Resting-state functional magnetic resonance imaging (fMRI) and structural MRI investigations offer complementary insights into the distinctive frontal functioning associated with high hypnotizability. Reviewed sStudies indicate that individuals with high hypnotizability display more pronounced reductions in activation of the medial prefrontal or dorsal anterior cingulate cortex following neutral hypnotic induction than their low hypnotizable ones. Moreover, research has disclosed that induction-specific reductions in the activation of the DMNefault Mode Network (DMN) regions correspond to spontaneous changes in cognitive and perceptual states, and the state of attentional absorption during a hypnotic induction has been associated with reduced DMN activity and increased prefrontal attentional system activity.
- functional neuroimaging
- EEG oscillations
- functional connectivity
- hypnosis
1. Introduction
2. Electroencephalography (EEG) Spectral Analysis and Functional Neuroimaging Techniques
The EEG and neuroimaging techniques involve registering electrical activity from the scalp using EEG electrodes mounted on a cap and signal processing through pre-amplification and amplification. These data are analyzed in temporal and frequency domains. Time domain analysis yields event-related potentials (ERPs) that reflect cortical activity linked to specific events and provide valuable insights into response timing of millisecond-level changes in cortical electrical activity. In the frequency domain, the EEG oscillatory activity is quantified by its frequency (i.e., the rate of repetition of the oscillatory event), amplitude or power (i.e., the magnitude of the oscillatory signal at a given frequency), and phase (a measure of its position in time relative to a pre-defined cycle or close to a reference oscillation at the same frequency). Resting-state oscillatory activity is traditionally defined as the superposition of oscillations falling within distinct frequency bands. The primary frequency bands of normative EEG oscillatory activity in adulthood are named delta (1–4 Hz), theta (4–8 Hz), alpha (8–13 Hz), beta (13–30 Hz), and gamma (>35 Hz). One of the most common signal frequency processing methods is a fast Fourier transform (FFT). An FFT provides the frequency power spectrum for a period, often averaged across a range of frequencies comprising a band (e.g., alpha). It also provides a phase spectrum. The power spectrum reflects the energy of each frequency determined by the squared amplitude of the wave. The phase spectrum reflects the phase in radians or degrees of the sine or cosine wave at each interval. Most frequency analyses focus exclusively on frequency power [37][21]. Relatively to the spectral power measurement, recent research exploring EEG has highlighted a significant factor that might surprise or complicate results in resting EEG. Specifically, the power distribution within EEG frequency bands comprises two key components: a rhythmic, periodic aspect and an irregular, overlapping, aperiodic component. This aperiodic activity showcases power across all frequencies under a 1/f power-law pattern. It reaches its peak at lower frequencies and steadily exponentially decreases as frequency rises [38][22]. The periodic component is characterized by the oscillatory peak’s bandwidth center frequency and relative amplitude. In contrast, the aperiodic component is defined by the slope and offset or intercept (i.e., the position where this slope starts) of the power spectrum. The intercept might indicate heightened spiking in neural populations [39,40][23][24]. Even if aperiodic activity does not offer meaningful information to the EEG signal, mixing it with periodic activity, as has been the case in the majority of the resting-EEG studies, can introduce noise effects in conventional EEG frequency analyses, as exemplified recently by Ouyang and colleagues [41][25]. These authors explored if cognitive processing speed correlated with alpha-range activity, as prior studies indicated. After separating periodic and aperiodic components within the alpha band, they found a link between mental processing speed and total alpha power before isolating them. However, this correlation vanished when considering only the actual periodic part of the signal. It persisted solely when the aperiodic portion was analyzed, i.e., measuring the aperiodic slope exponent and offset. As a result, both elements play a role in the overall power computed within distinct frequency bands, such as the alpha waves. This fact implies that the pure rhythmic activity is tied with non-oscillatory power that does not truly oscillate within that specific frequency range. This surprising outcome reshapes the understanding of earlier findings, emphasizing the psychological significance of aperiodic EEG data—challenging its classification as mere “noise.” These observations have highlighted the crucial need to differentiate between these distinct EEG activity patterns in research. Studies on EEG frequency oscillations obtained during several different behavioral tasks have outlined a link between EEG activity and various aspects of brain functions, encompassing sensory processing, perception, motor control, and cognitive activities such as attention, learning, memory, and emotional processing. These oscillations essentially serve as a functional code for the brain and are believed to facilitate communication between different brain regions and support associative processes [42,43,44,45][26][27][28][29].2.1. EEG and Neuroimaging
The interpolation of EEG signals into scalp tomography and source analysis aids in understanding temporal resolution. However, these methods lack precision in spatial resolution, failing to pinpoint the origin of the brain’s activity. This discrepancy, known as the inverse problem, arises because diverse cortical distributions could generate identical scalp activity patterns. Additionally, variations in skull thickness influence how brain activity manifests on the scalp. While EEG offers insights into underlying brain activity, techniques like dipole modeling simulate different brain generators to match theoretical scalp EEG patterns with actual recordings. However, for more precise localization, PET and MRI imaging techniques surpass EEG’s capabilities (for a detailed comparison, see [46][30]. Various classification methods exist to distinguish distinct topographies in multichannel EEG data, aiming to describe EEG as potential maps with different time courses [47,48][31][32]. Understanding large-scale brain network communication has become pivotal. Analytical methods estimating connectivity in networks, applicable to both fMRI and EEG, have gained traction due to EEG’s high temporal resolution and direct measurement of synchronized neuronal activity across various frequencies. Functional connectivity measures, such as cross-correlation and phase synchronization, are standard, while newer methods assess effective connectivity, capturing causal relationships within brain circuits [49,50,51][33][34][35]. Among multivariate methods for functional connectivity analysis, those using graph theory are providing appreciable improvement in understanding complex brain functioning. Graph theory provides the means for characterizing the available connections in the brain using a complex network model [52][36]. This method assumes that the brain is represented as a ‘graph’ composed of ‘nodes’ (brain regions or electrodes) connected by ‘edges’ (links or connections between these regions). Measurement of the strength of connections between nodes usually uses measures like the weighted Phase Lag Index (wPLI) to account for volume conduction effects on brain connectivity estimation [53][37].2.2. PET and fMRI Methods
PET and fMRI methods are fundamental for understanding brain activity. Positron emission tomography (PET) explicitly measures cerebral blood flow (CBF) changes related to brain functions, deriving energy from oxygen and glucose carried through blood flow. Analyzing these flow alterations in various brain regions helps identify active areas during specific tasks. PET involves participants inhaling or being injected with a radioactive isotope, providing the tracer recorded by the PET scanner. The general procedure is to make a measurement, enabled by a gamma ray detector, during a control task, which is subtracted from the measurement taken during an experimental task. While PET’s temporal resolution might be compromised due to the time it takes for readings (minutes), it excels in pinpointing active brain regions during different processing types. Its versatility in measuring radioactively labeled molecules facilitates insights into perfusion, metabolism, and neurotransmitter turnover. Functional magnetic resonance imaging (fMRI) also relies on blood flow changes in the cortex to identify active areas but uses a different technology from PET. An external magnet in fMRI detects local magnetic fields, analyzing hemoglobin’s magnetic properties pre- and post-oxygen absorption to map cortical blood changes and infer neuronal activity. Ogawa et al.’s groundbreaking research sparked significant enthusiasm for utilizing electrophysiology experiments to elucidate the neural basis of the blood-oxygen-level-dependent (BOLD) fMRI signal in human studies. In 1992, three distinct research groups separately achieved results in humans using the BOLD mechanism [59,60[38][39][40],61], initiating the surge of fMRI publications that have consistently emerged in scientific journals since then (for further details, see [62][41]). While fMRI boasts better spatial resolution and the ability to generate multiple images from a single individual compared to PET, its physical setup might be uncomfortable for some individuals and limits certain studies. There are advantages to combining fMRI and PET; using fMRI gives us clear structural brain scans, while PET allows us to index blood oxygen-dependent activity throughout the brain. However, while neuroimaging can specify the activated regions during mental states or cognitive tasks, it cannot determine the ‘necessary’ brain areas for the functions of interest. Traditionally, lesion studies offered insights, but purposely inducing lesions in humans is unethical. Transcranial magnetic stimulation (TMS) emerged as an alternative, allowing brief inhibition or facilitation of specific brain areas.3. Functional Neuroimaging Correlates of Resting Following Hypnotic Induction without Specific Suggestions
Several neuroimaging studies have demonstrated that hypnotic induction without task-specific or indirect suggestions induces plastic changes in neuronal activity by mainly engaging the frontal and thalamic areas (e.g., [22,30,64,65,66,67,68,69,70,71,72][42][43][44][45][46][47][48][49][50][51][52]). Figure 1 depicts the main neural networks, and the associated cortical brain regions found sensitive to hypnosis modulations and individual differences in hypnotizability. Above all, several reports indicate that hypnotic induction tunes higher-order neural systems involved in higher-order cognitive functions, supporting the top-down view of hypnosis. These observations are consistent across various experimental conditions, including the Stroop task, mental imagery, administration of noxious stimulations, as well as in resting state and neurophenomenological studies [9,64,65,70,73,74,75,76,77,78][8][44][45][50][53][54][55][56][57][58]. Dissociated control and cold control theories have resulted in being the most used models to explain experimental hypnosis data. These theoretical models served to design experiments well and formulate testable hypotheses (e.g., [79,80][59][60]).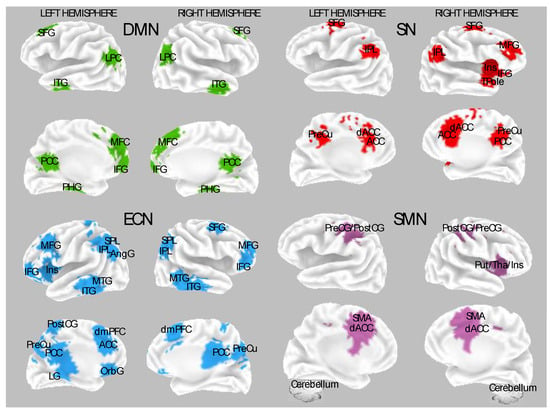
4. Structural and Functional Neuroimaging Correlates of Hypnotizability
5. EEG Oscillations and Their Associations with Hypnotizability and Hypnosis
Early investigations into EEG patterns during hypnosis reported an increased occurrence of occipital alpha waves in HHs compared to LHs [193,194,195,196,197,198][103][104][105][106][107][108]. Later studies have reported increased alpha activity in HHs during hypnosis [199][109], as well as after the hypnotic induction procedure [200,201,202][110][111][112]. However, findings from later studies failed to detect an increase in alpha activity with hypnosis [165,203][87][113]. However, Franz and co-workers [204][114] re-processed their-own EEG data from a previously published ERP odd-ball study [205][115] to investigate whether hypnotic suggestions of a visual obstruction would influence the amplitude of ERP components (N1, P2, and P3b amplitudes). The study also provided source reconstruction and spectro-temporal connectivity analysis of the P3b-related frequency oscillations within the conventional frequency bands (i.e., 1−30 Hz). In response to targets, P3b amplitudes and P3b source were significantly reduced in occipital and parietal brain areas (related to categorization and attention to stimuli). Additionally, both frontal and parietal electrodes were significantly reduced in effective connectivity within the sole P3-related alpha frequencies (10.5–12 Hz) during hypnosis compared to the control condition. These results provide preliminary evidence that hypnotic suggestions of a visual blockade are associated with a disruption of the coupling within the frontoparietal network implicated in top-down control. The most consistent relationship between EEG activity and hypnosis is reported in the theta band (4–8 Hz) [32,33,91][68][69][116]. Several studies have reported increased spectral power in the EEG-theta band during hypnosis [200,203,206,207,208][110][113][117][118][119]. Crawford and collaborators [209][120] said that HHs, compared to LHs, had significantly more significant hemispheric asymmetries (right greater than left) in the parietal region for all frequency bands usually associated with sustained attentional processing, in high-theta (5.5–7.45 Hz), high-alpha (11.5–13.45 Hz), and beta activity (16.5 and 25 Hz). Later, De Pascalis et al. [207][118] found that only HHs exhibited a higher low-band theta (4–6 Hz) amplitude in bilateral frontal and right posterior areas and a smaller alpha (8.25–10 Hz) amplitude bilaterally in the frontal cortex. There is also evidence of increased gamma activity (above 35 Hz) under hypnosis [213,214][121][122]. In an early study, Ulett and colleagues [198][108] measured a decrease in theta and an increase in alpha, beta, and gamma (40 Hz) activity in the right occipital cortex during hypnotic induction. Further, De Pascalis and colleagues [215,216][123][124] reported that HHs exhibited greater 40-Hz EEG amplitude density during emotional states than LHs in nonhypnotic and hypnotic conditions. However, these results were not confirmed by Crawford and coworkers’ [209][120] observations, although these authors reported a significant beta power increase in the right parietal region. Notably, the most pronounced differences in EEG patterns related to hypnotizability have been reported in the theta spectrum. However, these differences are primarily observed between HHs and LHs, excluding medium hypnotizable individuals (MHs). Crawford [219][125] proposed a dynamic neuropsychophysiological model of hypnosis involving the activation of the frontal-limbic attentional system. This model posits that attentional and disattentional processes are crucial in experiencing hypnosis, with low theta (3–6 Hz) and high theta (6–8 Hz) rhythms linked to these processes. Sabourin et al. [203][113] observed that during hypnosis, both LHs and HHs increased mean theta power (4–7.75 Hz), indicating intensified attentional processes and imagery enhancement. Further research consistently found that HHs tend to exhibit more significant slow-wave theta activity than LHs, both at baseline and during hypnosis, and both groups showed an increase in slow-wave activity after hypnotic inductions [202,206][112][117]. Despite these observations, recent studies have not found significant power changes in EEG frequency bands during hypnosis, questioning the hypothesis of theta rhythm as a definitive neurophysiological signature of hypnosis (e.g., [220,221][126][127]). Terhune and colleagues [127][128] also reported increased alpha2 (10.5–12 Hz) power during hypnosis but no significant differences in other EEG bands. Even studies comparing HHs to LHs in the waking state have shown mixed results, with some studies reporting higher theta power in HHs [6,200,222][6][110][129] and others noting no theta differences between the groups [168][90]. Williams and Gruzelier [202][112] reported increases in alpha power (7.5–13.5 Hz) in HHs at posterior regions during the transition from pre-hypnosis to hypnosis conditions, with decreases observed after hypnosis. The reverse pattern was seen in LHs. Schnyer and Allen [217][130] reported that HHs exhibited a greater density in the 36–44 Hz frequency band during posthypnosis recognition amnesia, indicating their enhanced ability to maintain focused attention outside hypnosis. Later, De Pascalis et al. [207][118] reported higher 40 Hz EEG amplitudes in HHs during resting hypnosis conditions compared to LHs.6. EEG Connectivity of Hypnosis and Hypnotizability
6.1. EEG Functional Connectivity Measure
The brain’s electrical activity results from dynamic interactions among distributed neural networks, displaying transient and quasi-stationary processes. The statistical dependency of physiological time series recorded from different brain areas, known as “functional connectivity”, encompasses synchronous oscillatory activity crucial for neural coordination across various cell assemblies involved in multiple systems and representations [237,238][131][132]. It is widely accepted that neural synchrony is pivotal in integrating information essential for perception, cognition, emotion, and the representation of consciousness aspects such as body ownership, self-consciousness, and identity [239,240][133][134]. One prevalent assumption in hypnosis-suggestion research is that induced alterations in experiential content are associated with distinct changes in functional connectivity. EEG functional connectivity and fMRI research have provided consistent findings showing that HH individuals exhibit distinctive patterns of neural network connectivity. Measures of dependence between multiple time series, whether linear (coherence, COH) or nonlinear (phase synchronization), can be expressed as combinations of lagged and instantaneous dependence. These measures, with values ranging from 0 to 1, indicate independence when they are zero. They are defined in the frequency domain and apply to stationary and non-stationary time series. These measures find utility in various fields, including neurophysiology, where they assess the connectivity of electric neuronal activity across different brain regions. However, it is essential to note that any measure of dependence in this context may include non-physiological contributions from volume conduction and limited spatial resolution [241][135]. A conventional measure of functional connectivity between two cortical regions is EEG coherence, believed to reflect the strength of interconnections between cortical areas [242][136]. EEG coherence between pairs of scalp locations offers valuable insights into brain states, indicating the competition between functional segregation and integration in brain dynamics. However, scalp-recorded EEG coherence has limitations, as even focal brain activity generates widespread EEG voltage patterns.6.2. EEG Functional Connectivity Findings under Resting Hypnosis
In a single case study by Fingelkurts and co-workers [248][137] using a sophisticated EEG functional connectivity measure (Index of Structural Synchrony), the study provided evidence of local and long-distance functional connectivity changes within the cortex after a neutral hypnotic induction and disruption in the functional synchronization among neural assemblies in the left frontal cortex and these changes remained stable even a year later. Cardeña and co-workers [156][78] recorded resting EEG during hypnosis by adopting a neurophenomenological approach to investigate neutral hypnosis (involving no specific suggestion other than to go into hypnosis). They found that hypnotic depth correlated positively with spectral power and power heterogeneity (i.e., the raw power of the Global Field Power curve) for the fast EEG frequencies of beta2 (18.5–21.0 Hz), beta3 (21.5–30 Hz), and gamma (35–44 Hz), but only among HHs, a finding seen consistent with the position reported in previous studies that hypnosis is associated with increased 40 Hz gamma activity, particularly among HHs (e.g., [249][138]). Following the neutral hypnotic induction, they observed that HHs had spontaneous imagery, positive affect, and anomalous perceptual states that were also associated with lower global functional connectivity during hypnosis. Imagery correlated positively with gamma power heterogeneity and negatively with alpha1 power heterogeneity. Generally, the HHs displayed an opposite pattern of correlations to that found for the Lows.6.3. EEG Functional Connectivity Correlates with Hypnotizability
Gruzelier [167][89] has proposed a working neurophysiological model of hypnosis and hypnotizability in which HHs under hypnosis are characterized by a reduced upper alpha band coherence between the left frontal and medial electrode pairs. In contrast, increased upper alpha coherence between the same electrode pairs marks the LHs, and decreased coherence within left frontal brain activity during hypnosis delineates the HHs. Advanced research by Egner and collaborators [64][44] used a combination of event-related fMRI and EEG coherence recordings with the Stroop task to test predictions from the dissociation theory that hypnosis dissociates executive control and monitoring processes [82,252][139][140]. These authors evaluated neural activity in the Stroop task after hypnotic induction without task-specific suggestions. The fMRI results revealed that conflict-related ACC activity interacted with hypnosis and hypnotizability since HHs displayed increased conflict-related neural activity in the ACC during hypnosis compared to baseline and LHs. Interestingly, activity in the DLPFC, associated with cognitive control, did not differ between hypnotizability groups and conditions. HHs exhibited a decrease in EEG gamma band coherence, from baseline to hypnosis, between frontal midline and left lateral scalp sites, while LHs showed a gamma band coherence increase. These findings indicate a decoupling between conflict monitoring and control processes during hypnosis and suggest a negative link between hypnotizability and efficiency of the frontal attention system. These findings align with the view that HH individuals are particularly prone to focusing attention at baseline. However, after hypnosis, their attention control ability appears weakened due to a disconnection between the conflict monitoring and cognitive control processes of the frontal lobe became disconnected. The study mentioned above findings by Egner and colleagues [64][44] fit well with the general model proposed by Jamieson and Woody [109][141], in which breakdowns in the functional integration between different components of executive control networks account for core features in the phenomenology of the hypnotic condition. Alternatively, these findings can also be explained as dissociation or shifts in subjective evaluations of one’s agency during the hypnotic experience, as proposed by the Cold Control theory of hypnosis [83][142]. The Cold Control theory suggests that hypnosis primarily involves a change in metacognition, where hypnotic responses occur due to a lack of awareness regarding the intentions that drive cognitive or sensorimotor actions, i.e., the activation of sensorimotor network.7. Conclusions
Resting-state fMRI and structural MRI investigations offer complementary insights into the distinctive frontal functioning associated with high hypnotizability. Reviewed studies indicate that individuals with high hypnotizability display more pronounced reductions in activation of the medial prefrontal or dorsal anterior cingulate cortex following neutral hypnotic induction than their low hypnotizable ones [9,67][8][47]. Moreover, research has disclosed that induction-specific reductions in the activation of the DMN regions correspond to spontaneous changes in cognitive and perceptual states, and the state of attentional absorption during a hypnotic induction has been associated with reduced DMN activity and increased prefrontal attentional system activity [73][53]. Furthermore, fMRI and EEG research using diverse functional connectivity methods has underscored the complexity of neural mechanisms during hypnosis. Specifically, HH individuals, who usually feel the most hypnotized, have shown the highest connectivity between the bilateral DLPFC (i.e., ECN) and ipsilateral insula (i.e., SN) during hypnosis compared to rest. At the same time, for LHs, there are no differences between conditions. This enhanced connectivity during hypnosis is accompanied by reduced connectivity within DMN regions, contributing to altered agency and self-consciousness [67,148][47][85]. The intricate relationship between SN, ECN, and DMN further delineates the neural correlates of hypnotizability. Some authors have reported the uncoupling of connectivity between the ECN and the DMN during hypnosis [67][47]. In contrast, others have found the contrary true [9][8]. Spiegel and colleagues’ work explained the dissociation between ECN and DMN in response to hypnotic induction as an engagement in the hypnotic state and the associated detachment from internal mental processes such as mind wandering and self-reflection. This explanation reinforces the idea of resting hypnosis as a different state of consciousness rather than a reduced level of arousal [96][80]. According to Demertzi and colleagues’ conceptualization [123][143], hypnosis alters resting-state fMRI networks by diminishing “extrinsic” lateral frontoparietal cortical connectivity, which might reflect a decreased sensory awareness. In this altered state, the default mode network (DMN) exhibits increased connectivity in its lateral parietal and middle frontal regions but reduced connectivity in its posterior midline and parahippocampal structures [74,123][54][143]. However, other studies reported that connectivity in hypnosis is not limited to the DMN, depending on the depth of hypnosis and the ongoing emotional involvement and mental content, making this conceptualization questionable [95,180][95][144]. Furthermore, the assessment of neuroimaging assays of hypnosis using the Activation Likelihood Estimation (ALE) method by Landry et al.’s [22][42] findings did not align with this perspective. Instead, the study demonstrated a stronger correlation between hypnotic responses and activation of the lingual gyrus, suggesting a potential association with mental imagery. Nevertheless, several encouraging research paths point to associations between hypnosis, hypnotizability with neuroimaging functional connectivity and EEG non-oscillatory aperiodic activity that may shed light on individual differences in hypnotic suggestibility and the mechanisms of suggestion. For example, DeSouza et al. [146][84], using structural neuroimaging measures, have reported a significant positive association between γ-aminobutyric acid (GABA) concentration in the anterior cingulate cortex (ACC) and hypnotizability [146][84], as well as an inverse relationship between trait hypnotizability and perseveration (served by executive control and the salience systems) [267][145]. This novel outcome indicates that hypnotizability is a temperamental trait predisposing individuals to be more or less responsive to suggestions. These observations would be highly beneficial for improving clinical treatments with hypnosis.References
- Kihlstrom, J.F. The domain of hypnosis, revisited. In The Oxford Handbook of Hypnosis: Theory, Research and Practice; Oxford University Press: New York, NY, USA, 2008; pp. 21–52.
- Halsband, U.; Mueller, S.; Hinterberger, T.; Strickner, S. Plasticity changes in the brain in hypnosis and meditation. Contemp. Hypn. 2009, 26, 194–215.
- Kihlstrom, J.F.; Glisky, M.L.; McGovern, S.; Rapcsak, S.Z.; Mennemeier, M.S. Hypnosis in the right hemisphere. Cortex 2013, 49, 393–399.
- Oakley, D.A.; Halligan, P.W. Hypnotic suggestion: Opportunities for cognitive neuroscience. Nat. Rev. Neurosci. 2013, 14, 565–576.
- Terhune, D.B.; Cleeremans, A.; Raz, A.; Lynn, S.J. Hypnosis and top-down regulation of consciousness. Neurosci. Biobehav. Rev. 2017, 81, 59–74.
- Vanhaudenhuyse, A.; Laureys, S.; Faymonville, M.E. Neurophysiology of hypnosis. Clin. Neurophysiol. 2014, 44, 343–353.
- Cox, R.E.; Bryant, R.A. Advances in hypnosis research: Methods, designs and contributions of intrinsic and instrumental hypnosis. In The Oxford Handbook of Hypnosis: Theory, Research, and Practice; Oxford University Press: New York, NY, USA, 2008; pp. 311–336.
- McGeown, W.J.; Mazzoni, G.; Venneri, A.; Kirsch, I. Hypnotic induction decreases anterior default mode activity. Conscious. Cogn. 2009, 18, 848–855.
- Oakley, D.A.; Halligan, P.W. Hypnotic suggestion and cognitive neuroscience. Trends Cogn. Sci. 2009, 13, 264–270.
- Rainville, P.; Price, D.D. Hypnosis Phenomenology and the Neurobiology of Consciousness. Int. J. Clin. Exp. Hypn. 2003, 51, 105–129.
- De Pascalis, V.; Scacchia, P.; Vecchio, A. Influences of hypnotic suggestibility, contextual factors, and EEG alpha on placebo analgesia. Am. J. Clin. Hypn. 2021, 63, 302–328.
- Cardeña, E.; Terhune, D.B. Hypnotizability, personality traits, and the propensity to experience alterations of consciousness. Psychol. Conscious. Theory Res. Pract. 2014, 1, 292–307.
- Piccione, C.; Hilgard, E.R.; Zimbardo, P.G. On the degree of stability of measured hypnotizability over a 25-year period. J. Personal. Soc. Psychol. 1989, 56, 289–295.
- Gorassini, D.R.; Spanos, N.P. A social-cognitive skills approach to the successful modification of hypnotic susceptibility. J. Personal. Soc. Psychol. 1986, 50, 1004–1012.
- Gorassini, D.R.; Spanos, N.P. The Carleton Skill Training Program for modifying hypnotic suggestibility: Original version and variations. In Clinical Hypnosis and Self-Regulation: Cognitive-Behavioral Perspectives; American Psychological Association: Washington, DC, USA, 1999; pp. 141–177.
- Rossi, E.L. Hypnosis and Ultradian Cycles: A New State(s) Theory of Hypnosis? Am. J. Clin. Hypn. 1982, 25, 21–32.
- Green, J.P.; Smith, R.J.; Kromer, M. Diurnal Variations in Hypnotic Responsiveness: Is There an Optimal Time to Be Hypnotized? Int. J. Clin. Exp. Hypn. 2015, 63, 171–181.
- Fassler, O.; Lynn, S.J.; Knox, J. Is hypnotic suggestibility a stable trait? Conscious. Cogn. 2008, 17, 240–253.
- Lynn, S.J.; Maxwell, R.; Green, J.P. The hypnotic induction in the broad scheme of hypnosis: A sociocognitive perspective. In Hypnotic Induction, 1st ed.; Routledge: London, UK, 2017; pp. 109–130.
- Lynn, S.J. Hypnotic reponsiveness can be modified after hypnotic training. In Proceedings of the Oral Presentation at XXI World Congress of Medical and Clinical Hypnosis, Montréal, QC, Canada, 22–25 August 2018.
- Gable, P.; Miller, M.; Bernat, E. The Oxford Handbook of EEG Frequency; Oxford University Press: New York, NY, USA, 2022.
- Donoghue, T.; Haller, M.; Peterson, E.J.; Varma, P.; Sebastian, P.; Gao, R.; Noto, T.; Lara, A.H.; Wallis, J.D.; Knight, R.T.; et al. Parameterizing neural power spectra into periodic and aperiodic components. Nat. Neurosci. 2020, 23, 1655–1665.
- Manning, J.R.; Jacobs, J.; Fried, I.; Kahana, M.J. Broadband Shifts in Local Field Potential Power Spectra Are Correlated with Single-Neuron Spiking in Humans. J. Neurosci. 2009, 29, 13613.
- Miller, K.J.; Hermes, D.; Honey, C.J.; Hebb, A.O.; Ramsey, N.F.; Knight, R.T.; Ojemann, J.G.; Fetz, E.E. Human motor cortical activity is selectively phase-entrained on underlying rhythms. PLoS Comput. Biol. 2012, 8, e1002655.
- Ouyang, G.; Hildebrandt, A.; Schmitz, F.; Herrmann, C.S. Decomposing alpha and 1/f brain activities reveals their differential associations with cognitive processing speed. NeuroImage 2020, 205, 116304.
- Güntekin, B.; Başar, E. A review of brain oscillations in perception of faces and emotional pictures. Neuropsychologia 2014, 58, 33–51.
- Buzsáki, G. Rhythms of the Brain; Oxford University Press Inc.: NewYork, NY, USA, 2006.
- Cohen, M.X. Analyzing Neural Time Series Data: Theory and Practice; MIT Press: Cambridge, MA, USA, 2014.
- Keil, A.; Thigpen, N. From Neural Oscillations to Cognitive Processes. In The Oxford Handbook of EEG Frequency; Gable, P.A., Miller, M.W., Bernat, E.M., Eds.; Oxford University Press: New York, NY, USA, 2022; pp. 40–64.
- Ray, W.J.; Oathes, D. Brain Imaging Techniques. Int. J. Clin. Exp. Hypn. 2003, 51, 97–104.
- Michel, C.M.; Murray, M.M. Towards the utilization of EEG as a brain imaging tool. NeuroImage 2012, 61, 371–385.
- De Lucia, M.; Michel, C.M.; Murray, M.M. Comparing ICA-based and Single-Trial Topographic ERP Analyses. Brain Topogr. 2010, 23, 119–127.
- Stephan, K.E.; Friston, K.J. Analyzing effective connectivity with functional magnetic resonance imaging. WIREs Cogn. Sci. 2010, 1, 446–459.
- Kiebel, S.J.; Garrido, M.I.; Moran, R.J.; Friston, K.J. Dynamic causal modelling for EEG and MEG. Cogn. Neurodynamics 2008, 2, 121–136.
- Kaminski, M.J.; Blinowska, K.J. A new method of the description of the information flow in the brain structures. Biol. Cybern. 1991, 65, 203–210.
- Bullmore, E.; Sporns, O. Complex brain networks: Graph theoretical analysis of structural and functional systems. Nat. Rev. Neurosci. 2009, 10, 186–198.
- Vinck, M.; Oostenveld, R.; van Wingerden, M.; Battaglia, F.; Pennartz, C.M.A. An improved index of phase-synchronization for electrophysiological data in the presence of volume-conduction, noise and sample-size bias. NeuroImage 2011, 55, 1548–1565.
- Kwong, K.K.; Belliveau, J.W.; Chesler, D.A.; Goldberg, I.E.; Weisskoff, R.M.; Poncelet, B.P.; Kennedy, D.N.; Hoppel, B.E.; Cohen, M.S.; Turner, R. Dynamic magnetic resonance imaging of human brain activity during primary sensory stimulation. Proc. Natl. Acad. Sci. USA 1992, 89, 5675–5679.
- Ogawa, S.; Tank, D.W.; Menon, R.; Ellermann, J.M.; Kim, S.G.; Merkle, H.; Ugurbil, K. Intrinsic signal changes accompanying sensory stimulation: Functional brain mapping with magnetic resonance imaging. Proc. Natl. Acad. Sci. USA 1992, 89, 5951–5955.
- Bandettini, P.A.; Wong, E.C.; Hinks, R.S.; Tikofsky, R.S.; Hyde, J.S. Time course EPI of human brain function during task activation. Magn. Reson. Med. 1992, 25, 390–397.
- Logothetis, N.K.; Pfeuffer, J. On the nature of the BOLD fMRI contrast mechanism. Magn. Reson. Imaging 2004, 22, 1517–1531.
- Landry, M.; Lifshitz, M.; Raz, A. Brain correlates of hypnosis: A systematic review and meta-analytic exploration. Neurosci. Biobehav. Rev. 2017, 81, 75–98.
- Halsband, U. Learning in trance: Functional brain imaging studies and neuropsychology. J. Physiol. Paris 2006, 99, 470–482.
- Egner, T.; Jamieson, G.; Gruzelier, J. Hypnosis decouples cognitive control from conflict monitoring processes of the frontal lobe. NeuroImage 2005, 27, 969–978.
- Faymonville, M.E.; Laureys, S.; Degueldre, C.; Del Fiore, G.; Luxen, A.; Franck, G.; Lamy, M.; Maquet, P. Neural mechanisms of antinociceptive effects of hypnosis. Anesthesiol. Phila. Then Hagerstown 2000, 92, 1257–1267.
- Grond, M.; Pawlik, G.; Walter, H.; Lesch, O.M.; Heiss, W.-D. Hypnotic catalepsy-induced changes of regional cerebral glucose metabolism. Psychiatry Res. Neuroimaging 1995, 61, 173–179.
- Jiang, H.; White, M.P.; Greicius, M.D.; Waelde, L.C.; Spiegel, D. Brain Activity and Functional Connectivity Associated with Hypnosis. Cereb. Cortex 2017, 27, 4083–4093.
- Kosslyn, S.M.; Thompson, W.L.; Costantini-Ferrando, M.F.; Alpert, N.M.; Spiegel, D. Hypnotic Visual Illusion Alters Color Processing in the Brain. Am. J. Psychiatry 2000, 157, 1279–1284.
- Maquet, P.; Faymonville, M.E.; Degueldre, C.; Delfiore, G.; Franck, G.; Luxen, A.; Lamy, M. Functional neuroanatomy of hypnotic state. Biol. Psychiatry 1999, 45, 327–333.
- Rainville, P. Brain mechanisms of pain affect and pain modulation. Curr. Opin. Neurobiol. 2002, 12, 195–204.
- Rainville, P.; Hofbauer, R.K.; Paus, T.; Duncan, G.H.; Bushnell, M.C.; Price, D.D. Cerebral mechanisms of hypnotic induction and suggestion. J. Cogn. Neurosci. 1999, 11, 110–125.
- Szechtman, H.; Woody, E.; Bowers, K.S.; Nahmias, C. Where the imaginal appears real: A positron emission tomography study of auditory hallucinations. Proc. Natl. Acad. Sci. USA 1998, 95, 1956–1960.
- Deeley, Q.; Oakley, D.A.; Toone, B.; Giampietro, V.; Brammer, M.J.; Williams, S.C.R.; Halligan, P.W. Modulating the Default Mode Network Using Hypnosis. Int. J. Clin. Exp. Hypn. 2012, 60, 206–228.
- Demertzi, A.; Soddu, A.; Faymonville, M.E.; Bahri, M.A.; Gosseries, O.; Vanhaudenhuyse, A.; Phillips, C.; Maquet, P.; Noirhomme, Q.; Luxen, A.; et al. Chapter 20-Hypnotic modulation of resting state fMRI default mode and extrinsic network connectivity. In Progress in Brain Research; Van Someren, E.J.W., Van Der Werf, Y.D., Roelfsema, P.R., Mansvelder, H.D., Lopes Da Silva, F.H., Eds.; Elsevier: Amsterdam, The Netherlands, 2011; Volume 193, pp. 309–322.
- Müller, K.; Bacht, K.; Prochnow, D.; Schramm, S.; Seitz, R.J. Activation of thalamus in motor imagery results from gating by hypnosis. NeuroImage 2013, 66, 361–367.
- Müller, K.; Bacht, K.; Schramm, S.; Seitz, R.J. The facilitating effect of clinical hypnosis on motor imagery: An fMRI study. Behav. Brain Res. 2012, 231, 164–169.
- Vanhaudenhuyse, A.; Boly, M.; Balteau, E.; Schnakers, C.; Moonen, G.; Luxen, A.; Lamy, M.; Degueldre, C.; Brichant, J.F.; Maquet, P.; et al. Pain and non-pain processing during hypnosis: A thulium-YAG event-related fMRI study. NeuroImage 2009, 47, 1047–1054.
- Rainville, P.; Streff, A.; Chen, J.-I.; Houzé, B.; Desmarteaux, C.; Piché, M. Hypnotic Automaticity in the Brain at Rest: An Arterial Spin Labelling Study. Int. J. Clin. Exp. Hypn. 2019, 67, 512–542.
- Dienes, Z.; Beran, M.; Brandl, J.L.; Perner, J.; Proust, J. Is hypnotic responding the strategic relinquishment of metacognition. In Foundations of Metacognition; Oxford University Press: New York, NY, USA, 2012; pp. 267–277.
- Jaffer, U.; Jamieson, G.A. Hypnosis and the neuroscience of cognitive and affective control. Aust. J. Clin. Exp. Hypn. 2012, 40, 1–20.
- Chambon, V.; Wenke, D.; Fleming, S.M.; Prinz, W.; Haggard, P. An Online Neural Substrate for Sense of Agency. Cereb. Cortex 2013, 23, 1031–1037.
- Haggard, P.; Chambon, V. Sense of agency. Curr. Biol. 2012, 22, R390–R392.
- Lush, P.; Naish, P.; Dienes, Z. Metacognition of intentions in mindfulness and hypnosis. Neurosci. Conscious. 2016, 2016, niw007.
- Buchner, R.; Andrews-Hanna, J.; Schacter, D. The brain’s default network. Ann. N. Y. Acad. Sci. 2008, 1124, 1–38.
- Sridharan, D.; Levitin, D.J.; Menon, V. A critical role for the right fronto-insular cortex in switching between central-executive and default-mode networks. Proc. Natl. Acad. Sci. USA 2008, 105, 12569–12574.
- Fox, M.D.; Raichle, M.E. Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat. Rev. Neurosci. 2007, 8, 700–711.
- Halsband, U.; Gerhard Wolf, T. Functional Changes in Brain Activity After Hypnosis: Neurobiological Mechanisms and Application to Patients with a Specific Phobia—Limitations and Future Directions. Int. J. Clin. Exp. Hypn. 2019, 67, 449–474.
- Jensen, M.P.; Adachi, T.; Hakimian, S. Brain Oscillations, Hypnosis, and Hypnotizability. Am. J. Clin. Hypn. 2015, 57, 230–253.
- Halsband, U.; Wolf, T.G. Current neuroscientific research database findings of brain activity changes after hypnosis. Am. J. Clin. Hypn. 2021, 63, 372–388.
- Wolf, T.G.; Faerber, K.A.; Rummel, C.; Halsband, U.; Campus, G. Functional Changes in Brain Activity Using Hypnosis: A Systematic Review. Brain Sci. 2022, 12, 108.
- Faymonville, M.E.; Boly, M.; Laureys, S. Functional neuroanatomy of the hypnotic state. J. Physiol.-Paris 2006, 99, 463–469.
- van Osch, M.J.P.; Teeuwisse, W.M.; Chen, Z.; Suzuki, Y.; Helle, M.; Schmid, S. Advances in arterial spin labelling MRI methods for measuring perfusion and collateral flow. J. Cereb. Blood Flow Metab. 2018, 38, 1461–1480.
- Blakemore, S.J.; Oakley, D.A.; Frith, C.D. Delusions of alien control in the normal brain. Neuropsychologia 2003, 41, 1058–1067.
- Darby, R.R.; Joutsa, J.; Burke, M.J.; Fox, M.D. Lesion network localization of free will. Proc. Natl. Acad. Sci. USA 2018, 115, 10792–10797.
- Jensen, M.P.; Jamieson, G.A.; Lutz, A.; Mazzoni, G.; McGeown, W.J.; Santarcangelo, E.L.; Demertzi, A.; De Pascalis, V.; Bányai, É.I.; Rominger, C.; et al. New directions in hypnosis research: Strategies for advancing the cognitive and clinical neuroscience of hypnosis. Neurosci. Conscious. 2017, 2017, nix004.
- Barnier, A.J.; Cox, R.E.; McConkey, K.M. The province of “highs”: The high hypnotizable person in the science of hypnosis and in psychological science. Psychol. Conscious. Theory Res. Pract. 2014, 1, 168–183.
- Heap, M.; Brown, R.J.; Oakley, D.A. The Highly Hypnotizable Person: Theoretical, Experimental and Clinical Issues; Routledge: London, UK, 2004.
- Cardeña, E.; Jönsson, P.; Terhune, D.B.; Marcusson-Clavertz, D. The neurophenomenology of neutral hypnosis. Cortex 2013, 49, 375–385.
- Pekala, R.J.; Kumar, V. An empirical-phenomenological approach to quantifying consciousness and states of consciousness: With particular reference to understanding the nature of hypnosis. In Hypnosis and Conscious States: The Cognitive Neuroscience Perspective; Jamieson, G.A., Ed.; Oxford University Press: New York, NY, USA, 2007; pp. 167–194.
- McGeown, W.J.; Mazzoni, G.; Vannucci, M.; Venneri, A. Structural and functional correlates of hypnotic depth and suggestibility. Psychiatry Res. Neuroimaging 2015, 231, 151–159.
- Huber, A.; Lui, F.; Duzzi, D.; Pagnoni, G.; Porro, C.A. Structural and functional cerebral correlates of hypnotic suggestibility. PLoS ONE 2014, 9, e93187.
- Horton, J.E.; Crawford, H.J.; Harrington, G.; Downs, J.H., III. Increased anterior corpus callosum size associated positively with hypnotizability and the ability to control pain. Brain 2004, 127, 1741–1747.
- Bloom, J.S.; Hynd, G.W. The Role of the Corpus Callosum in Interhemispheric Transfer of Information: Excitation or Inhibition? Neuropsychol. Rev. 2005, 15, 59–71.
- DeSouza, D.D.; Stimpson, K.H.; Baltusis, L.; Sacchet, M.D.; Gu, M.; Hurd, R.; Wu, H.; Yeomans, D.C.; Willliams, N.; Spiegel, D. Association between Anterior Cingulate NHuberbeurochemical Concentration and Individual Differences in Hypnotizability. Cereb. Cortex 2020, 30, 3644–3654.
- Hoeft, F.; Gabrieli, J.D.E.; Whitfield-Gabrieli, S.; Haas, B.W.; Bammer, R.; Menon, V.; Spiegel, D. Functional Brain Basis of Hypnotizability. Arch. Gen. Psychiatry 2012, 69, 1064–1072.
- Cojan, Y.; Piguet, C.; Vuilleumier, P. What makes your brain suggestible? Hypnotizability is associated with differential brain activity during attention outside hypnosis. NeuroImage 2015, 117, 367–374.
- Kihlstrom, J.F. Neuro-hypnotism: Prospects for hypnosis and neuroscience. Cortex 2013, 49, 365–374.
- Crawford, H.J.; Gruzelier, J.H. A midstream view of the neuropsychophysiology of hypnosis: Recent research and future directions. In Contemporary Hypnosis Research; Guilford Press: New York, NY, USA, 1992; pp. 227–266.
- Gruzelier, J.H. A working model of the neurophysiology of hypnosis: A review of evidence. Contemp. Hypn. 1998, 15, 3–21.
- De Pascalis, V. Psychophysiological correlates of hypnosis and hypnotic susceptibility. Int. J. Clin. Exp. Hypn. 1999, 47, 117–143.
- Kallio, S.; Revonsuo, A.; Hämäläinen, H.; Markela, J.; Gruzelier, J. Anterior brain functions and hypnosis: A test of the frontal hypothesis. Int. J. Clin. Exp. Hypn. 2001, 49, 95–108.
- Huber, A.; Lui, F.; Porro, C.A. Hypnotic susceptibility modulates brain activity related to experimental placebo analgesia. PAIN® 2013, 154, 1509–1518.
- Sheehan, P.; McConkey, K. Hypnosis and Experience (Psychology Revivals): The Exploration of Phenomena and Process; Routledge: London, UK, 2015.
- Bryant, R.A.; McConkey, K.M. Hypnotic blindness and the relevance of cognitive style. J. Personal. Soc. Psychol. 1990, 59, 756–761.
- Oakley, D.A. Hypnosis and Conversion Hysteria: A Unifying Model. Cogn. Neuropsychiatry 1999, 4, 243–265.
- Oakley, D.; Frasquilho, F. Hypnotic susceptibility, or F-bias: Its relevance to eating disorders. Contemp. Hypn. 1998, 15, 40–51.
- McConkey, K.M.; Glisky, M.L.; Kihlstrom, J.F. Individual differences among hypnotic virtuosos: A case comparison. Aust. J. Clin. Exp. Hypn. 1989, 17, 131–140.
- Santarcangelo, E.L. New views of hypnotizability. Front. Behav. Neurosci. 2014, 8, 224.
- Picerni, E.; Santarcangelo, E.L.; Laricchiuta, D.; Cutuli, D.; Petrosini, L.; Spalletta, G.; Piras, F. Cerebellar Structural Variations in Subjects with Different Hypnotizability. Cerebellum 2019, 18, 109–118.
- Santarcangelo, E.L.; Scattina, E. Responding to Sensorimotor Suggestions: From Endothelial Nitric Oxide to the Functional Equivalence between Imagery and Perception. Int. J. Clin. Exp. Hypn. 2019, 67, 394–407.
- Dienes, Z.; Hutton, S. Understanding hypnosis metacognitively: rTMS applied to left DLPFC increases hypnotic suggestibility. Cortex 2013, 49, 386–392.
- Coltheart, M.; Cox, R.; Sowman, P.; Morgan, H.; Barnier, A.; Langdon, R.; Connaughton, E.; Teichmann, L.; Williams, N.; Polito, V. Belief, delusion, hypnosis, and the right dorsolateral prefrontal cortex: A transcranial magnetic stimulation study. Cortex 2018, 101, 234–248.
- Bakan, P. Hypnotizability, Laterality of Eye-Movements and Functional Brain Asymmetry. Percept. Mot. Ski. 1969, 28, 927–932.
- Edmonston, W.E.; Grotevant, W.R. Hypnosis and Alpha Density. Am. J. Clin. Hypn. 1975, 17, 221–232.
- Engstrom, D.R.; London, P.; Hart, J.T. Hypnotic Susceptibility increased by EEG Alpha Training. Nature 1970, 227, 1261–1262.
- London, P.; Hart, J.T.; Leibovitz, M.P.; McDevitt, R.A. The psychophysiology of hypnotic susceptibility. In Psychophysiological Mechanisms of Hypnosis: An International Symposium Sponsored by the International Brain Research Organization and the Centre de Recherche, Institut de Psychiatrie La Rochefoucauld, Paris; Springer: Berlin/Heidelberg, Germany, 1969; pp. 151–172.
- Morgan, A.H.; Macdonald, H.; Hilgard, E.R. EEG Alpha: Lateral Asymmetry Related to Task, and Hypnotizability. Psychophysiology 1974, 11, 275–282.
- Ulett, G.A.; Akpinar, S.; Itil, T.M. Quantitative EEG analysis during hypnosis. Electroencephalogr. Clin. Neurophysiol. 1972, 33, 361–368.
- De Pascalis, V.; Palumbo, G. EEG Alpha Asymmetry: Task Difficulty and Hypnotizability. Percept. Mot. Ski. 1986, 62, 139–150.
- Graffin, N.F.; Ray, W.J.; Lundy, R. EEG concomitants of hypnosis and hypnotic susceptibility. J. Abnorm. Psychol. 1995, 104, 123–131.
- MacLeod-Morgan, C. Hypnotic susceptibility, EEG theta and alpha waves, and hemispheric specificity. In Hypnosis 1979; Elsevier: Amsterdam, The Netherlands, 1979.
- Williams, J.D.; Gruzelier, J.H. Differentiation of hypnosis and relaxation by analysis of narrow band theta and alpha frequencies. Int. J. Clin. Exp. Hypn. 2001, 49, 185–206.
- Sabourin, M.E.; Cutcomb, S.D.; Crawford, H.J.; Pribram, K. EEG correlates of hypnotic susceptibility and hypnotic trance: Spectral analysis and coherence. Int. J. Psychophysiol. 1990, 10, 125–142.
- Franz, M.; Schmidt, B.; Hecht, H.; Naumann, E.; Miltner, W.H.R. Suggested visual blockade during hypnosis: Top-down modulation of stimulus processing in a visual oddball task. PLoS ONE 2021, 16, e0257380.
- Schmidt, B.; Hecht, H.; Naumann, E.; Miltner, W.H.R. The Power of mind: Blocking visual perception by hypnosis. Sci. Rep. 2017, 7, 4889.
- De Pascalis, V. EEG oscillatory activity concomitant with hypnosis and hypnotizability. In International Handbook of Clinical Hypnosis; Linden, J.H., De Benedittis, G., Sugarman, L.I., Varga, K., Eds.; Routledge: London, UK, 2024; in press.
- Crawford, H.J. Cognitive and psychophysiological correlates of hypnotic responsiveness and hypnosis. In Creative Mastery in Hypnosis and Hypnoanalysis: A Festschrift for Erika Fromm; Plenum Press: New York, NY, USA, 1990; pp. 47–54.
- De Pascalis, V.; Ray, W.J.; Tranquillo, I.; D’Amico, D. EEG activity and heart rate during recall of emotional events in hypnosis: Relationships with hypnotizability and suggestibility. Int. J. Psychophysiol. 1998, 29, 255–275.
- Tebecis, A.K.; Provins, K.A.; Farnbach, R.W.; Pentony, P. Hypnosis and the EEG: A quantitative investigation. J. Nerv. Ment. Dis. 1975, 161, 1–17.
- Crawford, H.J.; Clarke, S.W.; Kitner-Triolo, M. Self-generated happy and sad emotions in low and highly hypnotizable persons during waking and hypnosis: Laterality and regional EEG activity differences. Int. J. Psychophysiol. 1996, 24, 239–266.
- De Benedittis, G. Neural mechanisms of hypnosis and meditation. J. Physiol.-Paris 2015, 109, 152–164.
- Jensen, M.P.; Adachi, T.; Tomé-Pires, C.; Lee, J.; Osman, Z.J.; Miró, J. Mechanisms of Hypnosis: Toward the Development of a Biopsychosocial Model. Int. J. Clin. Exp. Hypn. 2015, 63, 34–75.
- De Pascalis, V.; Marucci, F.S.; Penna, P.M.; Pessa, E. Hemispheric activity of 40 Hz EEG during recall of emotional events: Differences between low and high hypnotizables. Int. J. Psychophysiol. 1987, 5, 167–180.
- De Pascalis, V.; Marucci, F.S.; Penna, P.M. 40-Hz EEG asymmetry during recall of emotional events in waking and hypnosis: Differences between low and high hypnotizables. Int. J. Psychophysiol. 1989, 7, 85–96.
- Crawford, H.J. Brain dynamics and hypnosis: Attentional and disattentional processes. Int. J. Clin. Exp. Hypn. 1994, 42, 204–232.
- Jamieson, G.A.; Burgess, A.P. Hypnotic induction is followed by state-like changes in the organization of EEG functional connectivity in the theta and beta frequency bands in high-hypnotically susceptible individuals. Front. Hum. Neurosci. 2014, 8, 528.
- White, D.; Ciorciari, J.; Carbis, C.; Liley, D. EEG Correlates of Virtual Reality Hypnosis. Int. J. Clin. Exp. Hypn. 2009, 57, 94–116.
- Knauff, M.; Mulack, T.; Kassubek, J.; Salih, H.R.; Greenlee, M.W. Spatial imagery in deductive reasoning: A functional MRI study. Cogn. Brain Res. 2002, 13, 203–212.
- Kirenskaya, A.V.; Novototsky-Vlasov, V.Y.; Zvonikov, V.M. Waking EEG Spectral Power and Coherence Differences Between High and Low Hypnotizable Subjects. Int. J. Clin. Exp. Hypn. 2011, 59, 441–453.
- Schnyer, D.M.; Allen, J.J. Attention-related electroencephalographic and event-related potential predictors of responsiveness to suggested posthypnotic amnesia. Int. J. Clin. Exp. Hypn. 1995, 43, 295–315.
- Fingelkurts, A.A.; Fingelkurts, A.A.; Kähkönen, S. Functional connectivity in the brain—Is it an elusive concept? Neurosci. Biobehav. Rev. 2005, 28, 827–836.
- Lee, L.; Harrison, L.M.; Mechelli, A. A report of the functional connectivity workshop, Dusseldorf 2002. NeuroImage 2003, 19, 457–465.
- Varela, F.; Lachaux, J.-P.; Rodriguez, E.; Martinerie, J. The brainweb: Phase synchronization and large-scale integration. Nat. Rev. Neurosci. 2001, 2, 229–239.
- Tsakiris, M.; Hesse, M.D.; Boy, C.; Haggard, P.; Fink, G.R. Neural Signatures of Body Ownership: A Sensory Network for Bodily Self-Consciousness. Cereb. Cortex 2007, 17, 2235–2244.
- Pascual-Marqui, R.D. Instantaneous and lagged measurements of linear and nonlinear dependence between groups of multivariate time series: Frequency decomposition. arXiv 2007, arXiv:0711.1455.
- Bullock, T.H.; McClune, M.C.; Enright, J.T. Are the electroencephalograms mainly rhythmic? Assessment of periodicity in wide-band time series. Neuroscience 2003, 121, 233–252.
- Fingelkurts, A.A.; Fingelkurts, A.A.; Kallio, S.; Revonsuo, A. Cortex functional connectivity as a neurophysiological correlate of hypnosis: An EEG case study. Neuropsychologia 2007, 45, 1452–1462.
- De Pascalis, V. Phase-ordered gamma oscillations and the modulation of hypnotic experience. In Hypnosis and Conscious States: The Cognitive Neuroscience Perspective; Jamieson, G.A., Ed.; Oxford University Press: New York, NY, USA, 2007; pp. 67–89.
- Woody, E.Z.; Sadler, P. Dissociation theories of hypnosis. In The Oxford Handbook of Hypnosis: Theory, Research, and Practice; Oxford University Press Inc.: New York, NY, USA, 2008; pp. 81–110.
- Woody, E.Z.; Farvolden, P. Dissociation in Hypnosis and Frontal Executive Function. Am. J. Clin. Hypn. 1998, 40, 206–216.
- Jamieson, G.A.; Woody, E. Dissociated control as a paradigm for cognitive neuroscience research and theorizing in hypnosis. In Hypnosis and Conscious States: The Cognitive Neuroscience Perspective; Jamieson, G.A., Ed.; Oxford University Press: New York, NY, USA, 2007; pp. 111–129.
- Dienes, Z.; Perner, J. Executive control without conscious awareness: The cold control theory of hypnosis. In Hypnosis and Conscious States: The Cognitive Neuroscience Perspective; Jamieson, G.A., Ed.; Oxford University Press: New York, NY, USA, 2007; pp. 293–314.
- Demertzi, A.; Vanhaudenhuyse, A.; Noirhomme, Q.; Faymonville, M.-E.; Laureys, S. Hypnosis modulates behavioural measures and subjective ratings about external and internal awareness. J. Physiol.-Paris 2015, 109, 173–179.
- Lipari, S.; Baglio, F.; Griffanti, L.; Mendozzi, L.; Garegnani, M.; Motta, A.; Cecconi, P.; Pugnetti, L. Altered and asymmetric default mode network activity in a “hypnotic virtuoso”: An fMRI and EEG study. Conscious. Cogn. 2012, 21, 393–400.
- Faerman, A.; Spiegel, D. Shared cognitive mechanisms of hypnotizability with executive functioning and information salience. Sci. Rep. 2021, 11, 5704.
