Bovine leptospirosis is an important disease that affects the reproductive sphere. Due to its high relevance for the bovine production chain in a worldwide scenario, a better understanding of the disease is crucial to reduce its negative impacts. The main agents are strains from the Sejroe serogroup, such as Hardjo and Guaricura, which lead to renal and genital infection. The genital colonization causes a chronic, silent, and subclinical reproductive syndrome, called Bovine Genital Leptospirosis (BGL). Embryonic death, estrus repetition, subfertility, and abortions are the main signs of BGL condition in females. However, although leptospires have been identified in semen, the manifestation of BGL in bulls remains to be clarified.
- Leptospira
- reproduction
- abortion
- diagnosis
- control
- streptomycin
- vaccination
1. Agents and Epidemiological Aspects
2. Pathogenesis of Leptospiral Infection in Bovine
2.1. Pathogenesis and Clinical Manifestation in Females
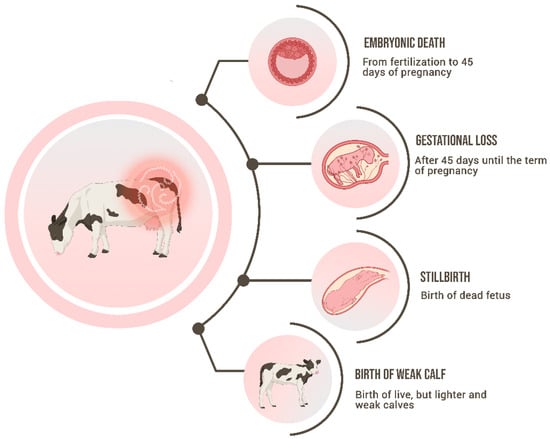
2.2. Aspects of Genital Infection in Males
References
- Zarantonelli, L.; Suanes, A.; Meny, P.; Buroni, F.; Nieves, C.; Salaberry, X.; Briano, C.; Ashfield, N.; Da Silva Silveira, C.; Dutra, F.; et al. Isolation of pathogenic Leptospira strains from naturally infected cattle in Uruguay reveals high serovar diversity, and uncovers a relevant risk for human leptospirosis. PLoS Negl. Trop. Dis. 2018, 13, e06694.
- Ellis, W.A. Animal leptospirosis. Curr. Top. Microbiol. Immunol. 2015, 387, 99–137.
- Vincent, A.T.; Schiettekatte, O.; Goarant, C.; Neela, V.K.; Bernet, E.; Thibeaux, R.; Ismail, N.; Mohd Khalid, M.K.N.; Amran, F.; Masuzawa, T.; et al. Revisiting the Taxonomy and Evolution of Pathogenicity of the Genus Leptospira through the Prism ofGenomics. PLoS Negl. Trop. Dis. 2019, 13, e0007270.
- Levett, P.N. Systematics of Leptospiraceae. Curr. Top. Microbiol. Immunol. 2015, 387, 11–20.
- Zhang, C.; Yang, H.; Li, X.; Cao, Z.; Zhou, H.; Zeng, L.; Xu, J.; Xu, Y.; Chang, Y.F.; Guo, X.; et al. Molecular Typing of Pathogenic Leptospira Serogroup Icterohaemorrhagiae Strains Circulating in China during the Past 50 Years. PLoS Negl. Trop. Dis. 2015, 9, e0003762.
- Loureiro, A.P.; Lilenbaum, W. Genital bovine leptospirosis: A new look for an old disease. Theriogenology 2020, 141, 41–47.
- Nally, J.E.; Hornsby, R.L.; Alt, D.P.; Bayles, D.; Wilson-Welder, J.H.; Palmquist, D.E.; Bauer, N.E. Isolation and characterization of pathogenic leptospires associated with cattle. Vet. Microbiol. 2018, 218, 25–30.
- Mazzanti, M.; Scialfa, E.; Rivero, M.; Passucci, J. Epidemiology of Leptospira spp. infection in a beef cattle area of Argentina. Front. Vet. Sci. 2023, 10, 1083024.
- Guedes, I.B.; Souza, G.O.; Rocha, K.S.; Cavalini, M.B.; Damasceno Neto, M.S.; Castro, J.F.P.; Souza Filho, A.F.; Negrão, M.P.; Cortez, A.; Moraes, C.C.G.; et al. Leptospira strains isolated from cattle in the Amazon region, Brazil, evidence of a variety of species and serogroups with a high frequency of the Sejroe serogroup. Comp. Immunol. Microbiol. Infect. Dis. 2021, 74, 101579.
- Fávero, J.F.; de Araújo, H.L.; Lilenbaum, W.; Machado, G.; Tonin, A.A.; Baldissera, M.D.; Stefani, L.M.; Da Silva, A.S. Bovine leptospirosis: Prevalence, associated risk factors for infection and their cause-effect relation. Microb. Pathog. 2017, 107, 149–154.
- Aymée, L.; Di Azevedo, M.I.N.; Borges, A.L.D.S.B.; Carvalho-Costa, F.A.; Lilenbaum, W. Leptospira spp. strains associated with Bovine Genital Leptospirosis (BGL). Microb. Pathog. 2022, 173, 105841.
- Pinto, P.S.; Barbosa, C.; Ferreira, A.M.R.; Lilenbaum, W. Uterine leptospiral infection is strongly associated to strains of serogroup Sejroe on experimentally infected hamsters. Microb. Pathog. 2020, 142, 104030.
- Chinchilla, D.; Nieves, C.; Gutiérrez, R.; Sordoillet, V.; Veyrier, F.J.; Picardeau, M. Phylogenomics of Leptospira santarosai, a prevalent pathogenic species in the Americas. PLoS Negl. Trop. Dis. 2023, 11, e0011733.
- Sohm, C.; Steiner, J.; Jöbstl, J.; Wittek, T.; Firth, C.; Steinparzer, R.; Desvars-Larrive, A. A systematic review on leptospirosis in cattle: A European perspective. One Health 2023, 17, 100608.
- Givens, M.D. Review: Risks of disease transmission through semen in cattle. Animals 2018, 12, 165–171.
- Barnabé, N.N.D.C.; Soares, R.R.; Barros, D.K.S.; Nogueira, D.B.; Costa, F.T.R.D.; Araújo Júnior, J.P.; Malossi, C.D.; Ullmann, L.S.; Costa, D.F.D.; Silva, M.L.C.R.; et al. Bovine Leptospirosis in Caatinga Biome, Brazil: New Insights into Diagnosis and Epidemiology. Trop. Med. Infect. Dis. 2023, 8, 177.
- Vinodh, R.; Raj, G.D.; Govindarajan, R.; Thiagarajan, V. Detection of Leptospira and Brucella genomes in bovine semen using polymerase chain reaction. Trop. Anim. Health Prod. 2008, 40, 323–329.
- Pimenta, C.L.R.M.; Costa, D.F.; Silva, M.L.C.R.; Pereira, H.D.; Júnior, J.P.A.; Malossi, C.D.; Ullmann, L.S.; Alves, C.J.; Azevedo, S.S. Strategies of the control of an outbreak of leptospiral infection in dairy cattle in Northeastern Brazil. Trop. Anim. Health Prod. 2019, 51, 237–241.
- Monahan, A.M.; Callanan, J.J.; Nally, J.E. Review paper: Host-pathogen interactions in the kidney during chronic leptospirosis. Vet. Pathol. 2009, 46, 792–799.
- Bielanski, A.; Surujballi, O.; Thomas, E.G.; Tanaka, E. Sanitary status of oocytes and embryos collected from heifers experimentally exposed to Leptospira borgpetersenii serovar hardjobovis. Anim. Reprod. Sci. 1998, 54, 65–73.
- Dos Santos Pereira, P.V.; Di Azevedo, M.I.N.; Dos Santos, B.B.A.L.; Loureiro, A.P.; Martins, G.; Carvalho-Costa, F.A.; Souza-Fabjan, J.M.G.; Lilenbaum, W. Bovine genital leptospirosis: Evidence of ovarian infection by Leptospira interrogans. Vet. Microbiol. 2022, 271, 109489.
- Dhaliwal, G.S.; Murray, R.D.; Dobson, H.; Montgomery, J.; Ellis, W.A. Presence of antigen and antibodies in serum and genital discharges of cows from dairy herds naturally infected with Leptospira interrogans serovar Hardjo. Res. Vet. Sci. 1996, 60, 163–167.
- Dhaliwal, G.S.; Murray, R.D.; Dobson, H.; Montgomery, J.; Ellis, W.A. Effect of Leptospira interrogans serovar Hardjo infection on milk yield in endemically infected dairy herds. Vet. Rec. 1996, 28, 319–320.
- Martins, G.; Guadelupe, B.; Aymée, L.; Balaro, M.F.A.; Pinto, P.H.; Di Azevedo, M.I.N.; Brandão, F.Z.; Lilenbaum, W. The efficacy of vaccination in the prevention of renal and genital leptospirosis in experimentally infected sheep. Trop. Med. Infect. Dis. 2022, 7, 321.
- Nogueira, D.B.; Da Costa, F.T.R.; Bezerra, C.S.; Silva, M.L.C.R.; Da Costa, D.F.; Viana, M.P.; Silva, J.D.; Júnior, J.P.A.; Malossi, C.D.; Ullmann, L.S.; et al. Use of serological and molecular techniques for detection of Leptospira sp. carrier sheep under semiarid conditions and the importance of genital transmission route. Acta Trop. 2020, 207, 105497.
- Ellis, W.A.; Thiermann, A.B. Isolation of leptospires from the genital tracts of Iowa cows. Am. J. Vet. Res. 1986, 47, 1694–1696.
- Soares, R.R.; Barnabé, N.N.D.C.; Nogueira, D.B.; da Silva, L.S.C.; Júnior, J.P.A.; Malossi, C.D.; Ullmann, L.S.; da Costa, D.F.; Silva, M.L.C.R.; Dos Santos Higino, S.S.; et al. Serological, molecular, and bacteriological approaches for detecting Leptospira sp. carrier rams maintained in semiarid conditions. Acta Trop. 2021, 21, 105759.
- Cilia, G.; Bertelloni, F.; Piredda, I.; Ponti, M.N.; Turchi, B.; Cantile, C.; Parisi, F.; Pinzauti, P.; Armani, A.; Palmas, B.; et al. Presence of pathogenic Leptospira spp. in the reproductive system and fetuses of wild boars (Sus scrofa) in Italy. PLoS Negl. Trop. Dis. 2020, 14, e0008982.
- Gonçalves, D.D.; Pastre, G.B.; Rey, L.M.R.; Fazoli, K.G.Z.; Silva, L.L.D.; Ferreira, L.R.P.; Fritzen, J.T.T.; Chideroli, R.T.; Durel, L.; Decuadro-Hansen, G.; et al. Leptospira spp. in Naturally Infected Dairy Cow from a Brazilian Border Region. Vector Borne Zoonotic Dis. 2021, 21, 864–869.
- Shetty, A.; Kundu, S.; Vernel-Pauillac, F.; Ratet, G.; Werts, C.; Gomes-Solecki, M. Transient Presence of Live Leptospira interrogans in Murine Testes. Microbiol. Spectr. 2022, 10, e0277521.
- Reese, S.T.; Franco, G.A.; Poole, R.K.; Hood, R.; Fernadez Montero, L.; Oliveira Filho, R.V.; Cooke, R.F.; Pohler, K.G. Pregnancy loss in beef cattle: A meta-analysis. Anim. Reprod. Sci. 2020, 212, 106251.
- Wiltbank, M.C.; Baez, G.M.; Garcia-Guerra, A.; Toledo, M.Z.; Monteiro, P.L.; Melo, L.F.; Ochoa, J.C.; Santos, J.E.; Sartori, R. Pivotal periods for pregnancy loss during the first trimester of gestation in lactating dairy cows. Theriogenology 2016, 86, 239–253.
- Pedrosa, J.; Ezepha, C.; Aymée, L.; Lilenbaum, W. Cellular inflammatory response in the bovine uterus by Leptospira infection may be related to embryo death and subfertility. Microb. Pathog. 2023, 185, 106449.
- Martins, T.; Pugliesi, G.; Sponchiado, M.; Gonella-Diaza, A.M.; Ojeda-Rojas, O.A.; Rodriguez, F.D.; Ramos, R.S.; Basso, A.C.; Binelli, M. Perturbations in the uterine luminal fluid composition are detrimental to pregnancy establishment in cattle. J. Anim. Sci. Biotechnol. 2018, 17, 9–70.
- Mathew, D.J.; Peterson, K.D.; Senn, L.K.; Oliver, M.A.; Ealy, A.D. Ruminant conceptus-maternal interactions: Interferon-tau and beyond. J. Anim. Sci. 2022, 100, 123.
- Husnain, A.; Arshad, U.; Zimpel, R.; Schmitt, E.; Dickson, M.J.; Perdomo, M.C.; Marinho, M.N.; Ashrafi, N.; Graham, S.F.; Bishop, J.V.; et al. Induced endometrial inflammation compromises conceptus development in dairy cattle. Biol. Reprod. 2023, 4, ioad088.
- Diskin, M.G.; Waters, S.M.; Parr, M.H.; Kenny, D.A. Pregnancy losses in cattle: Potential for improvement. Reprod. Fertil. Dev. 2016, 28, 83–93.
- Tomckowiack, C.; Matamala, S.; León, C.; Moroni, M.; Salgado, M. Detection of pathogenic Leptospira as a cause of abortion in cattle-observations on diagnosis. Austral J. Vet. Sci. 2022, 54, 77–81.
- Aymée, L.; Di Azevedo, M.I.N.; Reis, L.; Mendes, J.; Castro, F.D.F.A.D.; Carvalho-Costa, F.A.; Souza, G.N.D.; Lilenbaum, W. Unconventional sites for diagnosis of leptospirosis in bovine anicteric fetuses. Animals 2023, 13, 2832.
- Grégoire, L.; Bakinahe, R.; Petitjean, T.; Boarbi, S.; Delooz, L.; Fretin, D.; Saulmont, M.; Mori, M. Laboratory Diagnosis of Bovine Abortions Caused by Non-Maintenance Pathogenic Leptospira spp.: Necropsy, Serology and Molecular Study Out of a Belgian Experience. Pathogens 2020, 9, 413.
- Canisso, I.F.; Loux, S.C.; Lima, F.S. Biomarkers for placental disease in mares. Theriogenology 2020, 150, 302–307.
- Mock, T.; Mee, J.F.; Dettwiler, M.; Rodriguez-Campos, S.; Hüsler, J.; Michel, B.; Häfliger, I.M.; Drögemüller, C.; Bodmer, M.; Hirsbrunner, G. Evaluation of an investigative model in dairy herds with high calf perinatal mortality rates in Switzerland. Theriogenology 2020, 148, 48–59.
- Soares, R.R.; Barnabé, N.N.D.C.; Junior, J.P.A.; Malossi, C.D.; Ullmann, L.S.; Costa, D.F.; Silva, D.F.C.R.; Higino, S.S.S.; Azevedo, S.S.; Alves, C.J. Investigation of the Presence of Leptospira interrogans in urinary and genital tracts of male goats raised in the semiarid region of Brazil. Small Rumin. Res. 2023, 218, 106880.
- Diaz, E.A.; Luna, L.; Burgos-Mayorga, A.; Donoso, G.; Guzman, D.A.; Baquero, M.I.; Pearson, T.; Barragan, V.A. First detection of Leptospira santarosai in the reproductive track of a boar: A potential threat to swine production and public health. PLoS ONE 2022, 17, e0274362.
- Al-Busadah, K.A.; El-Bahr, S.M.; Khalafalla, A.I. Serum biochemical profile and molecular detection of pathogens in semen of infertile male dromedary camels (Camelus dromedarius). Anim. Reprod. Sci. 2017, 180, 58–65.
- Polo, C.; García-Seco, T.; Díez-Guerrier, A.; Briones, V.; Domínguez, L.; Pérez-Sancho, M. What about the bull? A systematic review about the role of males in bovine infectious infertility within cattle herds. Vet. Anim. Sci. 2023, 19, 100284.
- Jones, A.L. Sexually Transmitted Diseases of Bulls. Vet. Clin. N. Am. Food Anim. Pract. 2023, 40, 111–119.
- Maiolino, S.R.; Cortez, A.; Langoni, H.; Giuffrida, R.; Dos Santos, J.R.; de Nardi Júnior, G.; Lara, G.H.B.; Motta, R.G.; Chacur, M.G.M.; Monteiro, F.M.; et al. Sperm viability, serological, molecular, and modified seminal plasma agglutination tests in the diagnosis of Leptospira in the semen and serum of bovine bulls. Braz. J. Microbiol. 2021, 52, 2431–2438.
