Liver disease-related mortality is a major cause of death worldwide. Hepatic innate and adaptive immune cells play diverse roles in liver homeostasis and disease. Myeloid-derived suppressor cells (MDSCs) are a heterogeneous population of immature myeloid cells. MDSCs can be broadly divided into monocytic MDSCs and polymorphonuclear or granulocytic MDSCs, and they functionally interact with both liver parenchymal and nonparenchymal cells, such as hepatocytes and regulatory T cells, to impact liver disease progression. The infiltration and activation of MDSCs in liver disease can be regulated by inflammatory chemokines and cytokines, tumor-associated fibroblasts, epigenetic regulation factors, and gut microbiota during liver injury and cancer.
- myeloid-derived suppressor cells
- liver inflammation
- fibrosis
- hepatocellular carcinoma
- cell–cell interaction
1. Introduction
2. The Classification and Markers of MDSCs in Mouse and Human Livers
MDSCs are a heterogenous population of immature myeloid cells [17][23]. In mice, MDSCs (CD11b+GR-1+ cells) are broadly divided into two subpopulations (Figure 1): monocytic MDSCs (M-MDSCs, CD11b+Ly6G−Ly6Chigh cells) and polymorphonuclear or granulocytic MDSCs (PMN- or G-MDSCs, CD11b+Ly6G+Ly6Clow cells) [18][24]. In humans, MDSCs (CD11b+CD33+HLA-DR−Lin−) can also be further divided into two populations using biomarkers of CD15, CD14, CD66b, and interleukin/IL-4Rα [19][25]: M-MDSCs (CD15−CD14+CD66b−IL-4Rα+) and PMN-MDSCs (CD15+CD14−CD66b+IL-4Rα−) (Figure 1).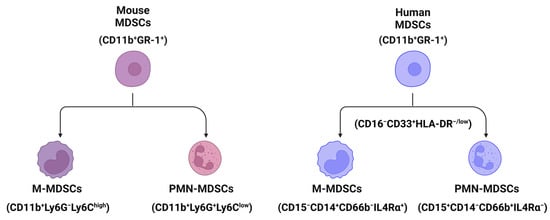
3. Pathogenesis of MDSCs in Liver Disease
3.1. MDSCs in Liver Inflammation
3.2. MDSCs in Hepatic Cell Death
Hepatic cell death happens in all different acute and chronic liver diseases with different types of cell death models [29][30][45,46], such as cell apoptosis, pyroptosis, ferroptosis, necrosis, and necroptosis. Bone marrow-derived MDSCs induced by the granulocyte-macrophage colony-stimulating factor (GM-CSF) after the stimulation of tumor necrosis factor-alpha (TNF-α) and lipopolysaccharide (LPS) display a protective effect against a lethal dose of acetaminophen (APAP)-induced liver failure by reducing liver infiltration of elastase-expressing neutrophils and inducing apoptosis of activated neutrophils [31][47].3.3. MDSCs in Liver Fibrosis and Cirrhosis
Bone marrow cells including CD11+Gr-1highF4/80− cells and CD11+Gr-1highF4/80+ cells can suppress the expression of collagen and α-smooth muscle actin in activated hepatic stellate cells (HSCs) in vitro and in vivo [32][48]. Accumulation of M-MDSCs (CD11b+Ly6G−Ly6C+ cells) in the livers of mice undergoing bile-duct ligation can inhibit the development of liver fibrosis [33][49]. The number of granulocytic MDSCs (G-MDSCs) has been shown to be increased in the livers of patients with alcoholic liver cirrhosis (ALC), which is positively correlated with the number of G-MDSCs in peripheral blood [34][50].3.4. MDSCs in Hepatocarcinogenesis
In mice with fatty liver and graft injury, arachidonic acid can activate nucleotide-binding oligomerization domain-like receptor family pyrin domain-containing 3 (NLRP3) inflammasome in MDSCs through fatty acid transport protein 2 (FATP2), which can increase IL-17 production in CD4+ T cells to cause tumor recurrence [35][51]. Accumulation of Toll-like receptor 4 (TLR4)-positive monocytic MDSCs in liver graft, which is driven by CXCL10-mediated mobilization, can increase the incidence of HCC recurrence after transplantation. In contrast, HCC recurrence can be suppressed by knocking down or suppressing the CXCL10 or TLR4 signaling pathways [36][52].4. The Interactions of MDSCs with Liver Parenchymal and Nonparenchymal Cells
4.1. Interaction with Parenchymal Cells
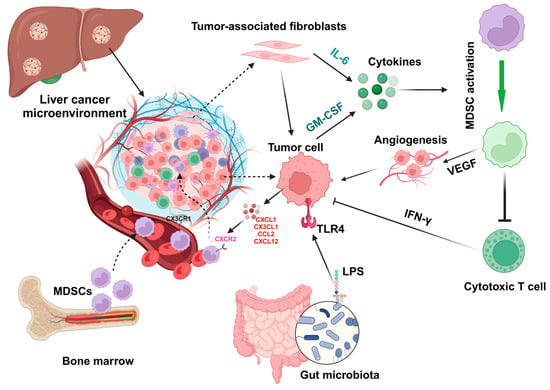
The accumulation of MDSCs in liver injury or cancer is driven by the chemokines/cytokines and their receptors. MDSCs express several chemokine receptors such as CCR2, CXCR2, CXCR4, and CXCR5, while liver tumor cells or malignant hepatocytes express chemokines such as CCL2, CCL5, CXCL1, CXCL5, and CXCL12, and the chemokine/its receptor axis mediates MDSC infiltration in the tumor microenvironment [37][38][39][55,56,57]. The upregulation of hepatic expression of CXCL1 and S100A9 protects fulminant hepatitis by inducing MDSC accumulation [40][58]. The function and infiltration of MDSCs can be changed in different HCC models, such as a diethylnitrosamine-induced HCC model and a subcutaneous tumor model induced by injection of tumor cells [41][59]. Cytokines such as granulocyte-colony stimulating factor (G-CSF) and GM-CSF secreted from tumor cells can activate MDSCs to express vascular endothelial growth factor (VEGF) and immunosuppressive factors, resulting in angiogenesis and suppression of immune cells [42][60].4.2. Interaction with Nonparenchymal Cells
In addition to hepatocytes, LSECs and HSCs can also express CXCL12 to attract the infiltration of MDSCs to the liver tumor microenvironment [38][43][56,61]. Activation of MDSCs induced by HSC-condition medium can suppress CD4+ and CD8+ T cell proliferation by upregulating the gene expression of inducible nitric oxide synthase (iNOS), arginase 1 (Arg-1), and IL-4Rα [43][61]. The interaction of HSCs with MDSCs is mediated by the molecular-binding prostaglandin E2 (PGE2) and its receptor 4 (EP4), which specifically induce the subset differentiation of G-MDSC [43][61]. Accumulation of tumor-infiltrating MDSCs including both G-MDSCs and M-MDSCs can also be regulated by chemokine CX3CL1 in HCC, which is upregulated by adoptive transfer of cytokine-induced killer (CIK) cells, a mixture of immune cells.5. Factors That Impact MDSC Infiltration and Function during Liver Injury
5.1. Inflammation
Proinflammatory cytokine IL-1β can induce overexpression of solute carrier family 7 member 11 (SLC7A11) in HCC cells to enhance tumor metastasis. The upregulation of SLC7A11 induces the infiltration of tumor-associated macrophages (TAMs) and MDSCs by activating the colony-stimulating factor 1 (CSF1)/colony-stimulating factor 1 receptor (CSF1R) axis [44][65]. Inflammatory mediators such as CX3CL1 and IL-13 in the HCC tumor microenvironment can regulate the infiltration of MDSCs (Figure 2) that contribute to the immunosuppressive function of cytokine-induced killer cells [45][62].