Probiotics and prebiotics have gained considerable attention as an eco-friendly alternative to antibiotics. This study investigated the effects of the probiotics Saccharomyces cerevisiae and Pediococcus acidilactici, alongside the prebiotic yeast cell wall, on the growth, intestinal morphology and liver and gills of common carp (Cyprinus carpio L.). The results showed considerable improvements in intestinal morphology (e.g. villi width and muscular layer thickness), with no pathological signs on the liver or gills and no changes to growth parameters. Additionally, a significant change in the gut microbiome was observed in fish that received a diet supplemented with the tested feed supplements at a concentration of 1 g/kg for 60 days in a recirculating aquaculture system.
- common carp
- Pediococcus acidilactici
- Saccharomyces cerevisiae
- yeast extract
- histomorphology
- oxford nanopore sequencing
- gut microbiota
1. Materials and Methods
- Materials and Methods
- Ethical issues
1.1. Ethical Issues
Experiments on common carp in the current study were conducted based on ethical guidelines and legal requirements. The Department of Animal Husbandry of the Institute of Animal Science, Biotechnology and Nature Conservation at the University of Debrecen obtained the necessary permit for these experiments from the Animal Welfare Committee of the University of Debrecen (15/2019/DE MÁB Kovács László). This permit, issued by the Hajdú-Bihar County Government Office on March 5, 2020, has a validity of five years (until March 5, 2025) and was authorized by Dr. Sándor Mihálka, Chief Veterinary Inspector.
- Feed composition
1.2. Feed Composition
The commercially available fish feed used in this study was Aqua Start (Garant Aqua, Austria). It is an extruded diet in the form of 1.5 mm pellets. According to the feed manufacturer, it was formulated to support optimal growth and health of juvenile fish. The composition of the feed, based on dry matter, included 52.0% crude protein, 18.0% crude fat, 0.5% crude fiber, 9.5% crude ash, 1.5% phosphorus, and 1.8% calcium. The feed was further supplemented with essential vitamins and minerals, including 15,000 IU/kg of vitamin A, 2,000 IU/kg of vitamin D₃, 200 mg/kg of vitamin E, and 250 mg/kg of stabilized vitamin C, and the major listed ingredients were wheat, fish meal, corn gluten, soybean protein concentrate, fish oil, and a vitamin–mineral premix. This feed acted as the basal diet for all experimental groups.
- Fish Preparation and PIT Tagging
1.3. Fish Preparation and PIT Tagging
Common carp (26.4 ± 5.2 g) were reared in a recirculating system, Fish Laboratory of the University of Debrecen, Hungary. After the acclimatization for 14 days, 120 fish were randomly distributed into 12 fish tanks and 4 treatments (10 fish per tank, 30 fish per treatment); the system was equipped with mechanical and aerated biofilters. To monitor fish performance during the experiment, individuals were assigned to PIT tags (Oregon RFID; Portland, OR, USA). Briefly, all fish were weighed prior to the experiment (g) and then individually injected with the PIT tag in front of the dorsal fin (Figure 1). After injection, tag function was checked using a hand-held PIT tag reader (iD Porte Ltd; Guernsey, Islands). The tagged fish were then left in the fish tanks for 72 h to allow the stress caused by the PIT injection to recover and then assigned to the experimental diets for 60 days.
- The experimental diets and animal housing
1.4. The Experimental Diets and Animal Housing
Three different feed additives, each produced by the Lallemand Animal Nutrition (Canada), were tested: Bactocell® is based on live lactic acid bacterium (Pediococcus acidilactici CNCM I-4622 MA 18/5M); Levucell® contains the probiotic Saccharomyces cerevisiae var. boulardii CNCM 1-1079 strain; YANG®, a commercial product from the yeast cell wall extract of Cyberlindnera jandinii and S. cerevisiae. The supplemented diets were prepared by mixing commercial feed pellets (Section 2.2) with each feed additive and sunflower oil (20 ml/ kg) in accordance with protocols by the product's manufacturer. The next step was to dry the mixture at 39°C for 24 h. After that, it was left at room temperature for daily use. The feeding trial has been conducted with 3 replicates (3 tanks/treatment) and 4 groups as follows: fish in the control group received a commercial feed only (C), whereas animals in the other 3 groups received diet supplemented with 1 g/kg of P. acidilactici (PA), S. cerevisiae (SC), and YANG® (YP), respectively. The water capacity of each tank was 350 liters. The RAS system was provided with an automated feeding system, and the feeding process was programmed to be three times a day (08:00, 14:00, and 20:00) with feeding rates of 3% of the total biomass. The lighting conditions were 12 h of light and 12 h of darkness. The removal of feces and uneaten feed was regular practice, performed as needed.

Figure 1. The injection of a Passive Integrated Transponder (PIT) tag to each fish for the purpose of individual identification. The white arrow shows the dorsal fin, and the yellow circle indicates the tagging site, which is located below the base of the dorsal fin.
- Water quality
1.5. Water Quality
Throughout the experimental period, the dissolved oxygen concentration (mg/L) was kept at 6.78 ± 0.3, the temperature (°C) at 25.9 ± 0.9, and the pH at 7.4 ± 0.14. Meanwhile, the concentrations of nitrate (NO₃⁻), nitrite (NO₂⁻) and ammonium (NH₄⁺) were monitored on a regular basis using a DR3900 Spectrophotometer (Hach, USA). The recorded values were within the optimal range: TDS concentration was 443 ± 6.7 ppm; nitrate concentration was 5.48 ± 2.97 mg/L; nitrite concentration was 0.24 ± 0.3 mg/L; and ammonium concentration was 0.37 ± 0.18 mg/L.
- Growth indices measurements
1.6. Growth Indices Measurements
Every couple of weeks, all the fish were collected then euthanized (Fernandes et al., 2016). The wet body weight (BW, g) was measured. feed conversion ratio (FCR, g/g); Survival rates (S%); and weight gain (WG, g); were calculated based on the following mathematical formulas:
- S (%) = (harvested individuals/stocked individuals) × 100
- WG (g) = Wf-Wi
- FCR) (g/g) = F/ (Wf - Wi)
where Wf is the final body weight (g), Wi is the initial wet body weight (g); F is feed intake (g) and t is the time (day),
- Histological examinations
1.7. Histological Examinations
After the feeding experiment ended (60 days), three carps from each group were selected randomly and anesthetized by bathing for 5 min in a clove oil solution (Fernandes et al., 2016) at 0.025 ml/liter at 23 ◦C. The fish intestine, liver, and liver were carefully withdrawn. The anterior, posterior, and mid-intestine of each fish are clearly identified. The intestinal and liver samples were then washed in PBS solution and fixed in 10% buffered formalin until analysis. The histochemical investigation was carried out as follows: samples were dehydrated in ethanol, cleaned, embedded and fixed in paraffin. Subsequently, the fixed tissues were sectioned at a thickness of 5 µm using a rotary microtome (Leica 2025, Germany). To examine the intestinal microstructure, hematoxylin-eosin (H&E) staining was conducted on 12 samples (n = 3 fish per treatment, 4 cross sections per individual). A light microscope (BX61, Olympus, Japan) connected with camera (DP71, Olympus, Japan) was used to capture images of stained cross sections (Fischer et al., 2008). The slides were observed under a light microscope with a magnification of 100x. Twelve villi per fish were randomly and carefully chosen from the cross-section per individual animal. The length of villi (LV), villus width (VW), crypt depth (CD), and the muscular layer thickness (ML) were measured (μm). Measurements were conducted with Olympus CellSens Entry Software (APA, MLA, Chicago, USA).
- Analysis of microbial communities of the intestine
1.8. Analysis of Microbial Communities of the Intestine
DNA Extraction1.8.1. DNA Extraction
The extraction of DNA was carried out in accordance with a standardized protocol. In summary, the samples were briefly melted at room temperature, mixed with lysis buffer (400 mM NaCl, 10 mM Tris-HCl, 100 mM EDTA, 0.4% SDS, and 24 μg/mL protease K, pH 8.0), and then incubated for 24 hours at 56°C. Following the incubation process, the samples were centrifuged at speed of 5,000 rpm. Subsequently, the upper layer of the samples was mixed with molecular-grade absolute ethanol and filtered through a silica column by means of centrifugation at a temperature equivalent to that of the surrounding environment for one minute at a speed of 8,500 revolutions per minute. Following two washing steps, the DNA was eluted in 30 μL of DEPC water. The DNA integrity was then evaluated through agarose gel electrophoresis. Subsequently, DNA concentration was measured in accordance with the manufacturer's guidelines. This was accomplished by employing the Qubit™ dsDNA HS Assay Kit (Thermo Scientific, USA). For the 16S rRNA gene amplification, the isolated DNA from all samples was utilized in a polymerase chain reaction (PCR) with primers designed to amplify the full-length 16S rRNA gene. The specific primers employed were 27F (5′-AGAGTTTGATCCTGGCTCAG-3′) and 1492R (5′-GGTTACCTTGTTACGACTT-3′), and Taq DNA polymerase LongAmp (New England Biolabs, USA) was used as the polymerase. The program for the polymerase chain reaction (PCR) included a denaturation step at 95°C for one minute, followed by 25 cycles of 95°C for 20 seconds, 56°C for 30 seconds, and 65°C for two minutes, and an extension at 65°C for five minutes. The 16S rRNA PCR amplicons were finally confirmed through agarose gel electrophoresis. Samples with 10 nonograms of high-molecular-weight were selected for library preparation.
Library Preparation1.8.2. Library Preparation
Library preparation was conducted using the 16S Barcoding Kit 24 V14 (SQK-16S114.24), following the manufacturer's instructions (Oxford Nanopore Technologies, UK). The process involved the following steps: Barcoded PCR Amplification: The 16S rRNA gene from each DNA sample was amplified using the barcoded primers included in the kit. Reactions were prepared in 0.2 ml thin-walled PCR tubes containing 15 µl of template DNA diluted in nuclease-free water and 25 µl of LongAmp™ Hot Start Taq 2X Master Mix (NEB, M0533S), for a total volume of 40 µl. A new pipette tip was used for each barcode to prevent cross-contamination. Thermal cycling conditions were as follows: initial denaturation at 95°C for 1 minute, followed by 25 cycles of 95°C for 20 seconds (denaturation), 55°C for 30 seconds (annealing), and 65°C for 2 minutes (extension), with a final extension step of 65°C for 5 minutes. The reaction was held at 4°C until further processing.
Bioinformatic analysis1.8.3. Bioinformatic Analysis
The base-calling of sequencing reads was conducted using Guppy (version 3.2.2, Oxford Nanopore Technologies, UK), and an initial quality filter was implemented to retain only those sequences that attained a Q-score of at least 7. The remaining reads were subsequently demultiplexed and trimmed using the Fastq 16S (Oxford Nanopore Technologies, UK) pipeline with default parameters. Filtered reads were initially annotated through Blastn against the NCBI RefSeq 16S ribosomal RNA database using EPI2ME software (Oxford Nanopore Technologies). For assigning each bacterial species, 16S rRNA gene reads that exceeded 1,400 nucleotides in length and demonstrated an accuracy level above 85% were subjected to further analysis. In the final list of identified bacterial species, species represented with less than ten reads were not considered. The Simpson's and Shannon's indexes were utilized to estimate alpha diversity, while Pielou's index was employed to estimate evenness.
- Statistical analysis
1.8.4. Statistical Analysis
The obtained data was subjected to the statistical analysis using IBM SPSS Statistics for Windows, Version 24.0 (Armonk, NY: IBM Corp.). One-way analysis of variance (ANOVA) was carried out, the variance equality was checked by Bartlett’s test and Tukey’s test was used for the post hoc tests. The data were also checked for normality by using the Shapiro-Wilk test. Evenness was calculated as the ratio of Shannon diversity to the natural logarithm of species richness. Samples with fewer than 5,000 reads were excluded from the analysis. Differences in alpha diversity between the four treatment groups were tested using the Kruskal–Walli’s test. Data were presented as means, and the significance level for differences was set at p< 0.05. Differences among groups were considered significant at p <0.05. Then the results were expressed as mean ± std (standard deviation of the mean).
2. Results
- Results
2.1. The Growth Performance
3.1. The growth performance
The results showed that fish in all treatment groups exhibited progressive increases in body weight (BW, g) and weight gain (WG, g) from 17th November 2023 to 18th January 2024, with the highest final BW recorded in the PA supplemented diet (106.8 ± 25.8 g), followed by YP (105.9 ± 25.3 g), the control (103.8 ± 30.8 g), and SC (99.9±25.5). Similarly, the highest WG was recorded in the YP (34.34 ± 16.2 g) and PA (36.1 ± 20.7 g) groups. Feed conversion ratio (FCR) improved over time and was similar across all groups at the end of the trial (1.0 ± 0.1), while survival rate (S%) remained at 100% in all groups throughout the study. The statistical analysis revealed no significant differences (p > 0.05) among the control and supplemented groups in terms of BW, WG, FCR, or S% at any sampling point. These findings indicate that none of the dietary supplements negatively affected growth performance or survival, and all feed additives tested were safe as the control diet under the given experimental conditions
Table 1.
Growth parameters of Common carp (
C. carpio
) received different dietary feed supplements over time (8-week trial)
|
Growth parameters |
Feed supplements |
17th Nov. 2023 |
4th Dec. 2023 |
18th Dec. 2023 |
2nd Jan. 2024 |
18th Jan. 2024 |
|
|
Control |
26.3±5.2 |
36.2±9 |
51.5±12 |
71.9±18 |
103.8±30.8 |
|
|
PA |
26.6±4.5 |
36.8±7.7 |
50.7±10.7 |
71.7±14 |
106.8±25.8 |
|
BW (g) |
SC |
27±5.6 |
35.2±8.3 |
50.5±12.9 |
69.4±17 |
99.9±25.5 |
|
|
YP |
26.4±5.2 |
35.9±8.6 |
51.1±10.3 |
71.6±16 |
105.9±25.3 |
|
|
P-value |
0.886 |
0.89 |
0.95 |
0.92 |
0.75 |
|
|
Control |
- |
9.93±5.6 |
14.94±6.3 |
21.11±15.5 |
31.86±19.8 |
|
|
PA |
- |
10.21±7.6 |
11.54±11.1 |
20.01±10.7 |
36.1±20.7 |
|
WG (g) |
SC |
- |
8.2±4.2 |
15.37±5.9 |
18.89±4.6 |
30.4±14.4 |
|
|
YP |
- |
9.94±5.4 |
16.2±6.6 |
19.53±7.7 |
34.34±16.2 |
|
|
P-value |
|
0.53 |
0.07 |
0.85 |
0.62 |
|
|
Control |
- |
1.9±0.4 |
1.4±0.1 |
0.9±0.1 |
1.0±0.2 |
|
|
PA |
- |
2.0±0.7 |
1.6±0.4 |
0.9±0.1 |
1.0±0.2 |
|
FCR (g/g) |
SC |
- |
2.1±0.8 |
1.4±0.2 |
1.0±0.01 |
1.0±0.1 |
|
|
YP |
- |
2.1±0.5 |
1.3±0.05 |
0.9±0.1 |
1.0±0.1 |
|
|
P-value |
|
0.97 |
0.54 |
0.71 |
0.9 |
|
|
Control |
- |
100±0 |
100±0 |
100±0 |
100±0 |
|
|
PA |
- |
100±0 |
100±0 |
100±0 |
100±0 |
|
S% |
SC |
- |
100±0 |
100±0 |
100±0 |
100±0 |
|
|
YP |
- |
100±0 |
100±0 |
100±0 |
100±0 |
“PA”:
Pediococcus acidilactici
, “SC”:
Saccharomyces cerevisia
, “YP”: Yeast prebiotic
2.2. The Intestinal Histoarchitecture
3.2. The intestinal histoarchitecture
Dietary treatments significantly influenced intestinal morphology in the anterior, mid, and posterior segments of C. carpio after 8 weeks (Table 2, Figure 2). Villi length, width, muscular thickness, and crypt depth were all significantly higher (p < 0.05) in the anterior intestine of all supplemented groups compared to the untreated control. The probiotics S. cerevisiae and P. acidilactici exhibited the highest villi length in the anterior intestine (1917.59 ± 301.68 µm and 1852.54 ± 217.84 µm, respectively) followed by other groups.
Regarding the mid intestine (Table 3), villi length and width significantly (p < 0.05) improved in S. cerevisiae supplemented diet, which had the longest villi (1350.20 ± 82.0 µm) and villi width (176.9±49.7 µm), however there were no significant effect (p < 0.05) between other groups. crypt depth and Muscular thickness did not differ significantly in the mid-intestine, though numerical changes were observed in supplemented groups.
Dietary supplementation with the probiotic S. cerevisiae had positive effects on intestinal morphology in the posterior intestine of C carpio (Table 4). Villus length was significantly differing (p = 0.023) between groups. The highest villus length was observed in the S. cerevisiae group (1,167.28 ± 150.96 µm), followed by the yeast prebiotic (947.68 ± 274.02 µm) and the P. acidilactici (784.41 ± 72.16 µm). The control group had the shortest villi (779.34 ± 158.73 µm). Villus width, muscular thickness, and crypt depth did not differ significantly among treatments (p > 0.05), but all supplemented groups had higher values than the control group. These results suggest that dietary probiotics and prebiotics improve posterior intestinal structure by enhancing villus length without negatively affecting other histomorphological parameters.
Table 2
. Effects of the tested feed supplements on the anterior intestine for
C. carpio
(Mean ± SD).
|
Parameters (µm) |
Control |
PA |
SC |
YP |
p-value |
|
Villi length |
1652.5±213.8a |
1917.9±301.8b |
1652.5±214.7bc |
1359.5±253.3bcd |
0.000 |
|
Villi width |
156.3±36.4a |
156.4±33.5a |
135.9±10.0ab |
140.1±24.3ab |
0.002 |
|
Muscular thickness |
110.4±29.3a |
103.8±30.4a |
94.3±14.7a |
140.1±19.1b |
0.000 |
|
Crypt depth |
39.3±5.4a |
69.7±13.2b |
67.9±27.8b |
66.3±39.2b |
0.031 |
Different letters within rows indicate significant differences (LSD,
p
< 0.05)
“PA”:
Pediococcus acidilactici
, “SC”:
Saccharomyces cerevisia, “YP”: Yeast prebiotic
, “YP”: Yeast prebiotic
Table 3
. Effects of the tested feed supplements on the midintestines for
C. carpio
(Mean ± SD).
|
Parameters (µm) |
Control |
PA |
SC |
YP |
p-value |
|
Villi length |
1042±175 a |
1069±300 a |
1350±202 b |
1132±182 a |
0.015 |
|
Villi width |
133.1±20.5 a |
110.7±22.1 a |
176.9±49.7 b |
139.1±43.5 a |
0.003 |
|
Muscular thickness |
77.9±27.8 a |
69.2±24.7 a |
93.6±32.4 a |
96.4±25.1 a |
0.107 |
|
Crypt depth |
46.8±10.6 a |
43.8±16.2 a |
48.0±6.0 a |
52.1±13.9 a |
0.509 |
Different letters within rows indicate significant differences (LSD,
p
< 0.05)
“PA”:
Pediococcus acidilactici
, “SC”:
Saccharomyces cerevisia
, “YP”: Yeast prebiotic
Table 4
. Effects of the tested feed supplements on the posterior intestine for
C. carpio
(Mean ± SD).
|
Parameters (µm) |
Control |
PA |
SC |
YP |
p-value |
|
Villi length |
779.3±188.7a |
784.4±171.2a |
1167.2±505.9b |
847.6±282.7a |
0.028 |
|
Villi width |
114.8±18.2a |
116.0±25.5a |
116.0±58.1a |
116.0±57.7a |
0.111 |
|
Muscular thickness |
67.0±26.1a |
71.1±16.1a |
78.0±18.5a |
74.2±23.4a |
0. 722 |
|
Crypt depth |
38.3±17.9a |
51.4±7.1a |
50.0±15.1a |
40.7±11.8a |
0.092 |
Different letters within rows indicate significant differences (LSD,
p
< 0.05)
“PA”:
Pediococcus acidilactici
, “SC”:
Saccharomyces cerevisia, “YP”: Yeast prebiotic
, “YP”: Yeast prebiotic
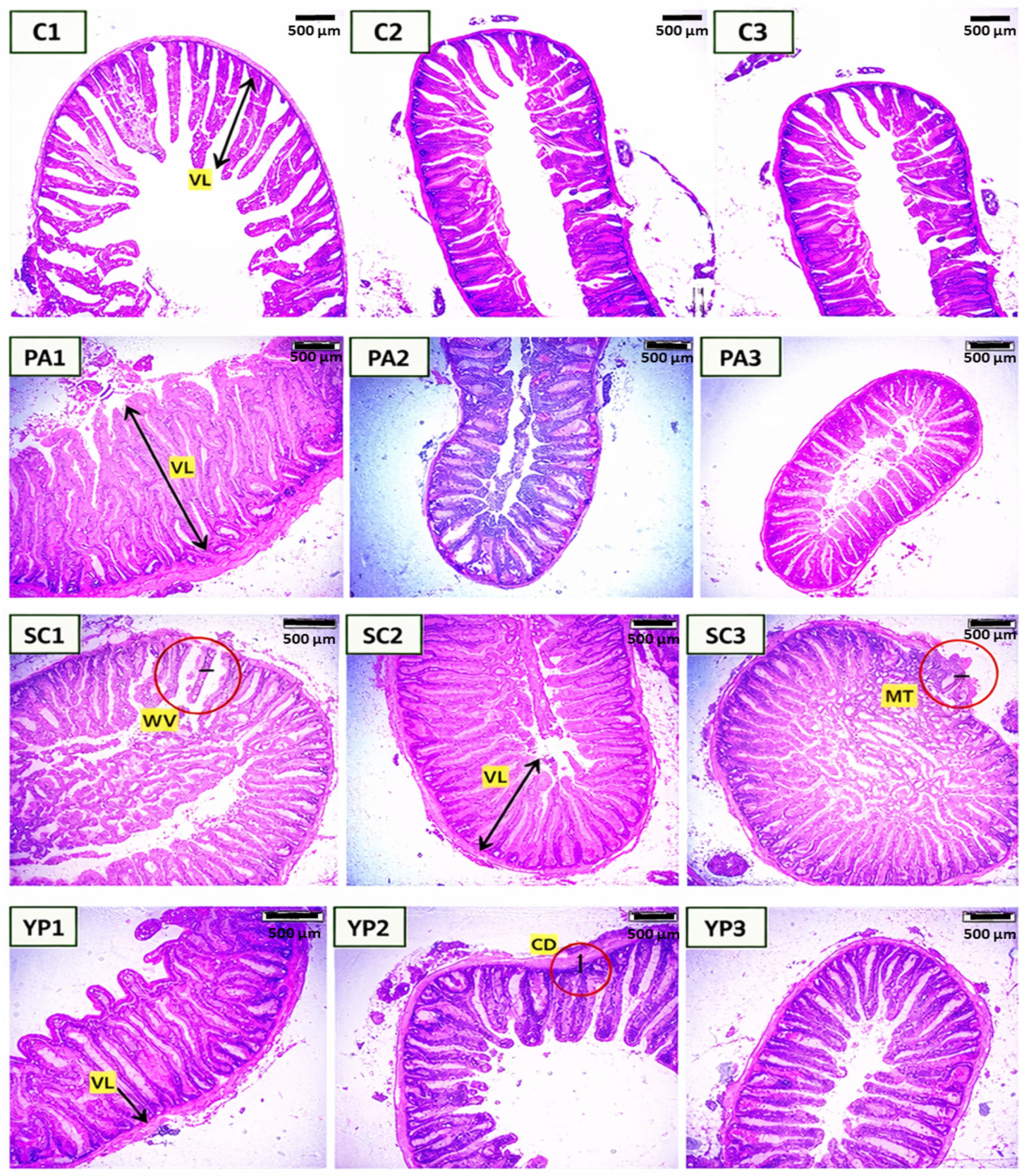
Figure 2. Histological cross sections (H&E) of anterior (1), mid (2), and posterior intestine (3) of common carp showed the effect of P. acidilactici (PA), S. cerevisiae (SC), and the yeast cell wall prebiotic (YP), in comparison with the control (C). VL: villus length, MT: Muscular layer thickness, VW: villi width, CD: crypt depth. bar= 500 μm.
2.3. Gills and Liver Histology
3.3. Gills and liver histology
Histological examination of the gills of common carp following the 60-day feeding trial showed no differences between the groups supplemented with yeast cell wall prebiotic, the probiotics S. cerevisiae or P. acidilactici, and the control (Figure 3). The gill lamellae and filaments in all groups exhibited normal architecture, with an unbroken epithelial lining and no hyperplasia, fusion or necrosis. The secondary lamellae were also clearly defined. In addition, Histological examination of liver (Figure 4) revealed no visible pathological changes in any of the treatment groups. Normal liver architecture, characterized by healthy hepatocytes, suggests that none of the tested feed additives had any negative effects on liver morphology.
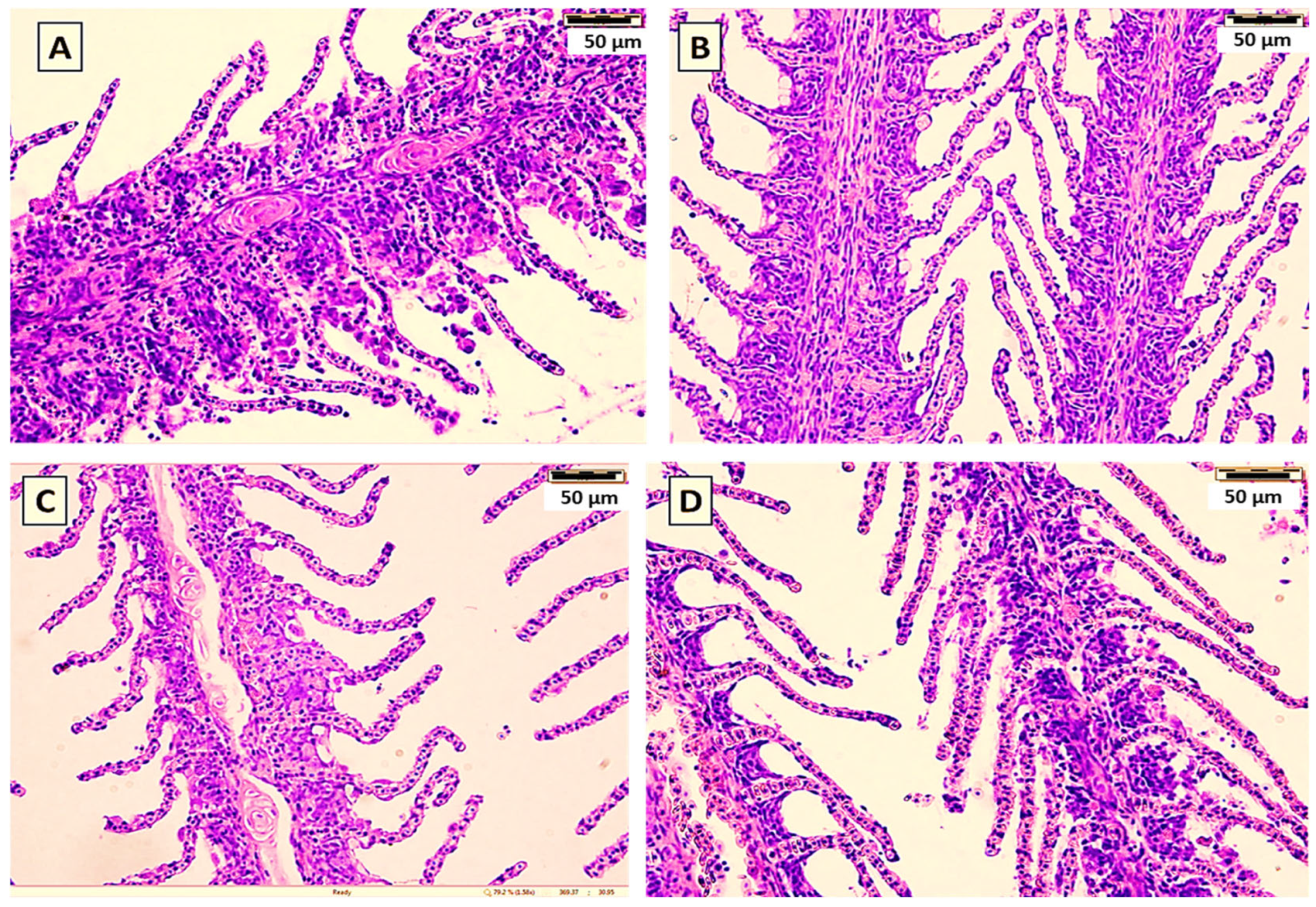
Figure 3. Histological sections (H&E) of the gills of common carp showed the effect of the yeast cell wall (A), S. cerevisiae (B), P. acidilactici (C) in comparison with the control (D) for 60-day feeding trial.
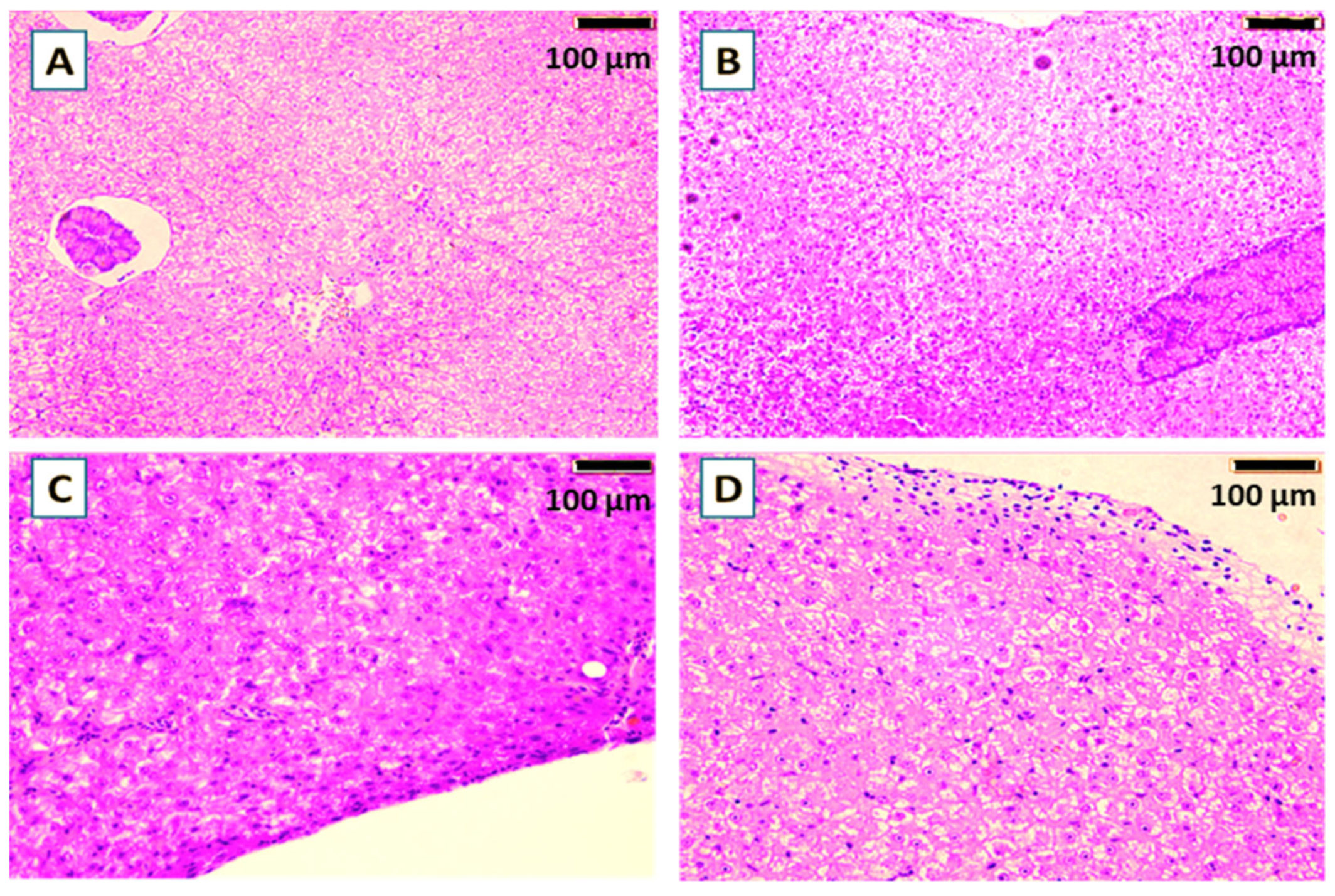
Figure 4. Histological sections (H&E) of the liver of common carp showed the effect of the yeast cell wall (A), S. cerevisiae (B), P. acidilactici (C) in comparison with the control (D) for 60-days trial.
3.4. Alteration in the intestinal microbiota2.4. Alteration in the Intestinal Microbiota
Across all dietary supplements and the control, the microbial community is predominantly composed of Proteobacteria and Fusobacteria (Figure 5), though their proportions vary notably between groups. PA exhibited a high abundance of Proteobacteria (~65%), followed by Fusobacteria (~26%), while the control and SC are dominated by Fusobacteria (>55%), with reduced proportions of Proteobacteria (< 43%). In addition, YP group showed a profile like PA, with Proteobacteria again comprising most of the bacterial community. Minor phyla including Firmicutes, Actinobacteria, Planctomycetes, and Tenericutes are present in low proportions across all groups, with Firmicutes being slightly more prominent in samples PA and YP. Notably, Planctomycetes appear only in PA group.
The genus-level bacterial composition across the four groups (PA, SC, YP, and the control), revealed a considerable variation in microbial community. In group PA, the bacterial community is primarily dominated by Cetobacterium, which accounts for approximately 26.5% of the total relative abundance (~0.265), closely followed by Aeromonas at around 26.2% (~0.262). Stenotrophomonas is the third most abundant genus, contributing to about 8.6% (~0.086), while Polynucleobacter and Nordella represent approximately 5.9% (~0.0593) and 4.6% (~0.046), respectively. Other genera, such as Mycoplasma, Romboutsia, and Staphylococcus, are present at lower abundances, each contributing less than 4% (~ 0.04) to the community structure.
In group SC, the bacterial community is predominantly composed of two genera: Cetobacterium and Aeromonas, which together account for nearly 85% of the total relative abundance. Cetobacterium is the most abundant genus, representing 50.5% (~0.505) of the community, followed by Aeromonas at 34.1% (~0.341). Stenotrophomonas contributes 4.9% (~0.049), while Mycoplasma is present at 3.1% (~0.031). All remaining genres each constitute less than 3% of the community. This composition indicates a low-diversity microbial profile heavily dominated by Cetobacterium, with moderate contributions from Aeromonas and minimal representation of other taxa, suggesting a highly specialized or selective microbial environment in group SC.
In group YP, the bacterial community is heavily dominated by Aeromonas, which comprises approximately 48.4% of the total abundance (~0.484), followed by Cetobacterium at around 31.6% (value = 0.316). Stenotrophomonas is the third most abundant genus, contributing about 7.6% (~0.076), while all remaining genera—such as Polynucleobacter, Mycoplasma, and Staphylococcus—each contribute less than 2% (< 0.02).
In group C, the bacterial community is overwhelmingly dominated by Cetobacterium, which constitutes approximately 56.2% of the total abundance (~0.562), indicating a strong single-genus dominance. This is followed by Aeromonas at 26.6% (~0.266), while Gemmobacter and Romboutsia contribute approximately 8.0% (~0.08) and 4.4% (~0.044), respectively. Other genres, including Plesiomonas and Shewanella, are present at lower levels, each accounting for less than 4% of the community (~0.04). This composition suggests a moderately diverse bacterial structure with a pronounced dominance by Cetobacterium, potentially reflecting specific environmental or host-associated factors shaping the microbiota in group C.
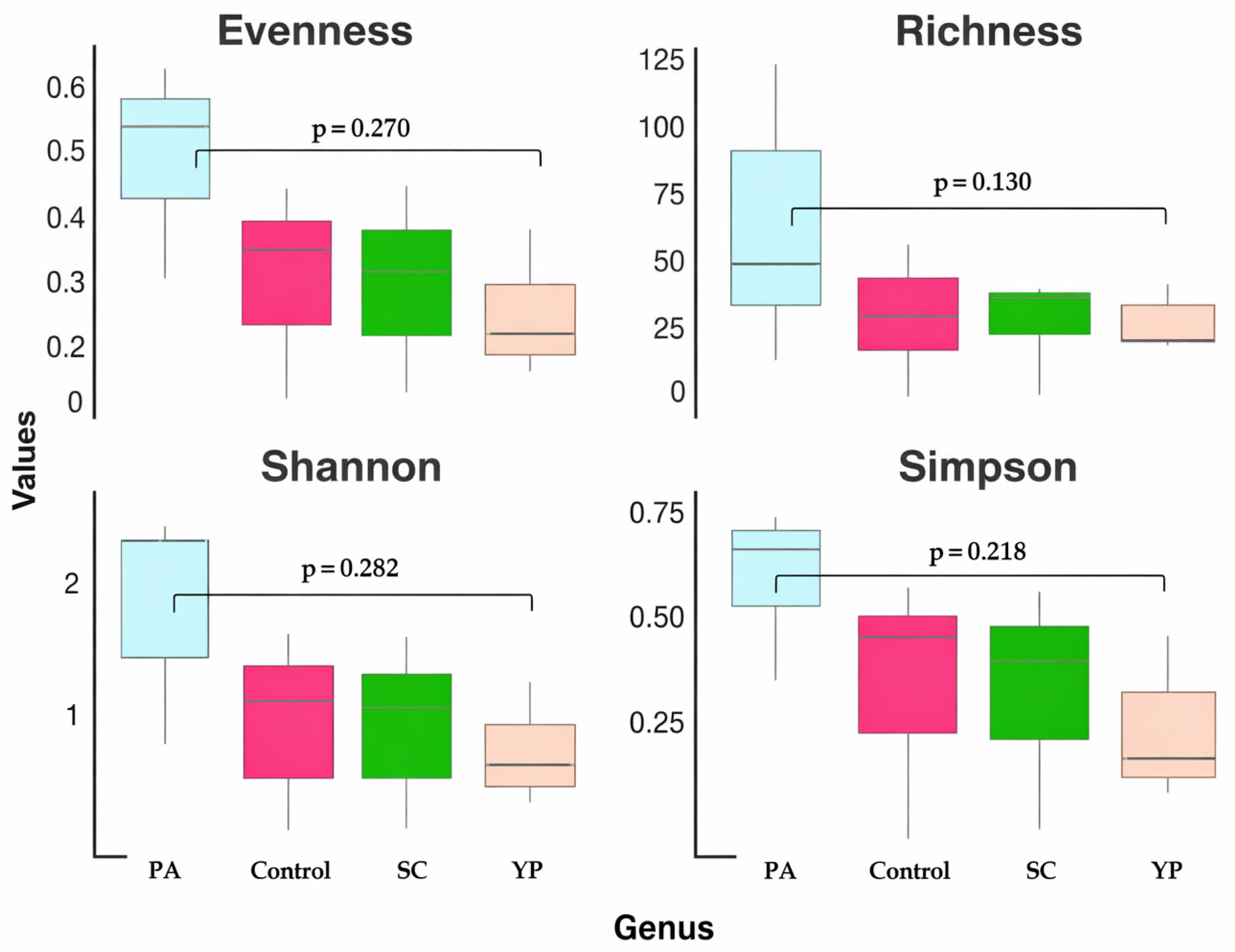
The alpha diversity metrics, including evenness, richness, Shannon, and Simpson indices, across the four groups (PA, SC, YP and the control group) at the genus level are shown in Figure 6. In PA, the highest metric across all feed supplements were recorded, including the highest evenness (~0.55–0.60), richness (~60–70), Shannon index (~2.2), and Simpson index (~0.75). In contrast, group YP shows the lowest diversity, with notably reduced evenness (~0.2), richness (~25), Shannon (~1.0), and Simpson (~0.35) indices, reflecting a community dominated by a few genera. Regarding the control and SC groups, intermediate diversity levels with similar evenness (~0.35–0.40), richness (~30–40), and diversity indices (Shannon ~1.2–1.4 and Simpson ~0.55–0.60) were recorded. These patterns could suggest that group PA supports the most complex and balanced microbiota, while group YP harbors the least diverse and most uneven bacterial community.
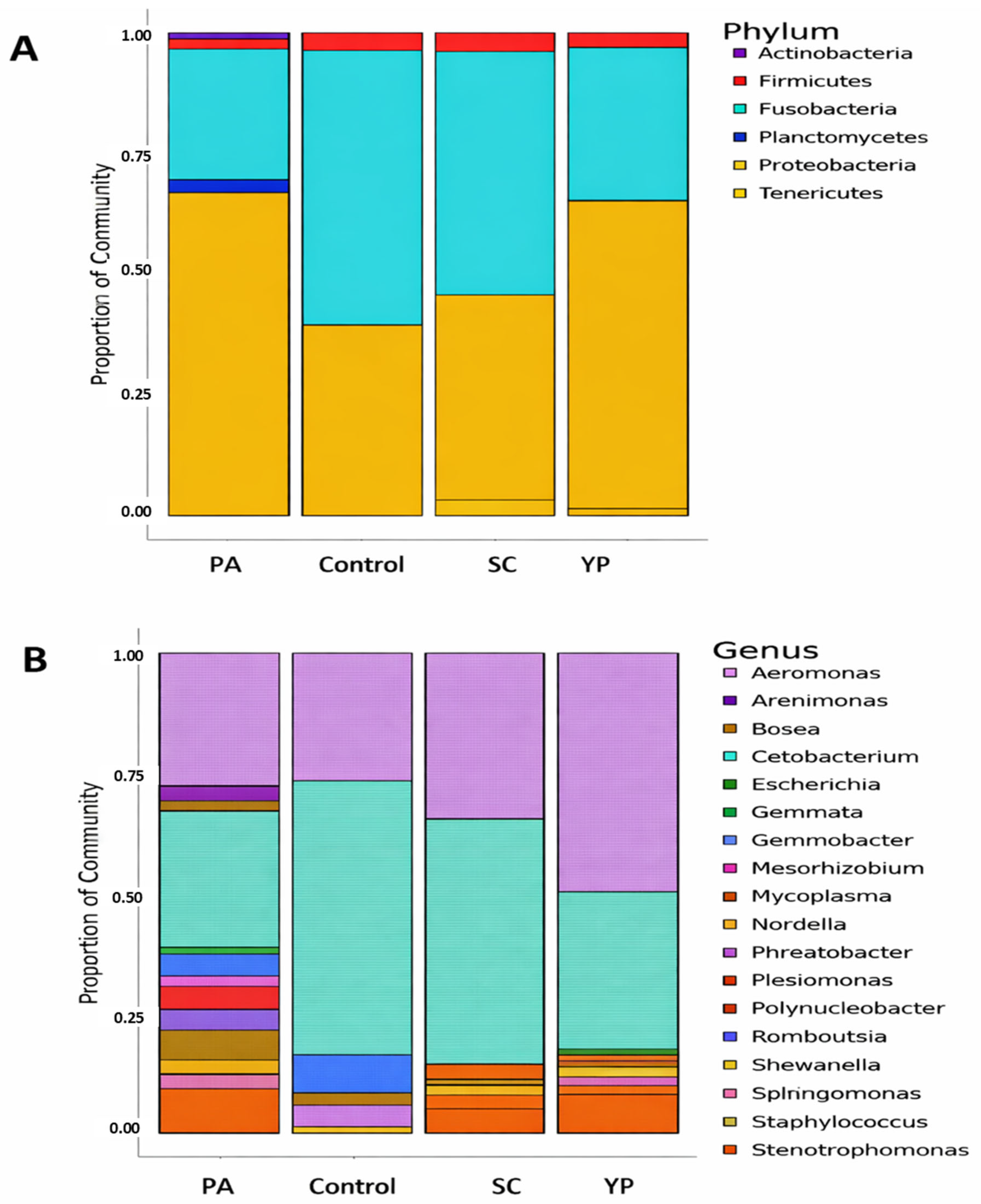
Figure 5. The relative abundance of bacterial phyla (A) and genera (B) in the intestine of C. carp was assessed after 8 weeks of receiving a diet supplemented with PA, SC, YP, or a control diet only. This assessment was conducted using 16S rRNA gene sequencing. "PA": Pediococcus acidilactici, "SC": Saccharomyces cerevisia, "YP": Yeast prebiotic
|
PA Control SC YP |
|
PA Control SC YP |
Figure 6. Boxplots showing four alpha diversity metrics (evenness, richness, Shannon index, and Simpson index) at the genus level for the bacterial communities in the intestines of C. carpio that received a prebiotic- and probiotic-supplemented diet for 8 weeks.
3. Discussion
- Discussion
The rapidly growing global population needs high-quality protein, and sustainable aquaculture practices are essential for producing it (Carrias et al., 2012)[1]. Probiotics and prebiotics have recently gained popularity in the aquaculture industry as an effective method of improving fish health (Dawood et al[2][3][4]., 2019; E. A. H Mohammed et al., 2025; Elshafia Ali Hamid Mohammed et al., 2025a). Generally, extensive studies have been conducted to investigate the effects of probiotics as promising technique in fish farming (Abdel-Tawwab, 2012; Abdel-Tawwab et al., 2020; Abou‐El‐Atta et al., 2019; Hendam et al., 2023; Huerta-Rábago et al., 2019; Elshafia Ali Hamid Mohammed et al., 2025b)[5][6][7][8][9][10]. In our research, using feed supplements containing probiotics and prebiotic for common carp produced promising results that could inform further research into enhancing growth, gut integrity and intestinal microbiota. Growth parameters are crucial in aquaculture as they indicate health and productivity of fish, and can be affected by factors such as the quality of the feed and environmental conditions (Oduleye, 1982)[11]. LAB-bacteria such as P. acidilactici were noticed to improve growth, survival (%), the feed digestion and the immune system of different aquatic animals (Huerta-Rábago et al[3][9]., 2019; E. A. H Mohammed et al., 2025). To the best of our knowledge, only a few studies have specifically examined the effects of S. cerevisiae and P. acidilactici, as well as the yeast cell wall-based prebiotic, on the growth indices, morphometric features of the intestine, and gut microbiota of cultured carp. However, there are no published studies that have examined the effect of yeast cell wall prebiotics on cultured common carp.
- Growth performance
3.1. Growth Performance
Studies have shown that prebiotics and probiotics can enhance the growth of various fish species. For instance, the yeast S. cerevisiae has been shown to improve growth indices of the European sea bass (Dicentrarchus labrax) (Oliva-Teles and Gonçalves, 2001)[12], striped catfish (P. hypophthalmus) (Boonanuntanasarn et al.[13], 2019), white shrimp (Litopenaeus vannamei) (Amin et al.[14], 2025), and Nile tilapia (O. niloticus) (Chen et al[15]., 2019). The growth-promoting effectiveness of P. acidilactici in improving growth performance has also been recorded in Nile tilapia (Eissa et[16] al., 2023a) and Sea bass (Eissa et al[17]., 2022).
In our study, we found that fish receiving a diet supplemented with S. cerevisiae or P. acidilactici, did not exhibit a notable effect in either body weight or survival. Our findings are not aligned with those of Yanbo et al. (Yanbo and Zirong[18], 2006), who observed that the growth parameters of common carp can be enhanced when received a diet supplemented with Bacillus sp. at 1 g/kg for 4 weeks. Despite this, the effectiveness of the tested feed supplements may be increased by raising the dose to over 1 g/kg. This was noted by Eissa et al. (Eissa et al[16]., 2023a).
- The intestinal histoarchitecture, liver, and gills
3.2. The Intestinal Histoarchitecture, Liver, and Gills
Probiotics and yeast prebiotics, particularly strains such as Pediococcus spp. and Saccharomyces cerevisiae, have been shown to significantly improve fish growth through various mechanisms. These mechanisms include improving nutrient digestibility, modulating gut microbiome, enhancing gut integrity, and supporting overall health.
In our study, the probiotic P. acidilactici showed significant improvements in the intestinal architecture especially the anterior intestine of the common carp, including villi length and width, muscular thickness, and crypt depth (p<0.05). These findings are consistent with Eissa et al. (Eissa[16] et al., 2023a) who noticed that the feed supplementation with P. acidilactici can positively enhance the development of internal organs, such as and length of intestinal villi and liver of Nile tilapia (O. niloticus). Additionally, our results are also somewhat consistent with those of Ashouri et al. (Ashouri et al., 2020)[19], Wang et al. (Wang et al.[20], 2020), and Zheng et al. (Zheng[21] et al., 2018) who detected improvement in villus surface, the width of villi, and crypt depth when lactic acid probiotics are used as a feed supplement. Considering the morphological parameters of intestinal villi, it is evident that they are directly correlated with nutrients absorption and digestion efficiency (Dawood et al., 2020)[22].
In this study, S. cerevisiae also considerably improved the width and length of the intestinal villi of common carp compared to the control group. The efficacy of S. cerevisiae has also been reported by Boonanuntanasarn et al. (Boonanuntanasarn[13] et al., 2019) who noticed that dietary supplementation with S. cerevisiae (1x10⁸ CFU) remarkably improved villus length (μm) in the anterior and mid intestines of catfish (Pangasianodon hypophthalmus). In addition, the efficacy of S. cerevisiae for improving intestinal architecture has been confirmed in Sea Bream (Sparus Aurata) (El-Bab[23] et al., 2022) and Atlantic Salmon (Salmo salar) (Momoh et al[24]., 2025). Increased villi length and width indicate an enlarged absorptive area of intestinal tissue, which improves growth, development, and feed utilization. However, the precise mechanisms through which probiotics and prebiotics enhance intestinal absorption are not fully understood. The cells at the tip of the villi are continuously sloughed off, and the intestinal epithelium's renewal rate is high enough to replace them (Aubin et al[25]., 2005). Additionally, S. cerevisiae's beneficial effect on intestinal histomorphology could be attributed to the monosaccharides that composing its cell wall. These monosaccharides have been shown to positively impact the intestinal villi and microvilli of various fish species (Aubin et al[25]., 2005). Additionally, the beneficial effect of S. cerevisiae on intestinal histomorphology could be attributed to the monosaccharides that compose its cell wall. These saccharides have been shown to positively impact the intestinal villi and microvilli of various fish species. Furthermore, several studies have shown that yeast-based feed supplements enhance the intestinal indices of Nile tilapia (Chen[15] et al., 2019) and Jian carp (C. carpio var. Jian) (Yuan et al[26]., 2019).
In gills histology, the lamellae in all groups exhibited normal architecture, with an unbroken epithelial lining and no hyperplasia, fusion or necrosis. These observations suggest that including probiotic and yeast-based supplements in the diet did not have any histopathological effects on gill tissue. Maintenance of normal gill structure suggests that the tested additives were non-toxic and well tolerated by common carp under experimental conditions. Similar findings have been reported that probiotics feed additives showed no harmful impact on the gill morphology of fish (Eissa et al[27]., 2023b). These findings further support the safety and suitability of these in aquaculture.
In liver histology, liver sections of common carp revealed no visible pathological changes in any of the treatment groups. Normal liver architecture, characterized by healthy hepatocytes, suggests that none of the tested feed additives had any negative effects on liver morphology. There have been similar reports that probiotic feed additives have no harmful impact on the liver morphology of fish (Eissa et al., 2023b)[27]. These results provide further support for the safety and suitability of these additives in aquaculture.
- The intestinal microbiota
3.3. The Intestinal Microbiota
Unlike traditional culture-dependent methods, which are limited to detecting only cultivable microorganisms and often overlook most gut-residing bacteria, Oxford Nanopore sequencing technology provides a comprehensive, high-resolution profile of both abundant and rare microbial taxa. The intestine of any aquatic animal provides an ideal environment for beneficial microbes to colonize and grow (Han et al[28]., 2010). The composition of the intestinal microbiome can influence host physiology, growth, and health (Elshafia Ali Hamid Mohammed et al[10]., 2025b). A diversity of intestinal microbes is generally considered beneficial for the health of the host. Therefore, an investigation was conducted to examine the impacts of probiotics, specifically S. cerevisiae, and P. acidilactici as well as yeast prebiotics, on the gut microbiome of C. carpio.
In our results, Actinobacteria has detected only with law abundance (~0.011) in the intestine of fish received diet supplemented with P. acidilactici. Actinobacteria, especially Streptomyces spp. has garnered interest as potential probiotics in aquaculture due to their numerous advantages, including the capacity to synthesize bioactive compounds and their involvement in nutrient cycling. Their contributions to fish health and growth performance are significant, making them notable players in sustainable aquaculture practices [29][30](Ghosh et al., 2023; Tan et al., 2016). For example, Arghideh et al. (Arghideh et al., 2022) reported that Streptomyces chartreusis (KU324443) enhanced the growth parameters, antioxidant activity, and serum immune parameters in common carp (C. carpio) fingerlings. In addition, Das et al. (Das[31] et al., 2006) found positive impacts on growth performance of black tiger shrimp fed diets supplemented with Streptomyces spp. probiotic. Furthermore, the beneficial effect of has been noticed on shrimp by García-Bernal et al. (García[32] Bernal et al., 2017) and AftabUddin et al. (AftabUddin et al[33]., 2018).
In our study, all groups recorded high abundance of Cetobacterium genus (26-50%). The Cetobacterium, which belongs to the Fusobacteriota phylum is known to be associated with polypeptide and protein digestion, benefits the host as a good source of vitamin B12, butyrate, and antibacterial compounds (Finegold et al[34]., 2003). Cetobacterium genus is found in a range of freshwater fish species (Li et al., 2018). Accordingly, this genus considered an indicator of healthy fish (Kim et al[35][36]., 2021; Li et al., 2017). For example, Ma et al. (Ma[37] et al., 2019) noticed that Cetobacterium decreased from relative abundance of 23% in healthy intestines to 0.5% in diseased intestines of Yunlong Grouper (Epinephelus moara × Epinephelus lanceolatus).
Aeromonas were the second largest genius in all groups but are generally recognized as pathogens and are commonly found in aquatic environments. Aeromonas spp. has also been recognized as a causative agent of human and animal diseases, including septicemia, which is characterized by the presence of A. septicemia (Praveen et al., 2016)[38]. Aeromonas spp. have been identified as significant bacterial contaminants that can compromise the integrity of aquatic ecosystems and organisms (Qian et al[39]., 2018). However, they are also constituents of the fish microbiome, which plays a substantial role for the fermentation of organic substances, the expression of antibacterial activity, and the degradation of cellulose (Van Kessel et al., 2011)[40].
- Conclusion
4. Conclusion
In the context of rising demand for seafood and animal protein, aquaculture has emerged as a prominent component of global food production systems. However, the combined effect of pathogens, poor fish management practices and adverse environmental conditions is detrimental to productivity, resulting in substantial economic losses for farmers. Antibiotics are often used in aquaculture to combat diseases and improve fish performance. However, continuous antibiotic use in aquaculture exerts selective pressure on the microbial community, leading to the emergence of antibiotic-resistant strains with the capacity to disseminate extensively. Considering these, probiotics and prebiotics are being explored as sustainable alternatives for aquaculture production.
Neither the probiotics S. cerevisiae and P. acidilactici and the yeast cell wall extract at a concentration of 1 g/kg for 8-week trial did not affect growth indices of common carp. However, all the tested feed supplements resulted in a significant improvement in the anterior intestine including villi length and width, muscular thickness, and the crypt depth. In addition, the yeast probiotic (Saccharomyces cerevisiae) exhibited a significant improvement of villi length in all parts of intestinal tissue of C. carpio (anterior, mid, and posterior intestine). However, other supplements did not exhibit significant effect on midintestines and posterior intestine of C. carpio.
The genus-level bacterial composition across the four groups revealed a considerable variation in microbial community. In P. acidilactici group, the bacterial community is dominated by Cetobacterium, Aeromonas, Stenotrophomonas, while Polynucleobacter, Nordella, Mycoplasma, Romboutsia, and Staphylococcus, are present at lower abundances, each contributing less than 9% (~ 0.09) to the community structure. In S. cerevisiae supplemented diet, the bacterial community is also predominantly composed of Cetobacterium and Aeromonas, which together account for nearly 85% of the total relative abundance. In the yeast cell wall prebiotic, the bacterial community is heavily dominated by Aeromonas, which comprises approximately 48.4% of the total abundance (~0.484), followed by Cetobacterium and Stenotrophomonas. However, Actinobacteria has detected only with law abundance (~0.011) in the intestine of fish received diet supplemented with P. acidilactici.
Author Contributions: Conceptualization, Elshafia Ali Hamid Mohammed, Milán Fehér, Levente Czeglédi and Károly Pál; Data curation, Elshafia Ali Hamid Mohammed, Abdelhakam Esmaeil Mohamed Ahmed, Christopher Teye-Gaga; Investigation, Elshafia Ali Hamid Mohammed; Methodology, Milán Fehér and Károly Pál; Project administration, Károly Pál; Resources, Milán Fehér, Péter Bársony, and Levente Czeglédi; Supervision, Károly Pál; Writing – original draft, Elshafia Ali Hamid Mohammed; Writing – review & editing, Milán Fehér, Péter Bársony, Abdelhakam Esmaeil Mohamed Ahmed, Christopher Teye-Gaga, Levente Czeglédi, Váradi Alex, Freytag Csongor, Elshafia Ali Hamid Mohammed and Károly Pál. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Data Availability Statement: The data that support the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest: All authors declare that there are no conflicts of interest regarding the publication.
References
Abdel-Tawwab, M., 2012. Interactive effects of dietary protein and live bakery yeast, Saccharomyces cerevisiae on growth performance of Nile tilapia, Oreochromis niloticus (L.) fry and their challenge against Aeromonas hydrophila infection. Aquacult Int 20, 317–331. https://doi.org/10.1007/s10499-011-9462-8
Abdel-Tawwab, M., Adeshina, I., Issa, Z.A., 2020. Antioxidants and immune responses, resistance to Aspergilus flavus infection, and growth performance of Nile tilapia, Oreochromis niloticus, fed diets supplemented with yeast, Saccharomyces serevisiae. Animal Feed Science and Technology 263, 114484. https://doi.org/10.1016/j.anifeedsci.2020.114484
Abou‐El‐Atta, M.E., Abdel‐Tawwab, M., Abdel‐Razek, N., Abdelhakim, T.M.N., 2019. Effects of dietary probiotic Lactobacillus plantarum and whey protein concentrate on the productive parameters, immunity response and susceptibility of Nile tilapia, Oreochromis niloticus (L.), to Aeromonas sobria infection. Aquacult Nutr 25, 1367–1377. https://doi.org/10.1111/anu.12957
AftabUddin, S., Roman, W.U., Hasan, C.K., Ahmed, M., Rahman, H., Siddique, M.A.M., 2018. First incidence of loose-shell syndrome disease in the giant tiger shrimp Penaeus monodon from the brackish water ponds in Bangladesh. Journal of Applied Animal Research 46, 210–217. https://doi.org/10.1080/09712119.2017.1285771
Amin, I., Can, E., Khan, T., Fatima, J., Khalid, M., 2025. Synergistic effects of combined probiotic supplementation on growth performance, survival, and health status in Labeo rohita. Aquacult Int 33, 340. https://doi.org/10.1007/s10499-025-02025-5
Arghideh, M., Hoseinifar, S.H., Ghorbani Nasrabadi, R., Mazandarani, M., El-Haroun, E., Van Doan, H., 2022. Evaluation of Soil-Derived Streptomyces chartreusis KU324443 Effects as Probiotic on Growth Performance, Antioxidant Enzyme Activity, Mucosal and Serum Immune Parameters, and Related Gene Expression in Common Carp (Cyprinus carpio) Fingerlings. Aquaculture Nutrition 2022, 1–9. https://doi.org/10.1155/2022/2278130
Ashouri, G., Mahboobi Soofiani, N., Hoseinifar, S.H., Jalali, S.A.H., Morshedi, V., Valinassab, T., Bagheri, D., Van Doan, H., Torfi Mozanzadeh, M., Carnevali, O., 2020. Influence of dietary sodium alginate and Pediococcus acidilactici on liver antioxidant status, intestinal lysozyme gene expression, histomorphology, microbiota, and digestive enzymes activity, in Asian sea bass (Lates calcarifer) juveniles. Aquaculture 518, 734638. https://doi.org/10.1016/j.aquaculture.2019.734638
Aubin, J., Gatesoupe, F.-J., Labbe, L., Lebrun, L., 2005. Trial of probiotics to prevent the vertebral column compression syndrome in rainbow trout (Oncorhynchus mykiss Walbaum). Aquac Research 36, 758–767. https://doi.org/10.1111/j.1365-2109.2005.01280.x
Boonanuntanasarn, S., Ditthab, K., Jangprai, A., Nakharuthai, C., 2019. Effects of Microencapsulated Saccharomyces cerevisiae on Growth, Hematological Indices, Blood Chemical, and Immune Parameters and Intestinal Morphology in Striped Catfish, Pangasianodon hypophthalmus. Probiotics & Antimicro. Prot. 11, 427–437. https://doi.org/10.1007/s12602-018-9404-0
Carrias, A., Ran, C., Terhune, J.S., Liles, M.R., 2012. Bacteria and bacteriophages as biological agents for disease control in aquaculture, in: Infectious Disease in Aquaculture. Elsevier, pp. 353–393. https://doi.org/10.1533/9780857095732.3.353
Chen, X.-Q., Zhao, W., Xie, S.-W., Xie, J.-J., Zhang, Z.-H., Tian, L.-X., Liu, Y.-J., Niu, J., 2019. Effects of dietary hydrolyzed yeast (Rhodotorula mucilaginosa) on growth performance, immune response, antioxidant capacity and histomorphology of juvenile Nile tilapia (Oreochromis niloticus). Fish & Shellfish Immunology 90, 30–39. https://doi.org/10.1016/j.fsi.2019.03.068
Das, S., Lyla, P.S., Ajmal Khan, S., 2006. Application of streptomyces as a probiotic in the laboratory culture of Penaeus monodon (Fabricius). Israeli Journal of Aquaculture - Bamidgeh 58. https://doi.org/10.46989/001c.20439
Dawood, M.A.O., Koshio, S., Abdel‐Daim, M.M., Van Doan, H., 2019. Probiotic application for sustainable aquaculture. Reviews in Aquaculture 11, 907–924. https://doi.org/10.1111/raq.12272
Dawood, M.A.O., Magouz, F.I., Essa, M., Mansour, M., 2020. Impact of Yeast Fermented Poultry by-Product Meal on Growth, Digestive Enzyme Activities, Intestinal Morphometry and Immune Response Traits of Common Carp ( Cyprinus carpio ). Annals of Animal Science 20, 939–959. https://doi.org/10.2478/aoas-2020-0021
Eissa, E.-S.H., Baghdady, E.S., Gaafar, A.Y., El-Badawi, A.A., Bazina, W.K., Abd Al-Kareem, O.M., Abd El-Hamed, N.N.B., 2022. Assessing the Influence of Dietary Pediococcus acidilactici Probiotic Supplementation in the Feed of European Sea Bass (Dicentrarchus labrax L.) (Linnaeus, 1758) on Farm Water Quality, Growth, Feed Utilization, Survival Rate, Body Composition, Blood Biochemical Parameters, and Intestinal Histology. Aquaculture Nutrition 2022, 1–11. https://doi.org/10.1155/2022/5841220
Eissa, M.E.H., Alaryani, F.S., Elbahnaswy, S., Khattab, M.S., Elfeky, A., AbouelFadl, K.Y., Eissa, E.-S.H., Ahmed, R.A., Van Doan, H., El-Haroun, E., 2023a. Dietary inclusion of Pediococcus acidilactici probiotic promoted the growth indices, hemato-biochemical indices, enzymatic profile, intestinal and liver histomorphology, and resistance of Nile Tilapia against Aspergillus flavus. Animal Feed Science and Technology 306, 115814. https://doi.org/10.1016/j.anifeedsci.2023.115814
Eissa, M.E.H., Alaryani, F.S., Elbahnaswy, S., Khattab, M.S., Elfeky, A., AbouelFadl, K.Y., Eissa, E.-S.H., Ahmed, R.A., Van Doan, H., El-Haroun, E., 2023b. Dietary inclusion of Pediococcus acidilactici probiotic promoted the growth indices, hemato-biochemical indices, enzymatic profile, intestinal and liver histomorphology, and resistance of Nile Tilapia against Aspergillus flavus. Animal Feed Science and Technology 306, 115814. https://doi.org/10.1016/j.anifeedsci.2023.115814
El-Bab, A.F.F., Saghir, S.A.M., El-Naser, I.A.A., El-Kheir, S.M.M.A., Abdel-Kader, M.F., Alruhaimi, R.S., Alqhtani, H.A., Mahmoud, A.M., Naiel, M.A.E., El-Raghi, A.A., 2022. The Effect of Dietary Saccharomyces cerevisiae on Growth Performance, Oxidative Status, and Immune Response of Sea Bream (Sparus aurata). Life 12, 1013. https://doi.org/10.3390/life12071013
Fernandes, I.M., Bastos, Y.F., Barreto, D.S., Lourenço, L.S., Penha, J.M., 2016. The efficacy of clove oil as an anaesthetic and in euthanasia procedure for small-sized tropical fishes. Braz. J. Biol. 77, 444–450. https://doi.org/10.1590/1519-6984.15015
Finegold, S.M., Vaisanen, M.-L., Molitoris, D.R., Tomzynski, T.J., Song, Y., Liu, C., Collins, M.D., Lawson, P.A., 2003. Cetobacterium somerae sp. nov. from Human Feces and Emended Description of the Genus Cetobacterium. Systematic and Applied Microbiology 26, 177–181. https://doi.org/10.1078/072320203322346010
Fischer, A.H., Jacobson, K.A., Rose, J., Zeller, R., 2008. Hematoxylin and Eosin Staining of Tissue and Cell Sections. Cold Spring Harb Protoc 2008, pdb.prot4986. https://doi.org/10.1101/pdb.prot4986
García Bernal, M., Trabal Fernández, N., Saucedo Lastra, P.E., Medina Marrero, R., Mazón-Suástegui, J.M., 2017. Streptomyces effect on the bacterial microbiota associated to Crassostrea sikamea oyster. J Appl Microbiol 122, 601–614. https://doi.org/10.1111/jam.13382
Ghosh, K., Harikrishnan, R., Mukhopadhyay, A., Ringø, E., 2023. Fungi and Actinobacteria: Alternative Probiotics for Sustainable Aquaculture. Fishes 8, 575. https://doi.org/10.3390/fishes8120575
Han, S., Liu, Y., Zhou, Z., He, S., Cao, Y., Shi, P., Yao, B., Ringø, E., 2010. Analysis of bacterial diversity in the intestine of grass carp (Ctenopharyngodon idellus) based on 16S rDNA gene sequences: Intestinal bacterial diversity in grass carp. Aquaculture Research 42, 47–56. https://doi.org/10.1111/j.1365-2109.2010.02543.x
Hendam, B.M., Munir, M.B., Eissa, M.E.H., El-Haroun, E., Doan, H.V., Chung, T.H., Eissa, E.-S.H., 2023. Effects of water additive probiotic, Pediococcus acidilactici on growth performance, feed utilization, hematology, gene expression and disease resistance against Aspergillus flavus of Nile tilapia (Oreochromis niloticus). Animal Feed Science and Technology 303, 115696. https://doi.org/10.1016/j.anifeedsci.2023.115696
Huerta-Rábago, J.A., Martínez-Porchas, M., Miranda-Baeza, A., Nieves-Soto, M., Rivas-Vega, M.E., Martínez-Córdova, L.R., 2019. Addition of commercial probiotic in a biofloc shrimp farm of Litopenaeus vannamei during the nursery phase: Effect on bacterial diversity using massive sequencing 16S rRNA. Aquaculture 502, 391–399. https://doi.org/10.1016/j.aquaculture.2018.12.055
Kim, P.S., Shin, N.-R., Lee, J.-B., Kim, M.-S., Whon, T.W., Hyun, D.-W., Yun, J.-H., Jung, M.-J., Kim, J.Y., Bae, J.-W., 2021. Host habitat is the major determinant of the gut microbiome of fish. Microbiome 9, 166. https://doi.org/10.1186/s40168-021-01113-x
Li, J., Liu, G., Li, C., Deng, Y., Tadda, M.A., Lan, L., Zhu, S., Liu, D., 2018. Effects of different solid carbon sources on water quality, biofloc quality and gut microbiota of Nile tilapia (Oreochromis niloticus) larvae. Aquaculture 495, 919–931. https://doi.org/10.1016/j.aquaculture.2018.06.078
Li, T., Li, H., Gatesoupe, F.-J., She, R., Lin, Q., Yan, X., Li, J., Li, X., 2017. Bacterial Signatures of “Red-Operculum” Disease in the Gut of Crucian Carp (Carassius auratus). Microb Ecol 74, 510–521. https://doi.org/10.1007/s00248-017-0967-1
Ma, C., Chen, C., Jia, L., He, X., Zhang, B., 2019. Comparison of the intestinal microbiota composition and function in healthy and diseased Yunlong Grouper. AMB Expr 9, 187. https://doi.org/10.1186/s13568-019-0913-3
Mohammed, Elshafia Ali Hamid, Ahmed, A.E.M., Kovács, B., Pál, K., 2025a. The Significance of Probiotics in Aquaculture: A Review of Research Trend and Latest Scientific Findings. Antibiotics 14, 242. https://doi.org/10.3390/antibiotics14030242
Mohammed, E. A. H, Ahmed, A.E.M., Teye-Gaga, C., Pál, K., 2025. The potential use of Pediococcus spp. probiotic in aquaculture: A review. Acta Agraria Debreceniensis 99–106. https://doi.org/doi.org/10.34101/actaagrar/1/15372
Mohammed, Elshafia Ali Hamid, Fehér, M., Bársony, P., Pál, K., 2025b. Alteration in Gut Microbiome of Common Carp (Cyprinus carpio L., 1758) Mediated by Probiotics and Yeast Prebiotic, in: IECA 2025. Presented at the IECA 2025, MDPI, p. 1. https://doi.org/10.3390/blsf2025045001
Momoh, T.A., Odu-Onikosi, S.G., Amulejoye, F.D., Wilson, J., Eynon, B., Kühlwein, H., Kuri, V., Merrifield, D.L., 2025. Brewers’ Yeast ( Saccharomyces cerevisiae ) Purified Functional Feed Additives Mitigate Soybean Meal‐Induced Enteritis in Atlantic Salmon ( Salmo salar ) Parr. Aquaculture Nutrition 2025, 8555658. https://doi.org/10.1155/anu/8555658
Oduleye, S.O., 1982. Growth and growth regulation in the cichlids. Aquaculture 27, 301–306. https://doi.org/10.1016/0044-8486(82)90065-5
Oliva-Teles, A., Gonçalves, P., 2001. Partial replacement of fishmeal by brewers yeast (Saccaromyces cerevisae) in diets for sea bass (Dicentrarchus labrax) juveniles. Aquaculture 202, 269–278. https://doi.org/10.1016/S0044-8486(01)00777-3
Praveen, P.K., Debnath, C., Shekhar, S., Dalai, N., Ganguly, S., 2016. Incidence of Aeromonas spp. infection in fish and chicken meat and its related public health hazards: A review. Vet World 9, 6–11. https://doi.org/10.14202/vetworld.2016.6-11
Qian, Y., Ye, J., Yang, S., Lin, Z., Cao, W., Xie, J., 2018. Evaluation of the spoilage potential of Shewanella putrefaciens , Aeromonas hydrophila , and Aeromonas sobria isolated from spoiled Pacific white shrimp ( Litopenaeus vannamei ) during cold storage. Journal of Food Safety 38, e12550. https://doi.org/10.1111/jfs.12550
Tan, L.T.-H., Chan, K.-G., Lee, L.-H., Goh, B.-H., 2016. Streptomyces Bacteria as Potential Probiotics in Aquaculture. Front. Microbiol. 7. https://doi.org/10.3389/fmicb.2016.00079
Van Kessel, M.A., Dutilh, B.E., Neveling, K., Kwint, M.P., Veltman, J.A., Flik, G., Jetten, M.S., Klaren, P.H., Op Den Camp, H.J., 2011. Pyrosequencing of 16S rRNA gene amplicons to study the microbiota in the gastrointestinal tract of carp (Cyprinus carpio L.). AMB Expr 1, 41. https://doi.org/10.1186/2191-0855-1-41
Wang, R., Guo, Z., Tang, Y., Kuang, J., Duan, Y., Lin, H., Jiang, S., Shu, H., Huang, J., 2020. Effects on development and microbial community of shrimp Litopenaeus vannamei larvae with probiotics treatment. AMB Expr 10, 109. https://doi.org/10.1186/s13568-020-01041-3
Yanbo, W., Zirong, X., 2006. Effect of probiotics for common carp (Cyprinus carpio) based on growth performance and digestive enzyme activities. Animal Feed Science and Technology 127, 283–292. https://doi.org/10.1016/j.anifeedsci.2005.09.003
Yuan, X.-Y., Jiang, G.-Z., Wang, C.-C., Abasubong, K.P., Zou, Q., Zhou, Y.-Y., Liu, W.-B., 2019. Effects of partial replacement of fish meal by yeast hydrolysate on antioxidant capability, intestinal morphology, and inflammation-related gene expression of juvenile Jian carp (Cyprinus carpio var. Jian). Fish Physiol Biochem 45, 187–197. https://doi.org/10.1007/s10695-018-0552-7
Zheng, X., Duan, Y., Dong, H., Zhang, J., 2018. Effects of Dietary Lactobacillus plantarum on Growth Performance, Digestive Enzymes and Gut Morphology of Litopenaeus vannamei. Probiotics & Antimicro. Prot. 10, 504–510. https://doi.org/10.1007/s12602-017-9300-z
References
- Carrias, A., Ran, C., Terhune, J.S., Liles, M.R., 2012. Bacteria and bacteriophages as biological agents for disease control in aquaculture, in: Infectious Disease in Aquaculture. Elsevier, pp. 353–393. https://doi.org/10.1533/9780857095732.3.353.
- Dawood, M.A.O., Koshio, S., Abdel‐Daim, M.M., Van Doan, H., 2019. Probiotic application for sustainable aquaculture. Reviews in Aquaculture 11, 907–924. https://doi.org/10.1111/raq.12272.
- Mohammed, E. A. H, Ahmed, A.E.M., Teye-Gaga, C., Pál, K., 2025. The potential use of Pediococcus spp. probiotic in aquaculture: A review. Acta Agraria Debreceniensis 99–106. https://doi.org/doi.org/10.34101/actaagrar/1/15372.
- Mohammed, Elshafia Ali Hamid, Ahmed, A.E.M., Kovács, B., Pál, K., 2025a. The Significance of Probiotics in Aquaculture: A Review of Research Trend and Latest Scientific Findings. Antibiotics 14, 242. https://doi.org/10.3390/antibiotics14030242.
- Abdel-Tawwab, M., 2012. Interactive effects of dietary protein and live bakery yeast, Saccharomyces cerevisiae on growth performance of Nile tilapia, Oreochromis niloticus (L.) fry and their challenge against Aeromonas hydrophila infection. Aquacult Int 20, 317–331. https://doi.org/10.1007/s10499-011-9462-8.
- Abdel-Tawwab, M., Adeshina, I., Issa, Z.A., 2020. Antioxidants and immune responses, resistance to Aspergilus flavus infection, and growth performance of Nile tilapia, Oreochromis niloticus, fed diets supplemented with yeast, Saccharomyces serevisiae. Animal Feed Science and Technology 263, 114484. https://doi.org/10.1016/j.anifeedsci.2020.114484.
- Abou‐El‐Atta, M.E., Abdel‐Tawwab, M., Abdel‐Razek, N., Abdelhakim, T.M.N., 2019. Effects of dietary probiotic Lactobacillus plantarum and whey protein concentrate on the productive parameters, immunity response and susceptibility of Nile tilapia, Oreochromis niloticus (L.), to Aeromonas sobria infection. Aquacult Nutr 25, 1367–1377. https://doi.org/10.1111/anu.12957.
- Hendam, B.M., Munir, M.B., Eissa, M.E.H., El-Haroun, E., Doan, H.V., Chung, T.H., Eissa, E.-S.H., 2023. Effects of water additive probiotic, Pediococcus acidilactici on growth performance, feed utilization, hematology, gene expression and disease resistance against Aspergillus flavus of Nile tilapia (Oreochromis niloticus). Animal Feed Science and Technology 303, 115696. https://doi.org/10.1016/j.anifeedsci.2023.115696.
- Huerta-Rábago, J.A., Martínez-Porchas, M., Miranda-Baeza, A., Nieves-Soto, M., Rivas-Vega, M.E., Martínez-Córdova, L.R., 2019. Addition of commercial probiotic in a biofloc shrimp farm of Litopenaeus vannamei during the nursery phase: Effect on bacterial diversity using massive sequencing 16S rRNA. Aquaculture 502, 391–399. https://doi.org/10.1016/j.aquaculture.2018.12.055.
- Mohammed, Elshafia Ali Hamid, Fehér, M., Bársony, P., Pál, K., 2025b. Alteration in Gut Microbiome of Common Carp (Cyprinus carpio L., 1758) Mediated by Probiotics and Yeast Prebiotic, in: IECA 2025. Presented at the IECA 2025, MDPI, p. 1. https://doi.org/10.3390/blsf2025045001.
- Oduleye, S.O., 1982. Growth and growth regulation in the cichlids. Aquaculture 27, 301–306. https://doi.org/10.1016/0044-8486(82)90065-5.
- Oliva-Teles, A., Gonçalves, P., 2001. Partial replacement of fishmeal by brewers yeast (Saccaromyces cerevisae) in diets for sea bass (Dicentrarchus labrax) juveniles. Aquaculture 202, 269–278. https://doi.org/10.1016/S0044-8486(01)00777-3.
- Boonanuntanasarn, S., Ditthab, K., Jangprai, A., Nakharuthai, C., 2019. Effects of Microencapsulated Saccharomyces cerevisiae on Growth, Hematological Indices, Blood Chemical, and Immune Parameters and Intestinal Morphology in Striped Catfish, Pangasianodon hypophthalmus. Probiotics & Antimicro. Prot. 11, 427–437. https://doi.org/10.1007/s12602-018-9404-0.
- Amin, I., Can, E., Khan, T., Fatima, J., Khalid, M., 2025. Synergistic effects of combined probiotic supplementation on growth performance, survival, and health status in Labeo rohita. Aquacult Int 33, 340. https://doi.org/10.1007/s10499-025-02025-5.
- Chen, X.-Q., Zhao, W., Xie, S.-W., Xie, J.-J., Zhang, Z.-H., Tian, L.-X., Liu, Y.-J., Niu, J., 2019. Effects of dietary hydrolyzed yeast (Rhodotorula mucilaginosa) on growth performance, immune response, antioxidant capacity and histomorphology of juvenile Nile tilapia (Oreochromis niloticus). Fish & Shellfish Immunology 90, 30–39. https://doi.org/10.1016/j.fsi.2019.03.068.
- Eissa, M.E.H., Alaryani, F.S., Elbahnaswy, S., Khattab, M.S., Elfeky, A., AbouelFadl, K.Y., Eissa, E.-S.H., Ahmed, R.A., Van Doan, H., El-Haroun, E., 2023a. Dietary inclusion of Pediococcus acidilactici probiotic promoted the growth indices, hemato-biochemical indices, enzymatic profile, intestinal and liver histomorphology, and resistance of Nile Tilapia against Aspergillus flavus. Animal Feed Science and Technology 306, 115814. https://doi.org/10.1016/j.anifeedsci.2023.115814.
- Eissa, E.-S.H., Baghdady, E.S., Gaafar, A.Y., El-Badawi, A.A., Bazina, W.K., Abd Al-Kareem, O.M., Abd El-Hamed, N.N.B., 2022. Assessing the Influence of Dietary Pediococcus acidilactici Probiotic Supplementation in the Feed of European Sea Bass (Dicentrarchus labrax L.) (Linnaeus, 1758) on Farm Water Quality, Growth, Feed Utilization, Survival Rate, Body Composition, Blood Biochemical Parameters, and Intestinal Histology. Aquaculture Nutrition 2022, 1–11. https://doi.org/10.1155/2022/5841220.
- Yanbo, W., Zirong, X., 2006. Effect of probiotics for common carp (Cyprinus carpio) based on growth performance and digestive enzyme activities. Animal Feed Science and Technology 127, 283–292. https://doi.org/10.1016/j.anifeedsci.2005.09.003.
- Ashouri, G., Mahboobi Soofiani, N., Hoseinifar, S.H., Jalali, S.A.H., Morshedi, V., Valinassab, T., Bagheri, D., Van Doan, H., Torfi Mozanzadeh, M., Carnevali, O., 2020. Influence of dietary sodium alginate and Pediococcus acidilactici on liver antioxidant status, intestinal lysozyme gene expression, histomorphology, microbiota, and digestive enzymes activity, in Asian sea bass (Lates calcarifer) juveniles. Aquaculture 518, 734638. https://doi.org/10.1016/j.aquaculture.2019.734638.
- Wang, R., Guo, Z., Tang, Y., Kuang, J., Duan, Y., Lin, H., Jiang, S., Shu, H., Huang, J., 2020. Effects on development and microbial community of shrimp Litopenaeus vannamei larvae with probiotics treatment. AMB Expr 10, 109. https://doi.org/10.1186/s13568-020-01041-3.
- Zheng, X., Duan, Y., Dong, H., Zhang, J., 2018. Effects of Dietary Lactobacillus plantarum on Growth Performance, Digestive Enzymes and Gut Morphology of Litopenaeus vannamei. Probiotics & Antimicro. Prot. 10, 504–510. https://doi.org/10.1007/s12602-017-9300-z.
- Dawood, M.A.O., Magouz, F.I., Essa, M., Mansour, M., 2020. Impact of Yeast Fermented Poultry by-Product Meal on Growth, Digestive Enzyme Activities, Intestinal Morphometry and Immune Response Traits of Common Carp ( Cyprinus carpio ). Annals of Animal Science 20, 939–959. https://doi.org/10.2478/aoas-2020-0021.
- El-Bab, A.F.F., Saghir, S.A.M., El-Naser, I.A.A., El-Kheir, S.M.M.A., Abdel-Kader, M.F., Alruhaimi, R.S., Alqhtani, H.A., Mahmoud, A.M., Naiel, M.A.E., El-Raghi, A.A., 2022. The Effect of Dietary Saccharomyces cerevisiae on Growth Performance, Oxidative Status, and Immune Response of Sea Bream (Sparus aurata). Life 12, 1013. https://doi.org/10.3390/life12071013.
- Momoh, T.A., Odu-Onikosi, S.G., Amulejoye, F.D., Wilson, J., Eynon, B., Kühlwein, H., Kuri, V., Merrifield, D.L., 2025. Brewers’ Yeast ( Saccharomyces cerevisiae ) Purified Functional Feed Additives Mitigate Soybean Meal‐Induced Enteritis in Atlantic Salmon ( Salmo salar ) Parr. Aquaculture Nutrition 2025, 8555658. https://doi.org/10.1155/anu/8555658.
- Aubin, J., Gatesoupe, F.-J., Labbe, L., Lebrun, L., 2005. Trial of probiotics to prevent the vertebral column compression syndrome in rainbow trout (Oncorhynchus mykiss Walbaum). Aquac Research 36, 758–767. https://doi.org/10.1111/j.1365-2109.2005.01280.x.
- Yuan, X.-Y., Jiang, G.-Z., Wang, C.-C., Abasubong, K.P., Zou, Q., Zhou, Y.-Y., Liu, W.-B., 2019. Effects of partial replacement of fish meal by yeast hydrolysate on antioxidant capability, intestinal morphology, and inflammation-related gene expression of juvenile Jian carp (Cyprinus carpio var. Jian). Fish Physiol Biochem 45, 187–197. https://doi.org/10.1007/s10695-018-0552-7.
- Eissa, M.E.H., Alaryani, F.S., Elbahnaswy, S., Khattab, M.S., Elfeky, A., AbouelFadl, K.Y., Eissa, E.-S.H., Ahmed, R.A., Van Doan, H., El-Haroun, E., 2023b. Dietary inclusion of Pediococcus acidilactici probiotic promoted the growth indices, hemato-biochemical indices, enzymatic profile, intestinal and liver histomorphology, and resistance of Nile Tilapia against Aspergillus flavus. Animal Feed Science and Technology 306, 115814. https://doi.org/10.1016/j.anifeedsci.2023.115814.
- Han, S., Liu, Y., Zhou, Z., He, S., Cao, Y., Shi, P., Yao, B., Ringø, E., 2010. Analysis of bacterial diversity in the intestine of grass carp (Ctenopharyngodon idellus) based on 16S rDNA gene sequences: Intestinal bacterial diversity in grass carp. Aquaculture Research 42, 47–56. https://doi.org/10.1111/j.1365-2109.2010.02543.x.
- Ghosh, K., Harikrishnan, R., Mukhopadhyay, A., Ringø, E., 2023. Fungi and Actinobacteria: Alternative Probiotics for Sustainable Aquaculture. Fishes 8, 575. https://doi.org/10.3390/fishes8120575.
- Tan, L.T.-H., Chan, K.-G., Lee, L.-H., Goh, B.-H., 2016. Streptomyces Bacteria as Potential Probiotics in Aquaculture. Front. Microbiol. 7. https://doi.org/10.3389/fmicb.2016.00079.
- Das, S., Lyla, P.S., Ajmal Khan, S., 2006. Application of streptomyces as a probiotic in the laboratory culture of Penaeus monodon (Fabricius). Israeli Journal of Aquaculture - Bamidgeh 58. https://doi.org/10.46989/001c.20439.
- García Bernal, M., Trabal Fernández, N., Saucedo Lastra, P.E., Medina Marrero, R., Mazón-Suástegui, J.M., 2017. Streptomyces effect on the bacterial microbiota associated to Crassostrea sikamea oyster. J Appl Microbiol 122, 601–614. https://doi.org/10.1111/jam.13382.
- AftabUddin, S., Roman, W.U., Hasan, C.K., Ahmed, M., Rahman, H., Siddique, M.A.M., 2018. First incidence of loose-shell syndrome disease in the giant tiger shrimp Penaeus monodon from the brackish water ponds in Bangladesh. Journal of Applied Animal Research 46, 210–217. https://doi.org/10.1080/09712119.2017.1285771.
- Finegold, S.M., Vaisanen, M.-L., Molitoris, D.R., Tomzynski, T.J., Song, Y., Liu, C., Collins, M.D., Lawson, P.A., 2003. Cetobacterium somerae sp. nov. from Human Feces and Emended Description of the Genus Cetobacterium. Systematic and Applied Microbiology 26, 177–181. https://doi.org/10.1078/072320203322346010.
- Kim, P.S., Shin, N.-R., Lee, J.-B., Kim, M.-S., Whon, T.W., Hyun, D.-W., Yun, J.-H., Jung, M.-J., Kim, J.Y., Bae, J.-W., 2021. Host habitat is the major determinant of the gut microbiome of fish. Microbiome 9, 166. https://doi.org/10.1186/s40168-021-01113-x.
- Li, T., Li, H., Gatesoupe, F.-J., She, R., Lin, Q., Yan, X., Li, J., Li, X., 2017. Bacterial Signatures of “Red-Operculum” Disease in the Gut of Crucian Carp (Carassius auratus). Microb Ecol 74, 510–521. https://doi.org/10.1007/s00248-017-0967-1.
- Ma, C., Chen, C., Jia, L., He, X., Zhang, B., 2019. Comparison of the intestinal microbiota composition and function in healthy and diseased Yunlong Grouper. AMB Expr 9, 187. https://doi.org/10.1186/s13568-019-0913-3.
- Praveen, P.K., Debnath, C., Shekhar, S., Dalai, N., Ganguly, S., 2016. Incidence of Aeromonas spp. infection in fish and chicken meat and its related public health hazards: A review. Vet World 9, 6–11. https://doi.org/10.14202/vetworld.2016.6-11.
- Qian, Y., Ye, J., Yang, S., Lin, Z., Cao, W., Xie, J., 2018. Evaluation of the spoilage potential of Shewanella putrefaciens , Aeromonas hydrophila , and Aeromonas sobria isolated from spoiled Pacific white shrimp ( Litopenaeus vannamei ) during cold storage. Journal of Food Safety 38, e12550. https://doi.org/10.1111/jfs.12550.
- Van Kessel, M.A., Dutilh, B.E., Neveling, K., Kwint, M.P., Veltman, J.A., Flik, G., Jetten, M.S., Klaren, P.H., Op Den Camp, H.J., 2011. Pyrosequencing of 16S rRNA gene amplicons to study the microbiota in the gastrointestinal tract of carp (Cyprinus carpio L.). AMB Expr 1, 41. https://doi.org/10.1186/2191-0855-1-41.
