Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Fanny Huang and Version 1 by Vassilis Inglezakis.
Exploration of Mars with a human crew is widely viewed as the ultimate culmination of planetary exploration. Initial human landing on Mars would be limited to the simple in situ resource utilization (ISRU) based on atmosphere only, which is immediately accessible without the challenge of remote, autonomous mining of regolith, transporting regolith, and processing regolith. In a second generation of missions, it might be possible to include processing regolith, either for hydrated minerals or for imbedded ice.
- Mars
- ISRU
- in situ resources on Mars
- in situ resource utilization (ISRU)
1. Introduction
Exploration of Mars with a human crew is widely viewed as the ultimate culmination of planetary exploration [1]. Portree (2001) wrote a superb history of planning activities for missions to send humans to Mars [2]. In addition, Platoff (2001) wrote a history covering 1952 to 1970 [3].
According to Portree: “More than 1000 piloted Mars mission studies were conducted inside and outside NASA between about 1950 and 2000. Many were the product of NASA and industry study teams, while others were the work of committed individuals or private organizations. Due to space limitations, only 50 mission studies (one per year, or less than 5 percent of the total) are described in this monograph. The studies included are believed to be representative of most of the technologies and techniques associated with piloted Mars exploration.”
Rapp (2023) extended Portree’s history another twenty years, from 2001 through 2022 [1]. As of about 2020, the consensus was that a long-stay human mission to Mars was the most feasible approach and provided by far the greatest return on investment. Ash, Dowler, and Varsi (1978) published a pivotal paper in which they pointed out the potential savings by producing rocket fuel in situ from Mars resources for ascent from Mars, rather than bringing the rocket fuel from Earth [4]. They coined the phrase “ISPP” (In Situ Propellant Production), that was commonly used until about 2000. In the late 1990s (and beyond), several technology tasks were carried out related to “ISPP” and Mars mission planners typically incorporated ISPP in their plans for long-stay Mars missions [5]. Starting somewhere around the year 2000, Jerry Sanders at NASA JSC emerged as a leading NASA advocate for ISPP, and he produced a series of presentations over the years in which he broadened the concepts for utilization of in situ resources beyond propellant production, and he coined the now universally accepted phrase: “In Situ Resource Utilization” (ISRU). He and co-workers continue to advocate for a wide realm of ISRU activities on the Moon and Mars [6]. While Sanders continued to inform NASA on a wide array of potential ISRU opportunities, NASA did not develop Mars ISRU in a systematic way, and funding for specific tasks over the years was sporadic. That was until a major new investment was decided upon in 2014, leading to the MOXIE Project, which reached completion 30 September 2023 [7]. Despite the change in reference from ISPP to ISRU, the reality is that propellant production on Mars offers the most practical near-term advance in ISRU with significant demonstrable benefits, and essentially all the technical accomplishments in Mars ISRU (including MOXIE) since the original paper by Ash, Dowler, and Varsi were aimed at propellant production on Mars. In the period 2020–2023, NASA revised its previous concepts for human exploration of Mars, substituting a speculative short-stay mission concept for accepted long-stay mission concepts. In the process, the Mars ISRU seems to have been sidestepped. It is not clear whether this mission concept will stand the test of time or be a temporary phase [8].
ISRU has much greater leverage for ascent propellants than lunar ISRU [1]. The “gear ratio” (mass in LEO required to deliver one mass unit of payload to destination) for transport of mass from Earth LEO to the lunar surface is about 2.5; each mass unit saved on the lunar surface saves 2.5 mass units in Earth LEO. The gear ratio for transport of mass from Earth LEO to Mars’ surface is about 8–10; each mass unit saved on the lunar surface saves 8–10 mass units in Earth LEO. Ascent from the Moon requires about 4–6 tons of O2 propellant, while ascent from Mars requires about 30 tons of O2 propellant. Combining gear ratios with propellant requirements for ascent, even if lunar ISRU and Mars ISRU were equally feasible, the total mass savings in Earth LEO per ascent is about:
Lunar: (4 to 6) (2.5) = 10 to 14 tons
Mars: (30) (8 to 10) = 240 to 300 tons
In addition to leverage, Mars atmospheric processing is far simpler, and more reliable and credible than lunar ISRU. Simply pull in atmosphere through a filter, compress it, and pass it through electrolysis cells. On the Moon, you must dig tons of soil, heat to >1650 °C via solar concentrators in the carbothermal process, or mine tons of icy regolith in deep polar crevasses where there is no available power. Rapp [1] estimated zero return on investment for both lunar processes compared to launch from Earth.
Figure 1 provides a simplified timeline of highlights from the history of Mars ISRU, relevant NASA studies, and relevant events in relevant terrestrial technology. The modern era began with the landmark paper by Ash, Dowler, and Varsi [4], lay dormant until the late 1990s, and then continued at low levels of funding thereafter until the advent of MOXIE in 2014, which completed in 2023. NASA DRA 5.0 [5] demonstrated the benefits of Mars ISRU for long-stay missions, but NASA reverted to a short-stay concept without ISRU around 2020. Terrestrial developments that benefit Mars ISRU are also included.
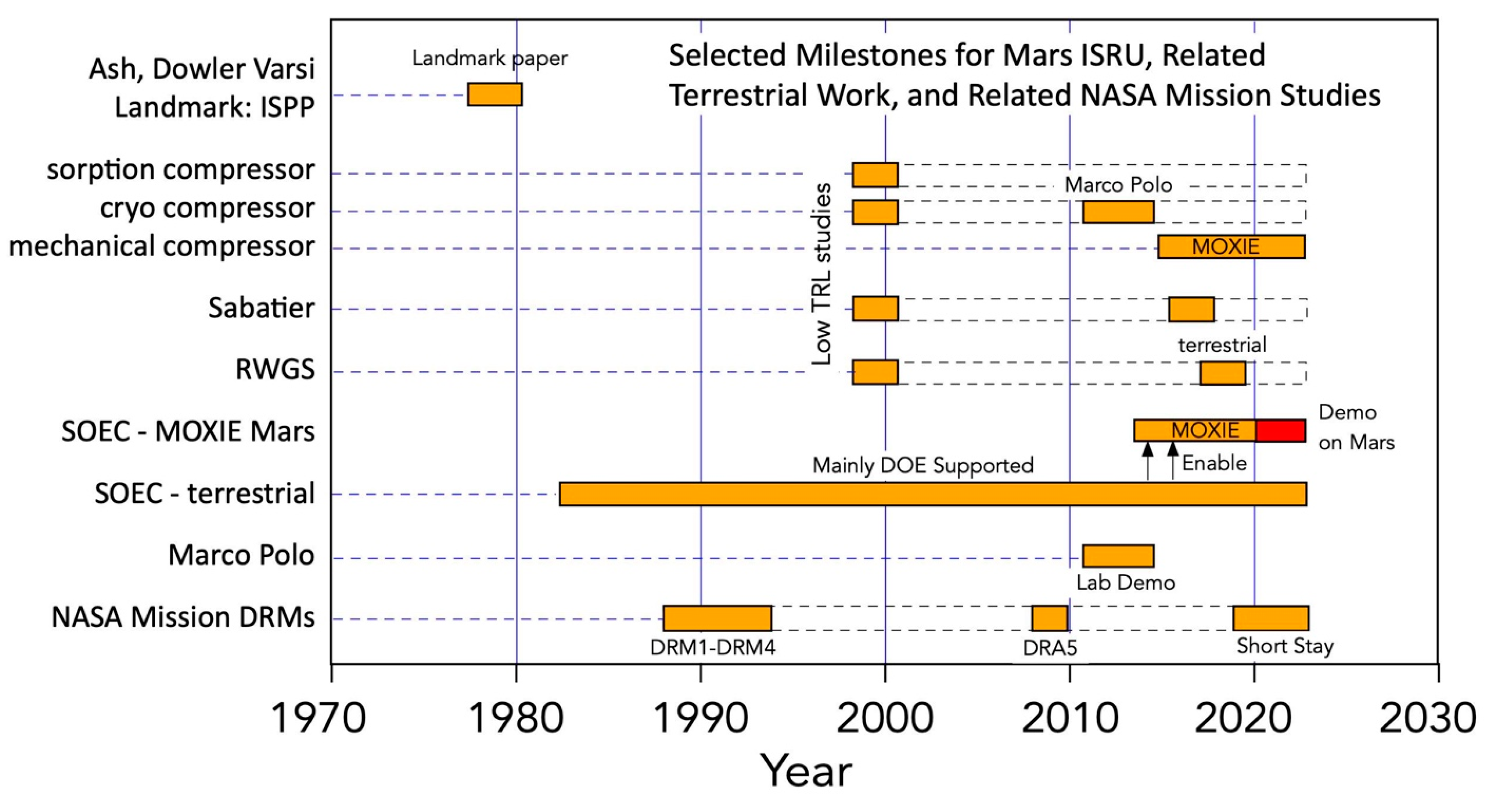
Figure 1. Simplified timeline of highlights from the history of Mars ISRU, relevant NASA studies, and relevant events in relevant terrestrial technology. Red bar highlights demonstration on Mars.
2. Mars In Situ Resource Utilization Technologies
The NASA Mars Architecture Team (MAT) describes the latest NASA concept for the first human mission to Mars, involving a very short round-trip duration, with only a 30-sol surface stay for two crew, minimal surface infrastructure, and ascent propellants brought from Earth [10][9]. This contrasts with previous architectures that showed benefits for a long stay with ISRU [5]. Researchers believe that a long stay mission will be implemented using Mars ISRU, whether in the first landing, or possibly only in secondary landings. The main feedstocks for processing on Mars are the atmosphere, ice imbedded in the regolith at suitably high latitudes, and regolith containing hydrated minerals. The main component of the Martian atmosphere is CO2, with smaller amounts of N2 and Ar, and traces of other gases.
Researchers' concept is that the initial human landing on Mars would be limited to the simple ISRU based on atmosphere only, which is immediately accessible without the challenge of remote, autonomous mining of regolith, transporting regolith, and processing regolith. In a second generation of missions, it might be possible to include processing regolith, either for hydrated minerals or for imbedded ice. There exist many Mars ISRU concepts, ranging from very likely practical, to likely second generation, to imaginative, futuristic ideas that might be pursued in the 22nd century or beyond. Here, researchers focus on ISRU processing for the first- and second-generation missions. It seems likely that the early application of Mars ISRU will be restricted to highly feasible atmospheric processing for production of ascent propellants and life support. The main product would be oxygen. Less likely, the inert gases in the atmosphere might be captured and the nitrogen might be used as a diluent or as a feedstock for processing. The second-generation missions in the campaign are likely to include processing atmosphere and water, obtained either from ice deposits, or hydrated minerals in the regolith. Several hydrated minerals on Mars have a high H2O content (10%) and subsurface ice is abundant, albeit at various depths at higher latitudes [1,11][1][10]. Availability of water would enable a range of possible chemical engineering processes involving carbon, hydrogen, and oxygen, leading to a variety of potential products (Figure 2).

Figure 2.
Potential Mars ISRU processes arranged by likely timing.
The abundant CO2 can be converted into O2 and CO by SOEC conversion or CO and H2O by reverse water-gas shift reaction (RWGS). The use of H2 in the RWGS process is internal. All the H2 is recycled. The O2 can then be used as an ascent propellant and for breathing, and if N2 is recovered, it can be processed, or used as a buffer gas. A significant amount of CO is produced, and in the likely simplest case, vented. If the SOEC process is utilized, there is no need to separate trace components from the CO2 in the Mars atmosphere since the inert gases simply pass through the electrolyzer. However, if cryogenic compression is used, separation is easily accomplished [11][10]. Studies of the RWGS process appear to have utilized pure CO2, so it is not clear whether pure CO2 is needed. Separation would be needed to acquire the N2 in the Mars atmosphere. Processing of nitrogen is likely to be delayed until the second generation of landings [12][11].
In second-generation applications of Mars ISRU, where indigenous H2O and CO2 are both acquired, the use of CO as a feedstock can be further pursued along several avenues. The Sabatier reaction will be important when hydrogen becomes available, converting H2O and CO2 to CH4 and O2 [11,13,14][10][12][13]. The Sabatier process was used in end-to-end lab demonstrations [15,16][14][15]. The CO is also used in the Fischer–Tropsch synthesis to produce hydrocarbons and methanol. The produced CH4, CO, and O2 can be used in a propellant mixture in rocket vehicles [15][14]. The N2 can be converted to NH3 by the Haber–Bosch synthesis [12][11]. These processes must be designed to work under Martian environmental and gravity conditions. An important aspect, sometimes overlooked, is that the above reactions produce mixtures that must be separated using processes such as distillation and membrane separation. Also, processing of Martian regolith is challenging but might also provide Fe, Al, and Si in future generations of landers. An early prototype of an integrated Mars atmosphere and soil processing system was developed during the MARCO POLO project [15][14]. Table 1 provides a summary of potential Mars ISRU technologies.
Table 1.
Chemical-based processes for ISRU on Mars.
| Process | Description | Conditions | Common Materials |
|---|---|---|---|
| Reactions | |||
| RWGS | CO2+H2 →CO2+H2 → CO + H2O | 700–900 °C 1 bar |
* Fe2O3@ Al2O3 |
| Sabatier | CO2+4H2 →CO2+4H2 → CH4 + H2O | 200–500 °C 10–30 bar |
* Ni@Al2O3 |
| Fischer–Tropsch | nCO+(2n+1)H2 →nCO+(2n+1)H2 → CnH2n+2 + nH2O nCO+2nH2 →nCO+2nH2 → CnH2n + nH2O |
300–350 °C 20–40 bar |
* Fe3O4@ Al2O3 |
| Methanol | CO+2H2 →CO+2H2 → CH3OH CO2+3H2 →CO2+3H2 → CH3OH + H2O |
200–300 °C 16–150 bar |
* CuO@Al2O3 |
| Haber–Bosch | N2+3H2 →N2+3H2 →2NH3 | 400–650 °C 200–400 bar |
* Fe3O4@ Al2O3 |
| Other | |||
| SOEC | CO2 →CO2 → CO + 0.5O2 H2O →H2O →0.5O2 + H2 Gas phase |
800–1000 °C | Zr2O Perovskite |
| Electrolysis | H2O →H2O → O2 + H2 Aqueous phase |
25 °C/1 bar | Polymers |
| Cryogenic separation | CO2 (v) →CO2 (v) → CO2 (s) | −78 °C–1 bar | - |
* A symbol such as Fe2O3@ Al2O3 means Fe2O3 supported on an alumina base.
In addition to all these chemical processes, other ISRU processes have been proposed, such as regolith usage for building and radiation shielding, crop production, etc.
References
- Rapp, D. Human Missions to Mars, 3rd ed.; Springer-Praxis Books; Springer: Heidelberg, Germany, 2023.
- Portree, D.S.F. Humans to Mars: Fifty Years of Mission Planning, 1950–2000; Monographs in Aerospace History, Series, Number 21; NASA History Division, Office of Policy and Plans; NASA Headquarters: Washington, DC, USA, 2001.
- Platoff, A. Eyes on the Red Planet: Human Mars Mission Planning, 1952–1970, NASA Report, NASA/CR-2001-208928, July 2001. Available online: https://escholarship.org/content/qt0dx7866r/qt0dx7866r_noSplash_0311d690a533bc3c9c94d74fb41c8f3a.pdf?t=nwvlqw (accessed on 29 November 2023).
- Ash, R.L.; Dowler, W.L.; Varsi, G. Feasibility of Rocket Propellant Production on Mars. Acta Astronaut. 1978, 5, 705–724.
- Drake, B.G. (Ed.) Human Exploration of Mars Design Reference Architecture 5.0. NASA Report. 2009. Available online: https://ntrs.nasa.gov/api/citations/20090012109/downloads/20090012109.pdf (accessed on 29 November 2023).
- Sanders, G.; Kleinhenz, J. In Situ Resource Utilization (ISRU) Strategy—Scope, Plans, and Priorities. In Proceedings of the NASA Advisory Council (NAC) Technology Innovation, and Engineering Committee, Washington DC, USA, 15 May 2023.
- Hecht, M.H.; Hoffman, J.; Rapp, D. Mars Oxygen ISRU Experiment (MOXIE). Space Sci. Rev. 2021, 217, 9.
- Rucker, M.A.; Craig, D.A.; Burke, L.M.; Chai, P.R.; Chappell, M.B.; Drake, B.G.; Troutman, P.A. NASA’s Strategic Analysis Cycle 2021 (SAC21) Human Mars Architecture. In Proceedings of the 2022 IEEE Conference Proceedings, Aerospace Conference, Big Sky, MT, USA, 5–12 March 2022. Also see Rapp (2023) loc cit.
- NASA. Reference Surface Activities for Crewed Mars Mission Systems and Utilization; Document No: HEOMD-415, 1/24/2022; NASA: Washington, DC, USA, 2022.
- Starr, S.O.; Muscatello, A.C. Mars in situ resource utilization: A review. Planet Space Sci. 2020, 182, 104824.
- Bezdek, M.J.; Chirik, P.J. A fresh approach to ammonia synthesis. Nature 2019, 568, 464–466.
- Yu, P.; Carpenter, J.; Woods, J.; Goberman, D.; Gavin, L.; Garr, J.; Ulrich, B. Poisoning Evaluation of On-Orbit Sabatier Assembly. In Proceedings of the International Conference on Environmental Systems, Changchun, China, 21–23 August 2020.
- Vogt, C.; Monai, M.; Kramer, G.J.; Weckhuysen, B.M. The renaissance of the Sabatier reaction and its applications on Earth and in space. Nat. Catal. 2019, 2, 188–197.
- Zubrin, R.M.; Muscatello, A.C.; Berggren, M. Integrated Mars In Situ Propellant Production System. J. Aerosp. Eng. 2013, 26, 43–56.
- Interbartolo, M.A.; Sanders, G.B.; Oryshchyn, L.; Lee, K.; Vaccaro, H.; Santiago-Maldonado, E.; Muscatello, A.C. Prototype Development of an Integrated Mars Atmosphere and Soil-Processing System. J. Aerosp. Eng. 2013, 26, 57–66.
More
