Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Lindsay Dong and Version 1 by Nyein Wint Yee Theik.
Neurotrophic tyrosine receptor kinase (NTRK) has been a remarkable therapeutic target for treating different malignancies, playing an essential role in oncogenic signaling pathways. Groundbreaking trials like NAVIGATE led to the approval of NTRK inhibitors by the Food and Drug Administration (FDA) to treat different malignancies, significantly impacting current oncology treatment. Accurate detection of NTRK gene fusion becomes very important for possible targeted therapy. Various methods to detect NTRK gene fusion have been applied widely based on sensitivity, specificity, and accessibility.
- NTRK gene fusion
- NTRK inhibitor
- malignancies
1. Introduction
Neurotrophic tyrosine receptor kinase (NTRK) inhibitors for certain tumors expressing these fusion proteins have recently become a new favored treatment [1]. The NTRK genes, including NTRK 1, 2, and 3, encode tropomyosin receptor kinase (TRK) receptor family proteins like TRKA, TRKB, and TRKC, known as TRK, responsible for neural cell progress and activities [2]. Any variations or rearrangements of the genes could lead to the activation of neural cells [3]. Among the NTRK genes, NTRK1 was initially recognized as an actionable oncogene in colon cancer by Mariano Barbacid and colleagues in 1982 [4]. Different NTRK translocations or gene fusions and TRK protein overexpression forms were eventually observed in solid tumors besides colon cancer, including other gastrointestinal, gynecological, thyroid, lung, and pediatric malignancies [5].
Among a total of 55 patients in phase 2 of the NAVIGATE trial, 55% of individuals remained progression-free after one year of follow-up, which demonstrated an excellent overall response rate (ORR) to larotrectinib (75% with a 95% confidence interval [CI] of 61–85%) [6]. This groundbreaking trial led to a first-generation NTRK inhibitor (larotrectinib) being approved by the Food and Drug Administration (FDA) that is histology agnostic, including treatment for adult and child malignancies. After that, another first-generation inhibitor (entrectinib) became the second inhibitor approved by the FDA [7].
2. Different Methods for NTRK Gene Fusion Detection
Diverse methods exist for detecting aberrant NTRK gene fusions. Each test has its characteristics, limitations, and benefits; however, the choice of test is typically determined by sample diversity and the test’s sensitivity and specificity. One molecular cytogenetics technique called fluorescence in situ hybridization (FISH) employs fluorescently labeled deoxyribonucleic acid (DNA) probes to detect abnormal rearrangements in fusion genes. Formalin-fixed, paraffin-embedded (FFPE) tumor specimens are typically used to visualize aberrant fusion genes. FISH is frequently used to diagnose certain cancers, such as secretory breast carcinoma (SBC) and infantile fibrosarcoma, due to the prevalence of NTRK genes in these conditions [8]. Reverse transcriptase polymerase chain reaction (RT-PCR) is generally helpful in detecting NTRK fusions by amplifying ribonucleic acid (RNA) molecules and is applied as a confirmation test after other tests show positive results for abnormal expression [9]. One of the most frequently used tests, Next-Generation Sequencing (NGS), is based on known NTRK fusion partners, validating the identification of fusion events. The benefit of the test is that it warrants comprehensive genomic profiling and can detect other actionable genetic alterations in addition to NTRK fusions and their various variants. Immunohistochemistry (IHC) is one of the most widely used and accessible assays due to its low cost and ability to detect abnormal NTRK protein expressions. The specificity and sensitivity are usually gene partner-dependent and tumor-dependent, making it an excellent screening test overall. However, confirmation tests are occasionally advised because its value is limited to screening [11][10]. Total transcriptome RNA sequencing necessitates sequencing the entire RNA transcriptome, which enables the identification of aberrant fusion events based on significant alignment patterns and breakpoints, even though it is pricey and laborious, and RNA technology is less commonly used than DNA NGS. In summary, DNA NGS in tissue is the best technology to detect NTRK fusions, and RNA NGS is the best complement to identify these fusions. Liquid biopsies are an alternative to DNA NGS; however, RNA NGS is not yet widely available in liquid biopsies.3. TRK Biology and Ontogenesis
NTRK belongs to the tyrosine receptor kinase family, which includes NTRK1, NTRK2, and NTRK3, encoding for TRKA, TRKB, and TRKC, respectively. NTRK1 was initially identified as an oncogene in 1982 by Mariano Barbacid and colleagues during gene transfer assays aimed at discovering genes with transforming capabilities in human tumor specimens, specifically those from colon cancer [4]. Subsequently, the cDNA of the NTRK1 proto-oncogene was isolated and characterized as TRKA, a 790-amino acid protein displaying features characteristic of cell surface receptor tyrosine kinases, in 1989 [12][11]. TRKA was found to be expressed in the nervous system in 1991 and recognized as a receptor for neurotrophic nerve growth factor (NGF) [13][12]. TRK receptors are capable of binding to different ligands such as NGF for TRKA, brain-derived neurotrophic factor (BDNF) or neurotrophin 4 (NT-4) for TRKB, and neurotrophin 3 (NT-3) for TRKC [14][13]. Neurotrophins were initially identified as survival molecules for neurons. The binding of neurotrophins to their alternative receptor, p75NTR, primarily results in the activation of JNK signaling cascades, as well as the P75NTR-interacting protein (NRAGE, also known as melanoma-associated antigen D1), and P75NTR-associated cell death executor (NADE) adaptors that promote cell cycle arrest and apoptosis directly [15,16,17][14][15][16]. TRK receptors can be activated by different mechanisms, such as the G-protein coupled receptor (GPCR) mechanism, without neurotrophins’ involvement. In order to increase the survival of neural cells through AKT activity, the TRK receptor can be activated by two GPCR ligands, adenosine, and pituitary adenylate cyclase-activating polypeptide (PACAP) [20][17]. Somatic NTRK mutation has been identified in various tumor types, including colorectal and lung cancers such as large cell neuroendocrine carcinoma and NSCLC, as well as melanoma and acute myeloid leukemia [21,22,23,24][18][19][20][21].4. Mechanisms of Action
Both first-generation NTRK inhibitors are orally administered and warranted to treat adult solid malignancies and some pediatric tumors. They bind selectively to the ATP-binding site of abnormal NTRK gene fusion proteins, inhibit ATP from binding to the fusion protein, and inhibit the action of signals in its downward path that collaborate in the growth of cells, survival, and differentiation. This inhibition disrupts the disruption of abnormal signaling cascades and encourages tumor regression (Figure 1) [25][22].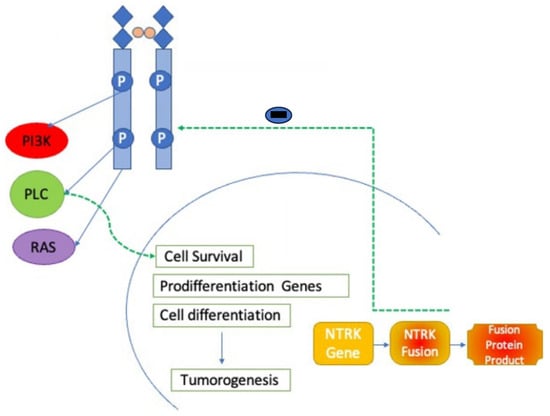
Figure 1. NTRK inhibitor mechanism of action. NTRK inhibitors block action through the NTRK receptor preventing downstream activation of phoshoinositide-3-kinase (PI3K), phospholipase C (PLC), and RAS pathways.
5. First-Generation NTRK Inhibitors
5.1. Larotrectinib
First-generation NTRK inhibitor trials predominantly adopt a basket trial design due to the infrequent occurrence of NTRK fusions within specific tumor types. Larotrectinib, the inaugural pan-TRK inhibitor, selectively targets TRK receptor proteins, namely TRKA, TRKB, and TRKC. Three pivotal clinical trials, namely LOXO-TRK-14001 (NCT02122913), SCOUT (NCT02637687), and NAVIGATE (NCT02576431), collectively enrolled 55 eligible patients across 21 distinct cancer types [6,28,29][6][24][25]. The cohort comprised both adult and pediatric patients administered with 100 mg of larotrectinib twice daily. The objective response rate (ORR) was calculated at 75% (with a 95% confidence interval of 67–85%) [6].5.2. Entrectinib
Entrectinib is a multikinase inhibitor that halts reactive oxygen species (ROS) oncogene 1 (ROS1) and anaplastic lymphoma kinase (ALK) in addition to blocking tyrosine receptor kinase A, B, and C [31][26]. Integrated analysis of four trials in 2019 included STARTRK-1, STARTRK-2, and ALKA-372-001, with 54 adult patients aged over 18 years who received entrectinib [32][27]. Common toxicities are noted in Grades 1 and 2, including neurologic complications such as dizziness (27% of patients), cognitive changes (4%), and weight gain, which is most commonly reported as Grade 3. No treatment-related mortality was noted. The study revealed a CR of 7% and a PR of 50%.6. Second-Generation NTRK Inhibitors
6.1. Repotrectinib
Due to the limited durability and resistant mutations against first-generation inhibitors, second-generation inhibitor trials have started to overcome the difficulties. Repotrectinib has a smaller molecular weight and a more compact macrocyclic structure than the first generation [34][28]. Repotrectinib is a small compound compared to other inhibitors created to accommodate the bulky, positively charged arginine side chain in the solvent front without steric clashes. In phases 1 and 2 of the TRIDENT-1 study (NCT03093116), with 40 populations who received repotrectinib, the ORR was 41–62% [35][29]. Common side effects from the study include mild Grade 1 ataxia, paresthesia, nausea, perioral numbness, and dysgeusia—the FDA approved repotrectinib to treat patients with advanced solid tumors due to the safety profile of the medication.6.2. Selitrectinib
Although the major clinical trials of selitretinib, including NCT03206931 and NT03215511 are ongoing, most case reports reported that selitrectinib might be an efficient medication for solid malignancies [36,37][30][31]. Additionally, patients resistant to entrectinib through the NTRK3 G623R mutation revealed good responses with selitrectinib per the 2021 report [38][32].6.3. Taletrectinib
Taletrectinib is a newly emerged ROS1/NTRK kinase inhibitor with high activity against the G2031R solvent-front mutation compared to other inhibitors [39][33]. According to the published report of the first human phase 1 study (TRUST) in 2020 with 46 patient populations, the confirmed objective response rate was 33.3% [40][34]. Most common treatment-related adverse events, such as nausea (47.8%), diarrhea (43.5%), and vomiting (32.6%), were noted.6.4. Other Agents
Several multi-target tyrosine kinase inhibitors (TKIs) with varying inhibitory activity against tropomyosin receptor kinase (TRK) have received approvals for indications beyond the treatment of patients with NTRK fusions. Crizotinib, initially designed as a MET inhibitor, was later recognized as an inhibitor of anaplastic lymphoma kinase (ALK), ROS1, and TRK. However, its affinity for TRK is significantly lower than for MET, ALK, and ROS1. It is approved for treating ALK- and ROS1-rearranged non-small cell lung cancer (NSCLC) [41][35]. Cabozantinib, approved by the FDA for renal cell carcinoma and medullary thyroid carcinoma, targets multiple receptor tyrosine kinases, including MET, RET, AXL, TRKA, and TRKB [42,43,44][36][37][38]. Ponatinib, originally developed as a BCR-ABL1 inhibitor for chronic myelogenous leukemia, has demonstrated efficacy against most BCR-ABL1 resistance mutations and has shown potential in suppressing the growth of NTRK fusion-positive tumors in preclinical trials [45,46][39][40]. Nintedanib, recognized as an anti-angiogenic drug and vascular endothelial growth factor receptor (VEGFR) tyrosine kinase inhibitor, has also exhibited inhibitory effects on platelet-derived growth factor receptor (PDGFR), fibroblast growth factor receptor (FGFR), and TRK kinases [47,48][41][42]..5. Mechanisms of Resistance to NTRK Inhibitors
6.5. Mechanisms of Resistance to NTRK Inhibitors
However, resistance to first-generation inhibitors was identified through the mutations of NTRK1 gene fusion proteins such as G667C and G595R [49][43]. In a 2021 report, one patient with mammary secretory carcinoma of the parotic gland and the ETV6-NTRK3 fusion reported secondary resistance to entrectinib through the NTRK3 G623R mutation [38][32]. Second-generation inhibitors, which include selitrectinib and repotrectinib, have emerged to overcome the mechanism of acquired resistance. However, recent trials indicated that resistance against the second generation develops through the mutation of TRK proteins such as xDFG [50][44].6.6. Overall Side Effects and Effects of TRK Inhibition
According to Smeyne RJ et al., in a 1994 trial, NTRK1-null mice lacking most sympathetic neurons did not exhibit nociceptive and temperature sensations and eventually passed away within a month due to severe sensory and sympathetic neuropathies [51][45]. In comparison, NTRK2 knockout mice, lacking motor neurons or dorsal root and trigeminal ganglia neurons, died perinatally due to a lack of eating [52][46]. NTRK3-null mice also exhibited deficits in the quality and quantity of motor neurons and deficits in a population of dorsal root ganglia neurons, resulting in abnormal movements and posture [53][47]. Homozygous disruption of the NTRK2 gene revealed an increase in apoptosis of endothelial cells and a decreased number of intramyocardial blood vessels.7. Role of NTRK Inhibitors in Various Types of Cancers and Adverse Effects
7.1. NTRK Gene Fusion in Lung Cancer
Among lung cancers, especially non-small cell lung cancer (NSCLC), NTRK gene fusions can be found only 0.5% of the time. However, gene fusion can also be found in other subtypes like neuroendocrine carcinoma and sarcomatoid tumors of the lung [57][48]. NTRK1 and NTRK2 are commonly expressed in squamous cell carcinoma compared to adenocarcinoma and small cell lung cancer. First-generation inhibitors have been proven to be a practical and durable therapy for managing NSCLC.7.2. NTRK Gene Fusion in Colorectal Cancer
In approximately 0.7% of colon cancers, NTRK gene fusion, especially NTRK1 and NTRK3, is found alongside APC and TP53 gene aberrations and RAS/BRAF gene alterations [61][49]. The TPM3-NTRK1 rearrangement is commonly seen [62][50]. NTRK is prevalent in females with a right-sided primary colorectal tumor, RAS/RAF WT status, and MSI phenotype, according to data from the phase 2 NAVIGATE trial, which produces various responses in patients to larotrectinib with the gene-positive fusion, and which is locally advanced, and in metastatic GI cancers, especially in colorectal cancer with an ORR which is 3% and a partial response rate of 30% [63,64][51][52]. Therefore, it is crucial to identify NTRK fusions in GI malignancy patients, especially those with high microsatellite instability rates [65][53].7.3. NTRK Gene Fusion in Central Nervous System Malignancies
Central Nervous System (CNS) tumors, such as low- and high-grade gliomas, are distinguished from other solid malignancies by the quantity of NTRK gene fusion [66][54]. The NTRK1/TRKA fusion has been linked to an improved prognosis and an increased likelihood of tumor regression. The NTRK2/TRKB fusion has been found to be detrimental in certain tumors [66][54]. Gliomas, characterized by NTRK gene expression, tend to affect multiple regions of the nervous system, particularly the hemisphere and frequently have an aggressive clinical course. The cohort study conducted by Torre et al. identified the detailed anatomical distribution of NTRK fusions, revealing a predominant occurrence in the hemispheric region (66.7%).7.4. NTRK Gene Fusion in Thyroid Cancers
Thyroid cancer, like other solid tumors, has few NTRK fusions. NTRK rearrangement in the TKD of the NTRK1 gene can arise in thyroid cancer, particularly papillary thyroid carcinoma. Furthermore, a gene mutation known as the ETV-NTRK3 fusion is present in around 14.5% of patients diagnosed with thyroid cancer due to radiation exposure following the Chornobyl nuclear accident [70][55]. IRF2BP2-NTRK3 is also spotted in papillary thyroid carcinoma [61][49].7.5. NTRK Gene Fusion in Hematological Malignancies
Hematological malignancies, including acute myeloid and lymphoblastic leukemia, chronic lymphocytic leukemia (CLL), and Philadelphia-positive ALL, frequently have the NTRK-ETV6 fusion. Taylor et al. reported that larotrectinib, a first-generation inhibitor, reduced expression of the fusion gene while inhibiting TRK activity in patients with NTRK2-ETV6 variants associated with histiocytosis and complex myeloma [72][56]. Notably, TP53 cell lines are more sensitive to the first-generation inhibitor [73][57]. Despite these promising results, TRK aberrations have been shown to have reactivity against the therapeutic target, limiting the efficacy of clinical benefits in certain hematological tumors.7.6. NTRK Gene Fusion in Sarcomas
Among soft tissue sarcomas, the predominant characterization of infantile fibrosarcoma and lipofibromatosis-like neural tumors revolves around ETV6-NTRK3 fusions and NTRK1 gene rearrangements [74,75][58][59]. In a minority of cases, NTRK3-negative infantile fibrosarcomas have been documented to exhibit NTRK1 gene rearrangements instead [76][60].7.7. NTRK Gene Fusion in Melanocytic Tumors
NTRK fusions have a diagnostic and classification impact among melanocytic tumors. Among melanocytic neoplasms, Spitz tumors are one subgroup with distinct morphological features that tend to affect young individuals [81,82][61][62]. Yeh et al. identified ETV6-NTRK3, MYO5A-NTRK3, and MYH9-NTRK3 fusions in Spitz tumors [83][63].7.8. NTRK Gene Fusion in Salivary Gland Tumors
The presence of NTRK gene fusions has been specifically observed in rare tumors, particularly in secretory carcinomas of the breast and salivary gland, indicating a unique prevalence. The most common fusion variant within the category of salivary gland carcinomas is ETV6-NTRK3. This fusion subtype has been consistently identified in different types of salivary gland carcinomas, including two cases of secretory carcinoma and one case of adenocarcinoma [89][64]. The frequent presence of the ETV6-NTRK3 fusion in salivary gland tumors highlights its importance as a possible diagnostic indicator and target for medical treatment.References
- National Cancer Institute. FDA Approves Entrectinib for NTRK Fusion Cancers. Cancer Currents Blog. 16 August 2019. Available online: https://www.cancer.gov/news-events/cancer-currents-blog/2019/fda-entrectinib-ntrk-fusion (accessed on 1 January 2020).
- Drilon, A.; Siena, S.; Ou, S.I.; Patel, M.; Ahn, M.J.; Lee, J.; Bauer, T.M.; Farago, A.F.; Wheler, J.J.; Liu, S.V.; et al. Safety and Antitumor Activity of the Multitargeted Pan-TRK, ROS1, and ALK Inhibitor Entrectinib: Combined Results from Two Phase I Trials (ALKA-372-001 and STARTRK-1). Cancer Discov. 2017, 7, 400–409.
- Lannon, C.L.; Sorensen, P.H. ETV6-NTRK3: A chimeric protein tyrosine kinase with transformation activity in multiple cell lineages. Semin. Cancer Biol. 2005, 15, 215–223.
- Pulciani, S.; Santos, E.; Lauver, A.V.; Long, L.K.; Aaronson, S.A.; Barbacid, M. Oncogenes in solid human tumours. Nature 1982, 300, 539–542.
- Ruiz-Cordero, R.; Ng, D.L. Neurotrophic receptor tyrosine kinase (NTRK) fusions and their role in cancer. Cancer Cytopathol. 2020, 128, 775–779.
- Drilon, A.; Laetsch, T.W.; Kummar, S.; DuBois, S.G.; Lassen, U.N.; Demetri, G.D.; Nathenson, M.; Doebele, R.C.; Farago, A.F.; Pappo, A.S.; et al. Efficacy of Larotrectinib in TRK Fusion-Positive Cancers in Adults and Children. N. Engl. J. Med. 2018, 378, 731–739.
- Marcus, L.; Donoghue, M.; Aungst, S.; Myers, C.E.; Helms, W.S.; Shen, G.; Zhao, H.; Stephens, O.; Keegan, P.; Pazdur, R. FDA Approval Summary: Entrectinib for the Treatment of NTRK gene Fusion Solid Tumors. Clin. Cancer Res. 2021, 27, 928–932.
- Connor, A.; Perez-Ordoñez, B.; Shago, M.; Skálová, A.; Weinreb, I. Mammary analog secretory carcinoma of salivary gland origin with the ETV6 gene rearrangement by FISH: Expanded morphologic and immunohistochemical spectrum of a recently described entity. Am. J. Surg. Pathol. 2012, 36, 27–34.
- Skálová, A.; Vanecek, T.; Simpson, R.H.; Laco, J.; Majewska, H.; Baneckova, M.; Steiner, P.; Michal, M. Mammary Analogue Secretory Carcinoma of Salivary Glands: Molecular Analysis of 25 ETV6 Gene Rearranged Tumors with Lack of Detection of Classical ETV6-NTRK3 Fusion Transcript by Standard RT-PCR: Report of 4 Cases Harboring ETV6-X Gene Fusion. Am. J. Surg. Pathol. 2016, 40, 3–13.
- Wong, D.; Yip, S.; Sorensen, P.H. Methods for Identifying Patients with Tropomyosin Receptor Kinase (TRK) Fusion Cancer. Pathol. Oncol. Res. 2020, 26, 1385–1399.
- Martin-Zanca, D.; Oskam, R.; Mitra, G.; Copeland, T.; Barbacid, M. Molecular and biochemical characterization of the human trk proto-oncogene. Mol. Cell Biol. 1989, 9, 24–33.
- Klein, R.; Jing, S.Q.; Nanduri, V.; O’Rourke, E.; Barbacid, M. The trk proto-oncogene encodes a receptor for nerve growth factor. Cell 1991, 65, 189–197.
- Soppet, D.; Escandon, E.; Maragos, J.; Middlemas, D.S.; Reid, S.W.; Blair, J.; Burton, L.E.; Stanton, B.R.; Kaplan, D.R.; Hunter, T.; et al. The neurotrophic factors brain-derived neurotrophic factor and neurotrophin-3 are ligands for the trkB tyrosine kinase receptor. Cell 1991, 65, 895–903.
- Frade, J.M.; Barde, Y.A. Nerve growth factor: Two receptors, multiple functions. BioEssays: News and reviews in molecular, cellular and developmental biology. Bioessays 1998, 20, 137–145.
- Patel, T.D.; Jackman, A.; Rice, F.L.; Kucera, J.; Snider, W.D. Development of sensory neurons in the absence of NGF/TrkA signaling in vivo. Neuron 2000, 25, 345–357, Erratum in Neuron 2003, 37, 183.
- Teng, H.K.; Teng, K.K.; Lee, R.; Wright, S.; Tevar, S.; Almeida, R.D.; Kermani, P.; Torkin, R.; Chen, Z.Y.; Lee, F.S.; et al. ProBDNF induces neuronal apoptosis via activation of a receptor complex of p75NTR and sortilin. J. Neurosci. Off. J. Soc. Neurosci. 2005, 25, 5455–5463.
- Rajagopal, R.; Chen, Z.Y.; Lee, F.S.; Chao, M.V. Transactivation of Trk neurotrophin receptors by G-protein-coupled receptor ligands occurs on intracellular membranes. J. Neurosci. 2004, 24, 6650–6658.
- Geiger, T.R.; Song, J.Y.; Rosado, A.; Peeper, D.S. Functional characterization of human cancer-derived TRKB mutations. PLoS ONE 2011, 6, e16871.
- Harada, T.; Yatabe, Y.; Takeshita, M.; Koga, T.; Yano, T.; Wang, Y.; Giaccone, G. Role and relevance of TrkB mutations and expression in non-small cell lung cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2011, 17, 2638–2645.
- Miranda, C.; Mazzoni, M.; Sensi, M.; Pierotti, M.A.; Greco, A. Functional characterization of NTRK1 mutations identified in melanoma. Genes Chromosomes Cancer 2014, 53, 875–880.
- Tomasson, M.H.; Xiang, Z.; Walgren, R.; Zhao, Y.; Kasai, Y.; Miner, T.; Ries, R.E.; Lubman, O.; Fremont, D.H.; McLellan, M.D.; et al. Somatic mutations and germline sequence variants in the expressed tyrosine kinase genes of patients with de novo acute myeloid leukemia. Blood 2008, 111, 4797–4808.
- Cocco, E.; Scaltriti, M.; Drilon, A. NTRK fusion-positive cancers and TRK inhibitor therapy. Nat. Rev. Clin. Oncol. 2018, 15, 731–747.
- Drilon, A.; Li, G.; Dogan, S.; Gounder, M.; Shen, R.; Arcila, M.; Wang, L.; Hyman, D.M.; Hechtman, J.; Wei, G.; et al. What hides behind the MASC: Clinical response and acquired resistance to entrectinib after ETV6-NTRK3 identification in a mammary analogue secretory carcinoma (MASC). Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2016, 27, 920–926.
- Doebele, R.C.; Davis, L.E.; Vaishnavi, A.; Le, A.T.; Estrada-Bernal, A.; Keysar, S.; Jimeno, A.; Varella-Garcia, M.; Aisner, D.L.; Li, Y.; et al. An Oncogenic NTRK Fusion in a Patient with Soft-Tissue Sarcoma with Response to the Tropomyosin-Related Kinase Inhibitor LOXO-101. Cancer Discov. 2015, 5, 1049–1057.
- Laetsch, T.W.; DuBois, S.G.; Mascarenhas, L.; Turpin, B.; Federman, N.; Albert, C.M.; Nagasubramanian, R.; Davis, J.L.; Rudzinski, E.; Feraco, A.M.; et al. Larotrectinib for paediatric solid tumours harbouring NTRK gene fusions: Phase 1 results from a multicentre, open-label, phase 1/2 study. Lancet Oncol. 2018, 19, 705–714.
- Rolfo, C.; Ruiz, R.; Giovannetti, E.; Gil-Bazo, I.; Russo, A.; Passiglia, F.; Giallombardo, M.; Peeters, M.; Raez, L. Entrectinib: A potent new TRK, ROS1, and ALK inhibitor. Expert Opin. Investig. Drugs 2015, 24, 1493–1500.
- Doebele, R.C.; Drilon, A.; Paz-Ares, L.; Siena, S.; Shaw, A.T.; Farago, A.F.; Blakely, C.M.; Seto, T.; Cho, B.C.; Tosi, D.; et al. Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours: Integrated analysis of three phase 1–2 trials. Lancet Oncol. 2020, 21, 271–282.
- Murray, B.W.; Rogers, E.; Zhai, D.; Deng, W.; Chen, X.; Sprengeler, P.A.; Zhang, X.; Graber, A.; Reich, S.H.; Stopatschinskaja, S.; et al. Molecular Characteristics of Repotrectinib That Enable Potent Inhibition of TRK Fusion Proteins and Resistant Mutations. Mol. Cancer Ther. 2021, 20, 2446–2456.
- Cho, B.C.; Drilon, A.E.; Doebele, R.C.; Kim, D.W.; Lin, J.J.; Lee, J.; Ahn, M.J.; Zhu, V.W.; Ejadi, S.; Camidge, D.R. Safety and preliminary clinical activity of repotrectinib in patients with advanced ROS1 fusion-positive non-small cell lung cancer (TRIDENT-1 study). J. Clin. Oncol. 2019, 37 (Suppl. 15), 9011.
- Hyman, D.; Kummar, S.; Farago, A.; Geoerger, B.; Mau-Sorensen, M.; Taylor, M.; Garralda, E.; Nagasubramanian, R.; Natheson, M.; Song, L.; et al. Abstract CT127: Phase I and expanded access experience of LOXO-195 (BAY 2731954), a selective next-generation TRK inhibitor (TRKi). Cancer Res. 2019, 79 (Suppl. 13), CT127.
- National Institutes of Health (NIH). ClinicalTrials.gov. A Study to Test the Safety of the Investigational Drug Selitrectinib in Children and Adults that May Treat Cancer. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03215511?cond=LOXO-195&rank=1 (accessed on 11 April 2019).
- Florou, V.; Nevala-Plagemann, C.; Whisenant, J.; Maeda, P.; Gilcrease, G.W.; Garrido-Laguna, I. Clinical Activity of Selitrectinib in a Patient with Mammary Analogue Secretory Carcinoma of the Parotid Gland with Secondary Resistance to Entrectinib. J. Natl. Compr. Cancer Netw. 2021, 19, 478–482.
- Katayama, R.; Gong, B.; Togashi, N.; Miyamoto, M.; Kiga, M.; Iwasaki, S.; Kamai, Y.; Tominaga, Y.; Takeda, Y.; Kagoshima, Y.; et al. The new-generation selective ROS1/NTRK inhibitor DS-6051b overcomes crizotinib resistant ROS1-G2032R mutation in preclinical models. Nat. Commun. 2019, 10, 3604.
- Zhou, C.; Fan, H.; Wang, Y.; Wu, H.; Yang, N.; Li, K.; Wang, X.; Qin, X.; Yu, Q.; Fang, Y.; et al. Taletrectinib (AB-106; DS-6051b) in metastatic non-small cell lung cancer (NSCLC) patients with ROS1 fusion: Preliminary results of TRUST. J. Clin. Oncol. 2021, 39 (Suppl. 15).
- Kazandjian, D.; Blumenthal, G.M.; Luo, L.; He, K.; Fran, I.; Lemery, S.; Pazdur, R. Benefit-Risk Summary of Crizotinib for the Treatment of Patients with ROS1 Alteration-Positive, Metastatic Non-Small Cell Lung Cancer. Oncologist 2016, 21, 974–980.
- Singh, H.; Brave, M.; Beaver, J.A.; Cheng, J.; Tang, S.; Zahalka, E.; Palmby, T.R.; Venugopal, R.; Song, P.; Liu, Q.I.; et al. Food and Drug Administration Approval: Cabozantinib for the Treatment of Advanced Renal Cell Carcinoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2017, 23, 330–335.
- Zou, H.Y.; Li, Q.; Lee, J.H.; Arango, M.E.; McDonnell, S.R.; Yamazaki, S.; Koudriakova, T.B.; Alton, G.; Cui, J.J.; Kung, P.P.; et al. An orally available small-molecule inhibitor of c-Met, PF-2341066, exhibits cytoreductive antitumor efficacy through antiproliferative and antiangiogenic mechanisms. Cancer Res. 2007, 67, 4408–4417.
- Bowles, D.W.; Kessler, E.R.; Jimeno, A. Multi-targeted tyrosine kinase inhibitors in clinical development: Focus on XL-184 (cabozantinib). Drugs Today 2011, 47, 857–868.
- Shamroe, C.L.; Comeau, J.M. Ponatinib: A new tyrosine kinase inhibitor for the treatment of chronic myeloid leukemia and Philadelphia chromosome-positive acute lymphoblastic leukemia. Ann. Pharmacother. 2013, 47, 1540–1546.
- O’Hare, T.; Shakespeare, W.C.; Zhu, X.; Eide, C.A.; Rivera, V.M.; Wang, F.; Adrian, L.T.; Zhou, T.; Huang, W.S.; Xu, Q.; et al. AP24534, a pan-BCR-ABL inhibitor for chronic myeloid leukemia, potently inhibits the T315I mutant and overcomes mutation-based resistance. Cancer Cell 2009, 16, 401–412.
- Karimi-Shah, B.A.; Chowdhury, B.A. Forced vital capacity in idiopathic pulmonary fibrosis--FDA review of pirfenidone and nintedanib. N. Engl. J. Med. 2015, 372, 1189–1191.
- Hilberg, F.; Roth, G.J.; Krssak, M.; Kautschitsch, S.; Sommergruber, W.; Tontsch-Grunt, U.; Garin-Chesa, P.; Bader, G.; Zoephel, A.; Quant, J.; et al. BIBF 1120: Triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res. 2008, 68, 4774–4782.
- Fuse, M.J.; Okada, K.; Oh-Hara, T.; Ogura, H.; Fujita, N.; Katayama, R. Mechanisms of Resistance to NTRK Inhibitors and Therapeutic Strategies in NTRK1-Rearranged Cancers. Mol. Cancer Ther. 2017, 16, 2130–2143.
- Russo, M.; Misale, S.; Wei, G.; Siravegna, G.; Crisafulli, G.; Lazzari, L.; Corti, G.; Rospo, G.; Novara, L.; Mussolin, B.; et al. Acquired Resistance to the TRK Inhibitor Entrectinib in Colorectal Cancer. Cancer Discov. 2016, 6, 36–44.
- Smeyne, R.J.; Klein, R.; Schnapp, A.; Long, L.K.; Bryant, S.; Lewin, A.; Lira, S.A.; Barbacid, M. Severe sensory and sympathetic neuropathies in mice carrying a disrupted Trk/NGF receptor gene. Nature 1994, 368, 246–249.
- Klein, R.; Smeyne, R.J.; Wurst, W.; Long, L.K.; Auerbach, B.A.; Joyner, A.L.; Barbacid, M. Targeted disruption of the trkB neurotrophin receptor gene results in nervous system lesions and neonatal death. Cell 1993, 75, 113–122.
- Klein, R.; Silos-Santiago, I.; Smeyne, R.J.; Lira, S.A.; Brambilla, R.; Bryant, S.; Zhang, L.; Snider, W.D.; Barbacid, M. Disruption of the neurotrophin-3 receptor gene trkC eliminates la muscle afferents and results in abnormal movements. Nature 1994, 368, 249–251.
- Farago, A.F.; Taylor, M.S.; Doebele, R.C.; Zhu, V.W.; Kummar, S.; Spira, A.I.; Boyle, T.A.; Haura, E.B.; Arcila, M.E.; Benayed, R.; et al. Clinicopathologic Features of Non-Small-Cell Lung Cancer Harboring an NTRK Gene Fusion. JCO Precis. Oncol. 2018, 2, 1–12.
- Wang, H.; Li, Z.W.; Ou, Q.; Wu, X.; Nagasaka, M.; Shao, Y.; Ou, S.I.; Yang, Y. NTRK fusion positive colorectal cancer is a unique subset of CRC with high TMB and microsatellite instability. Cancer Med. 2022, 11, 2541–2549.
- Zhao, X.; Kotch, C.; Fox, E.; Surrey, L.F.; Wertheim, G.B.; Baloch, Z.W.; Lin, F.; Pillai, V.; Luo, M.; Kreiger, P.A.; et al. NTRK Fusions Identified in Pediatric Tumors: The Frequency, Fusion Partners, and Clinical Outcome. JCO Precis. Oncol. 2021, 1, 204–214.
- Pietrantonio, F.; Di Nicolantonio, F.; Schrock, A.B.; Lee, J.; Tejpar, S.; Sartore-Bianchi, A.; Hechtman, J.F.; Christiansen, J.; Novara, L.; Tebbutt, N.; et al. ALK, ROS1, and NTRK Rearrangements in Metastatic Colorectal Cancer. J. Natl. Cancer Inst. 2017, 109, djx089.
- Garralda, E.; Hong, D.S.; Xu, R.; Deeken, J.; Italiano, A.; Liu, T.; Ferrandiz, A.; Patel, J.; Lee, D.; Chung, H.; et al. Long-term efficacy and safety of larotrectinib in patients with tropomyosin receptor kinase (TRK) fusion gastrointestinal (GI) cancer: An expanded dataset. Ann. Oncol. 2022, 33 (Suppl. 4), S370.
- Cohen, R.; Pudlarz, T.; Delattre, J.F.; Colle, R.; André, T. Molecular Targets for the Treatment of Metastatic Colorectal Cancer. Cancers 2020, 12, 2350.
- Forsythe, A.; Zhang, W.; Phillip Strauss, U.; Fellous, M.; Korei, M.; Keating, K. A systematic review and meta-analysis of neurotrophic tyrosine receptor kinase gene fusion frequencies in solid tumors. Ther. Adv. Med. Oncol. 2020, 12, 1758835920975613.
- LiVolsi, V.A.; Abrosimov, A.A.; Bogdanova, T.; Fadda, G.; Hunt, J.L.; Ito, M.; Rosai, J.; Thomas, G.A.; Williams, E.D. The Chernobyl thyroid cancer experience: Pathology. Clin. Oncol. 2011, 23, 261–267.
- Taylor, J.; Pavlick, D.; Yoshimi, A.; Marcelus, C.; Chung, S.S.; Hechtman, J.F.; Benayed, R.; Cocco, E.; Durham, B.H.; Bitner, L.; et al. Oncogenic TRK fusions are amenable to inhibition in hematologic malignancies. J. Clin. Investig. 2018, 128, 3819–3825.
- Qin, K.; Hou, H.; Liang, Y.; Zhang, X. Prognostic value of TP53 concurrent mutations for EGFR- TKIs and ALK-TKIs based targeted therapy in advanced non-small cell lung cancer: A meta-analysis. BMC Cancer 2020, 20, 328.
- Knezevich, S.R.; McFadden, D.E.; Tao, W.; Lim, J.F.; Sorensen, P.H. A novel ETV6-NTRK3 gene fusion in congenital fibrosarcoma. Nat. Genet. 1998, 18, 184–187.
- Agaram, N.P.; Zhang, L.; Sung, Y.S.; Chen, C.L.; Chung, C.T.; Antonescu, C.R.; Fletcher, C.D. Recurrent NTRK1 Gene Fusions Define a Novel Subset of Locally Aggressive Lipofibromatosis-like Neural Tumors. Am. J. Surg. Pathol. 2016, 40, 1407–1416.
- Kao, Y.C.; Fletcher, C.D.M.; Alaggio, R.; Wexler, L.; Zhang, L.; Sung, Y.S.; Orhan, D.; Chang, W.C.; Swanson, D.; Dickson, B.C.; et al. Recurrent BRAF Gene Fusions in a Subset of Pediatric Spindle Cell Sarcomas: Expanding the Genetic Spectrum of Tumors with Overlapping Features with Infantile Fibrosarcoma. Am. J. Surg. Pathol. 2018, 42, 28–38.
- Kiuru, M.; Jungbluth, A.; Kutzner, H.; Wiesner, T.; Busam, K.J. Spitz Tumors: Comparison of Histological Features in Relationship to Immunohistochemical Staining for ALK and NTRK1. Int. J. Surg. Pathol. 2016, 24, 200–206.
- Uguen, A. Spitz Tumors with NTRK1 Fusions: TRK-A and pan-TRK Immunohistochemistry as Ancillary Diagnostic Tools. Am. J. Surg. Pathol. 2019, 43, 1438–1439.
- Yeh, I.; Tee, M.K.; Botton, T.; Shain, A.H.; Sparatta, A.J.; Gagnon, A.; Vemula, S.S.; Garrido, M.C.; Nakamaru, K.; Isoyama, T.; et al. NTRK3 kinase fusions in Spitz tumours. J. Pathol. 2016, 240, 282–290.
- Silvertown, J.D.; Lisle, C.; Semenuk, L.; Knapp, C.; Jaynes, J.; Berg, D.; Kaul, N.; Lachapelle, J.; Richardson, L.; Speevak, M.; et al. Prevalence of NTRK Fusions in Canadian Solid Tumour Cancer Patients. Mol. Diagn. Ther. 2023, 27, 87–103.
More
