Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Jason Zhu and Version 1 by Ewa W.A. Szczepańska-Sadowska.
In each vascular bed, the blood flow depends on perfusion pressure, the rheologic properties of the blood, the structural and functional properties of the vascular wall, and the action of vasoconstrictory and vasodilatory factors produced locally or inflowing from the systemic circulation. The fundamental role of the heart in the maintenance of blood circulation requires the particularly precise regulation of the coronary blood flow (CBF). Rhythmic contractions of the heart exert direct mechanical effects on the coronary vessels that are associated with the production of metabolic factors in the cardiac myocytes and the smooth muscle cells of coronary vessels.
- angiotensin
- diabetes mellitus
- heart failure
- hypertension
1. Introduction
In each vascular bed, the blood flow depends on perfusion pressure, the rheologic properties of the blood, the structural and functional properties of the vascular wall, and the action of vasoconstrictory and vasodilatory factors produced locally or inflowing from the systemic circulation. The fundamental role of the heart in the maintenance of blood circulation requires the particularly precise regulation of the coronary blood flow (CBF). Rhythmic contractions of the heart exert direct mechanical effects on the coronary vessels that are associated with the production of metabolic factors in the cardiac myocytes and the smooth muscle cells of coronary vessels. The local cardiac vasoactive compounds regulate CBF in concert with cardiovascular compounds inflowing from the systemic circulation, and with the neurogenic control of the heart by the autonomic nervous system [1]. Considerable evidence shows that the mechanical performance of the cardiac muscle and the production of the vasoactive factors are significantly affected in cardiovascular and metabolic diseases [2,3,4][2][3][4]. Among factors that regulate CBF and cardiac metabolism, are components of the renin–angiotensin system (RAS), vasopressin system (VS), and insulin [5,6,7,8,9][5][6][7][8][9]. In addition, the activation of RAS generates oxidative stress, which causes the overproduction of reactive oxygen species (ROS), as well as significant disturbances of sarcolemma, sarcoplasmatic reticulum, and myofibrils that contribute to the development of cardiomyopathy [10,11,12,13][10][11][12][13]. An increasing number of studies provide evidence that RAS and VS participate in the regulation of metabolism and closely cooperate with insulin in this respect. Insulin resistance, cerebral glucose hypometabolism, cardiac autonomic neuropathy, and cardiovascular disturbances belong to serious complications of the diabetes mellitus of type 1 (T1DM) and type 2 (T2DM), alongside prediabetes (pre-DM) and metabolic (MetS) syndromes [14]; however, the causative links of these disorders are not yet fully understood [15,16,17,18][15][16][17][18].
2. Renin-Angiotensin System
2.1. Components of Renin-Angiotensin System
The RAS includes renin, angiotensinogen, and angiotensin peptides. Renin is a highly potent enzyme, acting on the N-terminal of angiotensinogen (AGT), which, in humans, is a molecule that is composed of 485 aminoacids. Renin removes a decapeptide angiotensin I [(Ang I, Ang-(1-10)] which can be converted to either the highly active angiotensin II [(Ang II, Ang-(1-8)] via the angiotensin-converting enzyme 1 (ACE1), or to the less active Ang-(1-9) via the angiotensin-converting enzyme 2 (ACE2). Ang-(1-7) can be cleaved either from Ang-(1-9) by ACE1 or from Ang-(1-8) by ACE2 (Figure 1). Other components of the RAS system [Ang III, Ang-(2-8), Ang IV, Ang-(3-8), Ang-(1-5), Ang-(5-8), Ang-(1-12)] are produced by enzymatic transformations that engage carboxypeptidases and endopeptidases.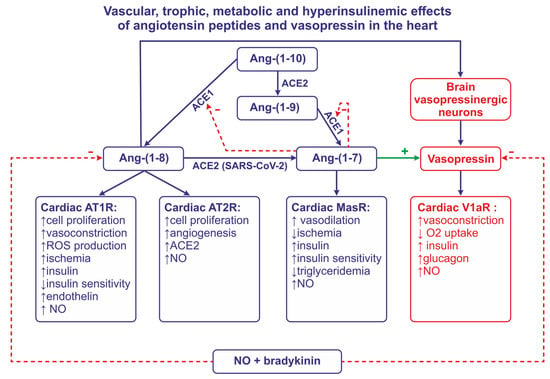
Figure 1. This figure illustrates the role of angiotensin converting enzyme -1 (ACE1), angiotensin converting enzyme-2 (ACE2), nitric oxide (NO), and bradykinin in formation and the release of angiotensin II (Ang-(1-8) and Ang-(1-7). This figure also summarizes effects of Ang-(1-8) and Ang-(1-7) on the release of vasopressin and insulin, and the effects of angiotensins and vasopressin on coronary vessels, insulin sensitivity, the formation of NO, and trophic and metabolic processes in the heart. Abbreviations: Ang—angiotensin, AT1R—angiotensin receptor of type 1, AT2R—angiotensin receptor of type 2, MasR—MAS receptor, SARS-CoV-2—severe acute respiratory syndrome associated coronavirus, V1aR—vasopressin receptor.
2.2. Cardiac Effects of Ang II
Thus far, the regulation of the functions of cardiomyocytes and coronary vessels by Ang II have been assessed, mainly in experimental studies.2.2.1. Action of Ang II on Cardiac Muscle
Angiotensin receptors are present in both the coronary vessels and the cardiac muscle (Figure 2). The studies demonstrating the autocrine release of Ang II from the mechanically stretched cardiomyocytes suggested that Ang II may be produced in the heart and may contribute to the development of stretch-induced hypertrophy [21]. The essential role of AT1R in the appropriate functions of the heart was confirmed in studies conducted on control mice and mice with AT1aR and AT1bR knockout; the knockout resulted in atrophic changes of the myocardium, in the abolishment of the Ang II–induced decrease of coronary blood flow, and in the reduction of the left ventricle pressure [22].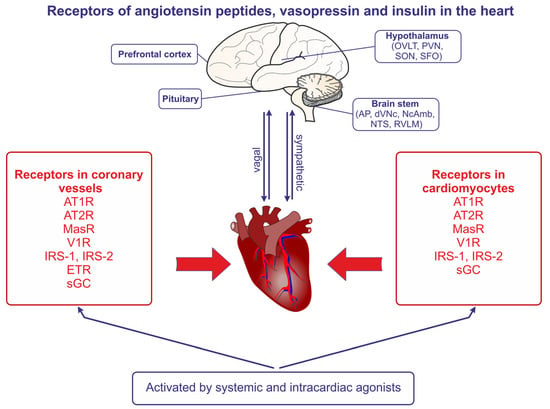
Figure 2. The presence of angiotensin II receptors of type 1 (AT1R) and type 2 (AT2R), the angiotensin- (1-7) receptor (MasR), the vasopressin V1 receptor (V1aR), the endothelin receptor (ETR), the nitric oxide receptor (sGC, soluble guanylyl cyclase), the insulin receptor substrate of type 1 (IRS-1), and the insulin receptor substrate of type 2 (IRS-2) in the coronary vessels and the cardiomyocytes of the heart. The regions of the brain expressing receptors of angiotensins and vasopressin are also indicated, and the bilateral sympathetic and vagal connections between the brain and the heart are shown. Abbreviations: AP—area postrema, dVNc—dorsal nucleus of vagus nerve, NcAmb—nucleus ambiguus, NTS—nucleus of the solitary tract, OVLT—organum vasculosum of the lamina terminalis, PVN—paraventricular nucleus, RVLM—rostral ventrolateral medulla, SFO—subfornical organ, SON—supraoptic nucleus.
2.2.2. Action of Ang II on Coronary Vessels
Historically, the first extensive study exploring the effects of the systemic administration of Ang II on coronary vessels was performed in 1976 by Giacomelli et al. [27] on Holtzman rats. The study demonstrated that Ang II, delivered in a hypertensive dose (1.7 μg/min/kg) for 4 h, induced the injury of coronary vessels; this was manifested by the increased permeability of the epicardial arteries and lesions of intramural arteries and arterioles. Next, experiments on Wistar Kyoto rats showed that the infusion of Ang II at the same rate for 2 h increased blood pressure and elicited the vasoconstriction of intracardiac intramural arteries and arterioles which were associated with endothelial cell vacuolization, smooth muscle cell fragmentation, and necrosis [28]. Experiments on trained rats revealed that the administration of Ang II prominently reduced the density of the left ventricle (LV) vessels of the trained animals performing repetitive exercises for 10 weeks [29]. Human in vitro studies. Experiments on the cultured coronary artery smooth muscle cells of humans showed that Ang II enhances the migration and proliferation of these cells and that this process can be blocked by AT1R antagonist valsartan [30]. There is evidence that the growth-promoting action of Ang II on the smooth muscle cells of human coronary vessels requires activation of the mammalian target of the rapamacine (mTOR)-sensitive signaling pathway, as well as of phosphatidylinositol 3-kinase, p70(s6k), and eukaryotic inhibitor factor-4E [31]. Human coronary artery endothelial cells (HCAECs) also respond to moderate concentrations of Ang II with the upregulation of the proteins that promote tube formation, such as signal transducers and the activator of transcription 3 (STAT3) and mir-21; it can also be presumed that Ag II is involved in the process of vasculogenesis [19,32][19][32]. Experimental evidence for the role of intracardiac RAS. With regard to the potential role of intracardiac Ang II in coronary vessels, there is evidence for the presence of some of the components of RAS in the heart. Experiments on cultured bovine aortic endothelial cells provided evidence that these cells are able to synthesize and secrete angiotensin peptides, and the authors have suggested that the coronary endothelial cells may possess similar properties [33]. Furthermore, the employment of quantitative in vitro autoradiography in the rat allowed for the demonstration of the presence of ACE in the endothelium and the smooth muscles of the coronary vessels, aorta, and pulmonary artery [34,35,36][34][35][36]. Studies on rat coronary endothelial cells (CES) and vascular smooth cells (VSMC) visualized AT1R in endothelial cells and AT2R in both types of cells. Ang II induced the AT1R-dependent proliferation of VSMC, whereas, in CES, the proliferative effect could be observed after blockade of AT2R. In addition, in CES the antiproliferative action could be observed after the administration of a selective agonist of AT2R (CGP 42112). Altogether, the results suggested that Ang II is able to promote the proliferation of VSMC and can also suppress the proliferation of CES [37]. Both AT1R and AT2R were demonstrated in the smooth muscle cells of porcine coronary artery explants, and both types of these receptors participated in the stimulation of the migration of smooth muscle cells by Ang II [38] (Figure 1). Connections of Ang II with NO and hypoxia. Strong evidence points to the important role of nitric oxide (NO) in the regulation of coronary vessels by Ang II. Earlier studies indicated that the long-term inhibition of NO synthesis via the administration of N(ω)-nitro-L-arginine methyl ester (L-NAME) in Wistar Kyoto rats enhanced the activation of ACE and induced the increase of the wall-to-lumen ratio and the enhancement of perivascular fibrolysis, thereby exhibiting the remodeling of coronary vessels. These effects were markedly reduced by the administration of an ACE inhibitor (tamocapril) and by the blockage of AT1R [39,40][39][40]. Other studies confirmed the important role of Bk and NO in the ACE-mediated vasodilation of coronary vessels [41,42][41][42]. In vitro studies on endothelial cells of male Wistar rats showed that Ang II increases NO production in the proliferating cells and that this effect is mediated both by AT1R and AT2R [43]. It is likely that the interaction between Ang II and NO plays a role in the adaptation of coronary endothelial cells to hypoxia. Experiments on mice provided evidence that, under hypoxic conditions, Ang II acting on AT2R induces endothelial sprout formation and that this effect is mediated by Bk and NO [44]. Furthermore, it has been shown that the exposure of human coronary endothelial cells to hypoxia increases the phosphorylation of JNK and the activity of hypoxia-inducible factor-1α (HIF-1α). Moreover, together with hypoxia, Ang II increases the secretion of visfatin, which is an adipocytokine with angiogenic properties. The authors found that hypoxia or hypoxia combined with an application of hyperbaric oxygen increases glucose uptake and promotes migration and tube formation in the cells. These effects could be blocked by the AT1R antagonist losartan [45]. Interaction of Ang II and endothelin. There is evidence that Ang II cooperates with endothelin (ET) in the regulation of CBF. The majority of endothelin-1 (ET-1) originates from endothelial cells where its synthesis is stimulated by Ang II, thrombin, and inflammatory cytokines [46,47][46][47]. It has been found that cultured endothelial coronary cells co-express Ang II and ET-1 and that the exposure of these cells to isoproterenol or high potassium concentrations increases the co-expression of these peptides, whereas exposure to sodium nitroprusside or S-nitroso-N-acetyl penicillamine (SNAP) decreases Ang II and ET-1 secretion [36]. Other studies have shown that the stimulation of α1 adrenergic receptors in rat cardiomyocytes increases the production of Ang II, which subsequently stimulates the formation of ET-1 through the activation of NADP oxidase [47]. In healthy awake swine, a separate blockade of AT1R and ETA/ETB receptors produced similar vasodilatory responses as did the combined blockade of these receptors; however, afterwards, the myocardial infarction responsiveness to both peptides was significantly altered [48].2.2.3. Ang II and Epicardial Adipose Tissue
The electrical and mechanical function of the heart as well as coronary blood flow can also be regulated by bioactive compounds generated in the epicardial adipose tissue (EAT) [49]. The pericardial fat is a source of adipokine, anti-inflammatory cytokine (IL-10), pro-inflammatory cytokine (IL-6), and other pro-inflammatory factors, (IFNγ, MCP1) regulating the function of the cardiac muscle and coronary blood vessels. In patients with cardiovascular diseases, the production of ROS in EAT is higher than in the subcutaneous adipose tissue [50]. There is also evidence for activation RAS in the EAT [51,52][51][52].2.3. Cardiac Effects of Ang-(1-7) and Other Angiotensins
In many instances, Ang-(1-7), when acting on MasR, exerts similar effects to those observed after the stimulation of AT2R by Ang II, and is involved in the cardioprotective factor (Figure 1). The mechanism of the cardioprotective action of Ang-(1-7) is not yet fully recognized; however, it appears that it depends on the generation of NO [53,54,55,56][53][54][55][56]. There is also evidence that Ang-(1-7) acts as an ACE1 inhibitor, and that it potentiates the vasodilatory effect of bradykinin via B2 receptors [57]. The possibility that the vasodilatory action of Ang-(1-7) in the coronary vessels is mediated by Bk and NO has been confirmed in experiments on hearts isolated from guinea pigs and Wistar rats [58,59][58][59]. Also, other angiotensin peptides [Ang-(1-2), Ang-(1-3), Ang-(1-4), Ang-(1-5)] reduce the pressure perfusion of the rat coronary vessels, and it is likely that their action depends on the stimulation of Mas receptors and the release of NO [60].2.4. Centrally Mediated Effects of Angiotensin Peptides
The local action of Ang II in the heart may be potentiated by the interaction of this peptide with cardiovascular neurons in the central nervous system. Although the systemic Ang II does not cross the blood–brain barrier, it can act in the subfornical organ (SFO), and it can activate neural pathways projecting from the SFO to the paraventricular nucleus (PVN), where it can stimulate AVP secretion and the activation of the sympathetic outflow [61,62,63][61][62][63]. The systemic administration of relatively small doses of Ang II in mice was found to elevate blood pressure, plasma AVP, and endothelin-1 (ET1) levels, and to decrease cerebral blood flow (CBF). As the above effects were significantly reduced by the intra-brain applications of the AT1R antagonist (losartan), the AVP V1aR antagonist (SR49059), and the ROS scavenger (MnTBAP), it is possible that they were mediated by Ang II, which penetrated to the brain, and through the stimulation of the AT1R-elevated release of AVP, the activation of V1aR, and the generation of ROS [64].2.5. Angiotensin Peptides in Regulation of the Heart in Cardiovascular Diseases
Multiple experimental studies provide evidence that the excessive activation of the RAS pathway importantly contributes to the initiation of pathogenic responses in the heart (Figure 2). The most advanced studies refer to the role of Ang II and Ang-(1-7).2.5.1. Experimental Evidence for the Negative Role of Ang II in the Heart in Myocardial Infarction and Hypertension
Experiments on sham-operated rats and rats with myocardial infarction revealed that the prolonged administration of ACE inhibitor quinapril helps to preserve high energy phosphate metabolism in the infarcted rats, which suggested that endogenously produced Ang II exerts a negative effect on the metabolism of the infarcted heart [65]. The administration of the ACE inhibitor (enalaprilat) in a porcine model of myocardial infarction significantly reduced the area of necrosis and reduced regional cell motion [66]. The positive effects of ACE inhibition and AT1R blockade during myocardial ischemia could also be caused by the increased production of NO and Bk [67,68,69][67][68][69]. Studies on dogs with pacing-induced heart failure revealed that the vasoconstrictory effect of AT1R stimulation on the coronary vessels is markedly attenuated in cardiac heart failure, which suggested the desensitization of the coronary vessels to the vasoconstrictory effect of Ang II. The authors suggested that the desensitization to Ang II may be caused by the accumulation of some vasodilatory compounds, for instance bradykinin and/or NO [70]. Indeed, the inhibition of ACE and blockade of AT1R increased cardiac bradykinin and NO levels, and elevated coronary blood flow in another study on dogs with myocardial ischemia induced by the reduction of coronary perfusion pressure [71]. The AT1R blockade restored endothelial NO synthase, contractile-type myosin heavy chain isoform SM2, and calponin and Gata-6 levels in the coronary vessels of SHR rats [72]. Subsequent studies showed that blockade of AT1R by losartan in rats with the myocardial infarction improved cardiac performance and reversed several negative biochemical processes, such as elevated sarcoplasmic reticulum Ca2+ uptake, the activation of Ca2+ pump protein, and phospholamban protein, as well as the stimulation of myofibrils (MF) Ca2+-activated ATPase, myosin heavy chain (α-MHC) mRNA, and β-MHC mRNA [73]. There is evidence that the myocardial infarction modulates interactions between AT1R and ETA/ETB receptors in the coronary vessels. For instance, it was shown that in healthy awake swine, the separate blockade of AT1R and ETA/ETB receptors produced similar vasodilatory responses as the combined blockade of these receptors [48]; however, 2–3 weeks after the myocardial infarction, the coronary vasodilatory responses to individual blockades of AT1R and ETA/ETB receptors were abolished in spite of that fact that the expression of AT1R and ETA receptors was not altered. In addition, blockade of ETA/ETB receptors in presence of blockade of AT1R was able to produce coronary vasodilation in the infarcted swine. Thus, the authors postulated that under control conditions, Ang II and endothelin act in the coronary vessels as independent vasoconstrictory peptides, whereas the myocardial infarction initiates cross-talk interactions between these compounds at the post-receptor level [48]. The effectiveness of the stimulation of AT1R may depend on the accessibility of the AT1R-associated protein (ATRAP), which is able to enhance the internalization of AT1R from the cell surface to cytoplasm and reduce the action of Ang II [74]. More recently, studies performed on non-culprit arteries harvested from the rabbit model of myocardial ischemia-reperfusion provided evidence that the expression of AT1R was higher in the ischemia-reperfusion group than in the sham group, but expressions of AT1R, connexin 43, and β-tubulin were lower in the ischemic postconditioning group than in the ischemia reperfusion group [75]. There is also evidence for the enhanced involvement of central AT1R in blood pressure regulation in hypertension. Blockade of central AT1R with losartan significantly reduced the hypertension of renin transgenic rats, although it was not effective in normotensive SD rats [76]. Furthermore, it was shown that cardiac sympathetic afferent reflex and renal sympathetic activity were enhanced in SD rats with the myocardial infarction, and that these effects were associated with the elevated stimulation of AT1R receptors by Ang II in the PVN [77]. The central effects of angiotensin peptides may play an especially significant role when the cardiovascular disease is associated with stress. For instance, it has been found that prolonged (2 weeks) restraint stress of C57BL/6J mice enhanced the expression of the mRNA levels of angiotensinogen, TNF-α, IL-6, monocyte chemoattractant protein-1 (MCP-1), insulin receptor substrate (IRS-1), and glucose transporter 4 (GLUT4), and could, in this way, promote the synthesis of angiotensin peptides and cytokines as well as the development of insulin insensitivity [78].2.5.2. Negative Role of Ang II in the Heart in Human Heart Failure
Positive effects of ACE inhibition and AT1R blockade were also found in human patients with cardiac failure. In patients undergoing coronary arteriography blockade of AT1R with losartan improved epicardial blood flow during a stress-induced cold pressure test and during exercise [79]. Moreover, prolonged therapy with ACE inhibitors promoted the development of collateral circulation in patients with coronary artery stenosis, presumably due to coronary artery angiogenesis [80]. Studies analyzing the expression of ACE in samples of coronary vessels obtained from patients suffering from coronary disease revealed the presence of ACE in the atherosclerotic plaques [81]. In addition, experiments on coronary arterioles obtained from patients suffering from atherosclerosis showed that the incubation of these vessels with an ACE inhibitor (Lisinopril) significantly ameliorated the vasodilatory responses to several endothelium-dependent agonists. The latter effect could be abolished by pretreatment with NO synthase inhibitor [82]. In patients with chronic heart failure, the combined application of an ACE1 inhibitor (enalapril) and losartan for 16 weeks significantly reduced plasma insulin levels, homeostatic model assessment of insulin resistance (HOMA-IR), TNF-α, interleukin-6, and MCP-1 levels. Correlations were found between the reduction of HOMA-IR and decreases of IL-1 and MCP-1, which suggested the significant role of activation ACE1 and AT1R in the regulation of cytokines and insulin secretion [83]. Studies in vitro using the model of endothelium-denuded coronary vessels revealed that levels of AT1R mRNA and AT2R mRNA were lower in the smooth muscle cells of patients with heart failure than in the control subjects [84]. The expression of AT2R was demonstrated in atherosclerotic plaques that were isolated from the internal coronary artery of human patients [85].2.5.3. Positive Aspects of Action of ACE2 and Ang-(1-7) in Heart Failure
There is evidence that cardiac failure causes the activation of ACE2 and the stimulation of Ang-(1-7) pathways (see Figure 1). Experimental studies. Experiments on control and infarcted Wistar rats, which were treated or non-treated with selective Ang-(1-7) agonist, Ang-(1-7) antagonist, and with N(G)nitro-l-arginine methyl ester (L-NAME), provided evidence that Ang-(1-7) improves the hemodynamic parameters of the heart and decreases the infarcted area through actions mediated by NO [86]. Prolonged blockade of ACE2 activity in male Wistar rats was found to increase the myocardial infarct size and reduce the left ventricle percentage shortening [87]. Human studies. Experiments on human coronary endothelial cells provided evidence that the stimulation of AT2R increases the expression of ACE2 and that the stimulation of ACE2 and AT2R results in the inhibition of stimulatory effects of TNF α on IkB and NF-kB signaling, which may suggest that the activation of ACE2 and AT2R exerts anti-inflammatory action in these cells [88]. Recently, it has been found that circulating levels of ACE2 and Ang-(1-7) are significantly elevated in patients suffering from coronary artery disease, and that the levels of ACE2 are significantly higher in the female groups than in the male groups of patients [89]. It should be noted that ACE2 acts as a receptor for SARS-CoV-2 (Figure 1) and that the altered activity of ACE2 and/or SARS-CoV-2 may interfere with the development of atherosclerotic lesions in COVID-19 infections; indeed, SARS-CoV-2 viral mRNA is present in atherosclerotic plaques [90]. Moreover, SARS-CoV-2 infection increases the production of pro-atherogenic and proinflammatory cytokines, such as IL-1β and IL-6 [90,91][90][91]. Recently, it has been reported that patients infected with SARS-CoV-2 produce autoantibodies that are able to inhibit ACE2 activity and intensify the severity of COVID-19 [92].2.5.4. Genotypes of ACE and Angiotensin Receptors as Determinants of Coronary Diseases
There is evidence that, to some extent, susceptibility to the myocardial infarction and coronary artery disease may be determined by the specific polymorphism of the ACE genotype. The study comparing the distribution of ACE genotypes in Korean patients suffering from acute coronary syndrome and in healthy subjects indicated that the genotype DD of the ACE gene may be an independent risk factor of acute coronary syndrome [93]. There is evidence for the association of the insertion/deletion (ID) polymorphism of ACE with responsiveness of coronary vessels to NO-mediated vasodilation, i.e., patients with the ACE DD genotype presented significantly lower vasodilatory responses to sodium nitroprusside (SNP) than the other groups of patients [94]. The detection of ACE polymorphism in the leukocytes of patients suffering from coronary artery disease provided evidence that ACE genotype polymorphism may be associated with the development of atherosclerotic plaques. Specifically, it has been shown that DD and ID genotypes manifest higher numbers of diseased coronary vessels, whereas genotype I shows smaller numbers of atherosclerotic lesions [95]. The D allele of the ACE gene was also found to be a strong and independent risk factor for coronary artery disease in patients with non-insulin-dependent diabetes mellitus [96]. The polymorphism of the angiotensinogen M235T genotype occurs more frequently in patients with acute myocardial infarction than in the healthy control subjects [97]. The role of the specific genotype of AT1R in susceptibility of human coronary vessels to vasoconstrictory factors was determined in patients undergoing arteriography, among whom it was found that the subjects with the CC genotype of AT1R manifest significantly greater responsiveness to methylergonovine maleate, which is a potent vasoconstrictor [98].2.6. Interaction of Angiotensin Peptides with Insulin
2.6.1. Interaction of Angiotensin II with Insulin
Experimental and clinical studies provide evidence for the complex interactions between angiotensin peptides and insulin in the regulation of the coronary blood flow and the myocardial function [6,99][6][99]. Thus, prolonged (7 weeks) hyperinsulinemia decreased the expression of AT1R and increased the expression of AT2R in the atrium of SD rats, whereas it enhanced the expression of both types of these receptors in the left ventricle [99]. Moreover, hyperinsulinemia elicited an increase of the LV mass and relative wall thickness and reduced the stroke volume and cardiac output. These changes were associated with the hypertrophy of myocytes, interstitial fibrosis, and increased phosphorylation of IRS-1, ERK 1/2, MEK1/2, Akt, and PI3K. It is likely that insulin could cooperate with the sympathetic system, because its effects could be significantly attenuated by the application of metoprolol, an antagonist of beta-adrenergic receptors [99]. On the other hand, Ang II was found to participate in the development of insulin resistance and endothelial dysfunction in atherosclerosis, whereas the inhibition of ACE exerted positive effects in animal models of cardiometabolic syndrome [100]. In SD rats maintained on a fructose-rich diet, which promotes the development of hypertension and insulin resistance, the administration of the ACE inhibitor or AT1R blocker (Olmesartan) significantly reduced blood pressure, improved insulin sensitivity, and reduced adipocyte size [101]. Further studies revealed that blockade of AT1R in insulin-treated rats significantly potentiated NO-mediated vasodilation [102].2.6.2. Interaction of Ang-(1-7) with Insulin
Ang-(1-7) appears to interact with insulin in the opposite way to Ang II. The subcutaneous (sc) infusion of Ang-(1-7) was found to reduce insulin resistance, hypertriglyceridemia, obesity, and hepatic fat accumulation in rats maintained on a high fructose/low-magnesium diet, which imitates the metabolic syndrome [103]. Similarly, the sc administration of Ang-(1-7) improved insulin sensitivity in C57BL/6J mice maintained on a high-fat diet and this effect was associated with increased glucose uptake into the skeletal muscle [104]. Ang-(1-7) also exerts other effects that may potently contribute to the regulation of metabolism via insulin. For instance, it has been found that it induces the proliferation of pancreatic β-cells, increases insulin secretion, ameliorates the sensitivity of skeletal muscles and adipose tissue to insulin, and improves the metabolic function of the liver [105].2.6.3. Central Effects of Insulin
It should be noted that insulin receptors and insulin-like growth factor 1 receptors are present in the central nervous system in the regions involved in the regulation of the sympathetic nervous system, metabolism, and blood pressure [106,107][106][107]. The systemic and central administration of insulin into the RVLM increases lumbar sympathetic nerve activity and this effect can be reversed by blockade of glutamatergic NMDA receptors [106]. Therefore, it is likely that the direct interaction of insulin with angiotensin peptides in the heart may be potentiated by its intra-brain action, promoting the stimulation of the sympathetic nervous system.2.7. The Role of Angiotensin Peptides in the Heart in Obesity, Diabetes Mellitus, and Hypertension
Multiple experimental and clinical studies report that a high-fat diet, obesity, and diabetes mellitus significantly influence the activity of the RAS.2.7.1. The Role of Ang II in Experimental Obesity, Diabetes Mellitus, and Hypertension
Enhanced ACE activity associated with diminished responsiveness to the vasodilatory action of bradykinin was found in the coronary arterioles of obese rats maintained on a high-fat diet [108]. The treatment of type 2 diabetic KK-Ay mice (an experimental model of T2DM) with an AT1R antagonist (valsartan) significantly improved insulin sensitivity and reduced the plasma glucose level. This was associated with the phosphorylation of IRS and with the translocation of GLUT 4 to the plasma membrane. In addition, the application of valsartan reduced the expression of TNF-α and the production of superoxide. The treatment of KK-Ay mice with an AT2R antagonist (PD123319) did not have significant effects on insulin sensitivity [109]; however, positive effects on metabolism by means of AT2R were found in another study on KK-AY mice, in which the intraperitoneal injections of AT2R agonist (compound 21, C21) reduced insulin resistance [110]. Experiments on db/db mice, which is another model of T2DM, provided evidence that activation AT1R plays a role in the remodeling of coronary vessels in diabetes mellitus. Thus, 16-week-old db/db mice manifested hyperglycemia, hyperlipidemia, obesity, insulin resistance, and coronary remodeling [111], whereas, after prolonged treatment with losartan in the dose which does not alter systemic blood pressure, the mice showed a significantly decreased number of VSMC, the reduced remodeling of the coronary vessels, and increased coronary flow reserve [112]. There is evidence for the enhanced involvement of angiotensin peptides in the development of insulin resistance and in the generation of oxidative stress in hypertension. In a model of Dahl-salt sensitive hypertension, the hypertensive animals manifested insulin resistance, increased the expression of AT1R mRNA and protein, and impaired Ach-induced endothelium-dependent relaxation (EDR). The administration of an AT1R blocker (candesartan) or an antioxidant compound (tempol) reduced arterial blood pressure and insulin resistance and normalized EDR and O2- in the wall of the aorta [113]. Furthermore, SHR rats that were maintained on a high-fat diet and were developing type II diabetes (SHRDI) manifested body-weight loss, hyperglycemia, reduced plasma insulin levels, decreased myocardial and brain capillary vascularization, and enhanced oxidative stress. The harmful effects of diabetes in SHRDI rats could be prevented by the chronic inhibition of ACE with enalapril or by blockade of an AT1R antagonist (olmesartan) [114]. Obesity, increased blood pressure, myocardial hypertrophy, and interstitial fibrosis, associated with elevated levels of ERK, PI3K, and Tyr-phosphorylated β-insulin receptor subunit (βIR) and with decreased myocardial JNK expressions, were observed in Wistar-Kyoto rats fed with a hypercaloric diet for 30 weeks. Several of these disorders, such as dyslipidemia and insulin resistance, could be efficiently reduced by the administration of losartan in drinking water [115]. It is likely that some of the detrimental effects of Ang II in obesity and diabetes mellitus are mediated by aldosterone, which closely cooperates with RAS and participates in the regulation of insulin secretion. Experiments on isolated pancreatic islets revealed that aldosterone acting on mineralocorticoid receptors (MR) enhances glucose-induced insulin secretion and ROS production. The activation of MR also decreased the sensitivity to insulin in adipocytes and in skeletal muscles [116], and promoted the development of inflammation, oxidative stress, lipid disorders, and insulin resistance, thereby impairing vascular insulin metabolic signaling [15,117,118][15][117][118].2.7.2. The Role of Ang II in Human Obesity, Diabetes Mellitus, and Hypertension
The activity of ACE is enhanced in the coronary arterioles of obese human patients [108]. Measurements of insulin sensitivity index (ISI) and evaluations of HOMA-IR in patients with impaired glucose tolerance (IGT) and in diabetes mellitus revealed that the administration of AT1R antagonist valsartan effectively reduces resting fasting insulin levels, elevates ISI and adiponectin levels, and decreases HOMA-IR and high sensitivity C-reactive protein (hsCRP) in the IGT group [119]. There is evidence that patients with prediabetes and type 2 diabetes have elevated pericardial and peri-aortic adipose tissue [120]. It has been shown that the production of prohypertensive components of the RAS, in particular angiotensinogen mRNA and ACE mRNA, is elevated in the epicardial adipose tissue of obese patients [51]. Elevated expressions of ACE2 and ADM17 (a disintegrin and metalloproteinase 17) genes have been found in the epicardial fat of patients with type II diabetes mellitus and in subjects with obesity [52]. As ADM17 is a membrane-bound enzyme, which participates in the proteolytic cleavage of proinflammatory cytokines, such as TNF-α [121], it is possible that ADM17 elevation in obesity and in type II diabetes may signalize inflammatory processes and other detrimental changes, which appear in the heart. in these diseases [122].2.7.3. The Role of ACE2 and Ang-(1-7) in Obesity, Diabetes Mellitus, and Hypertension
Experimental evidence from animals maintained on high-fat diets strongly suggests that ACE2 and Ang-(1-7) may partly oppose the effects of the activation of AT1R. Studies on ACE2 mutant (ACE2KO) mice and wild type mice provided evidence that a high-fat diet causes glucose intolerance, myocardial insulin resistance, cardiac steatosis, lipotoxicity, and the development of the proinflammatory phenotype of the adipose tissue. These effects were associated with a decrease in cardiac adiponectin, which is an anti-inflammatory adipokine of adipocytes, and could be partly removed by an administration of Ang-(1-7) that lasts for 4 weeks [123]. In the obese mice maintained on a high-fat diet, the administration of Ang-(1-7) for 28 days enlarged brown adipose tissue, upregulated thermogenesis, improved impaired glucose homeostasis, and enhanced expression UCP1 uncoupling protein-1 (UCP-1) in the brown adipose tissue [124].3. Vasopressin System
3.1. Components of Vasopressin System
Arginine vasopressin is synthesized in the neuroendocrine cells of the hypothalamus, located mainly in the supraoptic nucleus (SON), the PVN, and the suprachiasmatic nucleus (SCN) [125]. In addition, immunocytochemical and functional studies provided evidence for the synthesis of AVP in the heart and lungs [126,127][126][127]. In most mammals, AVP is a chief active peptide composed of Cys-Tyr-Phe-Gln-Asn-Cys-Pro-Arg-Gly-NH2. The AVP gene is found in 20 chromosomes and consists of exons encoding the sequence of the 145-aminoacid polypeptide precursor-forming N-terminal signal peptide, and sequences of AVP, neurophysin II (NPII), and copeptin. Copeptin is a glycopeptide, composed of 39 aminoacids released in equimolar quantities as AVP and is frequently used due to its more stable biomarker molecule [128]. Measurements of copeptin have been included in ESC guidelines on the management of non-ST elevation myocardial infarction [129]. Actions of AVP are mediated by V1 receptors (V1R), which include V1a receptors (V1aR), and V1b receptors (V1bR), and by V2 receptors (V2R), and oxytocin receptors (OTR) [20,130][20][130]. V1aR are found in the brain, the spinal cord, the heart and vessels, the kidneys, the lungs, and the digestive system (mainly in the liver and the pancreas) [131,132,133][131][132][133]. V1bR are present in the pituitary, the brain, the pancreatic gland, and the lungs [132,134][132][134]. Thus far, V2R have been located mainly in the kidney [131,135,136][131][135][136]; however, there is also some evidence for their presence in the heart [137].3.2. Regulation of Vasopressin Release
Under physiological conditions, the release of AVP is regulated largely by changes of blood osmolality. Among other stimulators are stress, hypovolemia, hypotension, hypoxia, hypoglycemia, and several neuroactive factors, whose significance increases in cardiovascular and metabolic diseases. Angiotensin peptides, in particular Ang II, and Ang-1-7 are potent stimulators of AVP secretion [9]. Furthermore, experiments on rats have shown that the intracerebroventricular (ICV) infusion of insulin increases plasma AVP in a dose-dependent manner and that this effect is not associated with systemic hypoglycemia [138]. In addition, hypoxia and hypoglycemia are able to stimulate AVP release, whereas vasopressin elevates blood glucose levels and increases brain glucose retention. There is evidence that these effects occur with the engagement of carotid chemoreceptors and NTS [139,140,141][139][140][141]. In experiments on conscious freely moving rats, it was found that the central administration of glucagon-like peptide-1 (7-36) amide (GLP1) activates the parvocellular and magnocellular neurons of the PVN and SON and elevates plasma AVP level [142]. In the PVN and SON, the vasopressinergic neurons and GLP-1 receptors are co-expressed [143].Inhibitory Role of Nitric Oxide in Vasopressin Release
Nitric oxide plays significant role in the regulation of AVP secretion. NO released in the PVN inhibits the secretion of AVP and plays an essential role in buffering pressor responses that are exerted by other hypertensive compounds, such as Ang II and endothelin-1 (ET-1) [63,144][63][144]. It is worth noting that the neuronal NO synthase (nNOS) is co-expressed with AT1R in the PVN and SON neurons and that Ang II stimulates the release of AVP and upregulates AVP mRNA levels in the PVN and SON neurons; these effects are potentiated by the inhibition of nNOS activity [145]. Earlier studies suggested that during LPS-induced endotoxemia, the secretion of AVP is inhibited by NO and CO [146,147,148][146][147][148]. More recently, experiments on the hypothalamic explants of rats provided evidence that NO reduces AVP release, whereas CO and hydrogen sulfide (H2S) can exert the opposite effect [149]. It is also likely that NO may inhibit the local cardiac release of AVP and buffer the vasoconstrictory action of this peptide in the heart [126]. In support of this assumption, experiments on pressure-overloaded hearts of the rat in which AVP mRNA and AVP peptide were detected mainly in endothelial cells and vascular smooth muscle cells [126].3.3. Cardiac Effects of Vasopressin
3.3.1. Experimental and Human Studies
Evidence for the involvement of vasopressin in the regulation of the healthy heart comes mainly from experimental studies. Earlier research provided evidence that AVP constricts coronary vessels with diameters less than 100 μm, whereas it does not exert significant effects or can even dilate larger vessels [150,151,152][150][151][152]. Experiments on the isolated hearts of male Long Evans rats showed that the application of AVP significantly reduced coronary blood flow and myocardial oxygen consumption, and that these effects were markedly attenuated during acute hypoxia. The latter finding suggested that hypoxia may significantly modulate the vasoconstrictory potency of AVP in the heart [153]. It has been shown that the vasoconstrictory effect of vasopressin on coronary vessels is mediated mainly by V1R (Figure 2) and that this can be demonstrated in humans [20,154][20][154]. However, it should be noted that coronary vasoconstriction was also observed in patients with spastic angina after the application of desmopressin (DDAVP), which is a selective agonist for V2R [155].3.3.2. The Interaction of Vasopressin with NO and Endocannabinoids in the Heart
Experimental studies provide evidence that the regulation of the coronary blood flow by vasopressin is significantly modulated by locally released NO. It has been shown that AVP and its V1a agonist [Phe2Ile3proOrn8] vasopressin significantly increase iNOS mRNA and NO production, as well as cytosolic free Ca2+ level in cultures of neonatal rat cardiac myocytes stimulated by IL-1β [156]. Studies on the isolated coronary arteries of monkeys demonstrated that acting on V1a receptors AVP can elicit vasodilation mediated by NO [157]. Further studies performed on the hearts of SD rats showed that AVP is able to stimulate inducible NO synthase (iNOS), nuclear factor kappa-B (NF-kappa B), and collagen synthesis. The results suggested that in the heart, AVP may participate in the development of cardiac fibrosis and inflammatory processes, and these effects are attenuated by the antifibrotic action of NO [158]. Negative effects of the excessive stimulation of V1aR in the heart of mice were demonstrated by Li et al. [159], who found that the selective overexpression of V1aR in the heart causes cardiac dysfunction and hypertrophy associated with ventricular dilation and an excessive activation of the Gαq/11-mediated pathway [159]. Among factors which are able to decrease the vasoconstrictory potency of vasopressin are cannabinoids, which are stimulating CB1 receptors (for instance anandamide) [160].3.4. Vasopressin in the Heart in Cardiovascular Diseases
Substantial evidence indicates that the responsiveness of coronary vessels to vasopressin is altered under pathological conditions.3.4.1. Experimental Studies
Measurements of CBF in conscious dogs with an experimentally induced area of hypoperfusion of the collateral-dependent myocardium showed that the systemic infusion of AVP during physical exercise decreased CBF in the collateral zone and that this effect was associated with an increase of resistance in the transcollateral and small coronary vessels. Under control conditions, the exercise did not have any significant effects on these parameters [161]. Recent studies have shown that during cardiac ischemia, AVP may play a beneficial role through the action exerted in mitochondria. For instance, experiments on male Wistar rats subjected to 30 min of ischemia followed by 120 min of reperfusion showed that the administration of AVP decreased the sensitivity of the mitochondrial permeability transition pore (MPTP) to the action of MPTP openers, thereby protecting mitochondria from swelling. In addition, the infusion of AVP significantly reduced the infarct size and plasma levels of lactate dehydrogenase, creatinine kinase-MB, and malondialdehyde [162].3.5. Role of Vasopressin in Obesity and Diabetes Mellitus
3.5.1. Clinical and Experimental Evidence for the Regulation of Glycemia by Vasopressin
Several studies provide evidence that vasopressin and insulin cooperate in the regulation of glycemia. It was found that the withdrawal of insulin in patients with diabetes mellitus elevated plasma vasopressin concentration [165][163], whereas the administration of AVP elicited hyperglycemia, which was associated with the stimulation of the sympathetic system [144,166][144][164]. Moreover, it has been shown that hypoglycemia stimulates the release of AVP and that the glucosensitive receptors engaged in the regulation of AVP release are located inside the blood–brain barrier [167][165]. The ICV infusion of insulin in doses that do not cause systemic hypoglycemia increased the secretion of AVP in a dose-dependent manner [138]. There is evidence that AVP participates in the regulation of glycemic responses during hypoglycemia. For instance, the central administration of vasopressin in rats was found to reduce the hyperglycemic and hyperglucagonemic responses to hypoglycemia induced by the administration of 2-deoxy-D-glucose [168][166].3.5.2. The Role of Vasopressin in the Regulation of Insulin and Glucagon Release
It has been shown that AVP actively participates in the regulation of insulin and glucagon release and closely cooperates with these hormones in the regulation of glycemia. Vasopressin has been detected in human and rat pancreatic extracts [169][167] and AVP receptors have been found in pancreatic islets of several species, including human beings. Moreover, it has been found that AVP stimulates the release of glucagon and insulin [132,170,171][132][168][169]. Interestingly, AVP was able to release insulin from beta pancreatic cells and glucagon from alpha pancreatic cells; however, the type of the cell stimulated was strongly dependent on the level of glycemia. At elevated glucose concentrations, AVP increased insulin secretion, whereas, at reduced glucose concentrations, it promoted the release of glucagon [172][170]. The latter finding suggested that AVP may play an essential role in the regulation of glucagon release during hypoglycemia. The pancreatic alpha cells secreting glucagon express high levels of the V1bR gene, and blockade of V1bR reduces the AVP-dependent release of glucagon. The effectiveness of the stimulation of glucagon release by AVP appears to be significantly impaired in patients with type 1 diabetes mellitus [173][171]. Frequently, hypoglycemia and stress stimulate the release of AVP together with CRH [167,174,175,176][165][172][173][174]. It has been also shown that the increased release of AVP during hypoglycemia significantly contributes to the stimulation of ACTH secretion [177,178,179][175][176][177]. It is likely that AVP plays an essential role in the regulation of ACTH secretion during hypoglycemia and stress, because AVP-deficient Brattleboro rats manifest reduced elevations of plasma ACTH and corticosterone levels during hypoglycemia and exposure to various types of stressors [180][178].3.5.3. The Role of Vasopressin in Diabetes Mellitus and Obesity
There is evidence that the insulin-dependent release of AVP may be markedly dysregulated in diabetes mellitus and obesity. Experimental studies. Higher numbers of AVP mRNA expressing cells were found in the PVN of nonobese diabetic mice than in the PVN of nondiabetic mice and control C57B1/6 mice [181][179]. There is evidence that the responsiveness of coronary vessels to vasopressin may significantly differ in diabetic males and females. In this respect, it has been shown that isolated coronary vessels of control female rats and female rats with streptozotocin-induced diabetes are less sensitive to the vasoconstrictive action of AVP than the corresponding vessels of control and diabetic male rats. The study provided evidence that the reduced vasoconstrictory responses to AVP in females could result from an increased production of NO [182][180]. Engagement of the AVP-V1aR pathway in the regulation of glucose homeostasis was confirmed by experiments on receptor-deficient mice (V1aR(-/-), which showed significant disturbances in glucose metabolism, such as hyperglycemia, higher hepatic glucose production, and decreased liver glycogen content. The mutant mice responded with elevation of plasma AVP levels and developed overt obesity when they were maintained on a high-caloric intake, while the same diet was not effective in wild-type mice [183][181]. Furthermore, experiments performed on the V1aR knockout mice and double V1aR/V1bR knockout mice, as well as on obese Zucker rats, revealed that the inappropriate activation of V1R induces significant disturbances in glucose and lipid metabolism, which may have an indirect detrimental effect on glucose and lipid supply to the heart [184,185][182][183]. Studies on Zucker diabetic fatty (ZDF) rats, which serve as an experimental model of type 2 diabetes, demonstrated higher levels of AVP in the PVN and SON of ZDF rats than in the PVN and SON of Zucker lean control (ZLC) rats and the pre-diabetic rats. Thus, the results provided direct evidence that type 2 diabetes is associated with the increased secretion of AVP in the hypothalamic nuclei [186][184]. Clinical studies. It has been shown that the administration of insulin elicits significantly higher elevations of plasma AVP and oxytocin levels in diabetic patients than in healthy subjects [187][185]. Higher plasma copeptin levels and higher insulin plasma concentrations were found in obese patients and patients with metabolic syndromes [188,189,190][186][187][188]. On the other hand, the diabetic patients with asymptomatic hypoglycemia responded with smaller increases of blood AVP and lower elevations of heart rate in response to hypoglycemia than the healthy subjects. These findings suggested that the appropriate secretion of AVP may play an essential role in the appropriate sensation of glycemia [191][189]. The decreased expression of hepatic V1aR mRNA was found in rats with streptozotocin-induced diabetes mellitus [192][190].3.5.4. Genotypes of Vasopressin Receptors as Risk Factors of Coronary Diseases
Certain variations of the human AVPR1A gene are associated with specific abnormalities of metabolism. It has been found that the subjects carrying the T allele of rs 1042615 and male subjects carrying rs 1042615 allele demonstrate lower concentrations of triglycerides and higher fasting blood glucose concentrations than CC carriers [193][191]. It has also been reported that the allele of rs35810727 AVPR1B gene is associated with elevated body mass index, and that the human subjects carrying this variance of the AVPR1B gene are more prone to develop obesity and diabetes mellitus [194][192].References
- Bakkar, N.Z.; Dwaib, H.S.; Fares, S.; Eid, A.H.; Al-Dhaheri, Y.; El-Yazbi, A.F. Cardiac Autonomic Neuropathy: A Progressive Consequence of Chronic Low-Grade Inflammation in Type 2 Diabetes and Related Metabolic Disorders. Int. J. Mol. Sci. 2020, 21, 9005.
- Shimabukuro, M. Cardiac adiposity and global cardiometabolic risk: New concept and clinical implication. Circ. J. 2009, 73, 27–34.
- Petrie, J.R.; Guzik, T.J.; Touyz, R.M. Diabetes, Hypertension, and Cardiovascular Disease: Clinical Insights and Vascular Mechanisms. Can. J. Cardiol. 2017, 34, 575–584.
- Severino, P.; D’Amato, A.; Pucci, M.; Infusino, F.; Adamo, F.; Birtolo, L.I.; Netti, L.; Montefusco, G.; Chimenti, C.; Lavalle, C.; et al. Ischemic Heart Disease Pathophysiology Paradigms Overview: From Plaque Activation to Microvascular Dysfunction. Int. J. Mol. Sci. 2020, 21, 8118.
- Anderson, E.A.; Hoffman, R.P.; Balon, T.W.; Sinkey, C.A.; Mark, A.L. Hyperinsulinemia produces both sympathetic neural activation and vasodilation in normal humans. J. Clin. Investig. 1991, 87, 2246–2252.
- Favre, G.A.; Esnault, V.L.; Van Obberghen, E. Modulation of glucose metabolism by the renin-angiotensin-aldosterone system. Am. J. Physiol. Endocrinol. Metab. 2015, 308, E435–E449.
- Szczepanska-Sadowska, E.; Czarzasta, K.; Cudnoch-Jedrzejewska, A. Dysregulation of the Renin-Angiotensin System and the Vasopressinergic System Interactions in Cardiovascular Disorders. Curr. Hypertens. Rep. 2018, 20, 19.
- Szczepanska-Sadowska, E. The Heart as a Target of Vasopressin and Other Cardiovascular Peptides in Health and Cardiovascular Diseases. Int. J. Mol. Sci. 2022, 23, 14414.
- Szczepanska-Sadowska, E.; Żera, T.; Kowara, M.; Cudnoch-Jedrzejewska, A. The contribution of angiotensin peptides to cardiovascular regulation in health and disease. In Angiotensin from the Kidney to Coronavirus. Molecular Mediators in Health and Disease: How Cells Communicate; Academic Press-Elsevier: Amsterdam, The Netherlands, 2023; pp. 21–76. ISBN 978-0-323-99618-1.
- Dhalla, N.S.; Shah, A.K.; Tappia, P.S. Role of Oxidative Stress in Metabolic and Subcellular Abnormalities in Diabetic Cardiomyopathy. Int. J. Mol. Sci. 2020, 21, 2413.
- Ghorbanzadeh, V.; Jafarpour, A.; Pirnia, A.; Pajouhi, N.; Khaksarian, M.; Veiskarami, S.; Nazari, A. The role of vasopressin V1A and oxytocin OTR receptors in protective effects of arginine vasopressin against H2O2-induced oxidative stress in H9C2 cells. Arch. Physiol. Biochem. 2022, 128, 830–835.
- González, P.; Lozano, P.; Ros, G.; Solano, F. Hyperglycemia and Oxidative Stress: An Integral, Updated and Critical Overview of Their Metabolic Interconnections. Int. J. Mol. Sci. 2023, 24, 9352.
- Masenga, S.K.; Kabwe, L.S.; Chakulya, M.; Kirabo, A. Mechanisms of Oxidative Stress in Metabolic Syndrome. Int. J. Mol. Sci. 2023, 24, 7898.
- Williams, S.M.; Eleftheriadou, A.; Alam, U.; Cuthbertson, D.J.; Wilding, J.P.H. Cardiac Autonomic Neuropathy in Obesity, the Metabolic Syndrome and Prediabetes: A Narrative Review. Diabetes Ther. 2019, 10, 1995–2021.
- Jia, G.; Lockette, W.; Sowers, J.R. Mineralocorticoid receptors in the pathogenesis of insulin resistance and related disorders: From basic studies to clinical disease. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2021, 320, R276–R286.
- Kenny, H.C.; Abel, E.D. Heart Failure in Type 2 Diabetes Mellitus. Circ. Res. 2019, 124, 121–141.
- Blázquez, E.; Hurtado-Carneiro, V.; LeBaut-Ayuso, Y.; Velázquez, E.; García-García, L.; Gómez-Oliver, F.; Ruiz-Albusac, J.M.; Ávila, J.; Pozo, M.Á. Significance of Brain Glucose Hypometabolism, Altered Insulin Signal Transduction, and Insulin Resistance in Several Neurological Diseases. Front. Endocrinol. 2022, 13, 873301.
- Lee, S.H.; Park, S.Y.; Choi, C.S. Insulin Resistance: From Mechanisms to Therapeutic Strategies. Diabetes Metab. J. 2022, 46, 15–37.
- Mehta, J.K.; Kaur, G.; Buttar, H.S.; Bagabir, H.A.; Bagabir, R.A.; Bagabir, S.A.; Haque, S.; Tuli, H.S.; Telessy, I.G. Role of the renin-angiotensin system in the pathophysiology of coronary heart disease and heart failure: Diagnostic biomarkers and therapy with drugs and natural products. Front. Physiol. 2023, 14, 1034170.
- Szczepanska-Sadowska, E.; Zera, T.; Sosnowski, P.; Cudnoch-Jedrzejewska, A.; Puszko, A.; Misicka, A. Vasopressin and related peptides; potential value in diagnosis, prognosis and treatment of clinical disorders. Curr. Drug Metab. 2017, 18, 306–345.
- Sadoshima, J.; Xu, Y.; Slayter, H.S.; Izumo, S. Autocrine release of angiotensin II mediates stretch-induced hypertrophy of cardiac myocytes in vitro. Cell 1993, 75, 977–984.
- van Esch, J.H.; Gembardt, F.; Sterner-Kock, A.; Heringer-Walther, S.; Le, T.H.; Lassner, D.; Stijnen, T.; Coffman, T.M.; Schultheiss, H.P.; Danser, A.H.; et al. Cardiac phenotype and angiotensin II levels in AT1a, AT1b, and AT2 receptor single, double, and triple knockouts. Cardiovasc. Res. 2010, 86, 401–409.
- Ricci, C.; Pastukh, V.; Schaffer, S.W. Involvement of the mitochondrial permeability transition pore in angiotensin II-mediated apoptosis. Exp. Clin. Cardiol. 2005, 10, 160–164.
- Ide, T.; Tsutsui, H.; Hayashidani, S.; Kang, D.; Suematsu, N.; Nakamura, K.; Utsumi, H.; Hamasaki, N.; Takeshita, A. Mitochondrial DNA damage and dysfunction associated with oxidative stress in failing hearts after myocardial infarction. Circ. Res. 2001, 88, 529–535.
- Dai, D.F.; Johnson, S.C.; Villarin, J.J.; Chin, M.T.; Nieves-Cintrón, M.; Chen, T.; Marcinek, D.J.; Dorn, G.W., 2nd; Kang, Y.J.; Prolla, T.A.; et al. Mitochondrial oxidative stress mediates angiotensin II-induced cardiac hypertrophy and Galphaq overexpression-induced heart failure. Circ. Res. 2011, 108, 837–846.
- Bhullar, S.K.; Dhalla, N.S. Angiotensin II-Induced Signal Transduction Mechanisms for Cardiac Hypertrophy. Cells 2022, 11, 3336.
- Giacomelli, F.; Anversa, P.; Wiener, J. Effect of angiotensin-induced hypertension on rat coronary arteries and myocardium. Am. J. Pathol. 1976, 84, 111–138.
- Bhan, R.D.; Giacomelli, F.; Wiener, J. Adrenoreceptor blockade in angiotensin-induced hypertension: Effect on rat coronary arteries and myocardium. Am. J. Pathol. 1982, 108, 60–71.
- Belabbas, H.; Zalvidea, S.; Casellas, D.; Molès, J.P.; Galbes, O.; Mercier, J.; Jover, B. Contrasting effect of exercise and angiotensin II hypertension on in vivo and in vitro cardiac angiogenesis in rats. Am. J. Physiol. Regu. Integr. Comp. Physiol. 2008, 295, R1512–R1518.
- Kohno, M.; Ohmori, K.; Nozaki, S.; Mizushige, K.; Yasunari, K.; Kano, H.; Minam, M.; Yoshikawa, J. Effects of valsartan on angiotensin II-induced migration of human coronary artery smooth muscle cells. Hypertens. Res. 2000, 23, 677–681.
- Hafizi, S.; Wang, X.; Chester, A.H.; Yacoub, M.H.; Proud, C.G. ANG II activates effectors of mTOR via PI3-K signaling in human coronary smooth muscle cells. Am. J. Physiol. Heart Circ. Physiol. 2004, 287, H1232–H1238.
- Mehta, J.L.; Mercanti, F.; Stone, A.; Wang, X.; Ding, Z.; Romeo, F.; Khaidakov, M. Gene and microRNA transcriptional signatures of angiotensin II in endothelial cells. J. Cardiovasc. Pharmacol. 2015, 65, 123–129.
- Kifor, I.; Dzau, V.J. Endothelial renin-angiotensin pathway: Evidence for intracellular synthesis and secretion of angiotensins. Circ. Res. 1987, 60, 422–428.
- Pinto, J.E.; Viglione, P.; Saavedra, J.M. Autoradiographic localization and quantification of rat heart angiotensin converting enzyme. Am. J. Hypertens. 1991, 4 Pt 1, 321–326.
- Yamada, H.; Fabris, B.; Allen, A.M.; Jackson, B.; Johnston, C.I.; Mendelsohn, A.O. Localization of angiotensin converting enzyme in rat heart. Circ. Res. 1991, 68, 141–149.
- Kusaka, Y.; Kelly, R.A.; Williams, G.H.; Kifor, I. Coronary microvascular endothelial cells cosecrete angiotensin II and endothelin-1 via a regulated pathway. Am. J. Physiol. Heart Circ. Physiol. 2000, 279, H1087–H1096.
- Stoll, M.; Steckelings, U.M.; Paul, M.; Bottari, S.P.; Metzger, R.; Unger, T. The angiotensin AT2-receptor mediates inhibition of cell proliferation in coronary endothelial cells. J. Clin. Investig. 1995, 95, 651–657.
- Louis, S.; Saward, L.; Zahradka, P. Both AT1 and AT2 receptors mediate proliferation and migration of porcine vascular smooth muscle cells. Am. J. Physiol. Heart Circ. Physiol. 2011, 301, H746–H756.
- Takemoto, M.; Egashira, K.; Tomita, H.; Usui, M.; Okamoto, H.; Kitabatake, A.; Shimokawa, H.; Sueishi, K.; Takeshita, A. Chronic angiotensin-converting enzyme inhibition and angiotensin II type 1 receptor blockade: Effects on cardiovascular remodeling in rats induced by the long-term blockade of nitric oxide synthesis. Hypertension 1997, 30, 1621–1627.
- Takemoto, M.; Egashira, K.; Usui, M.; Numaguchi, K.; Tomita, H.; Tsutsui, H.; Shimokawa, H.; Sueishi, K.; Takeshita, A. Important role of tissue angiotensin-converting enzyme activity in the pathogenesis of coronary vascular and myocardial structural changes induced by long-term blockade of nitric oxide synthesis in rats. J. Clin. Investig. 1997, 99, 278–287.
- Kuga, T.; Mohri, M.; Egashira, K.; Hirakawa, Y.; Tagawa, T.; Shimokawa, H.; Takeshita, A. Bradykinin-induced vasodilation of human coronary arteries in vivo: Role of nitric oxide and angiotensin-converting enzyme. J. Am. Coll. Cardiol. 1997, 30, 108–112.
- Prasad, A.; Husain, S.; Quyyumi, A.A. Abnormal flow-mediated epicardial vasomotion in human coronary arteries is improved by angiotensin-converting enzyme inhibition: A potential role of bradykinin. J. Am. Coll/Cardiol. 1999, 33, 796–804.
- Bayraktutan, U.; Ulker, S. Effects of angiotensin II on nitric oxide generation in proliferating and quiescent rat coronary microvascular endothelial cells. Hypertens. Res. 2003, 26, 749–757.
- Munk, V.C.; Sanchez de Miguel, L.; Petrimpol, M.; Butz, N.; Banfi, A.; Eriksson, U.; Hein, L.; Humar, R.; Battegay, E.J. Angiotensin II induces angiogenesis in the hypoxic adult mouse heart in vitro through an AT2-B2 receptor pathway. Hypertension 2007, 49, 1178–1185.
- Chiu, C.Z.; Wang, B.W.; Yu, Y.J.; Shyu, K.G. Hyperbaric oxygen activates visfatin expression and angiogenesis via angiotensin II and JNK pathway in hypoxic human coronary artery endothelial cells. J. Cell Mol. Med. 2020, 24, 2434–2443.
- Rubanyi, G.M.; Polokoff, M.A. Endothelins: Molecular biology, biochemistry, pharmacology, physiology, and pathophysiology. Pharmacol. Rev. 1994, 46, 325–415.
- Yamaguchi, O.; Kaneshiro, T.; Saitoh, S.; Ishibashi, T.; Maruyama, Y.; Takeishi, Y. Regulation of coronary vascular tone via redox modulation in the alpha1-adrenergic-angiotensin-endothelin axis of the myocardium. Am. J. Physiol. Heart Circ. Physiol. 2009, 296, H226–H232.
- de Beer, V.J.; Sorop, O.; Pijnappels, D.A.; Dekkers, D.H.; Boomsma, F.; Lamers, J.M.; Duncker, D.J.; Merkus, D. Integrative control of coronary resistance vessel tone by endothelin and angiotensin II is altered in swine with a recent myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 2008, 294, H2069–H2077.
- Patel, V.B.; Shah, S.; Verma, S.; Oudit, G.Y. Epicardial adipose tissue as a metabolic transducer: Role in heart failure and coronary artery disease. Heart Fail. Rev. 2017, 22, 889–902.
- Salgado-Somoza, A.; Teijeira-Fernández, E.; Fernández, A.L.; González-Juanatey, J.R.; Eiras, S. Proteomic analysis of epicardial and subcutaneous adipose tissue reveals differences in proteins involved in oxidative stress. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, H202–H209.
- Roubíček, T.; Dolinková, M.; Bláha, J.; Haluzíková, D.; Bošanská, L.; Mráz, M.; Kremen, J.; Haluzík, M. Increased angiotensinogen production in epicardial adipose tissue during cardiac surgery: Possible role in a postoperative insulin resistance. Physiol Res. 2008, 57, 911–917.
- Couselo-Seijas, M.; Almengló, C.; Agra-Bermejo, R.M.; Luis Fernandez, Á.; Alvarez, E.; R González-Juanatey, J.; Eiras, S. Higher ACE2 expression levels in epicardial cells than subcutaneous stromal cells from patients with cardiovascular disease: Diabetes and obesity as possible enhancer. Eur. J. Clin. Investig. 2021, 51, e13463.
- Pörsti, I.; Bara, A.T.; Busse, R.; Hecker, M. Release of nitric oxide by angiotensin-(1–7) from porcine coronary endothelium: Implications for a novel angiotensin receptor. Br. J. Pharmacol. 1994, 111, 652–654.
- Ueda, S.; Masumori-Maemoto, S.; Ashino, K.; Nagahara, T.; Gotoh, E.; Umemura, S.; Ishii, M. Angiotensin-(1–7) attenuates vasoconstriction evoked by angiotensin II but not by noradrenaline in man. Hypertension 2000, 35, 998–1001.
- Ferreira, A.J.; Santos, R.A. Cardiovascular actions of angiotensin-(1–7). Braz. J. Med. Biol. Res. 2005, 38, 499–507.
- Westermeier, F.; Bustamante, M.; Pavez, M.; García, L.; Chiong, M.; Ocaranza, M.P.; Lavandero, S. Novel players in cardioprotection: Insulin like growth factor-1, angiotensin-(1–7) and angiotensin-(1–9). Pharmacol. Res. 2015, 101, 41–55.
- Tom, B.; de Vries, R.; Saxena, P.R.; Danser, A.H. Bradykinin potentiation by angiotensin-(1–7) and ACE inhibitors correlates with ACE C- and N-domain blockade. Hypertension 2001, 38, 95–99.
- Almeida, A.P.; Frábregas, B.C.; Madureira, M.M.; Santos, R.J.; Campagnole-Santos, M.J.; Santos, R.A. Angiotensin-(1–7) potentiates the coronary vasodilatatory effect of bradykinin in the isolated rat heart. Braz. J. Med. Biol. Res. 2000, 33, 709–713.
- Kozlovski, V.I.; Lomnicka, M.; Fedorowicz, A.; Chlopicki, S. On the mechanism of coronary vasodilation induced by angiotensin-(1–7) in the isolated guinea pig heart. Basic Clin. Pharmacol. Toxicol. 2007, 100, 361–365.
- Moraes, P.L.; Kangussu, L.M.; da Silva, L.G., Jr.; Castro, C.H.; Santos, R.A.S.; Ferreira, A.J. Cardiovascular effects of small peptides of the renin angiotensin system. Physiol. Rep. 2017, 5, e13505.
- Anderson, J.W.; Smith, P.M.; Ferguson, A.V. Subfornical organ neurons projecting to paraventricular nucleus: Whole-cell properties. Brain Res. 2001, 921, 78–85.
- McKinley, M.J.; McAllen, R.M.; Davern, P.; Giles, M.E.; Penschow, J.; Sunn, N.; Uschakov, A.; Oldfield, B.J. The sensory circumventricular organs of the mammalian brain. In Advances in Anatomy, Embryology and Cell Biology; Springer: Berlin/Heidelberg, Germany, 2003; Volume 172, pp. 1–122.
- McKinley, M.J.; McAllen, R.M.; Pennington, G.L.; Smardencas, A.; Weisinger, R.S.; Oldfield, B.J. Physiological actions of angiotensin II mediated by AT1 and AT2 receptors in the brain. Clin. Exp. Pharmacol. Physiol. 1996, 23 (Suppl. S3), S99–S104.
- Capone, C.; Faraco, G.; Peterson, J.R.; Coleman, C.; Anrather, J.; Milner, T.A.; Pickel, V.M.; Davisson, R.L.; Iadecola, C. Central cardiovascular circuits contribute to the neurovascular dysfunction in angiotensin II hypertension. J. Neurosci. 2012, 32, 4878–4886.
- Horn, M.; Neubauer, S.; Frantz, S.; Hugel, S.; Hu, K.; Gaudron, P.; Schnackerz, K.; Ertl, G. Preservation of left ventricular mechanical function and energy metabolism in rats after myocardial infarction by the angiotensin-converting enzyme inhibitor quinapril. J. Cardiovasc. Pharmacol. 1996, 27, 201–210.
- Lazar, H.L.; Volpe, C.; Bao, Y.; Rivers, S.; Vita, J.A.; Keaney, J.F., Jr. Beneficial effects of angiotensin-converting enzyme inhibitors during acute revascularization. Ann. Thorac. Surg. 1998, 66, 487–492.
- Danser, A.H.; Tom, B.; de Vries, R.; Saxena, P.R. L-NAME-resistant bradykinin-induced relaxation in porcine coronary arteries is NO-dependent: Effect of ACE inhibition. Br. J. Pharmacol. 2000, 131, 195–202.
- Tom, B.; Dendorfer, A.; de Vries, R.; Saxena, P.R.; Jan Danser, A.H. Bradykinin potentiation by ACE inhibitors: A matter of metabolism. Br. J. Pharmacol. 2002, 137, 276–284.
- Gauthier, K.M.; Cepura, C.J.; Campbell, W.B. ACE inhibition enhances bradykinin relaxations through nitric oxide and B1 receptor activation in bovine coronary arteries. Biol. Chem. 2013, 394, 1205–1212.
- Oikawa, Y.; Maehara, K.; Saito, T.; Tamagawa, K.; Maruyama, Y. Attenuation of angiotensin II-mediated coronary vasoconstriction and vasodilatory action of angiotensin-converting enzyme inhibitor in pacing-induced heart failure in dogs. J. Am. Coll. Cardiol. 2001, 38, 1188–1194.
- Kitakaze, M.; Asanuma, H.; Funaya, H.; Node, K.; Takashima, S.; Sanada, S.; Asakura, M.; Ogita, H.; Kim, J.; Hori, M. Angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers synergistically increase coronary blood flow in canine ischemic myocardium: Role of bradykinin. J. Am. Coll. Cardiol. 2002, 40, 162–166.
- Kubo, M.; Umemoto, S.; Fujii, K.; Itoh, S.; Tanaka, M.; Kawahara, S.; Matsuzaki, M. Effects of angiotensin II type 1 receptor antagonist on smooth muscle cell phenotype in intramyocardial arteries from spontaneously hypertensive rats. Hypertens. Res. 2004, 27, 685–693.
- Babick, A.; Chapman, D.; Zieroth, S.; Elimban, V.; Dhalla, N.S. Reversal of subcellular remodelling by losartan in heart failure due to myocardial infarction. J. Cell Mol. Med. 2012, 16, 2958–2967.
- Tanaka, Y.; Tamura, K.; Koide, Y.; Sakai, M.; Tsurumi, Y.; Noda, Y.; Umemura, M.; Ishigami, T.; Uchino, K.; Kimura, K.; et al. The novel angiotensin II type 1 receptor (AT1R)-associated protein ATRAP downregulates AT1R and ameliorates cardiomyocyte hypertrophy. FEBS Lett. 2005, 579, 1579–1586.
- Wang, J.; He, S.Y. Effect of ischemic postconditioning on cell apoptosis and expression of relevant genes in non-culprit coronary arteries. J. Investig. Med. 2020, 68, 1276–1281.
- Paczwa, P.; Szczepańska-Sadowska, E.; Loń, S.; Ganten, S.L.; Ganten, D. Role of central AT1 and V1 receptors in cardiovascular adaptation to hemorrhage in SD and renin TGR rats. Am. J. Physiol. 1999, 276, H1918–H1926.
- Zhu, G.Q.; Gao, L.; Patel, K.P.; Zucker, I.H.; Wang, W. ANG II in the paraventricular nucleus potentiates the cardiac sympathetic afferent reflex in rats with heart failure. J. Appl. Physiol. 2004, 97, 1746–1754.
- Hayashi, M.; Tsutamoto, T.; Wada, A.; Maeda, K.; Mabuchi, N.; Tsutsui, T.; Matsui, T.; Fujii, M.; Matsumoto, T.; Yamamoto, T.; et al. Relationship between transcardiac extraction of aldosterone and left ventricular remodeling in patients with first acute myocardial infarction: Extracting aldosterone through the heart promotes ventricular remodeling after acute myocardial infarction. J. Am. Coll. Cardiol. 2001, 38, 1375–1382.
- Prasad, A.; Halcox, J.P.; Waclawiw, M.A.; Quyyumi, A.A. Angiotensin type 1 receptor antagonism reverses abnormal coronary vasomotion in atherosclerosis. J. Am. Coll. Cardiol. 2001, 38, 1089–1095.
- Miura, S.; Nishikawa, H.; Zhang, B.; Matsuo, Y.; Kawamura, A.; Tsuchiya, Y.; Matsuo, K.; Saku, K. Angiotensin-converting enzyme inhibitor promotes coronary collateral circulation in patients with coronary artery disease. Circ. J. 2003, 67, 535–538.
- Staniloae, C.; Schwab, A.J.; Simard, A.; Gallo, R.; Dyrda, I.; Gosselin, G.; Lesperance, J.; Ryan, J.W.; Dupuis, J. In vivo measurement of coronary circulation angiotensin-converting enzyme activity in humans. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H17–H22.
- Tiefenbacher, C.P.; Friedrich, S.; Bleeke, T.; Vahl, C.; Chen, X.; Niroomand, F. ACE inhibitors and statins acutely improve endothelial dysfunction of human coronary arterioles. Am. J. Physiol. Heart Circ. Physiol. 2004, 286, H1425–H1432.
- Ogino, K.; Kato, M.; Furuse, Y.; Kinugasa, Y.; Kaetsu, Y.; Mizuta, E.; Sugihara, S.; Ishida, K.; Yanagihara, K.; Hisatome, I.; et al. Addition of losartan to angiotensin-converting enzyme inhibitors improves insulin resistance in patients with chronic heart failure treated without β-blockers. Circ. J. 2010, 74, 2346–2352.
- Wackenfors, A.; Pantev, E.; Emilson, M.; Edvinsson, L.; Malmsjö, M. Angiotensin II receptor mRNA expression and vasoconstriction in human coronary arteries: Effects of heart failure and age. Basic Clin. Pharmacol. Toxicol. 2004, 95, 266–272.
- Johansson, M.E.; Fagerberg, B.; Bergström, G. Angiotensin type 2 receptor is expressed in human atherosclerotic lesions. J. Renin Angiotensin Aldosterone Syst. 2008, 9, 17–21.
- Ferreira, A.J.; Jacoby, B.A.; Araújo, C.A.; Macedo, F.A.; Silva, G.A.; Almeida, A.P.; Caliari, M.V.; Santos, R.A. The nonpeptide angiotensin-(1–7) receptor Mas agonist AVE-0991 attenuates heart failure induced by myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 2007, 292, H1113–H1119.
- Kim, M.A.; Yang, D.; Kida, K.; Molotkova, N.; Yeo, S.J.; Varki, N.; Iwata, M.; Dalton, N.D.; Peterson, K.L.; Siems, W.E.; et al. Effects of ACE2 inhibition in the post-myocardial infarction heart. J. Card. Fail. 2010, 16, 777–785.
- Zhu, L.; Carretero, O.A.; Xu, J.; Harding, P.; Ramadurai, N.; Gu, X.; Peterson, E.; Yang, X.P. Activation of angiotensin II type 2 receptor suppresses TNF-α-induced ICAM-1 via NF-κB: Possible role of ACE. Am. J. Physiol. Heart. Circ. Physiol. 2015, 309, H827–H834.
- Zhou, X.; Zhang, P.; Liang, T.; Chen, Y.; Liu, D.; Yu, H. Relationship between circulating levels of angiotensin-converting enzyme 2-angiotensin-(1–7)-MAS axis and coronary heart disease. Heart Vessels 2020, 35, 153–161.
- Drucker, D.J. Diabetes, obesity, metabolism, and SARS-CoV-2 infection: The end of the beginning. Cell Metab. 2021, 33, 479–498.
- Eberhardt, N.; Noval, M.G.; Kaur, R.; Sajja, S.; Amadori, L.; Das, D.; Cilhoroz, B.; Stewart, O.; Fernandez, D.M.; Shamailova, R.; et al. SARS-CoV-2 infection triggers pro-atherogenic inflammatory responses in human coronary vessels. Nat. Cardiovasc. Res. 2023, 2, 899–916.
- Xie, L.; Zhang, Z.; Wang, Q.; Chen, Y.; Lu, D.; Wu, W. COVID-19 and Diabetes: A Comprehensive Review of Angiotensin Converting Enzyme 2, Mutual Effects and Pharmacotherapy. Front. Endocrinol. 2021, 12, 772865.
- Park, H.Y.; Kwon, H.M.; Kim, D.; Jang, Y.; Shim, W.H.; Cho, S.Y.; Kim, H.S. The angiotensin converting enzyme genetic polymorphism in acute coronary syndrome--ACE polymorphism as a risk factor of acute coronary syndrome. J. Korean Med. Sci. 1997, 12, 391–397.
- Prasad, A.; Narayanan, S.; Waclawiw, M.A.; Epstein, N.; Quyyumi, A.A. The insertion/deletion polymorphism of the angiotensin-converting enzyme gene determines coronary vascular tone and nitric oxide activity. J. Am. Coll. Cardiol. 2000, 36, 1579–1586.
- Guney, A.I.; Ergec, D.; Kirac, D.; Ozturhan, H.; Caner, M.; Koc, G.; Kaspar, C.; Ulucan, K.; Agirbasli, M. Effects of ACE polymorphisms and other risk factors on the severity of coronary artery disease. Genet. Mol. Res. 2013, 12, 6895–6906.
- Ruiz, J.; Blanché, H.; Cohen, N.; Velho, G.; Cambien, F.; Cohen, D.; Passa, P.; Froguel, P. Insertion/deletion polymorphism of the angiotensin-converting enzyme gene is strongly associated with coronary heart disease in non-insulin-dependent diabetes mellitus. Proc. Natl. Acad. Sci. USA 1994, 91, 3662–3665.
- Mehri, S.; Mahjoub, S.; Farhati, A.; Bousaada, R.; Ben Arab, S.; Baudin, B.; Hammami, M. Angiotensinogen gene polymorphism in acute myocardial infarction patients. J. Renin Angiotensin Aldosterone Syst. 2011, 12, 42–47.
- Amant, C.; Hamon, M.; Bauters, C.; Richard, F.; Helbecque, N.; McFadden, E.P.; Escudero, X.; Lablanche, J.M.; Amouyel, P.; Bertrand, M.E. The angiotensin II type 1 receptor gene polymorphism is associated with coronary artery vasoconstriction. J. Am. Coll. Cardiol. 1997, 29, 486–490.
- Samuelsson, A.M.; Bollano, E.; Mobini, R.; Larsson, B.M.; Omerovic, E.; Fu, M.; Waagstein, F.; Holmäng, A. Hyperinsulinemia: Effect on cardiac mass/function, angiotensin II receptor expression, and insulin signaling pathways. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, H787–H796.
- Lyu, J.; Imachi, H.; Fukunaga, K.; Sato, S.; Ibata, T.; Kobayashi, T.; Dong, T.; Yoshimoto, T.; Yonezaki, K.; Nagata, H.; et al. Angiotensin II induces cholesterol accumulation and impairs insulin secretion by regulating ABCA1 in beta cells. J. Lipid. Res. 2018, 59, 1906–1915.
- Liu, Z. The renin-angiotensin system and insulin resistance. Curr. Diab. Rep. 2007, 7, 34–42.
- Furuhashi, M.; Ura, N.; Takizawa, H.; Yoshida, D.; Moniwa, N.; Murakami, H.; Higashiura, K.; Shimamoto, K. Blockade of the renin-angiotensin system decreases adipocyte size with improvement in insulin sensitivity. J. Hypertens. 2004, 22, 1977–1982.
- Marcus, Y.; Shefer, G.; Sasson, K.; Kohen, F.; Limor, R.; Pappo, O.; Nevo, N.; Biton, I.; Bach, M.; Berkutzki, T.; et al. Angiotensin 1–7 as means to prevent the metabolic syndrome: Lessons from the fructose-fed rat model. Diabetes 2013, 62, 1121–1130.
- Williams, I.M.; Otero, Y.F.; Bracy, D.P.; Wasserman, D.H.; Biaggioni, I.; Arnold, A.C. Chronic Angiotensin-(1–7) Improves Insulin Sensitivity in High-Fat Fed Mice Independent of Blood Pressure. Hypertension 2016, 67, 983–991.
- Zhao, S.; Sun, W.; Jiang, P. Role of the ACE2/Ang-(1–7)/Mas axis in glucose metabolism. Rev. Cardiovasc. Med. 2021, 22, 769–777.
- Bardgett, M.E.; McCarthy, J.J.; Stocker, S.D. Glutamatergic receptor activation in the rostral ventrolateral medulla mediates the sympathoexcitatory response to hyperinsulinemia. Hypertension 2010, 55, 284–290.
- Ogundele, O.M.; Lee, C.C.; Francis, J. Age-dependent alterations to paraventricular nucleus insulin-like growth factor 1 receptor as a possible link between sympathoexcitation and inflammation. J. Neurochem. 2016, 139, 706–721.
- Feher, A.; Cassuto, J.; Szabo, A.; Patel, V.; Vinayak Kamath, M.; Bagi, Z. Increased tissue angiotensin-converting enzyme activity impairs bradykinin-induced dilation of coronary arterioles in obesity. Circ. J. 2013, 77, 1867–1876.
- Shiuchi, T.; Iwai, M.; Li, H.S.; Wu, L.; Min, L.J.; Li, J.M.; Okumura, M.; Cui, T.X.; Horiuchi, M. Angiotensin II type-1 receptor blocker valsartan enhances insulin sensitivity in skeletal muscles of diabetic mice. Hypertension 2004, 43, 1003–1010.
- Ohshima, K.; Mogi, M.; Jing, F.; Iwanami, J.; Tsukuda, K.; Min, L.J.; Higaki, J.; Horiuchi, M. Roles of interleukin 17 in angiotensin II type 1 receptor-mediated insulin resistance. Hypertension 2012, 59, 493–499.
- Katz, P.S.; Trask, A.J.; Souza-Smith, F.M.; Hutchinson, K.R.; Galantowicz, M.L.; Lord, K.C.; Stewart, J.A., Jr.; Cismowski, M.J.; Varner, K.J.; Lucchesi, P.A. Coronary arterioles in type 2 diabetic (db/db) mice undergo a distinct pattern of remodeling associated with decreased vessel stiffness. Basic Res. Cardiol. 2011, 106, 1123–1134.
- Husarek, K.E.; Katz, P.S.; Trask, A.J.; Galantowicz, M.L.; Cismowski, M.J.; Lucchesi, P.A. The angiotensin receptor blocker losartan reduces coronary arteriole remodeling in type 2 diabetic mice. Vascul. Pharmacol. 2016, 76, 28–36.
- Zhou, M.S.; Schulman, I.H.; Raij, L. Role of angiotensin II and oxidative stress in vascular insulin resistance linked to hypertension. Am. J. Physiol. Heart Circ. Physiol. 2009, 296, H833–H839.
- Estato, V.; Obadia, N.; Carvalho-Tavares, J.; Freitas, F.S.; Reis, P.; Castro-Faria Neto, H.; Lessa, M.A.; Tibiriçá, E. Blockade of the renin-angiotensin system improves cerebral microcirculatory perfusion in diabetic hypertensive rats. Microvasc. Res. 2013, 87, 41–49.
- Oliveira-Junior, S.A.; Martinez, P.F.; Guizoni, D.M.; Campos, D.H.; Fernandes, T.; Oliveira, E.M.; Okoshi, M.P.; Okoshi, K.; Padovani, C.R.; Cicogna, A.C. AT1 receptor blockade attenuates insulin resistance and myocardial remodeling in rats with diet-induced obesity. PLoS ONE 2014, 9, e86447.
- Luther, J.M. Effects of aldosterone on insulin sensitivity and secretion. Steroids 2014, 91, 54–60.
- Ren, J.; Sowers, J.R.; Zhang, Y. Metabolic Stress, Autophagy, and Cardiovascular Aging: From Pathophysiology to Therapeutics. Trends Endocrinol. Metab. 2018, 29, 699–711.
- Igbekele, A.E.; Jia, G.; Hill, M.A.; Sowers, J.R.; Jia, G. Mineralocorticoid Receptor Activation in Vascular Insulin Resistance and Dysfunction. Int. J. Mol. Sci. 2022, 23, 8954.
- Pscherer, S.; Heemann, U.; Frank, H. Effect of Renin-Angiotensin system blockade on insulin resistance and inflammatory parameters in patients with impaired glucose tolerance. Diabetes Care 2010, 33, 914–919.
- Yang, F.S.; Yun, C.H.; Wu, T.H.; Hsieh, Y.C.; Bezerra, H.G.; Liu, C.C.; Wu, Y.J.; Kuo, J.Y.; Hung, C.L.; Hou, C.J.; et al. High pericardial and peri-aortic adipose tissue burden in pre-diabetic and diabetic subjects. BMC Cardiovasc. Disord. 2013, 13, 98.
- Nelson, K.K.; Schlöndorff, J.; Blobel, C.P. Evidence for an interaction of the metalloprotease-disintegrin tumour necrosis factor alpha convertase (TACE) with mitotic arrest deficient 2 (MAD2), and of the metalloprotease-disintegrin MDC9 with a novel MAD2-related protein, MAD2beta. Biochem. J. 1999, 343, 673–680.
- Ormazabal, V.; Nair, S.; Elfeky, O.; Aguayo, C.; Salomon, C.; Zuñiga, F.A. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc Diabetol. 2018, 17, 122.
- Patel, V.B.; Basu, R.; Oudit, G.Y. ACE2/Ang 1–7 axis: A critical regulator of epicardial adipose tissue inflammation and cardiac dysfunction in obesity. Adipocyte 2016, 5, 306–311.
- Morimoto, H.; Mori, J.; Nakajima, H.; Kawabe, Y.; Tsuma, Y.; Fukuhara, S.; Kodo, K.; Ikoma, K.; Matoba, S.; Oudit, G.Y.; et al. Angiotensin 1–7 stimulates brown adipose tissue and reduces diet-induced obesity. Am. J. Physiol. Endocrinol. Metab. 2018, 314, E131–E138.
- Buijs, R.M.; De Vries, G.J.; Van Leeuwen, F.W.; Swaab, D.F. Vasopressin and oxytocin: Distribution and putative functions in the brain. Prog. Brain Res. 1983, 60, 115–122.
- Hupf, H.; Grimm, D.; Riegger, G.A.; Schunkert, H. Evidence for a vasopressin system in the rat heart. Circ. Res. 1999, 84, 365–370.
- Chen, X.; Lu, G.; Tang, K.; Li, Q.; Gao, X. The secretion patterns and roles of cardiac and circulating arginine vasopressin during the development of heart failure. Neuropeptides 2015, 51, 63–73.
- Morgenthaler, N.G.; Struck, J.; Alonso, C.; Bergmann, A. Assay for the measurement of copeptin, a stable peptide derived from the precursor of vasopressin. Clin. Chem. 2006, 52, 112–119.
- Roffi, M.; Patrono, C. CardioPulse: ’Ten Commandments’ of 2015 European Society of Cardiology Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation (NSTE-ACS). Eur. Heart J. 2016, 37, 208.
- Song, Z.; Albers, H.E. Cross-talk among oxytocin and arginine-vasopressin receptors: Relevance for basic and clinical studies of the brain and periphery. Front. Neuroendocrinol. 2018, 51, 14–24.
- Ostrowski, N.L.; Lolait, S.J.; Bradley, D.J.; O’Carroll, A.M.; Brownstein, M.J.; Young, W.S., 3rd. Distribution of V1a and V2 vasopressin receptor messenger ribonucleic acids in rat liver, kidney, pituitary and brain. Endocrinology 1992, 131, 533–535.
- Folny, V.; Raufaste, D.; Lukovic, L.; Pouzet, B.; Rochard, P.; Pascal, M.; Serradeil-Le Gal, C. Pancreatic vasopressin V1b receptors: Characterization in In-R1-G9 cells and localization in human pancreas. Am. J. Physiol. Endocrinol. Metab. 2003, 285, E566–E576.
- Monstein, H.J.; Truedsson, M.; Ryberg, A.; Ohlsson, B. Vasopressin receptor mRNA expression in the human gastrointestinal tract. Eur. Surg. Res. 2008, 40, 34–40.
- Corbani, M.; Marir, R.; Trueba, M.; Chafai, M.; Vincent, A.; Borie, A.M.; Desarménien, M.G.; Ueta, Y.; Tomboly, C.; Olma, A.; et al. Neuroanatomical distribution and function of the vasopressin V1B receptor in the rat brain deciphered using specific fluorescent ligands. Gen. Comp. Endocrinol. 2018, 258, 15–32.
- Góźdź, A.; Szczepańska-Sadowska, E.; Szczepańska, K.; Maśliński, W.; Luszczyk, B. Vasopressin V1a, V1b and V2 receptors mRNA in the kidney and heart of the renin transgenic TGR(mRen2)27 and Sprague Dawley rats. J. Physiol. Pharmacol. 2002, 53, 349–357.
- Morel, A.; Lolait, S.J.; Brownstein, M.J. Molecular cloning and expression of rat V1a and V2 arginine vasopressin receptors. Regul. Pept. 1993, 45, 53–59.
- Gutkowska, J.; Miszkurka, M.; Danalache, B.; Gassanov, N.; Wang, D.; Jankowski, M. Functional arginine vasopressin system in early heart maturation. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, H2262–H2270.
- Nishimura, M.; Takahashi, H.; Matsusawa, M.; Ikegaki, I.; Nakanishi, T.; Hirabayashi, M.; Yoshimura, M. Intracerebroventricular infusions of insulin increase vasopressin release in rats. Clin. Exp. Pharmacol. Physiol. 1990, 17, 763–768.
- Yarkov, A.; Montero, S.; Lemus, M.; Roces de Alvarez-Buylla, E.; Alvarez-Buylla, R. Arginine-vasopressin in nucleus of the tractus solitarius induces hyperglycemia and brain glucose retention. Brain Res. 2001, 902, 212–222.
- Montero, S.A.; Yarkov, A.; Lemus, M.; Mendoza, H.; Valles, V.; de Alvarez-Buylla, E.R.; Alvarez-Buylla, R. Enhancing effect of vasopressin on the hyperglycemic response to carotid body chemoreceptor stimulation. Role of metabolic variables. Adv. Exp. Med. Biol. 2003, 536, 95–107.
- Montero, S.; Mendoza, H.; Valles, V.; Lemus, M.; Alvarez-Buylla, R.; de Alvarez-Buylla, E.R. Arginine-vasopressin mediates central and peripheral glucose regulation in response to carotid body receptor stimulation with Na-cyanide. J. Appl. Physiol. 2006, 100, 1902–1909.
- Larsen, P.J.; Tang-Christensen, M.; Jessop, D.S. Central administration of glucagon-like peptide-1 activates hypothalamic neuroendocrine neurons in the rat. Endocrinology 1997, 138, 4445–4455.
- Zueco, J.A.; Esquifino, A.I.; Chowen, J.A.; Alvarez, E.; Castrillón, P.O.; Blázquez, E. Coexpression of glucagon-like peptide-1 (GLP-1) receptor, vasopressin, and oxytocin mRNAs in neurons of the rat hypothalamic supraoptic and paraventricular nuclei: Effect of GLP-1(7–36)amide on vasopressin and oxytocin release. J. Neurochem. 1999, 72, 10–16.
- Rossi, R.; Scharrer, E. Vasopressin increases blood glucose in pygmy goats (Capra hircus) by an alpha-adrenergic mechanism. Comp. Biochem. Physiol. A Comp. Physiol. 1991, 100, 863–866.
- Zhang, L.; Tong, M.; Xiao, M.; Li, L.; Ding, J. Nitric oxide mediates feedback inhibition in angiotensin II-induced upregulation of vasopressin mRNA. Peptides 2009, 30, 913–917.
- Martínez, M.A.; Fernández, N.; García-Villalón, A.L.; Monge, L.; Diéguez, G. Comparison of the in vivo coronary action of endothelin-1 and vasopressin role of nitric oxide and prostanoids. Vascul. Pharmacol. 2003, 40, 247–252.
- Giusti-Paiva, A.; Elias, L.L.; Antunes-Rodrigues, J. Inhibitory effect of gaseous neuromodulators in vasopressin and oxytocin release induced by endotoxin in rats. Neurosci. Lett. 2005, 381, 320–324.
- Corrêa, P.B.; Pancoto, J.A.; de Oliveira-Pelegrin, G.R.; Cárnio, E.C.; Rocha, M.J. Participation of iNOS-derived NO in hypothalamic activation and vasopressin release during polymicrobial sepsis. J. Neuroimmunol. 2007, 183, 17–25.
- Coletti, R.; de Lima, J.B.M.; Vechiato, F.M.V.; de Oliveira, F.L.; Debarba, L.K.; Almeida-Pereira, G.; Elias, L.L.K.; Antunes-Rodrigues, J. Nitric oxide acutely modulates hypothalamic and neurohypophyseal carbon monoxide and hydrogen sulphide production to control vasopressin, oxytocin and atrial natriuretic peptide release in rats. J. Neuroendocrinol. 2019, 31, e12686.
- Peters, K.G.; Marcus, M.L.; Harrison, D.G. Vasopressin and the mature coronary collateral circulation. Circulation 1989, 79, 1324–1331.
- Marcus, M.L.; Chilian, W.M.; Kanatsuka, H.; Dellsperger, K.C.; Eastham, C.L.; Lamping, K.G. Understanding the coronary circulation through studies at the microvascular level. Circulation 1990, 82, 1–7.
- Maturi, M.F.; Martin, S.E.; Markle, D.; Maxwell, M.; Burruss, C.R.; Speir, E.; Greene, R.; Ro, Y.M.; Vitale, D.; Green, M.V.; et al. Coronary vasoconstriction induced by vasopressin. Production of myocardial ischemia in dogs by constriction of nondiseased small vessels. Circulation 1991, 83, 2111–2121.
- Boyle, W.A., 3rd; Segel, L.D. Attenuation of vasopressin-mediated coronary constriction and myocardial depression in the hypoxic heart. Circ. Res. 1990, 66, 710–721.
- Kam, P.C.; Williams, S.; Yoong, F.F. Vasopressin and terlipressin: Pharmacology andw its clinical relevance. Anaesthesia 2004, 59, 993–1001.
- Adachi, Y.; Sakakura, K.; Akashi, N.; Wada, H.; Momomura, S.I.; Fujita, H. Coronary Spastic Angina Induced after Oral Desmopressin (DDAVP) Administration. Intern. Med. 2016, 55, 3603–3606.
- Yamamoto, K.; Ikeda, U.; Okada, K.; Saito, T.; Kawahara, Y.; Okuda, M.; Yokoyama, M.; Shimada, K. Arginine vasopressin increases nitric oxide synthesis in cytokine-stimulated rat cardiac myocytes. Hypertension 1997, 30, 1112–1120.
- Okamura, T.; Ayajiki, K.; Fujioka, H.; Toda, N. Mechanisms underlying arginine vasopressin-induced relaxation in monkey isolated coronary arteries. J. Hypertens. 1999, 17, 673–678.
- Fan, Y.H.; Zhao, L.Y.; Zheng, Q.S.; Xue, Y.S.; Yang, X.D.; Tian, J.W.; Xu, L. Arginine vasopressin-induced nitric oxide content changes in cultured cardiac fibroblasts and its relation to nuclear factor-kappaB. Sheng Li Xue Bao 2003, 55, 417–421.
- Li, X.; Chan, T.O.; Myers, V.; Chowdhury, I.; Zhang, X.Q.; Song, J.; Zhang, J.; Andrel, J.; Funakoshi, H.; Robbins, J.; et al. Controlled and cardiac-restricted overexpression of the arginine vasopressin V1A receptor causes reversible left ventricular dysfunction through Gαq-mediated cell signaling. Circulation 2011, 124, 572–581.
- Wagner, J.A.; Abesser, M.; Karcher, J.; Laser, M.; Kunos, G. Coronary vasodilator effects of endogenous cannabinoids in vasopressin-preconstricted unpaced rat isolated hearts. J. Cardiovasc. Pharmacol. 2005, 46, 348–355.
- Foreman, B.W.; Dai, X.Z.; Bache, R.J. Vasoconstriction of canine coronary collateral vessels with vasopressin limits blood flow to collateral-dependent myocardium during exercise. Circ. Res. 1991, 69, 657–664.
- Nazari, A.; Sadr, S.S.; Faghihi, M.; Azizi, Y.; Hosseini, M.J.; Mobarra, N.; Tavakoli, A.; Imani, A. Vasopressin attenuates ischemia-reperfusion injury via reduction of oxidative stress and inhibition of mitochondrial permeability transition pore opening in rat hearts. Eur. J. Pharmacol. 2015, 760, 96–102.
- Milles, J.J.; Baylis, P.H.; Wright, A.D. Plasma vasopressin during insulin withdrawal in insulin-dependent diabetes. Diabetologia 1981, 20, 607–611.
- Baylis, P.H.; Zerbe, R.L.; Robertson, G.L. Arginine vasopressin response to insulin-induced hypoglycemia in man. J. Clin. Endocrinol. Metab. 1981, 53, 935–940.
- Chiodera, P.; Volpi, R.; Capretti, L.; Speroni, G.; Marcato, A.; Rossi, G.; Coiro, V. Hypoglycemia-induced arginine vasopressin and oxytocin release is mediated by glucoreceptors located inside the blood-brain barrier. Neuroendocrinology 1992, 55, 655–659.
- Nagai, N.; Nagai, K.; Takezawa, K.; Chun, S.J.; Nakagawa, H. Suppressive effect of vasopressin on the hyperglycemic response to intracranial injection of 2-deoxy-D-glucose. Neurosci. Lett. 1995, 190, 187–190.
- Amico, J.A.; Finn, F.M.; Haldar, J. Oxytocin and vasopressin are present in human and rat pancreas. Am. J. Med. Sci. 1988, 296, 303–307.
- Mineo, H.; Ito, M.; Muto, H.; Kamita, H.; Hyun, H.S.; Onaga, T.; Yanaihara, N. Effects of oxytocin, arginine-vasopressin and lysine-vasopressin on insulin and glucagon secretion in sheep. Res. Vet. Sci. 1997, 62, 105–110.
- Mohan, S.; Moffett, R.C.; Thomas, K.G.; Irwin, N.; Flatt, P.R. Vasopressin receptors in islets enhance glucose tolerance, pancreatic beta-cell secretory function, proliferation and survival. Biochimie 2019, 158, 191–198.
- Abu-Basha, E.A.; Yibchok-Anun, S.; Hsu, W.H. Glucose dependency of arginine vasopressin-induced insulin and glucagon release from the perfused rat pancreas. Metabolism 2002, 51, 1184–1190.
- Kim, A.; Knudsen, J.G.; Madara, J.C.; Benrick, A.; Hill, T.G.; Abdul Kadir, L.; Kellard, J.A.; Mellander, L.; Miranda, C.; Lin, H.; et al. Arginine-vasopressin mediates counter-regulatory glucagon release and is diminished in type 1 diabetes. eLife 2021, 10, e72919.
- Engler, D.; Pham, T.; Fullerton, M.J.; Ooi, G.; Funder, J.W.; Clarke, I.J. Studies of the secretion of corticotropin-releasing factor and arginine vasopressin into the hypophysial-portal circulation of the conscious sheep. I. Effect of an audiovisual stimulus and insulin-induced hypoglycemia. Neuroendocrinology 1989, 49, 367–381.
- Ellis, M.J.; Schmidli, R.S.; Donald, R.A.; Livesey, J.H.; Espiner, E.A. Plasma corticotrophin-releasing factor and vasopressin responses to hypoglycaemia in normal man. Clin. Endocrinol. 1990, 32, 93–100.
- Tallroth, G.; Ryding, E.; Ekman, R.; Agardh, C.D. The response of regulatory peptides to moderate hypoglycaemia of short duration in type 1 (insulin-dependent) diabetes mellitus and in normal man. Diabetes Res. 1992, 20, 73–85.
- Watabe, T.; Tanaka, K.; Kumagae, M.; Itoh, S.; Takeda, F.; Morio, K.; Hasegawa, M.; Horiuchi, T.; Miyabe, S.; Shimizu, N. Hormonal responses to insulin-induced hypoglycemia in man. J. Clin. Endocrinol. Metab. 1987, 65, 1187–1191.
- Muret, L.; Priou, A.; Oliver, C.; Grino, M. Stimulation of adrenocorticotropin secretion by insulin-induced hypoglycemia in the developing rat involves arginine vasopressin but not corticotropin-releasing factor. Endocrinology 1992, 130, 2725–2732.
- Raff, H.; Papanek, P.E.; Liard, J.F.; Cowley, A.W., Jr. The effect of intracarotid vasopressin infusion on ACTH release in neurohypophysectomized, conscious dogs. Am. J. Physiol. 1994, 267, R653–R658.
- Zelena, D.; Domokos, A.; Jain, S.K.; Jankord, R.; Filaretova, L. The stimuli-specific role of vasopressin in the hypothalamus-pituitary-adrenal axis response to stress. J. Endocrinol. 2009, 202, 263–278.
- Saravia, F.E.; Gonzalez, S.L.; Roig, P.; Alves, V.; Homo-Delarche, F.; De Nicola, A.F. Diabetes increases the expression of hypothalamic neuropeptides in a spontaneous model of type I diabetes, the nonobese diabetic (NOD) mouse. Cell Mol. Neurobiol. 2001, 21, 15–27.
- García-Villalón, A.L.; Sanz, E.; Monge, L.; Fernández, N.; Martínez, M.A.; Climent, B.; Diéguez, G. Vascular reactivity to vasopressin during diabetes: Gender and regional differences. Eur. J. Pharmacol. 2003, 459, 247–254.
- Nakamura, K.; Aoyagi, T.; Hiroyama, M.; Kusakawa, S.; Mizutani, R.; Sanbe, A.; Yamauchi, J.; Kamohara, M.; Momose, K.; Tanoue, A. Both V(1A) and V(1B) vasopressin receptors deficiency result in impaired glucose tolerance. Eur. J. Pharmacol. 2009, 613, 182–188.
- Nakamura, K.; Velho, G.; Bouby, N. Vasopressin and metabolic disorders: Translation from experimental models to clinical use. J. Intern. Med. 2017, 282, 298–309.
- Taveau, C.; Chollet, C.; Bichet, D.G.; Velho, G.; Guillon, G.; Corbani, M.; Roussel, R.; Bankir, L.; Melander, O.; Bouby, N. Acute and chronic hyperglycemic effects of vasopressin in normal rats: Involvement of V1A receptors. Am. J. Physiol. Endocrinol. Metab. 2017, 312, E127–E135.
- Yi, S.S.; Hwang, I.K.; Kim, Y.N.; Kim, I.Y.; Pak, S.I.; Lee, I.S.; Seong, J.K.; Yoon, Y.S. Enhanced expressions of arginine vasopressin (Avp) in the hypothalamic paraventricular and supraoptic nuclei of type 2 diabetic rats. Neurochem. Res. 2008, 33, 833–841.
- Fisher, B.M.; Baylis, P.H.; Thornton, S.; Frier, B.M. Arginine vasopressin and oxytocin responses to insulin-induced hypoglycemia in type 1 (insulin-dependent) diabetes. J. Clin. Endocrinol. Metab. 1989, 68, 688–692.
- Enhörning, S.; Struck, J.; Wirfält, E.; Hedblad, B.; Morgenthaler, N.G.; Melander, O. Plasma copeptin, a unifying factor behind the metabolic syndrome. J. Clin. Endocrinol. Metab. 2011, 96, E1065–E1072.
- Asferg, C.L.; Andersen, U.B.; Linneberg, A.; Goetze, J.P.; Jeppesen, J.L. Copeptin, a surrogate marker for arginine vasopressin secretion, is associated with higher glucose and insulin concentrations but not higher blood pressure in obese men. Diabet. Med. 2014, 31, 728–732.
- Ding, C.; Magkos, F. Oxytocin and Vasopressin Systems in Obesity and Metabolic Health: Mechanisms and Perspectives. Curr. Obes. Rep. 2019, 8, 301–316.
- Wolf, J.P.; Massol, J.; Nguyen, N.U.; Berthelay, S. Arginine vasopressin response to insulin-induced hypoglycemia in insulin-dependent diabetics with asymptomatic hypoglycemia. Horm. Metab. Res. 1990, 22, 232–236.
- Phillips, P.A.; Risvanis, J.; Hutchins, A.M.; Burrell, L.M.; MacGregor, D.; Gundlach, A.L.; Johnston, C.I. Down-regulation of vasopressin V1a receptor mRNA in diabetes mellitus in the rat. Clin. Sci. 1995, 88, 671–674.
- Enhörning, S.; Leosdottir, M.; Wallström, P.; Gullberg, B.; Berglund, G.; Wirfält, E.; Melander, O. Relation between human vasopressin 1a gene variance, fat intake, and diabetes. Am. J. Clin. Nutr. 2009, 89, 400–406.
- Enhörning, S.; Sjögren, M.; Hedblad, B.; Nilsson, P.M.; Struck, J.; Melander, O. Genetic vasopressin 1b receptor variance in overweight and diabetes mellitus. Eur. J. Endocrinol. 2016, 174, 69–75.
More
