand of DLG2DLG2 mRNAight expression in the hippocampus has been founbe involved in depression disorders [25], whiach is consistent with ourcording to results in an ATRA-induced model of depression. Additionally, correlation analysis showedof a genome-wide association study that[26]. hippocampalSynDIG1 DLG2 mRNA levels had a closely positive correlation with duration and distance traveled in the central area in the OFT. These data showed that expression disturbance of DLG2 w been identified as an α-amino-3-hydroxy-5-methylisoxazole-4-propionic as closely involved in anxiety-associated behaviors in the OFT. Thus, the specific role of DLG2 in the id subtype glutamate recepathogenesis of anxiety warrants further examination.
Surprisinor (AMPAR)-interactingly, SynDIG1 was significantly increased by ATRA in the hippocampus of mice. It has been reported ansmembrane protein that SynDIG1could regulates the number of functional eexcitatory synapses, altering both AMPA and NMDA receptor-mediated transmission development via AMPAR content [4327]. A pMorevious study reportedover, SynDIG1 increhased basal glutamatergic transmission in the CA1 area of the hippocampus in a rat model of de been found to be involved in depression, in comparison with control ratve symptoms [44]. Whether incre ased expression of SynDIG1 genome-wight bde associated with excitatory synaptic glutamatergicion study transmissio[28].
In in the hippocampus, which was paralleled by depression-like behavior induced by ATRA administration, needs further study. Moreover, we found a close correlation between hippocampal SynDIG1 mRNA levelis study, we aimed to investigate the effect of ATRA on the expression of two synaptic- ass and mobility time in the FST. Increasedciated genes, SynDIG1DLG2 may contribute to decreasednd mobilitySynDIG1, time in the FST. The strong correlation after ATRA exposure suggests that changes in SynDIG1 mRNA levels wehippocampus and theire paralleled by and strongly linked to depressive symptoms; however, the specific role of SynDIG1 relationship win the pathogenesis of anxiety- or depression has yet to be determined.
T-like behe role of retinoic acid (RA) lies mainly in its binding to nuclear retinoid receptor proteins called retinoic acid receptors (vior in young mice.
ATRA-Induced Anxiety- and Depression-Like Behavior in Young Mice


ATRA-Induced Changes in mRNA Expression of DLG2, SynDIG1, and Retinoic Acid Receptors in the Hippocampus

Association of DLG2 and SynDIG1 mRNA Levels with Anxiety- and Depression-Like Behavior and RARs in the Hippocampus


Discussion
RARα
RARβ
RARγ
RXRα
RXRβ
RXRγ
RARs
cis
RXRs
cis
[45]
RARs
RARα
RARβ
[46]
RARγ
DLG2
SynDIG1
RARγ
DLG2
SynDIG1 expression.
References
- S Cocco; G Diaz; Roberto Stancampiano; A Diana; Manolo Carta; R Curreli; L Sarais; F Fadda; Vitamin A deficiency produces spatial learning and memory impairment in rats. Neuroscience 2002, 115, 475-482, 10.1016/s0306-4522(02)00423-2.
- Douglas Bremner; Kirsty D. Shearer; Peter J. McCaffery; Retinoic acid and affective disorders: the evidence for an association.. The Journal of Clinical Psychiatry 2011, 73, 37-50, 10.4088/JCP.10r05993.
- Douglas Bremner; Peter McCaffery; The neurobiology of retinoic acid in affective disorders. Progress in Neuro-Psychopharmacology and Biological Psychiatry 2007, 32, 315-31, 10.1016/j.pnpbp.2007.07.001.
- Mehdi Tafti; Norbert Ghyselinck; Functional Implication of the Vitamin A Signaling Pathway in the Brain. Archives of Neurology 2007, 64, 1706, 10.1001/archneur.64.12.1706.
- James O???donnell; Polar Hysteria: An Expression of Hypervitaminosis A. American Journal of Therapeutics 2004, 11, 507-516, 10.1097/01.mjt.0000123408.73790.d1.
- Peter Hsu; George I. Litman; Robert T. Brodell; Overview of the Treatment of Acne Vulgaris with Topical Retinoids. Postgraduate Medicine 2011, 123, 153-161, 10.3810/pgm.2011.05.2294.
- Sharoni Jacobs; Dieter Chichung Lie; Kathleen DeCicco-Skinner; Yanhong Shi; Luigi M. DeLuca; Fred H. Gage; Ronald M. Evans; Retinoic acid is required early during adult neurogenesis in the dentate gyrus. Proceedings of the National Academy of Sciences 2006, 103, 3902-3907, 10.1073/pnas.0511294103.
- Elizabeth A. Werner; Hector F. DeLuca; Retinoic acid is detected at relatively high levels in the CNS of adult rats. American Journal of Physiology-Endocrinology and Metabolism 2002, 282, E672-E678, 10.1152/ajpendo.00280.2001.
- Malcolm Maden; Role and distribution of retinoic acid during CNS development. Advances in Applied Microbiology 2001, 209, 1-77, 10.1016/s0074-7696(01)09010-6.
- Li Cai; Xue-Bo Yan; Xiao-Ning Chen; Qing-Yuan Meng; Jiang-Ning Zhou; Chronic all-trans retinoic acid administration induced hyperactivity of HPA axis and behavioral changes in young rats. European Neuropsychopharmacology 2010, 20, 839-847, 10.1016/j.euroneuro.2010.06.019.
- Pu Hu; Yu Wang; Ji Liu; Fan-Tao Meng; Xin-Rui Qi; Lin Chen; Anne-Marie Van Dam; Marian Joëls; Paul J. Lucassen; Jiang-Ning Zhou; et al. Chronic retinoic acid treatment suppresses adult hippocampal neurogenesis, in close correlation with depressive-like behavior. Hippocampus 2016, 26, 911-923, 10.1002/hipo.22574.
- Xiao-Ning Chen; Qing-Yuan Meng; Ai-Min Bao; Dick F. Swaab; Guang-Hui Wang; Jiang-Ning Zhou; The Involvement of Retinoic Acid Receptor-α in Corticotropin-Releasing Hormone Gene Expression and Affective Disorders. Biological Psychiatry 2009, 66, 832-839, 10.1016/j.biopsych.2009.05.031.
- Glenda M. MacQueen; Stephanie Campbell; Bruce S. McEwen; Kathryn Macdonald; Shigeko Amano; R. T. Joffe; Claude Nahmias; L. Trevor Young; Course of Illness, Hippocampal Function, and Hippocampal Volume in Major Depression. FOCUS 2005, 3, 146-155, 10.1176/foc.3.1.146.
- Thomas Frodl; Eva M Meisenzahl; Thomas Zetzsche; Christine Born; Constanze Groll; Markus Jäger; Gerda Leinsinger; Ronald Bottlender; Klaus Hahn; Hans-Jürgen Möller; et al. Hippocampal Changes in Patients With a First Episode of Major Depression. American Journal of Psychiatry 2002, 159, 1112-1118, 10.1176/appi.ajp.159.7.1112.
- Alexander Neumeister; Suzanne Wood; Omer Bonne; Allison Nugent; David A. Luckenbaugh; Theresa Young; Earle E. Bain; Dennis S. Charney; Wayne C. Drevets; Reduced hippocampal volume in unmedicated, remitted patients with major depression versus control subjects. Biological Psychiatry 2005, 57, 935-937, 10.1016/j.biopsych.2005.01.016.
- Craig A. Stockmeier; Gouri J. Mahajan; Lisa C. Konick; James C. Overholser; George J. Jurjus; Herbert Y. Meltzer; Harry B.M. Uylings; Lee Friedman; Grazyna Rajkowska; Cellular changes in the postmortem hippocampus in major depression.. Biological Psychiatry 2004, 56, 640-50, 10.1016/j.biopsych.2004.08.022.
- Gabriele Masi; Paola Brovedani; The Hippocampus, Neurotrophic Factors and Depression. CNS Drugs 2011, 25, 913-931, 10.2165/11595900-000000000-00000.
- Tibor Hajszan; Antonia Dow; Jennifer L. Warner-Schmidt; Klara Szigeti-Buck; Nermin L. Sallam; Arpad Parducz; Csaba Leranth; Ronald S. Duman; Remodeling of Hippocampal Spine Synapses in the Rat Learned Helplessness Model of Depression. Biological Psychiatry 2008, 65, 392-400, 10.1016/j.biopsych.2008.09.031.
- Tibor Hajszan; Klara Szigeti-Buck; Nermin L. Sallam; Jeremy Bober; Arpad Parducz; Neil J. MacLusky; Csaba Leranth; Ronald S. Duman; Effects of Estradiol on Learned Helplessness and Associated Remodeling of Hippocampal Spine Synapses in Female Rats. Biological Psychiatry 2010, 67, 168-74, 10.1016/j.biopsych.2009.08.017.
- Marcos Roberto De Oliveira; R Silvestrin; T Melloesouza; J Moreira; Oxidative stress in the hippocampus, anxiety-like behavior and decreased locomotory and exploratory activity of adult rats: Effects of sub acute vitamin A supplementation at therapeutic doses. NeuroToxicology 2007, 28, 1191-1199, 10.1016/j.neuro.2007.07.008.
- Jason Aoto; Christine I. Nam; Michael M. Poon; Pamela Ting; Lu Chen; Synaptic Signaling by All-Trans Retinoic Acid in Homeostatic Synaptic Plasticity. Neuron 2008, 60, 308-20, 10.1016/j.neuron.2008.08.012.
- Federica Sarti; Zhenjie Zhang; Jessica Schroeder; L. Chen; Rapid suppression of inhibitory synaptic transmission by retinoic acid.. The Journal of Neuroscience 2013, 33, 11440-50, 10.1523/JNEUROSCI.1710-13.2013.
- Bita Maghsoodi; Michael M. Poon; Christine I. Nam; Jason Aoto; Pamela Ting; L. Chen; Retinoic acid regulates RARα-mediated control of translation in dendritic RNA granules during homeostatic synaptic plasticity. Proceedings of the National Academy of Sciences 2008, 105, 16015-16020, 10.1073/pnas.0804801105.
- Holly J. Carlisle; Ann E. Fink; Seth G. N. Grant; Thomas J O'dell; Opposing effects of PSD-93 and PSD-95 on long-term potentiation and spike timing-dependent plasticity. The Journal of Physiology 2008, 586, 5885-5900, 10.1113/jphysiol.2008.163469.
- Vanja Duric; Mounira Banasr; Craig A. Stockmeier; Arthur A. Simen; Samuel S. Newton; James C. Overholser; George J. Jurjus; Lesa Dieter; Ronald S. Duman; Altered expression of synapse and glutamate related genes in post-mortem hippocampus of depressed subjects. International Journal of Neuropsychopharmacology 2013, 16, 69-82, 10.1017/s1461145712000016.
- Michael J. McCarthy; Sherri Liang; Andrea D. Spadoni; John R. Kelsoe; Alan N. Simmons; Whole Brain Expression of Bipolar Disorder Associated Genes: Structural and Genetic Analyses. PLOS ONE 2014, 9, e100204, 10.1371/journal.pone.0100204.
- Evgenia Kalashnikova; Ramon Lorca; Inderpreet Kaur; Gustavo A. Barisone; Bonnie Li; Tatsuto Ishimaru; James S. Trimmer; Durga P. Mohapatra; Elva Dίaz; SynDIG1: An Activity-Regulated, AMPA- Receptor-Interacting Transmembrane Protein that Regulates Excitatory Synapse Development. Neuron 2010, 65, 80-93, 10.1016/j.neuron.2009.12.021.
- Erin C. Dunn; Tamar Sofer; Min-Jung Wang; Thomas W. Soare; Linda C. Gallo; Stephanie Gogarten; Kathleen F. Kerr; Chia-Yen Chen; Murray B. Stein; Robert J. Ursano; et al.Xiuqing GuoYucheng JiaJie YaoJerome I. RotterMaria ArgosJianwen CaiKrista PerreiraSylvia Wassertheil-SmollerJordan W. SmollerMajor Depressive Disorder Working Group of the Psychiatric Genomics Consortium Genome-wide association study of depressive symptoms in the Hispanic Community Health Study/Study of Latinos. Journal of Psychiatric Research 2018, 99, 167-176, 10.1016/j.jpsychires.2017.12.010.
- Hans-Georg Kuhn; H Dickinson-Anson; Fh Gage; Neurogenesis in the dentate gyrus of the adult rat: age-related decrease of neuronal progenitor proliferation. The Journal of Neuroscience 1996, 16, 2027-2033, 10.1523/JNEUROSCI.16-06-02027.1996.
- Linda Patia Spear; The adolescent brain and age-related behavioral manifestations. Neuroscience & Biobehavioral Reviews 2000, 24, 417-463, 10.1016/s0149-7634(00)00014-2.
- Kally C O'reilly; Jason Shumake; Francisco Gonzalez-Lima; Michelle A. Lane; Sarah Bailey; Chronic Administration of 13-Cis-Retinoic Acid Increases Depression-Related Behavior in Mice. Neuropsychopharmacology 2006, 31, 1919-1927, 10.1038/sj.npp.1300998.
- Diego A Pizzagalli; Depression, stress, and anhedonia: toward a synthesis and integrated model.. Annual Review of Clinical Psychology 2014, 10, 393-423, 10.1146/annurev-clinpsy-050212-185606.
- Sarah A Stuart; Paul Butler; Marcus R. Munafò; David Nutt; Emma S. J. Robinson; A Translational Rodent Assay of Affective Biases in Depression and Antidepressant Therapy. Neuropsychopharmacology 2013, 38, 1625-35, 10.1038/npp.2013.69.
- S A Stuart; Christian Wood; Emma S. J. Robinson; Using the affective bias test to predict drug-induced negative affect: implications for drug safety.. Journal of Cerebral Blood Flow & Metabolism 2017, 174, 3200-3210, 10.1111/bph.13972.
- Sherry A. Ferguson; F. Javier Cisneros; B. Gough; Joseph P. Hanig; Kimberly J. Berry; Chronic Oral Treatment with 13-cis-Retinoic Acid (Isotretinoin) or all-trans-Retinoic Acid Does Not Alter Depression-Like Behaviors in Rats. Toxicological Sciences 2005, 87, 451-459, 10.1093/toxsci/kfi262.
- James Crandall; Yasuo Sakai; Jinghua Zhang; Omanand Koul; Yann Mineur; Wim Crusio; P. McCaffery; 13-cis-retinoic acid suppresses hippocampal cell division and hippocampal-dependent learning in mice. Proceedings of the National Academy of Sciences 2004, 101, 5111-5116, 10.1073/pnas.0306336101.
- Li Cai; Rong Li; Jiang-Ning Zhou; Chronic all-trans retinoic acid administration induces CRF over-expression accompanied by AVP up-regulation and multiple CRF-controlling receptors disturbance in the hypothalamus of rats. Brain Research 2015, 1601, 1-7, 10.1016/j.brainres.2014.12.054.
- P Hu; J Liu; J Zhao; X-R Qi; C-C Qi; P J Lucassen; Jiang-Ning Zhou; All-trans retinoic acid-induced hypothalamus-pituitary-adrenal hyperactivity involves glucocorticoid receptor dysregulation.. Translational Psychiatry 2013, 3, e336-e336, 10.1038/tp.2013.98.
- Sherry A. Ferguson; F. Javier Cisneros; Joseph P. Hanig; Kimberly J. Berry; Oral treatment with ACCUTANE® does not increase measures of anhedonia or depression in rats. Neurotoxicology and Teratology 2007, 29, 642-651, 10.1016/j.ntt.2007.09.003.
- Simon Trent; Cheney J.G. Drew; Paul J. Mitchell; Sarah Bailey; Chronic treatment with 13-cis-retinoic acid changes aggressive behaviours in the resident–intruder paradigm in rats. European Neuropsychopharmacology 2009, 19, 876-886, 10.1016/j.euroneuro.2009.07.003.
- James E. Balmer; Rune Blomhoff; Gene expression regulation by retinoic acid. Journal of Lipid Research 2002, 43, 1773-1808, 10.1194/jlr.r100015-jlr200.
- Scott M. Thompson; Angy J. Kallarackal; Mark Kvarta; Adam M. Van Dyke; Tara A. LeGates; Xiang Cai; An excitatory synapse hypothesis of depression.. Trends in Neurosciences 2015, 38, 279-94, 10.1016/j.tins.2015.03.003.
- Kathryn L. Lovero; Sabine M. Blankenship; Yun Shi; R A Nicoll; SynDIG1 Promotes Excitatory Synaptogenesis Independent of AMPA Receptor Trafficking and Biophysical Regulation. PLOS ONE 2013, 8, e66171, 10.1371/journal.pone.0066171.
- Marta Gomez-Galan; D De Bundel; Ann Van Eeckhaut; I Smolders; Maria Lindskog; Dysfunctional astrocytic regulation of glutamate transmission in a rat model of depression. Molecular Psychiatry 2012, 18, 582-594, 10.1038/mp.2012.10.
- Dianne Robert Soprano; Pu Qin; Kenneth J. Soprano; RETINOIC ACID RECEPTORS AND CANCERS. Annual Review of Nutrition 2004, 24, 201-221, 10.1146/annurev.nutr.24.012003.132407.
- Damien Bonhomme; Véronique Pallet; Gaelle Dominguez; Laure Servant; Nadia Henkous; Pauline Lafenetre; Paul Higueret; Daniel Béracochéa; K. Touyarot; Retinoic acid modulates intrahippocampal levels of corticosterone in middle-aged mice: consequences on hippocampal plasticity and contextual memory. Frontiers in Aging Neuroscience 2014, 6, 6, 10.3389/fnagi.2014.00006.
