Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Axel Nierhaus and Version 2 by Lindsay Dong.
Various techniques for extracorporeal blood purification can decrease levels of elevated proinflammatory cytokines in septic shock, potentially mitigating the severity of the systemic inflammatory response. Some methods are effective in removing endotoxins, especially in sepsis caused by Gram-negative bacteria, which may aid in stabilizing the patient’s condition. Blood purification can enhance hemodynamic stability and reduce the need for vasopressors, crucial for managing septic shock. Techniques like continuous renal replacement therapy (CRRT) offer simultaneous management of acute kidney injury—a frequent complication in septic shock—alongside the removal of toxins and cytokines.
- sepsis
- septic hyperinflammation
- blood purification
- immune response
- cytokines
- endotoxi
1. Introduction
1.1. Septic Hyperinflammation
Sepsis is a life-threatening clinical condition with extensive physiological and biochemical abnormalities. Each year, approximately 49 million people worldwide are affected by sepsis, and it is estimated that 11 million deaths can be attributed to this syndrome. This accounts for up to 19.7% of all global deaths [1]. Although there appears to be a global decline in the average mortality rate, the current mortality rate for sepsis can still reach up to 25%. In the case of septic shock, which is a subset of sepsis characterized by profound circulatory, cellular, and metabolic disturbances, the hospital mortality rate approaches almost 60% [2].
Over recent decades, the definition of “sepsis” has continuously evolved, adapting to the expanding scope of knowledge. The current definition, established by the Third International Consensus (Sepsis-3), characterizes sepsis as “organ dysfunction caused by a dysregulated host response to infection” [3]. This definition notably emphasizes, for the first time, the critical role of both the innate and adaptive immune responses in the development of the clinical syndrome. Sepsis, unlike an uncomplicated and localized infection, involves a complex disruption of the finely tuned balance between pro- and anti-inflammatory processes. Although understanding of the development, pathophysiology, and immunological mechanisms of sepsis has advanced significantly over the last three decades, the syndrome’s complexity—with its myriad interactions and effects on various organs—means that the opportunities for successful and specific therapeutic interventions remain limited.
Even approaches within the realm of personalized or “precision medicine”, where treatments are tailored to predefined conditions or the specific needs of individual patients, have yet to achieve widespread success.
Sepsis necessitates timely and effective treatment strategies that vary across its continuum, from early sepsis to sepsis syndrome/severe sepsis and septic shock. The treatment modalities are multidisciplinary and escalate in intensity with the progression of the disease.
1.2. Immune Response Mechanisms in Sepsis: From Recognition to Regulation
Both adaptive and innate immunity rely on a variety of intracellular, membrane-bound, and soluble receptors. These include pattern recognition receptors (PRRs) which detect not only pathogen-associated molecular markers (PAMPs, e.g., endo- and exotoxins, DNA, lipids) from foreign invaders but also endogenous, host-derived danger signals (damage-associated molecular patterns, DAMPs). The recognition of PAMPs or DAMPs triggers a cascade aimed at both containing and destroying invasive pathogens, as well as repairing damaged tissue. The resulting upregulation of pro- and anti-inflammatory signaling pathways leads to a systemic release of cytokines, mediators, and pathogen-related molecules. This, in turn, activates coagulation and complement cascades, contributing to the immune response [4][7]. Toll-like receptors (TLRs), a subclass of PRRs, are located on the outer membranes as well as in intracellular vesicles of antigen-presenting cells (APCs) and monocytes [5][8]. Their interaction with PAMPs and DAMPs (e.g., extracellular LPS or intracellular nucleic acids) initiates signal transduction, which triggers a translocation of the nuclear factor-kappa-light-chain-enhancer of activated B cells (NF-κB) into the cell nucleus. This, in turn, leads to the expression of “early activation genes”. These genes include proinflammatory interleukins (IL), such as IL-1, IL-12, IL-18, along with tumor necrosis factor (TNF) and interferons (IFN). These proinflammatory substances then promote the activation of complement and coagulation pathways and stimulate the release of further cytokines (e.g., IL-6, IL-8, IFN-γ). This excessive and widespread increase in pro- and anti-inflammatory cytokines, resulting from the upregulation of both proinflammatory and anti-inflammatory signaling pathways, is a classic hallmark of sepsis. It leads to progressive tissue damage in the host and can ultimately escalate into multiorgan dysfunction. In the later stages of sepsis, the downregulation of activating cell surface molecules, increased apoptosis of immune cells, and T-cell exhaustion often result in emerging immunosuppression, a phenomenon known as “immune paralysis”. This condition renders affected patients susceptible to nosocomial infections, viral reactivation, and opportunistic pathogens [6][7][10,13].1.3. Neutrophils in Sepsis: Roles in Defense, Hyperinflammation, and Organ Damage
The immunological characterization of sepsis is complicated by its highly variable influence on the immunological phenotype, which can manifest itself either as hypo- or hyperreactive, or as a mixed form. Neutrophil granulocytes, a key component of the innate immune system, play a crucial role in the primary defense against pathogens. They contribute to hyperinflammation in sepsis through the release of proteases and reactive oxygen species. In response to severe bacterial infections, both mature and immature forms of neutrophils are released from the bone marrow in a process known as emergency granulopoiesis. However, when activated by interaction with PAMPs or DAMPs, immature neutrophils show reduced phagocytosis and limited oxidative burst capacity [8][9][10][14,15,16]. Neutrophil granulocytes are capable of releasing neutrophil extracellular traps (NETs) [11][17]. NETs are diffuse extracellular structures composed of decondensed chromatin with granular and nuclear proteins (histones) that can bind to endothelial or epithelial cells, potentially causing cell damage. This may promote the formation of intravascular thrombi and contribute to multiple organ damage [12][13][14][18,19,20]. NETs are also known for their ability to immobilize a wide range of pathogens. In addition to various cytokines like IL-8, IL-1β, and TNF, the release of NETs can also be triggered by platelet agonists such as adenosine diphosphate (ADP), arachidonic acid, collagen, thrombin, and some antibodies [11][12][15][16][17,18,23,24].1.4. Endothelial Dysfunction and Thromboinflammation in Hyperinflammatory Diseases
The endothelium, along with its protective layer of glycoprotein polysaccharides known as the glycocalyx, plays a significant role in the progression of diseases associated with hyperinflammation. Both are key targets in various mechanisms that perpetuate the inflammatory response [17][18][26,27]. In such conditions, endothelial cells may lose their antithrombotic properties. For example, the expression of surface-bound thrombomodulin can be reduced, leading to an increase in tissue factor (TF) expression. This, in combination with leukocytic microparticles and monocytes that also carry TF, triggers the activation of the coagulation cascade [19][28]. Feedback mechanisms consequently lead to progressive vascular hyperpermeability, increased recruitment of inflammatory cells, pronounced expression of adhesion molecules, and the release of additional cytokines. The binding of released TF to activated platelets and neutrophils, among others, further intensifies the prothrombotic situation. Simultaneously, the activity of antithrombotic factors, including antithrombin, the protein C system, and the tissue factor pathway inhibitor (TFPI), is reduced [20][30].1.5. Complement System Activation and Immunothrombosis in Sepsis and Systemic Inflammation
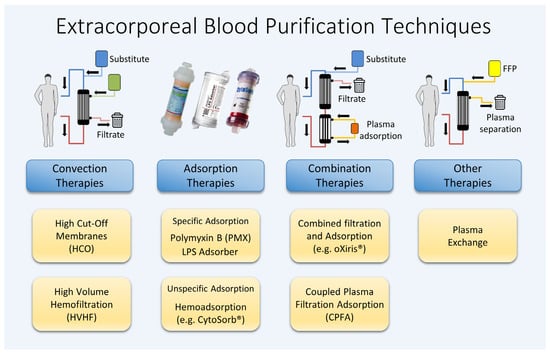
The complement system is a crucial component of innate immunity. In the initial phase of systemic hyperinflammation, elevated levels of activated complement factors such as the proinflammatory peptide fragments C3a, C4a, and C5a can be detected [16][24]. These anaphylatoxins, particularly C5a, intensify various responses, ranging from triggering apoptosis to the functional deactivation of neutrophils and the amplification of the hyperinflammatory response. C5a is known for its role in neutrophil chemotaxis; neutrophils, upon binding to the C5a receptor (C5aR), acquire the ability to migrate to and invade inflamed tissues. There, through the binding of PAMPs and DAMPs, they become activated and release granular enzymes, reactive oxygen species, and NETs [21][31]. Evolutionarily, the complement and coagulation systems share a common origin. The release of the proinflammatory complement factors C3a and C5a simultaneously not only leads to the recruitment but also the activation of platelets, endothelial cells, and leukocytes. Coagulation can be activated by coagulation factor XI, or, alternatively, through the cleavage of kininogen with release of bradykinin and antimicrobial peptides. Subsequent research suggests that, under certain conditions, thrombosis may play a significant physiological role in immune defense. Clinically, coagulopathy is a frequent complication of sepsis, and can be detected in up to one-third of critically ill patients. The International Society of Thrombosis and Haemostasis (ISTH) describes disseminated intravascular coagulopathy (DIC) as a syndrome “characterized by the intravascular activation of coagulation with loss of localization arising from different causes. It can originate from and cause damage to the microvasculature, which, if sufficiently severe, can produce organ dysfunction” [22][36]. The occurrence of DIC in sepsis is attributed to consumptive coagulopathy, driven by system-wide coagulation activation and accompanied by suppressed fibrinolysis. Alongside organ dysfunction due to systemic inflammation, decreased platelets, and increased PT-INR, the term “sepsis-induced coagulopathy (SIC)” has been introduced to describe this condition [23][37]. To summarize, many of the complex and diverse processes associated with septic hyperinflammation occur in the plasma. The various substances and messengers involved are systemically elevated and are present in a dissolved form, making them potential targets for treatment through blood purification. Despite numerous approaches being explored over the past decades, no single procedure nor combination of techniques has yet been identified that significantly improves the survival rates of patients with sepsis. Figure 1 illustrates a range of different techniques for extracorporeal blood purification, which are examined in this article for their application in treating sepsis and septic shock.
Figure 1.
Various extracorporeal blood purification methods available.
Figure 2 provides a schematic overview of the potential clearance properties of different blood purification methods based on the molecular weight of various mediators and toxins. Due to the large number of different commercially available membranes and technical settings, there is a wide variance in the actual effectiveness of the respective techniques.
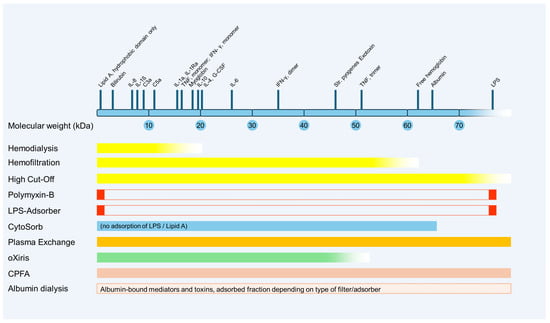

Figure 2. Schematic representation of potential clearance properties of blood purification methods based on the molecular weight of various mediators and toxins. CPFA, coupled plasma filtration adsorption; G-CSF, granulocyte-colony stimulating factor; IFN, interferon; IL, interleukin; kDa, kilodalton; LPS, lipopolysaccharide; TNF, tumor necrosis factor.
2. Renal Replacement Therapies (RRTs)
2.1. High-Cut-Off Membranes
Unlike standard high-flux membranes, high-cut-off (HCO) membranes feature increased pore size (20 nm instead of 10 nm), which theoretically allows for a more effective elimination of inflammatory mediators. The use of HCO membranes is similar to standard renal replacement therapy with a prescribed dose ranging between 25 and 40 mL/kg/h, as recommended by Kidney Disease Improving Global Outcomes (KDIGO). HCO membranes are employed in sepsis as well as in other conditions, such as acute kidney injury in the context of rhabdomyolysis or cast nephropathy in multiple myeloma. Initial studies on patients with sepsis-induced acute kidney injury predominantly indicated a more effective clearance of proinflammatory cytokines using HCO filters compared to classical high-flux filters. In a clinical trial involving 24 patients with sepsis-induced acute renal failure, Morgera et al. found that while the HCO membrane was effective in removing inflammatory mediators such as IL-1, IL-6, and TNF through convection, it also resulted in significant albumin loss compared to diffusion-based modalities [24][46]. Another study confirmed the higher sieving coefficient and mass removal rate of ultrafiltration for certain cytokines but failed to demonstrate a reduction in cytokine plasma levels in critically ill patients with acute kidney injury (AKI) within the first 72 h of therapy [25][47]. Following a small (n = 16) retrospective observational study that suggested a positive effect on mortality (37.5% mortality with HCO filter vs. 87.5% with continuous veno-venous hemodiafiltration, p = 0.03), these results were to be verified by the randomized High Cut-Off Sepsis Study (HICOSS) [26][48]. However, this trial was discontinued after a planned interim analysis showed no benefit in 28-day mortality (31% for the HCO group vs. 33% for the conventional group) or reductions in catecholamine use, days on mechanical ventilation, or duration of intensive care unit (ICU) stay. When diffusive modalities were used, albumin levels did not differ significantly. In summary, at present, there is no evidence supporting a positive effect of HCO filters in sepsis beyond established indications such as rhabdomyolysis.
2.2. High-Volume Hemofiltration
Continuous hemodialysis or hemodiafiltration with high filtration volume is likely the oldest method for extracorporeal removal of small molecules. Hemofiltration operates through convection, where dissolved substances are transported along with a solvent across a semipermeable membrane (ultrafiltration), driven by a positive transmembrane pressure gradient. The clearance in this process depends on the ultrafiltration rate, the sieving properties of the membrane for the solute, and the molecular size of the solute. According to the consensus definition, high-volume hemofiltration (HVHF) uses a convective target dose of more than 35 mL/kg/h, while a target dose of more than 45 mL/kg/h is classified as very-high-volume hemofiltration (VHVHF) [27][49]. As these methods do not require additional elements to be added to the standard circuit, they can be readily implemented as long as there is experience in the use of continuous renal replacement therapies. These techniques have been employed for immunomodulation in sepsis by aiming to eliminate inflammatory mediators through convection. Although most inflammatory molecules are medium-molecular substances and, in theory, can be removed by this technique, their endogenous release rate in sepsis is significantly higher compared to uremic toxins. Various studies have investigated the effects of different therapeutic regimens on outcome in sepsis and septic shock, using different target doses (HVHF and VHVHF) as well as comparing intermittent versus continuous usage [28][29][30][31][50,51,52,53]. Although a meta-analysis indicated lower mortality and improved hemodynamics, characterized by a lower heart rate and higher mean arterial pressure, it did not demonstrate a significant impact on disease severity or oxygenation index. Furthermore, most of the RCTs included in the meta-analysis were not of high quality, leading to questionable reliability of findings for various parameters (e.g., IL-6, mean arterial pressure) [32][54]. Therefore, the data available to date are insufficient for a conclusive assessment. Future studies should focus on exploring alternative extracorporeal therapies, rather than concentrating solely on HVHF as an adjunctive therapy for sepsis.
