Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Peter Tang and Version 1 by Naveed Ahmed Khan.
The unicellular eukaryotic Acanthamoeba spp. are ubiquitous free-living protists and can survive in various environments, such as water, soil and air. Although major strides have been made in developing and testing various anti-acanthamoebic drugs, recurrent infections, inadequate treatment outcomes, health complications, and side effects associated with the use of currently available drugs necessitate the development of more effective and safe therapeutic regimens. For any new anti-acanthamoebic drugs to be more effective, they must have either superior potency and safety or at least comparable potency and an improved safety profile compared to the existing drugs.
- Acanthamoeba
- combination therapy
- alternative treatment
- drug discovery
- natural products
- drug repurposing
- parasites
- Protozoa
- Pathogens
1. Introduction
The unicellular eukaryotic Acanthamoeba spp. are ubiquitous free-living protists and can survive in various environments, such as water, soil and air. During its life cycle (Figure 1), Acanthamoeba can undergo a phenotypic transition, under stressful conditions, from being an active trophozoite to a dormant cyst and vice versa [1]. The trophozoite is irregular in shape with pseudopods for locomotion and spike-like protrusions called acanthopodia [2]. The latter mediate the adhesion of Acanthamoeba to biological and inert surfaces [3]. The subcellular features of trophozoite include nucleus, mitochondria, endoplasmic reticulum (ER), Golgi complex, digestive vacuoles, and contractile vacuoles (for osmoregulation), which are enclosed by a thin plasma membrane [4]. The cyst stage, besides being round shaped, is enclosed by a distinctive thick wall, which is made mainly of cellulose [5,6][5][6].
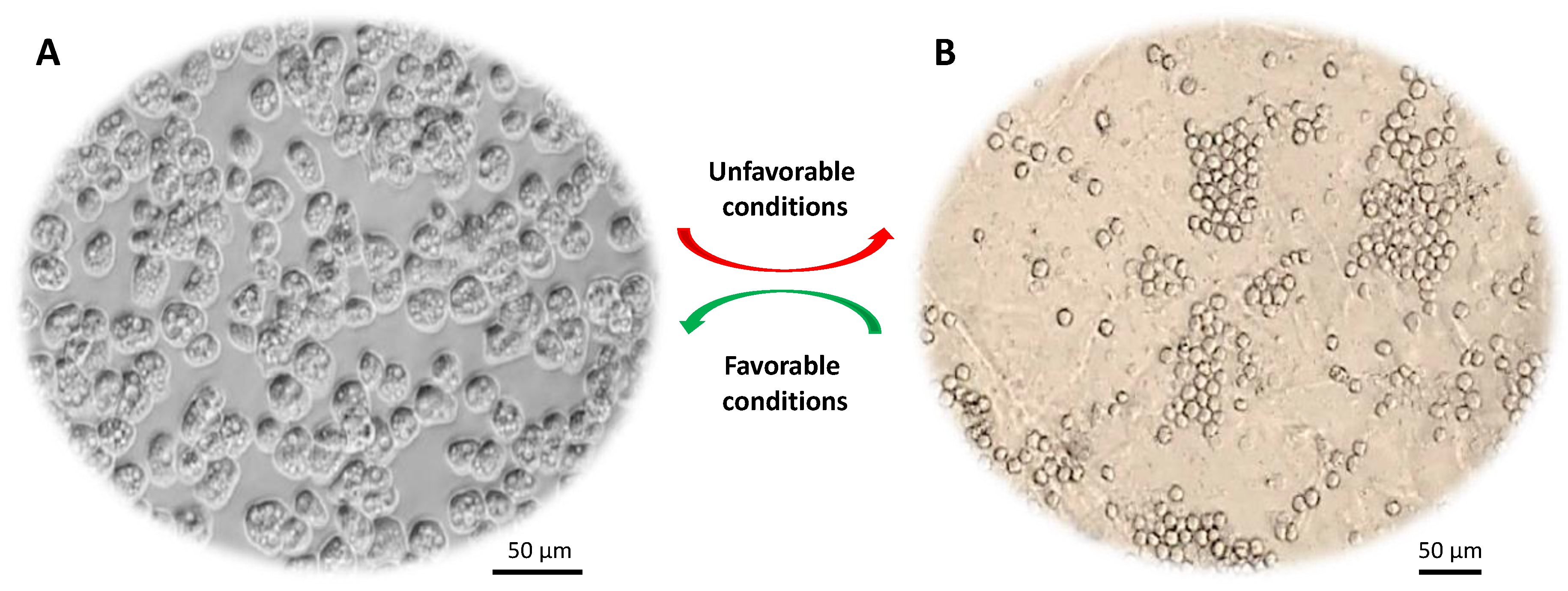
Figure 1. Life cycle of Acanthamoeba spp. (A) Trophozoite form that divides actively via binary fission. (B) Cyst form that represents the dormant stage. Under harsh conditions (e.g., food deprivation, extremes in pH, temperature and osmolarity) trophozoites transform into dormant cysts.
Acanthamoeba trophozoites (15–45 μm) are metabolically and reproductively active in the presence of appropriate environmental conditions, such as sufficient nutrients and favorable osmolarity, pH and temperature. However, this organism has a remarkable ability to transform into a dormant cyst stage (10–25 μm) under stressful conditions, such as adverse temperature, high osmolarity, high salination, extreme pH and scarcity of nutrients or drought [7]. Acanthamoeba cysts have the ability to persist in the environment for many years without losing their virulence and they can be airborne. During this encystation process, Acanthamoeba surrounds itself with a unique capsule that consists of two dense layers (inner endocyst and outer ectocyst). A recent study showed that cyst wall proteins are largely composed of three groups of cellulose binding lectins and that cyst wall formation is a well-orchestrated process whereby lectins bind with glycopolymers to form a well-developed cyst wall supported with an endocyst layer [8]. This protective cyst wall is what makes Acanthamoeba cysts tolerant to antibiotics and biocides including chlorination [7], and can survive under extreme physiological, radiological and chemical conditions. Once conditions become favorable, cysts switch back to their trophozoite form, a process known as excystation. Cysts are primarily responsible for prolonged treatment of Acanthamoeba infections. The majority of available drugs target functional aspects such as synthesis of DNA, RNA, protein, cell wall, or metabolic activity of the pathogen.
2. Repurposed Drugs
Recent studies have shown that existing drugs used clinically for other diseases may have the potential to target Acanthamoeba infections. With anti-acanthamoebic effects, they can progress to randomized, controlled clinical trials to evaluate their effectiveness against Acanthamoeba infections, which would save time and drug development costs. For example, three FDA-approved marketed drugs, amlodipine, loperamide, and prochlorperazine were shown to exhibit potent trophicidal effects [69][9]. Amlodipine is a dihydropyridine calcium channel blocker used in the treatment of hypertension and angina pectoris. Loperamide is a widely used antidiarrheal drug that acts primarily through activation of opioid receptors. Prochlorperazine, a drug of the same class as trifluoperazine, exhibited potent amoebicidal effects. Haloperidol and prochlorperazine act primarily as dopamine receptor blockers and have been used as anti-psychotic drugs. It was interesting to note that prochlorperazine showed potent trophicidal as well as cysticidal effects, while haloperidol was effective against trophozoites but not cysts. A combination of chlorpromazine and rokitamycin exhibited synergistic trophistatic , trophicidal, and cysticidal activities against A. castellanii suggesting their usefulness as chemotherapeutic agents against Acanthamoeba infections. The precise mode of action of prochlorperazine against Acanthamoeba is unclear but it may involve inhibition of amoeba calcium regulatory proteins, or lipophilic action on the amoeba plasma membrane. Prochlorperazine is thought to exert its anti-psychotic effects by blocking dopamine receptors but also has moderate anti-cholinergic and alpha-adrenergic receptor-blocking activity, as compared to haloperidol, which is a weak anti-cholinergic, muscarinic M1 (silent antagonist) at 10 µM [70][10]. Another anti-cholinergic agent, procyclidine, which is widely used as anti-parkinsonian agents because of its anti-cholinergic action showed trophicidal effects. Digoxin is a potent inhibitor of the active transport of sodium and potassium across cell membranes and showed trophicidal effects, possibly through a combination of lytic and apoptotic signaling induction. As there is limited availability of effective drugs to treat Acanthamoeba infections, clinically available drugs offer potential agents in managing AK and GAE. Repurposing drugs suggests the presence of a pharmacophore with microbicidal activity [71][11] providing an incentive for further investigation into compounds with similar structures. Exploring novel indications for existing drugs is an attractive short-term strategy offering major savings in development time and expense. Similarly, corifungin (a water-soluble polyene macrolide) and tigecycline (a third-generation tetracycline) were found to reduce Acanthamoeba growth (73% and 46% inhibition at 100 µM, respectively) by degenerating cytoplasm architecture and dysfunctioning the mitochondria of A. castellanii trophozoites [72,73,74][12][13][14]. Drugs that target G-protein coupled receptors (GPCRs) i.e., dopamine, muscarinic receptors, α- or β-adrenergic receptors and 5HT receptors have also been used due to their critical role in cellular signaling. Inhibition of β adrenergic receptor by propranolol affected A. castellanii growth, encystation and viability [75][15]. Combinations of prochlorperazine plus loperamide, prochlorperazine plus apomorphine and procyclidine plus loperamide were proved to be amoebicidal against A. castellanii [21][16]. Moreover, chloroquine, an anti-malarial drug, was able to inhibit autophagy, a type of programmed cell death (PCD), which was highly stimulated during encystation of Acanthamoeba, leading to reduction in the survival of A. castellanii [73][13]. Atorvastatin, fluvastatin, simvastatin and voriconazole could also induce PCD in A. castellanii [76][17].3. Improvements in Existing Drugs
The reformulation or analogues of drugs can improve compliance, pharmacodynamics and pharmacokinetics, making current medicines more clinically effective. High throughput screening of structural analogues of miltefosine such as heterocyclic alkylphosphocholines (APCs) [77][18] and oleylphosphocholine (OlPC) [78][19] demonstrated similar or stronger in vivo efficacy compared to miltefosine. It is worth noting that heterocyclic alkylphosphocholines possess ability to cross the BBB [79][20] and have potential in treating GAE due to Acanthamoeba and possibly other brain-infecting amoebae. Recently several studies have shown that the efficacy of marketed drugs can be enhanced by conjugation with metals such as gold or silver to synthesize drug-conjugated metal nanoparticles and can be repurposed as potential drugs for treating infections due to pathogenic free-living amoebae [80][21]. However, the use of metal nanoparticles can be associated with cytotoxicity [81][22] and side effects [82][23]. To develop a holistic approach to control AK certain measures should be included to augment pharmacotherapy, for example, by reducing incidence of eye infection by including new compounds with proven efficacies against trophozoites and cysts in cleaning solutions of contact lenses.4. siRNA-Based Therapeutics
Small interfering RNA molecules (siRNAs) have highlighted the benefits of existing statins against AK [86][24]. siRNA molecules can be synthesized artificially to silence or knockdown a particular mRNA. They could be used as a potent therapeutic option or a method for target validation in drug discovery. Specific targets investigated include extracellular serine protease [87][25], xylose isomerase [88][26], encystation-mediating serine proteinase (EMSP) [89][27], protein kinase C [90][28], cellulose synthase [91][29], protein arginine methyltransferase 1 [92][30], cysteine protease inhibitor (AcStefin) [93][31], and 3-Hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) [86][24]. It was found that after silencing the aforementioned targets by siRNA, encystation was prevented. It was also proposed that a combination of two gene-specific siRNAs (one targeting serine proteases and one targeting glycogen phosphorylase) could affect the growth rate and survival of amoeba [94][32]. Later, Zorzi et al. [95][33] designed siRNA-loaded liposomes for the successful treatment of a murine model of ocular keratitis caused by Acanthamoeba, further highlighting siRNAs as a promising future therapeutic approach.5. Alternative Therapeutics
The effectiveness of a murine monoclonal anti-idiotypic antibody and a synthetic killer mimotope (resembling a yeast toxin) in inhibiting and damaging Acanthamoeba growth on contact lenses has been shown [96][34]. Therefore, these biologics show clinical potential for development in order to prevent Acanthamoeba growth on contact lenses. These would consist of an A. castellanii-specific Fab portion that is specific for its surface, covalently linked to the A chain of the diphtheria toxin [97][35]. Similarly, photodynamic chemotherapy is a novel intervention that involves the use of Acanthamoeba-specific antibodies linked to photosensitizers like phthalocyanine (RLP068) or Hypocrellin B [53][36]. Photodynamic therapy may be advantageous over conventional methods. It is a treatment that involves the use of light-sensitive medication and a light source to destroy cells. Incubation of cysts with compounds and irradiation with 600–700nm light has been found to cause rapid and extensive damage, but its clinical validity is yet to be confirmed [98][37]. Photochemotherapeutic strategy was proposed to target Acanthamoeba infections. As mannose-binding protein is expressed on the surface membranes, photosensitizing compound porphyrin conjugated with mannose could achieve more specific drug targeting. Pre-treatment with this could reduce host cell cytotoxicity from 97% to 4.9% [99][38]. A recent study showed that Rose bengal photodynamic antimicrobial therapy can reduce parasitic load and diminished clinical severity of AK in a rabbit model [100][39].6. Natural Compounds
Traditionally many drugs with biological activity were discovered from natural compounds. Among a plethora of natural compounds tested, a few examples include Ipomoea spp., Kaempferia galanga, Cananga odorata [101][40], oakmoss (a natural fragrance ingredient) [102][41], a hexane fraction of Pterocaulon polystachyum (Asteraceae) [103][42], ethyl acetate extract of Limouni olive leaf [104][43], resveratrol and curcuminoids [105][44] were found to be amoebicidal. Of note, resveratrol is of high interest for further investigation because it can prevent amoeba binding to the human brain microvascular endothelial cells (hBMECs) and it is selective to A. castellanii, but not the hBMECs [105][44]. Interestingly, resveratrol could also act as a topoisomerase II inhibitor [106][45] that prevents DNA ligation and subsequently leads to cell apoptosis. Plant extracts from Rubus chamaemorus, Pueraria lobate and Solidago virgaurea could also be used in combination with other GAE drugs as they were found to extend the survival of Acanthamoeba-infected animals [107][46]. Plant-derived artemisinin and artesunate have caused 93% reduction in the trophozoite growth [108][47]. They exert their amoebistatic activity via induction of reactive oxygen species and lipid peroxidation, leading to oxidative stress and apoptosis [25][48]. Magainins, peptides produced by the skin of the African frog, had both trophistatic and trophicidal activities in vitro. Their anti-acanthamoebic effects are attributed to interruption of the ion conductance across the cell membrane [109][49]. A recent study showed that a number of natural compounds of plant or commercial origins (e.g., quercetin, kolavenic acid extracted from plant Polyalthia longifolia var pendula and crude plant methanolic extract of Caesalpinia pulcherrima) exhibited considerable suppression of the amoebae growth [110][50]. Conjugation of the plant-derived compounds (e.g., quercetin) with silver nanoparticles increased their anti-acanthamoebic effect, and reduced the encystation and excystation of A. castellanii, without exhibiting toxicity against human cells [110][50]. Taken collectively, this broad range of nutraceuticals display promising anti-acanthamoebic potential, making natural products interesting drug leads in the foreseeable future.7. Potential Targets for New Anti-Acanthamoeba Therapy
Many pharmacological targets have been discovered over the last few years [30][51]. For example, the myosin superfamily includes 18 different classes of motor proteins. Of the many classes that are expressed in Acanthamoeba, myosin-I and -II have been studied most extensively. The function of both myosins is based on the use of ATP hydrolysis to generate forces required for cellular functions. Acanthamoeba expresses three types of myosin-I subtypes, myosin-IA, -IB, and -IC. Myosin-IA functions in cytoplasmic vesicle transport, myosin-IB functions in pseudopod extension and phagocytosis, and myosin-IC is the only subtype that functions in contractile vacuole [25][48]. Acanthamoeba myosin-IC was of particular interest as it performs functions that human myosin-IC lacks and it is only 44% homologous to human myosin-IC [111][52]. The contractile vacuole is highly important in Acanthamoeba because it maintains homeostasis by regulating the amount of water within amoeba. It absorbs water by osmosis from the cytoplasm and moves to the surface of the amoeba and undergoes exocytosis when full. If myosin-IC is blocked, Acanthamoeba would be unable to regulate its internal water content and ultimately lead to cell lysis. Pentachloropseudilin (PCIP), a non-competitive, reversible myosin-IC inhibitor, is the only specific myosin-IC inhibitor developed and tested on Hela cells [112][53] but has not been tested on Acanthamoeba. It works by reducing the coupling between actin and nucleotide blinding sites [111][52]. Looking at the effects of PCIP analogues to inhibit or kill trophozoites might provide additional insight to current therapy. Moxifloxacin has shown limited efficacy against Acanthamoeba and exerts its effect by inhibition of DNA gyrase, a type II topoisomerase, and topoisomerase IV, which is required for DNA replication. Topoisomerase I inhibitors (e.g., camptothecin, irinotecan, topotecan) and topoisomerase II inhibitors (e.g., doxorubicin, amsacrine, etoposide) are generally used as anti-cancer drugs to induce apoptosis and death of cancer cells, and may have a potential activity against Acanthamoeba. High levels of elastase activity were found in A. culbertsoni [18][54]. Hence, it will be important to examine the potential of elastase inhibitors to inhibit Acanthamoeba elaborate elastase, a type of protease that degrades connective tissue proteins and causes cellular damage. The majority of the cyst wall structure is cellulose; thus, targeting cellulose biosynthesis can prevent encystment. The 2,6-dichlorobenzonitrile (DCB), a cellulose synthesis inhibitor, blocked Acanthamoeba encystment [113][55]. Using cellulase to the degrade cyst wall might make amoeba more susceptible to therapeutic compounds [114][56]. Alkaline phosphates have been identified in Acanthamoeba contractile vacuoles [115][57] and can be targeted by compounds, such as polyoxometalates. A. castellanii was found to have a novel complement of shikimate pathway enzymes [116][58]. Using (6S)-6-fluoroshikimic acid (antibacterial) and glyphosate (herbicide) can be a new approach to inhibit the shikimate pathway enzymes. Agents that affect membrane sterols, which are present in trophozoites and cysts, but absent from the host cells, have the potential to selectively suppress the amoeba growth. This assumption motivated Shing et al. [117][59] to examine the anti-acanthamoebic potential of the FDA-approved antifungal conazoles, which target sterol 14-demethylase (CYP51). Isavuconazole and posaconazole showed high efficacies against A. castellanii trophozoites. Additionally, isavuconazole damaged trophozoites within a day and suppressed excystation. Given the high safety of isavuconazole and its ability to block A. castellanii excystation, this drug was suggested as a cost-effective option for the treatment of primary and repeated AK. Reyes-Batlle et al., [118][60] identified new N-substituted quinolin-2(1H)-ones compounds with selective toxicity against trophozoites and cysts. The compounds’ toxicity was attributed to their ability to significantly lower the levels of ATP, without increasing the permeability of the cell membrane, leading to apoptosis and death of the amoeba. The promising potential of N-acyl substituted quinolin-2(1H)-ones suggests that these compounds may serve as a new scaffold for the identification of novel and better anti-acanthamoebic drugs.8. New Anti-Acanthamoebic Approaches
The application of riboflavin (B2) and concurrent ultraviolet light A (UVA) exposure to the cornea has shown potential as a new approach for AK therapy. UV irradiation of B2 produces free radicals that cause oxidation and cross-linking of the corneal collagen [119,120][61][62]. This prevents further tissue damage and parasitic reproduction by damaging their nucleic acid material [121][63]. Despite the potential, there has been no confirmation from clinical trials to incorporate this as a mainstay therapy. The use of drug-carriers improves the penetration of existing drugs into cystic forms in ocular or nasal drug administration. Current drugs; propamidine isethionate 0.1%, neomycin 1% or miconazole 1% lack cysticidal activity, however when combined with dimethyl-sulfoxide 30%, propamidine isethionate 0.1% exhibited better cysticidal activity. As dimethyl-sulfoxide itself has been used topically in the past, it can be considered clinically safe [122][64]. Furthermore, liposomal carriers of pentamidine isethionate improve the drug’s potency in vitro [123][65] and chitosan microspheres have been found to improve rokitamycin’s anti-amoebic activity and dissolution rate, providing a controlled-drug release [124][66]. More recently, drug conjugation with metals such as gold or silver to synthesize drug-conjugated metal nanoparticles have shown tremendous potential in the improved killing of parasites in vitro [80][21].9. Improved Drug Delivery to the Blood–Brain Barrier
Effective treatment depends heavily on the drug’s ability to cross the BBB as drug transport is often hampered by highly selective BBB. The majority of drugs that target the brain in clinical practice are lipid soluble small molecules (i.e., antibiotics) with molecular weight < 400 Da [125][67]. Among all the drugs that were used to treat GAE, none were delivered specifically to the CNS and limited research is done to improve their delivery to the BBB. Although a liposomal delivery is an available drug delivery option for amphotericin B (Ambisome) to enhance its lipid solubility, it was generally unable to cross the BBB [126][68]. In addition, despite the presence of rifampin and pyrimethamine in effective concentrations in the CSF (another gateway of drug transport to the brain), they transport poorly to the brain [125][67]. The development of improved drug delivery methods for current anti-acanthamoebic drugs is crucial to enhance the action of existing drugs until novel compounds with increased potency are available. Four main approaches are currently used to deliver drugs to the CNS [25][48]: i) transcranial drug delivery (injection in the cranium), ii) intranasal drug delivery that provide direct drug transport to the CSF, iii) transient modification of BBB i.e., increase BBB permeability using ultrasound or electromagnetic heating of nanoparticles [127][69] and iv) modification of physiochemical properties of drugs such as the lipidization of small molecules, lipid-based nanotherapeutics for the delivery of siRNA, and drug-conjugation with metals such as gold or silver to form drug-conjugated metal nanoparticles [128][70] and microspheres encapsulation to improve and prolong in vitro anti-amoebic activity. An example of microsphere was rokitamycin, an anti-acanthamoebic macrolide, loaded in microspheres showed better solubility, penetration, and enhancement [124][66].10. Theranostics as A Potential Strategy
The theranostic approaches, which combine therapeutic and diagnostic methods in one platform, have the potential to overcome conventional diagnostic and therapeutic limitations associated with the management of neglected diseases such as Acanthamoeba infections. Recent studies have proposed the theranostic strategy against infections due to pathogenic free-living amoebae [129][71]. It is hoped that such development can help expedite timely and sensitive diagnosis augmented with effective therapeutic capabilities. However, it requires development of smart materials for improved laboratory and point-of-care testing. Nanomaterials have already shown promising theranostic applications in non-communicable diseases and these can provide a breakthrough against Acanthamoeba infections.References
- Neff, R.J.; Neff, R.H. The biochemistry of amoebic encystment. Symp. Soc. Exp. Biol. 1969, 23, 51–81.
- Lorenzo-Morales, J.; Khan, N.A.; Walochnik, J. An update on Acanthamoeba keratitis: Diagnosis, pathogenesis and treatment. Parasite 2015, 22, 10.
- Niederkorn, J.Y.; Alizadeh, H.; Leher, H.; McCulley, J.P. The pathogenesis of Acanthamoeba keratitis. Microbes Infect. 1999, 1, 437–443.
- Bowers, B.; Korn, E.D. The fine structure of Acanthamoeba castellanii. I. the trophozoite. J. Cell Biol. 1968, 39, 95–111.
- Khan, N.A. Acanthamoeba: Biology and increasing importance in human health. FEMS Microbiol. Rev. 2006, 30, 564–595.
- Iovieno, A.; Gore, D.M.; Carnt, N.; Dart, J.K. Acanthamoeba sclerokeratitis: Epidemiology, clinical features, and treatment outcomes. Ophthalmology 2014, 121, 2340–2347.
- Bowers, B.; Korn, E.D. The fine structure of Acanthamoeba castellanii (Neff strain) II Encystment. J. Cell Biol. 1969, 41, 786–805.
- Magistrado-Coxen, P.; Aqeel, Y.; Lopez, A.; Haserick, J.R.; Urbanowicz, B.R.; Costello, C.E.; Samuelson, J. The most abundant cyst wall proteins of Acanthamoeba castellanii are lectins that bind cellulose and localize to distinct structures in developing and mature cyst walls. PLoS Negl. Trop. Dis. 2019, 13, e0007352.
- Baig, A.M.; Iqbal, J.; Khan, N.A. In vitro efficacies of clinically available drugs against growth and viability of an Acanthamoeba castellanii keratitis isolate belonging to the T4 genotype. Antimicrob. Agents Chemother. 2013, 57, 3561–3567.
- Kroeze, W.K.; Hufeisen, S.J.; Popadak, B.A.; Renock, S.M.; Steinberg, S.; Ernsberger, P.; Jayathilake, K.; Meltzer, H.Y.; Roth, B.L. H1-histamine receptor affinity predicts short-term weight gain for typical and atypical antipsychotic drugs. Neuropsychopharmacology 2003, 28, 519–526.
- Palit, P.P.; Mandal, S.C.; Mandal, N.B. Reuse of old existing, marketed non-antibiotic drugs as antimicrobial agents: A new emerging therapeutic approach In Microbial Pathogens and Strategies for Combating Them: Science, Technology and Education; Méndez-Vilas, A., Ed.; Formatex Research Center: Badajoz, Spain, 2013; Volume 3, pp. 1883–1892.
- Debnath, A.; Tunac, J.B.; Silva-Olivares, A.; Galindo-Gómez, S.; Shibayama, M.; McKerrow, J.H. In vitro efficacy of corifungin against Acanthamoeba castellanii trophozoites and cysts. Antimicrob. Agents Chemother. 2014, 58, 1523–1528.
- Jha, B.K.; Jung, H.J.; Seo, I.; Kim, H.A.; Suh, S.I.; Suh, M.H.; Baek, W.K. Chloroquine has a cytotoxic effect on Acanthamoeba encystation through modulation of autophagy. Antimicrob. Agents Chemother. 2014, 58, 6235–6241.
- Jha, B.K.; Seo, I.; Kong, H.H.; Suh, S.I.; Suh, M.H.; Baek, W.K. Tigecycline inhibits proliferation of Acanthamoeba castellanii. Parasitol. Res. 2015, 114, 1189–1195.
- Aqeel, Y.; Siddiqui, R.; Anwar, A.; Shah, M.R.; Khoja, S.; Khan, N.A. Photochemotherapeutic strategy against Acanthamoeba infections. Antimicrob. Agents Chemother. 2015, 59, 3031–3041.
- Kulsoom, H.; Baig, A.M.; Siddiqui, R.; Khan, N.A. Combined drug therapy in the management of granulomatous amoebic encephalitis due to Acanthamoeba spp., and Balamuthia mandrillaris. Exp. Parasitol. 2014, 145, S115–S120.
- Martín-Navarro, C.M.; López-Arencibia, A.; Sifaoui, I.; Reyes-Batlle, M.; Valladares, B.; Martínez-Carretero, E.; Piñero, J.E.; Maciver, S.K.; Lorenzo-Morales, J. Statins and voriconazole induce programmed cell death in Acanthamoeba castellanii. Antimicrob. Agents Chemother. 2015, 59, 2817–2824.
- Garajová, M.; Mrva, M.; Timko, L.; Lukáč, M.; Ondriska, F. Cytomorphological changes and susceptibility of clinical isolates of Acanthamoeba spp. to heterocyclic alkylphosphocholines. Exp. Parasitol. 2014, 145, S102–S110.
- Fortin, A.; Hendrickx, S.; Yardley, V.; Cos, P.; Jansen, H.; Maes, L. Efficacy and tolerability of oleylphosphocholine (OlPC) in a laboratory model of visceral leishmaniasis. J. Antimicrob. Chemother. 2012, 67, 2707–2712.
- Palusinska-Szysz, M.; Kania, M.; Turska-Szewczuk, A.; Danikiewicz, W.; Russa, R.; Fuchs, B. Identification of unusual phospholipid fatty acyl compositions of Acanthamoeba castellanii. PLoS ONE 2014, 9, e101243.
- Anwar, A.; Mungroo, M.R.; Anwar, A.; Sullivan, W.J., Jr.; Khan, N.A.; Siddiqui, R. Repositioning of Guanabenz in conjugation with gold and silver nanoparticles against pathogenic amoebae Acanthamoeba castellanii and Naegleria fowleri. ACS Infect. Dis. 2019, 5, 2039–2046.
- Kim, T.H.; Kim, M.; Park, H.S.; Shin, U.S.; Gong, M.S.; Kim, H.W. Size-dependent cellular toxicity of silver nanoparticles. J. Biomed. Mater. Res. A 2012, 100, 1033–1043.
- Greish, K.; Alqahtani, A.A.; Alotaibi, A.F.; Abdulla, A.M.; Bukelly, A.T.; Alsobyani, F.M.; Alharbi, G.H.; Alkiyumi, I.S.; Aldawish, M.M.; Alshahrani, T.F.; et al. The Effect of silver nanoparticles on learning, memory and social interaction in BALB/C mice. Int. J. Environ. Res. Public Health 2019, 16, 148.
- Martín-Navarro, C.M.; Lorenzo-Morales, J.; Machin, R.P.; López-Arencibia, A.; García-Castellano, J.M.; de Fuentes, I.; Loftus, B.; Maciver, S.K.; Valladares, B.; Piñero, J.E. Inhibition of 3-hydroxy-3-methylglutaryl-coenzyme A reductase and application of statins as a novel effective therapeutic approach against Acanthamoeba infections. Antimicrob. Agents Chemother. 2013, 57, 375–381.
- Lorenzo-Morales, J.; Ortega-Rivas, A.; Foronda, P.; Abreu-Acosta, N.; Ballart, D.; Martínez, E.; Valladares, B. RNA interference (RNAi) for the silencing of extracellular serine proteases genes in Acanthamoeba: Molecular analysis and effect on pathogenecity. Mol. Biochem. Parasitol. 2005, 144, 10–15.
- Aqeel, Y.; Siddiqui, R.; Khan, N.A. Silencing of xylose isomerase and cellulose synthase by siRNA inhibits encystation in Acanthamoeba castellanii. Parasitol. Res. 2013, 112, 1221–1227.
- Moon, E.K.; Chung, D.I.; Hong, Y.C.; Kong, H.H. Characterization of a serine proteinase mediating encystation of Acanthamoeba. Eukaryot Cell 2008, 7, 1513–1517.
- Moon, E.K.; Chung, D.I.; Hong, Y.; Kong, H.H. Protein kinase C signaling molecules regulate encystation of Acanthamoeba. Exp. Parasitol. 2012, 132, 524–529.
- Moon, E.K.; Hong, Y.; Chung, D.I.; Goo, Y.K.; Kong, H.H. Potential value of cellulose synthesis inhibitors combined with PHMB in the treatment of Acanthamoeba Keratitis. Cornea 2015, 34, 1593–1598.
- Moon, E.K.; Kong, H.H.; Hong, Y.; Lee, H.A.; Quan, F.S. Identification and characterization of protein arginine methyltransferase 1 in Acanthamoeba castellanii. Korean J. Parasitol. 2017, 55, 109–114.
- Lee, J.Y.; Song, S.M.; Moon, E.K.; Lee, Y.R.; Jha, B.K.; Danne, D.B.; Cha, H.J.; Yu, H.S.; Kong, H.H.; Chung, D.I.; et al. Cysteine protease inhibitor (AcStefin) is required for complete cyst formation of Acanthamoeba. Eukaryot. Cell 2013, 12, 567–574.
- Lorenzo-Morales, J.; Martín-Navarro, C.M.; López-Arencibia, A.; Santana-Morales, M.A.; Afonso-Lehmann, R.N.; Maciver, S.K.; Valladares, B.; Martínez-Carretero, E. Therapeutic potential of a combination of two gene-specific small interfering RNAs against clinical strains of Acanthamoeba. Antimicrob. Agents Chemother. 2010, 54, 5151–5155.
- Zorzi, G.K.; Schuh, R.S.; Maschio, V.J.; Brazil, N.T.; Rott, M.B.; Teixeira, H.F. Box Behnken design of siRNA-loaded liposomes for the treatment of a murine model of ocular keratitis caused by Acanthamoeba. Colloids Surf. B Biointerfaces 2019, 173, 725–732.
- Clarke, D.W.; Niederkorn, J.Y. The pathophysiology of Acanthamoeba keratitis. Trends Parasitol. 2006, 22, 175–180.
- Villemez, C.L.; Carlo, P.L. Preparation of an immunotoxin for Acanthamoeba castellanii. Biochem. Biophys. Res. Commun. 1984, 125, 25–29.
- Siddiqui, R.; Khan, N.A. Biology and pathogenesis of Acanthamoeba. Parasit. Vectors 2012, 5, 6.
- Ferro, S.; Coppellotti, O.; Roncucci, G.; Ben Amor, T.; Jori, G. Photosensitized inactivation of Acanthamoeba palestinensis in the cystic stage. J. Appl. Microbiol. 2006, 101, 206–212.
- Aqeel, Y.; Siddiqui, R.; Manan, Z.; Khan, N.A. The role of G protein coupled receptor-mediated signaling in the biological properties of Acanthamoeba castellanii of the T4 genotype. Microb. Pathog. 2015, 81, 22–27.
- Atalay, H.T.; Uysal, B.S.; Sarzhanov, F.; Usluca, S.; Yeşilırmak, N.; Özmen, M.C.; Erganiş, S.; Tefon, A.B.; Dogruman-Al, F.; Bilgihan, K. Rose Bengal-mediated photodynamic antimicrobial treatment of Acanthamoeba keratitis. Curr. Eye Res. 2020, 1–6.
- Chu, D.M.; Miles, H.; Toney, D.; Ngyuen, C.; Marciano-Cabral, F. Amebicidal activity of plant extracts from Southeast Asia on Acanthamoeba spp. Parasitol. Res. 1998, 84, 746–752.
- Nomura, H.; Isshiki, Y.; Sakuda, K.; Sakuma, K.; Kondo, S. Effects of oakmoss and its components on Acanthamoeba castellanii ATCC 30234 and the uptake of Legionella pneumophila JCM 7571 (ATCC 33152) into A. castellanii. Biocontrol Sci. 2015, 20, 59–65.
- Ródio, C.; da Rocha Vianna, D.; Kowalski, K.P.; Panatieri, L.F.; von Poser, G.; Rott, M.B. In vitro evaluation of the amebicidal activity of Pterocaulon polystachyum (Asteraceae) against trophozoites of Acanthamoeba castellanii. Parasitol. Res. 2008, 104, 191–194.
- Sifaoui, I.; López-Arencibia, A.; Ticona, J.C.; Martín-Navarro, C.M.; Reyes-Batlle, M.; Mejri, M.; Lorenzo-Morales, J.; Jiménez, A.I.; Valladares, B.; Lopez-Bazzocchi, I.; et al. Bioassay guided isolation and identification of anti-Acanthamoeba compounds from Tunisian olive leaf extracts. Exp. Parasitol. 2014, 145, S111–S114.
- Aqeel, Y.; Iqbal, J.; Siddiqui, R.; Gilani, A.H.; Khan, N.A. Anti-acanthamoebic properties of resveratrol and demethoxycurcumin. Exp. Parasitol. 2012, 132, 519–523.
- Leone, S.; Basso, E.; Polticelli, F.; Cozzi, R. Resveratrol acts as a topoisomerase II poison in human glioma cells. Int. J. Cancer 2012, 131, E173–E178.
- Derda, M.; Hadaś, E.; Thiem, B. Plant extracts as natural amoebicidal agents. Parasitol. Res. 2009, 104, 705–708.
- Nacapunchai, D.; Phadungkul, K.; Kaewcharus, S. In vitro effect of artesunate against Acanthamoeba spp. Southeast Asian J. Trop. Med. Public Health 2002, 33 (Suppl. 3), 49–52.
- Khan, N.A. Acanthamoeba Biology and Pathogenesis; Caister Academic Press: Norfolk, UK, 2009.
- Schuster, F.L.; Jacob, L.S. Effects of magainins on amoeba and cyst stages of Acanthamoeba polyphaga. Antimicrob. Agents Chemother. 1992, 36, 1263–1271.
- Anwar, A.; Ting, E.L.S.; Anwar, A.; Ain, N.U.; Faizi, S.; Shah, M.R.; Khan, N.A.; Siddiqui, R. Antiamoebic activity of plant-based natural products and their conjugated silver nanoparticles against Acanthamoeba castellanii (ATCC 50492). AMB Expr. 2020, 10, 24.
- Roberts, C.W.; Henriquez, F.L. Drug identification, validation, characterisation and exploitation for treatment of Acanthamoeba (species) infections. Exp. Parasitol. 2010, 126, 91–96.
- Martín-Navarro, C.M.; Lorenzo-Morales, J.; López-Arencibia, A.; Reyes-Batlle, M.; Piñero, J.E.; Valladares, B.; Maciver, S.K. Evaluation of Acanthamoeba myosin-IC as a potential therapeutic target. Antimicrob. Agents Chemother. 2014, 58, 2150–2155.
- Chinthalapudi, K.; Taft, M.H.; Martin, R.; Heissler, S.M.; Preller, M.; Hartmann, F.K.; Brandstaetter, H.; Kendrick-Jones, J.; Tsiavaliaris, G.; Gutzeit, H.O.; et al. Mechanism and specificity of pentachloropseudilin-mediated inhibition of myosin motor activity. J. Biol. Chem. 2011, 286, 29700–29708.
- Marciano-Cabral, F.; Cabral, G. Acanthamoeba spp. as agents of disease in humans. Clin. Microbiol. Rev. 2003, 16, 273–307.
- Dudley, R.; Alsam, S.; Khan, N.A. Cellulose biosynthesis pathway is a potential target in the improved treatment of Acanthamoeba keratitis. Appl. Microbiol. Biotechnol. 2007, 75, 133–140.
- Lakhundi, S.; Siddiqui, R.; Khan, N. Cellulose degradation: A therapeutic strategy in the improved treatment of Acanthamoeba infections. Parasit. Vectors 2015, 8, 23.
- Raza, R.; Matin, S.; Sarwar, M.; Barsukova-Stuckart, M.; Ibrahim, M.; Kortz, U.; Iqbal, J. Polyoxometalates as potent and selective inhibitors of alkaline phosphatases with profound anticancer and amoebicidal activities. Dalton Trans. 2012, 41, 14329–14336.
- Henriquez, F.L.; Campbell, S.J.; Sundararaj, B.K.; Cano, A.; Muench, S.P.; Roberts, C.W. The Acanthamoeba shikimate pathway has a unique molecular arrangement and is essential for aromatic amino acid biosynthesis. Protist 2015, 166, 93–105.
- Shing, B.; Singh, S.; Podust, L.M.; McKerrow, J.H.; Debnath, A. The antifungal drug isavuconazole is both amebicidal and cysticidal against Acanthamoeba castellanii. Antimicrob. Agents Chemother. 2020, 64.
- Reyes-Batlle, M.; Freijo, M.B.; López-Arencibia, A.; Lorenzo-Morales, J.; McNaughton-Smith, G.; Piñero, J.E.; Abad-Grillo, T. Identification of N-acyl quinolin-2(1H)-ones as new selective agents against clinical isolates of Acanthamoeba keratitis. Bioorg. Chem. 2020, 99, 103791.
- Khan, Y.A.; Kashiwabuchi, R.T.; Martins, S.A.; Castro-Combs, J.M.; Kalyani, S.; Stanley, P.; Flikier, D.; Behrens, A. Riboflavin and ultraviolet light a therapy as an adjuvant treatment for medically refractive Acanthamoeba keratitis: Report of 3 cases. Ophthalmology 2011, 118, 324–331.
- Spoerl, E.; Huhle, M.; Seiler, T. Induction of cross-links in corneal tissue. Exp. Eye Res. 1998, 66, 97–103.
- Joshi, P.C. Comparison of the DNA-damaging property of photosensitised riboflavin via singlet oxygen (1O2) and superoxide radical (O2-) mechanism. Toxicol. Lett. 1985, 26, 211–217.
- Saunders, P.P.; Proctor, E.M.; Rollins, D.F.; Richards, J.S. Enhanced killing of Acanthamoeba cysts in vitro using dimethylsulfoxide. Ophthalmology 1992, 99, 1197–1200.
- Siddiqui, R.; Syed, A.; Tomas, S.; Prieto-Garcia, J.; Khan, N.A. Effect of free versus liposomal-complexed pentamidine isethionate on biological characteristics of Acanthamoeba castellanii in vitro. J. Med. Microbiol. 2009, 58, 327–330.
- Rassu, G.; Gavini, E.; Jonassen, H.; Zambito, Y.; Fogli, S.; Breschi, M.C.; Giunchedi, P. New chitosan derivatives for the preparation of rokitamycin loaded microspheres designed for ocular or nasal administration. J. Pharm. Sci. 2009, 98, 4852–4865.
- Pardridge, W.M. Drug transport across the blood-brain barrier. J. Cereb. Blood Flow Metab. 2012, 32, 1959–1972.
- Takemoto, K.; Yamamoto, Y.; Ueda, Y. Influence of the progression of cryptococcal meningitis on brain penetration and efficacy of AmBisome in a murine model. Chemotherapy 2006, 52, 271–278.
- Tabatabaei, S.N.; Girouard, H.; Carret, A.S.; Martel, S. Remote control of the permeability of the blood-brain barrier by magnetic heating of nanoparticles: A proof of concept for brain drug delivery. J. Control. Release 2015, 206, 49–57.
- Schroeder, A.; Levins, C.G.; Cortez, C.; Langer, R.; Anderson, D.G. Lipid-based nanotherapeutics for siRNA delivery. J. Intern. Med. 2010, 267, 9–21.
- Anwar, A.; Siddiqui, R.; Khan, N.A. Importance of theranostics in rare brain-eating amoebae infections. ACS Chem. Neurosci. 2019, 10, 6–12.
More
