The term “Biofertilizer”, also named as bioinoculants or bioformulations, encompasses organic products comprising beneficial microorganisms, either in their active or inactive forms, able to colonize the rhizosphere or the internal tissues of plants. These microorganisms enhance a plant’s ability to uptake essential nutrients such as nitrogen, phosphorus, and potassium, promoting nutrient availability and uptake capacity, which results in increased crop yields. Thus, biofertilizers have been suggested as a safe and eco-friendly alternative to chemical fertilizers. Legal regulations and social environmental concerns are converging towards the promotion of more sustainable agriculture based on organic compounds and soil preservation.
- biofertilizers
- foliar application
- mulch
- spray application
1. Introduction
2. New Possibilities in Biofertilizer Application Techniques
2.1. Biofertilizers: Inception
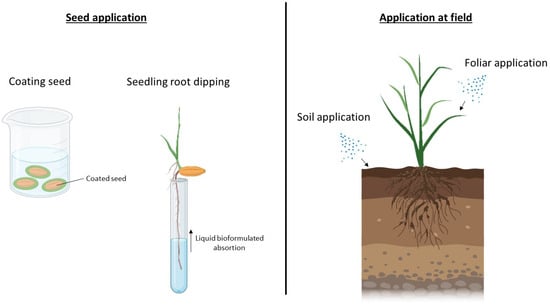
2.2. Microencapsulation and Nanobiofertilizers
2.3. Sprays and Foliar Application
2.4. Mulch Application
References
- Food and Agriculture Organization of the United Nations. Agricultural Production Statistics 2000–2021; Food and Agriculture Organization of the United Nations: Rome, Italy, 2022.
- Chaudhary, P.; Singh, S.; Chaudhary, A.; Sharma, A.; Kumar, G. Overview of Biofertilizers in Crop Production and Stress Management for Sustainable Agriculture. Front. Plant Sci. 2022, 13, 930340.
- Allouzi, M.M.A.; Allouzi, S.M.A.; Keng, Z.X.; Supramaniam, C.V.; Singh, A.; Chong, S. Liquid Biofertilizers as a Sustainable Solution for Agriculture. Heliyon 2022, 8, e12609.
- Nosheen, S.; Ajmal, I.; Song, Y. Microbes as Biofertilizers, a Potential Approach for Sustainable Crop Production. Sustainability 2021, 13, 1868.
- Ibáñez, A.; Garrido-Chamorro, S.; Barreiro, C. Microorganisms and Climate Change: A Not so Invisible Effect. Microbiol. Res. 2023, 14, 918–947.
- Adeyinka Fasusi, O.; Cruz, C.; Oluranti Babalola, O.; Agricultural, O.; Hasanuzzaman, M.; Ahmad Khan, E. Agricultural Sustainability: Microbial Biofertilizers in Rhizosphere Management. Agriculture 2021, 11, 163.
- Mahmud, A.A.; Upadhyay, S.K.; Srivastava, A.K.; Bhojiya, A.A. Biofertilizers: A Nexus between Soil Fertility and Crop Productivity under Abiotic Stress. Curr. Res. Environ. Sustain. 2021, 3, 100063.
- Mehra, A. Biofertilizers Market Worth $5.2 Billion by 2028; MarketsandMarkets: Pune, India, 2020.
- Pirttilä, A.M.; Tabas, H.M.P.; Baruah, N.; Koskimäki, J.J. Biofertilizers and Biocontrol Agents for Agriculture: How to Identify and Develop New Potent Microbial Strains and Traits. Microorganisms 2021, 9, 817.
- Mitter, E.K.; Tosi, M.; Obregón, D.; Dunfield, K.E.; Germida, J.J. Rethinking Crop Nutrition in Times of Modern Microbiology: Innovative Biofertilizer Technologies. Front. Sustain. Food Syst. 2021, 5, 606815.
- Pacheco-Aguirre, J.A.; Ruíz-Sánchez, E.; Ballina-Gómez, H.S.; Alvarado-López, C.J. Does Polymer-Based Encapsulation Enhance Performance of Plant Growth Promoting Microorganisms? A Meta-Analysis View. Agrociencia 2017, 51, 173–187.
- Sohaib, M.; Zahir, Z.A.; Khan, M.Y.; Ans, M.; Asghar, H.N.; Yasin, S.; Al-Barakah, F.N.I. Comparative Evaluation of Different Carrier-Based Multi-Strain Bacterial Formulations to Mitigate the Salt Stress in Wheat. Saudi J. Biol. Sci. 2020, 27, 777–787.
- Thomas, L.; Singh, I. Microbial Biofertilizers: Types and Applications. In Biofertilizers for Sustainable Agriculture and Environment; Springer: Berlin/Heidelberg, Germany, 2019; pp. 1–19.
- John, R.P.; Tyagi, R.D.; Brar, S.K.; Surampalli, R.Y.; Prévost, D. Bio-Encapsulation of Microbial Cells for Targeted Agricultural Delivery. Crit. Rev. Biotechnol. 2011, 31, 211–226.
- Balla, A.; Silini, A.; Cherif-Silini, H.; Chenari Bouket, A.; Alenezi, F.N.; Belbahri, L. Recent Advances in Encapsulation Techniques of Plant Growth-Promoting Microorganisms and Their Prospects in the Sustainable Agriculture. Appl. Sci. 2022, 12, 9020.
- Mącik, M.; Gryta, A.; Frąc, M. Biofertilizers in Agriculture: An Overview on Concepts, Strategies and Effects on Soil Microorganisms. In Advances in Agronomy; Springer: Berlin/Heidelberg, Germany, 2020; pp. 31–87.
- Gewin, V. How Peat Could Protect the Planet. Nature 2020, 578, 204–208.
- Dey, A. Liquid Biofertilizers and Their Applications: An Overview. In Environmental and Agricultural Microbiology; Wiley: Hoboken, NJ, USA, 2021; pp. 275–292.
- Patel, C.; Singh, J.; Karunakaran, A.; Ramakrishna, W. Evolution of Nano-Biofertilizer as a Green Technology for Agriculture. Agriculture 2023, 13, 1865.
- Singh, D.; Thapa, S.; Geat, N.; Mehriya, M.L.; Rajawat, M.V.S. Biofertilizers: Mechanisms and Application. In Biofertilizers; Elsevier: Amsterdam, The Netherlands, 2021; pp. 151–166.
- Singh, G.; Paithankar, I. Encapsulation of Biofertilizers, Biopesticides and Biocontrol Agents. In Sustainable Agriculture Reviews; Singh, N.K., Chattopadhyay, A., Lichtfouse, E., Eds.; Springer: Cham, Switzerland, 2023; pp. 121–150.
- Sudheer, S.; Bai, R.G.; Usmani, Z.; Sharma, M. Insights on Engineered Microbes in Sustainable Agriculture: Biotechnological Developments and Future Prospects. Curr. Genom. 2020, 21, 321–333.
- Young, C.; Rekha, P.D.; Lai, W.; Arun, A.B. Encapsulation of Plant Growth-promoting Bacteria in Alginate Beads Enriched with Humic Acid. Biotechnol. Bioeng. 2006, 95, 76–83.
- Saberi-Rise, R.; Moradi-Pour, M. The Effect of Bacillus subtilis Vru1 Encapsulated in Alginate—Bentonite Coating Enriched with Titanium Nanoparticles against Rhizoctonia solani on Bean. Int. J. Biol. Macromol. 2020, 152, 1089–1097.
- Panichikkal, J.; Prathap, G.; Nair, R.A.; Krishnankutty, R.E. Evaluation of Plant Probiotic Performance of Pseudomonas Sp. Encapsulated in Alginate Supplemented with Salicylic Acid and Zinc Oxide Nanoparticles. Int. J. Biol. Macromol. 2021, 166, 138–143.
- Amiet-Charpentier, C.; Gadille, P.; Benoit, J.P. Rhizobacteria Microencapsulation: Properties of Microparticles Obtained by Spray-Drying. J. Microencapsul. 1999, 16, 215–229.
- Thirugnanasambandan, T. Advances and Trends in Nano-Biofertilizers. SSRN Electron. J. 2018.
- Akhtar, N.; Ilyas, N.; Meraj, T.A.; Pour-Aboughadareh, A.; Sayyed, R.Z.; Mashwani, Z.-R.; Poczai, P. Improvement of Plant Responses by Nanobiofertilizer: A Step towards Sustainable Agriculture. Nanomaterials 2022, 12, 965.
- Garg, D.; Sridhar, K.; Stephen Inbaraj, B.; Chawla, P.; Tripathi, M.; Sharma, M. Nano-Biofertilizer Formulations for Agriculture: A Systematic Review on Recent Advances and Prospective Applications. Bioengineering 2023, 10, 1010.
- Li, C.-C.; Dang, F.; Li, M.; Zhu, M.; Zhong, H.; Hintelmann, H.; Zhou, D.-M. Effects of Exposure Pathways on the Accumulation and Phytotoxicity of Silver Nanoparticles in Soybean and Rice. Nanotoxicology 2017, 11, 699–709.
- Alshaal, T.; El-Ramady, H. Foliar Application: From Plant Nutrition to Biofortification. Environ. Biodivers. Soil Secur. 2017, 1, 71–83.
- Hong, J.; Wang, C.; Wagner, D.C.; Gardea-Torresdey, J.L.; He, F.; Rico, C.M. Foliar Application of Nanoparticles: Mechanisms of Absorption, Transfer, and Multiple Impacts. Environ. Sci. Nano 2021, 8, 1196–1210.
- Guedes, W.A.; Araújo, R.H.C.R.; Rocha, J.L.A.; de Lima, J.F.; Dias, G.A.; de Oliveira, Á.M.F.; de Lima, R.F.; Oliveira, L.M. Production of Papaya Seedlings Using Spirulina platensis as a Biostimulant Applied on Leaf and Root. J. Exp. Agric. Int. 2018, 28, 1–9.
- Paul, K.; Sorrentino, M.; Lucini, L.; Rouphael, Y.; Cardarelli, M.; Bonini, P.; Miras Moreno, M.B.; Reynaud, H.; Canaguier, R.; Trtílek, M.; et al. A Combined Phenotypic and Metabolomic Approach for Elucidating the Biostimulant Action of a Plant-Derived Protein Hydrolysate on Tomato Grown Under Limited Water Availability. Front. Plant Sci. 2019, 10, 493.
- Kapoore, R.V.; Wood, E.E.; Llewellyn, C.A. Algae Biostimulants: A Critical Look at Microalgal Biostimulants for Sustainable Agricultural Practices. Biotechnol. Adv. 2021, 49, 107754.
- Filipczak, J.; Żurawicz, E.; Paszt, L.S. Influence of Selected Biostimulants on the Growth and Yielding of “Elkat” Strawberry Plants. Zesz. Nauk. Inst. Ogrod. 2016, 24, 43–58.
- Oancea, F.; Velea, S.; Fatu, V.; Mincea, C.; Ilie, L. Micro-Algae Based Plant Biostimulant and Its Effect on Water Stressed Tomato Plants. Rom. J. Plant Prot. 2013, 6, 104–117.
- López-Padrón, I.; Martínez-González, L.; Pérez-Domínguez, G.; Cedeño-Rodríguez, L.; Reyes-Guerrero, Y.; Cárdenas-Travieso, R.M.; Núñez-Vázquez, M.; Cabrera-Rodríguez, J.A. Effects of Bioactive Products on Cicer arietinum L. Plants. Cultiv. Trop. 2021, 42, e06.
- Dias, G.A.; Rocha, R.H.C.; Araújo, J.L.; de Lima, J.F.; Guedes, W.A. Growth, Yield, And Postharvest Quality In Eggplant Produced Under Different Foliar Fertilizer (Spirulina platensis) Treatments. Semin. Cienc. Agrarias. Univ. Estadual De Londrina 2016, 37, 3893–3902.
- Abbasi, P.A.; Weselowski, B. Influence of Foliar Sprays of Bacillus subtilis QST 713 on Development of Early Blight Disease and Yield of Field Tomatoes in Ontario. Can. J. Plant Pathol. 2014, 36, 170–178.
- Abbasi, P.A.; Weselowski, B. Efficacy of Bacillus subtilis QST 713 Formulations, Copper Hydroxide, and Their Tank Mixes on Bacterial Spot of Tomato. Crop Prot. 2015, 74, 70–76.
- El Habbasha, E.S.; Mekhail Tawkik, M.; El Kramany, M.F. Comparative Efficacy of Different Bio-Chemical Foliar Applications on Growth, Yield and Yield Attributes of Some Wheat Cultivars. World J. Agric. Sci. 2013, 9, 345–353.
- Costa, R.R.G.F.; Quirino, G.D.S.F.; Naves, D.C.D.F.; Santos, C.B.; Rocha, A.F.D.S. Efficiency of Inoculant with Azospirillum brasilense on the Growth and Yield of Second-Harvest Maize. Pesqui. Agropecu. Trop. 2015, 45, 304–311.
- Preininger, C.; Sauer, U.; Bejarano, A.; Berninger, T. Concepts and Applications of Foliar Spray for Microbial Inoculants. Appl. Microbiol. Biotechnol. 2018, 102, 7265–7282.
- Abbate, C.; Scavo, A.; Pesce, G.R.; Fontanazza, S.; Restuccia, A.; Mauromicale, G. Soil Bioplastic Mulches for Agroecosystem Sustainability: A Comprehensive Review. Agriculture 2023, 13, 197.
- Huang, F.; Zhang, Q.; Wang, L.; Zhang, C.; Zhang, Y. Are Biodegradable Mulch Films a Sustainable Solution to Microplastic Mulch Film Pollution? A Biogeochemical Perspective. J. Hazard. Mater. 2023, 459, 132024.
- Lightfoot, D.R. Morphology and Ecology of Lithic-Mulch Agriculture. Geogr. Rev. 1994, 84, 172.
- Vinci, G.; Ruggieri, R.; Billi, A.; Pagnozzi, C.; Di Loreto, M.V.; Ruggeri, M. Sustainable Management of Organic Waste and Recycling for Bioplastics: A LCA Approach for the Italian Case Study. Sustainability 2021, 13, 6385.
- Skoczinski, P.; Carus, M.; de Guzman, D.; Käb, H.; Chinthapalli, R.; Ravenstijn, J.; Baltus, W.; Raschka, A. Bio-Based Building Blocks and Polymers—Global Capacities, Production and Trends 2020–2025; Language: Hürt, Germany, 2021.
- ASTM D5488-94DE1; Standard Terminology of Environmental Labeling of Packaging Materials and Packages. ASTM: West Conshohocken, PA, USA, 2002.
- Mansoor, Z.; Tchuenbou-Magaia, F.; Kowalczuk, M.; Adamus, G.; Manning, G.; Parati, M.; Radecka, I.; Khan, H. Polymers Use as Mulch Films in Agriculture—A Review of History, Problems and Current Trends. Polymers 2022, 14, 5062.
- Lasmini, S.A.; Idham; Pagiu, S.; Yusuf, R.; Hayati, N.; Yunus, M.; Pasaru, F.; Nasir, B.H.; Rosmini; Khasanah, N.; et al. Application of Mulch and Soil Microbes to Increase Growth and Yield of Chili Pepper. AIP Conf. Proc. 2023, 2606, 040001.
