Dairy wastewater (DWW) has a high fat content along with a high chemical oxygen demand (COD), which makes it problematic to dispose of. Biological treatment processes have shown great success in the remediation of this water. However, these are not without their shortfalls. A variety of biological processes have been listed here as well as suggestions to improve their effectiveness. To improve the treatment efficiency, there are two main lines of thinking: First, to optimise the process through the manipulation of the physical parameters of the systems (e.g., temperature, pH, hydraulic retention time (HRT), agitation, etc.). Second, to add either pre-treatment or post-treatment processes to the main process to increase the effectiveness of the entire process overall.
- dairy
- wastewater
- biological
- optimization
- treatment
1. Introduction
The disposal of wastewater is a topic of great importance as improper disposal may lead to a detrimental effect on the lives of people as well as the degradation of the environment. Many laws and regulations have been put in place to ensure that the wastewater being released from various industrial processes meets a minimum standard of safety [6][1]. Different industries release varying quantities of wastewater with different types of contamination. The release of dairy wastewater (DWW) into water systems without proper treatment causes an array of environmental issues due to its high organic content. The decomposition of DWW in a water source causes the dissolved oxygen (O2) inside the water body to drop as the microbes (especially algae) flourish in the nutrient-rich environment. This results in a hypoxic environment which is unsuitable for aquatic animal and plant life. The increased algal growth produces elevated levels of CO2 which lowers the pH of the body of water, which in turn slows the growth of fish and shellfish. The addition of DWW into fresh water also provides a breeding ground for various insects, which can upset the balance of the surrounding ecosystem. The degradation of casein also causes the formation of a sludge which is harmful to aquatic organisms [7][2]. This impacts the health of aquatic life and the potential for fishing which has a negative economic impact. In addition to these environmental implications, the processing of dairy products and the release of DWW can lead to several social impacts. These stem from two main sources, namely, the discharge of untreated or partially treated wastewater into nearby water bodies and the odour produced by the plant and effluents. The contamination caused by the release of DWW can promote the spread of bacteria such as Salmonella and E. coli, which can cause illness in humans and animals which use the water sources. Once contracted, these illnesses can easily be spread throughout the community.
2. Treatment Options
2.1. Aerobic Treatment Processes
Aerobic treatment occurs when the microbes are exposed to air and thus oxygen whilst anaerobic occurs without the presence of air. Aerobic treatment is effectively used to reduce the BOD5 as well as the phosphorous and nitrogen content in dairy wastewater. Aerobic processes are additionally more cost-effective than anaerobic ones for the removal of fats from DWW [21][3]. Importantly, the removal of nitrogen is largely associated with the oxidation of ammonia into nitrates, which in turn reduces the odour of the DWW. Aerobic systems are associated with high biomass growth and are susceptible to clogging if solely used to treat DWW [5][4].2.2. Activated Sludge
Activated sludge treatment methods are processes which introduce microbes into the wastewater stream to both treat the water and stimulate the growth of microbes. The microbial colonies are then separated from the bulk stream using a clarifier or filter, with a portion of the sludge being reintroduced to the reactor. A schematic representation of an activated sludge system can be seen in Figure 1. These methods are preferred for the removal of carbon, nitrates, and ammonia [4][5]. The constant recirculation of sludge allows for the microbes to acclimatize to the waste stream composition and increases the performance of the system, the longer it is operated for. These systems are also effective at breaking down complicated compounds such as lactose, FOG, and proteins [29][6].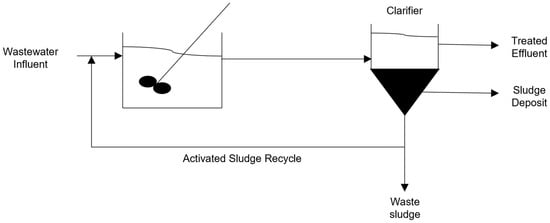
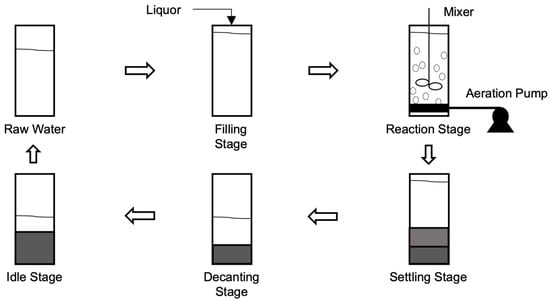
2.3. Sequencing Batch Reactors
2.3. Sequencing Batch Reactors
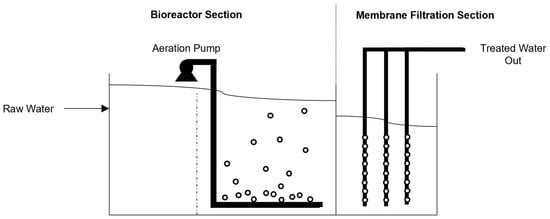
2.4. Membrane Bioreactor
2.5. Anaerobic Processes
2.6. Anaerobic Filters
2.6. Anaerobic Filters
Anaerobic filters (AFs) are reactors that are packed with a porous support media with biomass in the packing void. AF reactors contain at least two filtration chambers within the system, with the filter media used to prevent biomass from passing through. A side effect of these filtration chambers is enhanced solid removal, which can be observed [7][2]. A schematic representation of an AF system can be seen in Figure 4.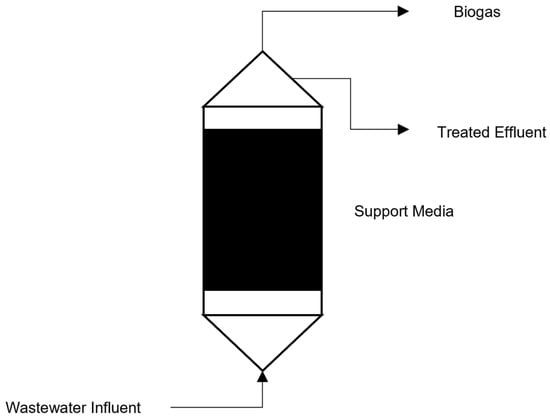
2.7. Anaerobic Packed Bed Reactors
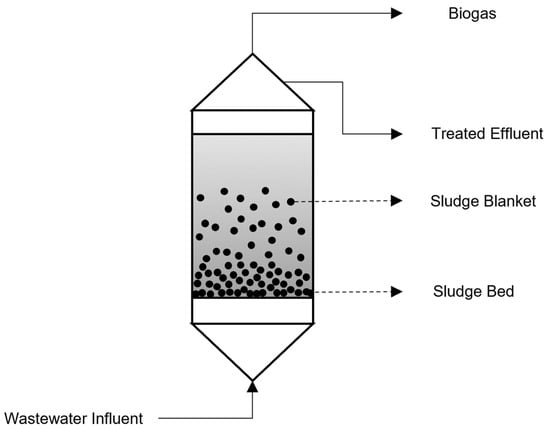
2.8. Up-Flow Anaerobic Sludge Blanket Reactors
2.9. Aerobic–Anaerobic Systems
3. The Effects of Experimental Parameters on the Performance of Biological Treatment Systems
3.1. pH Adjustment
3.2. Dissolved O
2
Levels
Aerobic digestion is enhanced by higher levels of dissolved O2; it therefore follows that anaerobic digestion will be enhanced by the removal of dissolved O2 from the wastewater. One straightforward approach to eliminate dissolved oxygen is to pass another gas through the wastewater. Some potential deoxygenating agents for anaerobic digestion are nitrogen or biogas. Biogas is a convenient option as an amount of CO2 and methane is produced by anaerobic digestion. However, the use of biogas poses an environmental issue due to methane being a greenhouse gas. Nitrogen gas is a suitable gas to use as it is non-reactive and does not pose any inherent environmental risks [39][15].3.3. Hydraulic Retention Time
Hydraulic retention time (HRT) refers to the average time that a liquid substrate will spend within a reactor. HRT is largely dependent on the type of reactor being used and the organic load of the wastewater being treated. A small HRT can sometimes result in high biomass washout, whereas a long HRT can require large reactor volumes [39][15]. The growth of the various groups of microorganisms used in AD are favoured by different HRTs. Methanogens prefer a longer HRT period as opposed to acidogenic bacteria, which prefer low HRTs. The determination of optimal hydraulic retention time (HRT) is a complex decision that requires the consideration of both the process and the desired outcomes, and it should be evaluated on a case-by-case basis.3.4. Aeration and Agitation
An important factor in aerobic treatments is the aeration which is introduced into the system. It is the driving force behind the reactions which aerobic digestion is utilized for, namely, the oxidation of ammonia and the breakdown of complex organic compounds. Therefore, an optimal aeration regime is essential for the performance of any treatment method used. At lower aeration levels, it was observed that there was a significant decrease in COD removal efficiency and an even more drastic decrease in the efficiency of ammonia removal [40][16]. This indicates that the available oxygen is being used to oxidise the carbon substrates in the wastewater as opposed to the ammonia. The most likely explanation is that ammonia requires a larger amount of O2 when compared to the organic components.3.5. Temperature
The temperature plays a critical role in the reactions involved in wastewater remediation. Different temperature ranges can have both favourable and unfavourable effects on the performance of a bioreactor. High temperatures can cause the denaturation of proteins within the microbes, causing them to lose their enzymatic activity and die. Low temperatures can cause the microbes to become dormant, inactive, or die. It is, therefore, essential that the temperatures within a bioreactor are carefully controlled and are not allowed to go beyond the range within which the microbes are able to thrive. However, choosing an optimal temperature for which to run a system at depends on the specific microorganisms used within it. Most anaerobic digesters used for wastewater treatment typically operate at mesophilic conditions, between 35 °C and 37 °C. Temperature needs to be carefully controlled as large fluctuations can be detrimental for these processes [44][17].4. Accessory Treatment Options
4.1. Hydrolysis
Anaerobic systems generally have difficulty in degrading FOGs and ammonia, as these are oxidation reactions which occur. High concentrations of FOGs can contribute to clogging within the reactor as they are poorly broken down through AD. An effective solution to these problems is a hydrolysis stage before the anaerobic digestor. An up-flow ASB reactor (UASB) was observed to be able to effectively treat wastewaters with high FOG contents which had been hydrolysed before treatment. It was noted that when fed unhydrolyzed wastewater, the UASB showed unstable COD removal and a tendency to accumulate some of the fats within the sludge [45][18]. This suggests that the inclusion of a separate hydrolysis stage can mitigate the risk of clogging within the reactor.4.2. Coagulation
Coagulation/Flocculation are currently the most commonly used processes for the removal of suspended and dissolved solids, colloids, and organic components in industrial wastewater [47][19]. There are two types of coagulants commonly used in wastewater treatment, namely, inorganic and organic coagulants. Inorganic coagulants are usually metal-based salts, usually containing aluminium or iron. The use of Alum (X × Al(SO4)2 × 12H2O, where X is a metal ion such as potassium or sodium) as a coagulant in the treatment of DWW was observed to reduce the turbidity of the water by 95% and reduce COD by 68% [48][20]. This was further enhanced by the addition of polymeric coagulants, resulting in a reduction in COD by 85%. However, inorganic coagulants produce large amounts of metal-rich floc, which must be further treated before it is disposed of. They can also alter the pH of the water requiring pH control and corrosion-resistant equipment. When trying to integrate inorganic coagulants with biological systems, it is important to evaluate whether the microbial colonies will be able to function unhindered. Organic coagulants can either be polymeric or natural coagulants. Polymeric coagulants produce longer polymer chains without any metals or hydroxides and produce smaller volumes of floc. They also do not impact the pH of the water. They also produce low-density floc, which does not always settle well. Natural coagulants are being investigated widely and there have been many studies which show their effectiveness in the treatment of wastewater. An extract made from the bark of Guazuma ulmifolia was used as a coagulant for DWW (3037 mgCOD/L & 1283 mgBOD5/L) and was observed to remove 95.8% of the turbidity, 76% of the COD and 81.2% of the BOD5 [49][21]. Natural coagulants’ low cost and eco-friendliness make them a suitable alternative to the more commonly used synthetic coagulants.4.3. Membrane Filtration
Membrane filtration methods such as microfiltration are effective at significantly reducing the TSS within the wastewater. However, it has little effect on the TN, COD, and BOD5 content and is commonly used as a pre-treatment step in a wastewater treatment process. Reverse osmosis (RO) is a viable option for dairy wastewater treatment and has been observed to reduce the TN and TOC by 94% and 84%, respectively [50][22]. Nanofiltration has been observed to be effective at reducing COD and TSS levels but does not remove all ions of interest, such as phosphates and nitrates, from the wastewater stream [51][23]. There have been some applications of nanofiltration and RO being used in conjunction with a bioreactor, which was observed to greatly improve the overall ion removal in addition to enhancing the reduction in COD and TSS. There are issues associated with RO and membrane filtration, which affect the long-term uses of this type of system. RO is an extremely energy-intensive process and is sometimes impractical to operate at the specifications required [52][24]. The replacement of this technology can be costly in the event of physical damage or fouling. In addition, this process produces a highly concentrated retentate which needs to be disposed of. This is a major concern as the disposal of the concentrate that is formed is more problematic than that of the wastewater itself.References
- Molewa, B.E. Revision of General Authorisations in Terms of Section 39 of the National Water Act, 1998 (Act No. 36 of 1998) (the Act). In Government Gazette No. 36820; Government Gazette of South Africa: Pretoria, South Africa, 2013.
- Joshiba, G.J.; Kumar, P.S.; Femina, C.C.; Jayashree, E.; Racchana, R.; Sivanesan, S. Critical review on biological treatment strategies of dairy wastewater. Desalination Water Treat. 2019, 160, 94–109.
- Zhao, K.; Wu, Y.W.; Young, S.; Chen, X.J. Biological Treatment of Dairy Wastewater: A Mini Review. J. Environ. Inform. Lett. 2020, 4, 22–31.
- Van Heerdan, B. Suid-Afrikaanse Melkprosesseer South African Milk Processors’ Organisation dersorganisasie A Milk SA publication compiled by the Milk Producers’ Organisation. 2022. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwiu5OLrtNWBAxXTWEEAHaPiAYYQFnoECBQQAQ&url=https%3A%2F%2Fmilksa.co.za%2Fsites%2Fdefault%2Ffiles%2F2018-09%2FLAC004%2520Nov%25202017.pdf&usg=AOvVaw07hNM16NuvQXnHrUDjHDjB&opi=89978449 (accessed on 10 July 2023).
- Water Access in South Africa|Water for All. Available online: http://www.12.000.scripts.mit.edu/mission2017/case-studies/water-access-in-south-africa/ (accessed on 2 May 2023).
- Bae, T.-H.; Han, S.-S.; Tak, T.-M. Membrane sequencing batch reactor system for the treatment of dairy industry wastewater. Process. Biochem. 2003, 39, 221–231.
- Slavov, A.K. General characteristics and treatment possibilities of dairy wastewater—A review. Food Technol. Biotechnol. 2017, 55, 14–28.
- Goli, A.; Shamiri, A.; Khosroyar, S.; Talaiekhozani, A.; Sanaye, R.; Azizi, K. A review on different aerobic and anaerobic treatment methods in dairy industry wastewater. J. Environ. Treat. Tech. 2019, 7, 113–141.
- Meyo, H.B.; Njoya, M.; Basitere, M.; Ntwampe, S.K.O.; Kaskote, E. Treatment of poultry slaughterhouse wastewater (Psw) using a pretreatment stage, an expanded granular sludge bed reactor (egsb), and a membrane bioreactor (mbr). Membranes 2021, 11, 345.
- Bal, A.S.; Dhagat, N.N. Upflow anaerobic sludge blanket reactor—A review. Indian J. Environ. Health 2001, 43, 1–82.
- Hanrahan, G. Aqueous Chemistry. In Key Concepts in Environmental Chemistry; Academic Press: Cambridge, MA, USA, 2012; pp. 73–106.
- Pascale, N.C.; Chastinet, J.J.; Bila, D.M.; Sant̀anna, G.L.; Quitério, S.L.; Vendramel, S.M.R. Enzymatic hydrolysis of floatable fatty wastes from dairy and meat food-processing industries and further anaerobic digestion. Water Sci. Technol. 2019, 79, 985–992.
- Omil, F.; Garrido, J.M.; Arrojo, B.; Méndez, R. Anaerobic filter reactor performance for the treatment of complex dairy wastewater at industrial scale. Water Res. 2003, 37, 4099–4108.
- Demirer, G.; Chen, S. Effect of retention time and organic loading rate on anaerobic acidification and biogasification of dairy manure. J. Chem. Technol. Biotechnol. 2004, 79, 1381–1387.
- Bella, K.; Rao, P.V. Anaerobic digestion of dairy wastewater: Effect of different parameters and co-digestion options—A review. Biomass-Convers. Biorefinery 2021, 13, 2527–2552.
- Tocchi, C.; Federici, E.; Fidati, L.; Manzi, R.; Vincigurerra, V.; Petruccioli, M. Aerobic treatment of dairy wastewater in an industrial three-reactor plant: Effect of aeration regime on performances and on protozoan and bacterial communities. Water Res. 2012, 46, 3334–3344.
- Goff, H.D.; Hill, A.; Ferrer, M.A. Milk Lipids-Chemical Properties; Physical Properties; Structure and Fat Globules; Func-tional Properties. In Dairy Science and Technology eBook; CRC Press: Boca Raton, FL, USA, 2005.
- Leal, M.C.; Freire, D.M.; Cammarota, M.C.; Sant’anna, G.L. Effect of enzymatic hydrolysis on anaerobic treatment of dairy wastewater. Process. Biochem. 2006, 41, 1173–1178.
- Mateus, G.A.P.; Formentini-Schmitt, D.M.; Nishi, L.; Fagundes-Klen, M.R.; Gomes, R.G.; Bergamasco, R. Coagulation/Flocculation with Moringa oleifera and Membrane Filtration for Dairy Wastewater Treatment. Water Air Soil Pollut. 2017, 228, 342.
- Loloei, M.; Nekonam, G.; Alidadi, H.; Kor, Y. Study of the coagulation process in wastewater treatment of dairy industries. Int. J. Environ. Health Sci. Eng. 2014, 3, 12.
- Muniz, G.L.; Da Silva, T.C.F.; Borges, A.C. Assessment and optimization of the use of a novel natural coagulant (Guazuma ulmifolia) for dairy wastewater treatment. Sci. Total. Environ. 2020, 744, 140864.
- Bortoluzzi, A.C.; Faitão, J.A.; Di Luccio, M.; Dallago, R.M.; Steffens, J.; Zabot, G.L.; Tres, M.V. Dairy wastewater treatment using integrated membrane systems. J. Environ. Chem. Eng. 2017, 5, 4819–4827.
- Andrade, L.; Mendes, F.; Espindola, J.; Amaral, M. Nanofiltration as tertiary treatment for the reuse of dairy wastewater treated by membrane bioreactor. Sep. Purif. Technol. 2014, 126, 21–29.
- Zhao, S.; Liao, Z.; Fane, A.; Li, J.; Tang, C.; Zheng, C.; Lin, J.; Kong, L. Engineering antifouling reverse osmosis membranes: A review. Desalination 2021, 499, 114857.
