Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Gabriel Fornieles and Version 2 by Rita Xu.
Head and neck cancer (HNC) is among the ten most frequent tumours, with 5-year survival rates varying from 30% to 70% depending on the stage and location of the tumour. HNC is traditionally known as head and neck squamous cell carcinoma (HNSCC), since 90% arises from epithelial cells. Metastasis remains a major cause of mortality in patients with HNSCC. HNSCC patients with metastatic disease have an extremely poor prognosis with a survival rate of less than a year. Matrix metalloproteinases (MMPs) have been described as biomarkers that promote cell migration and invasion.
- head and neck cancer (HNC)
- matrix metalloproteinases (MMPs)
- prognosis
1. Introduction
Head and neck cancer (HNC) is among the ten most frequent types of tumours, occupying the sixth place. Worldwide, HNC accounts for more than 890,000 new cases and 450,000 deaths annually due to this cancer [1]. It is defined as a malignant neoplasm that is developed from the mucosal epithelium in the oral cavity, pharynx and larynx. There are many types of HNCs, which are categorized by their anatomical location following the International Classification of Diseases (ICD-10) from the World Health Organization (WHO). HNC is traditionally known as head and neck squamous cell carcinoma (HNSCC), since 90% arises from epithelial cells [2][3][2,3]. Its incidence has decreased significantly in countries within Asia, North America, Australia, and south and east of Europe. But it is increasing in several countries located in Eastern and Northern Europe among men, and in Southern and Western Europe among women, reflecting the tobacco epidemic. This fact contrasts with the decline in other Western countries where smoking cessation has been earlier. However, an increased incidence in oral cancer in sites related to HPV infection was found, which could be attributed to changes in sexual behaviour, tobacco, alcohol and diet [4][5][4,5]. According to EUROCARE, considering head and neck tumours as the most lethal tumours, are those located in the hypopharynx and larynx with 25% and 59%, respectively. This is followed by the oropharynx with 39%, tongue with 43%, oral cavity with 45% and nasopharynx with 49% [6].
Smoking is the most important risk factor for oral cancer. Tobacco consumption has become a global epidemic. One billion men smoke worldwide, with 35% in developed countries and 50% in developing countries. And over 250 million women are smokers, with 22% in developed countries and 9% in developing countries. This trend indicates that men are smoking less and women are increasing their consumption [7]. Alcohol consumption, the second most important risk factor, is associated with the risk of suffering from HNSCC [8]. It is widespread throughout the history of humanity and across the planet. All alcoholic products carry with them an increased risk of HNSCC [9].
Tumour location is one of the most important prognostic factors because it influences cancer’s ability to metastasize. Tumours of the floor of the mouth and those of the posterior 2/3 of the tongue have a greater capacity for migration through lymph nodes than those that originate in the gums and buccal mucosa. Those originating in the lips, both in the vermilion and dermal portions, have a low metastatic behaviour, compared to tongue cancer. However, the location is not the only determining factor and others are important, such as the stage of the tumour [10].
The treatment alternatives are surgery resecting the tumour, radiotherapy (external radiotherapy or brachytherapy or a combination of these treatments) and chemotherapy, depending on the stage at which the oral cancer is found and after evaluating the variables of the tumour size and its possibilities of propagation to distance. Early diagnosis is essential so that therapy is as simple and effective as possible, directly influencing the success of treatment [3].
Locoregional recurrence is a major cause of patient mortality and morbidity. Although radiation eradicates a large proportion of tumour cells, selected groups of tumour cells (clonogens) are able to survive and repopulate the irradiated areas. Tumours are known as radioresistant if recurrences are observed within six months after the first course of radiation. Tumours can become resistant to radiotherapy for several reasons. Cells proliferate very rapidly and recolonise between treatments and/or tumour cells exhibit mechanisms to become resistant to radiation, such as hypoxia (as oxygen is needed to increase the damage of radiotherapy to the cell’s DNA). In these cases, systemic treatment can be added, improving the results of radiotherapy [11]. Radioresistance is also associated with a high degree of molecular heterogeneity. Other possible molecular mechanisms of radioresistance are enhanced DNA damage repair capacity, increased reactive oxygen species (ROS) scavenging capacity, epithelial–mesenchymal transition (EMT) and abnormal regulation of programmed cell death, which have revealed new targets for the therapy of different types of tumours, including oral squamous cell carcinoma (OSCC) [12]. The role of cancer stem cells (CSCs) in radioresistance has also been described [12][13][12,13].
2. MMPs and HNC
2.1. Invasion and Metastasis
The role of MMPs in invasion and metastasis has been evaluated by various authors [14][15][19,20]. For invasion to occur, a critical step must take place, which is the signalling of the initiation of the metastatic cascade through the interaction of the cells of the tumour with BM. Tumour cells initiate ECM degradation behaviour through BM proteolysis, leading to tumour cell propagation [3][15][3,20]. The degradation of the ECM also leads to an important change in the phenotypic change of the cells, acquiring a mesenchymal profile due to a phenomenon called the epithelial–mesenchymal transition, controlled mainly by MMPs [16][21]. Görögh et al. [15][20] concluded that MMP-2 overexpression was correlated with metastasis and that high levels of TIMP-1and -2 decrease tumour growth. Ren et al. [17][22] found that the mRNAs of MMP-7, MMP-13 and MMP-10 were regulated and that MMP-12 and MMP-9 were not regulated in metastatic tumours compared to non-metastatic ones. This suggests that there are genes that play important roles in metastasis through the regulation of MMP-7 and MMP-13. Nishio et al. [18][23] found that there are higher levels of MMP-2 and MMP-9 at metastatic sites compared to those of the primary tumour. Also, there were statistical differences found in both T1 and T2 cases. As compared to the higher expression of MMPs in metastatic regions, tumour-associated macrophages (TAMs) were in the primary regions. Thus, it can be concluded that the number of TAM and MMP expression levels are expected to have an inverse relationship between the primary and metastatic regions. Chakraborty et al. [19][24] observed that in 56.2% cases, the expression of MMP-9 was more correlated with the presence of lymph node metastases, an advanced stage of cancer and grade of tumour. De Vicente et al. [20][25], in an observational study, also concluded that MMP-2 and MMP-9 are overexpressed in patients with lymph node involvement and that MMP-9 is related to a low survival rate and therefore a poor prognosis. Targeting the remodelling of the HNSCC microenvironment has been suggested as a therapeutic approach to preventing lymph node metastasis. In this sense, it has been found that MMP-9 and MMP-14 were upregulated in the metastatic lymph nodes and closely positively correlated with the level of ALG-2 interacting protein X (ALIX), a protein that promotes the migration and invasion of HNSCC cells. The expression of ALIX in HNSCC was analysed and demonstrated to be statistically higher than in normal mucosae. MMP-9 and MMP-14 were found in the cytoplasm of cancer cells and were significantly enhanced in metastatic lymph nodes compared to the primary tumour. The degradation of extracellular matrix caused by MMPs is critical and the results suggest that ALIX could contribute to increased MMP-9 and MMP-14, leading to lymph node metastasis. Data from The Cancer Genome Atlas (TCGA) further verified the correlation between ALIX and MMP-14, although there was no correlation with MMP-9 [21][26].2.2. Epithelial–Mesenchymal Transition (EMT)
MMPs are a family of endopeptidases responsible for dissolving the ECM and the BM. Figure 1 summarises the main processes in which MMPs are involved. They are produced by various inflammatory and connective tissue cells such as the following: fibroblasts, lymphocytes, endothelial cells and macrophages. And its synthesis is strictly regulated by hormones, growth factors, cytokines and TIMPs [22][27].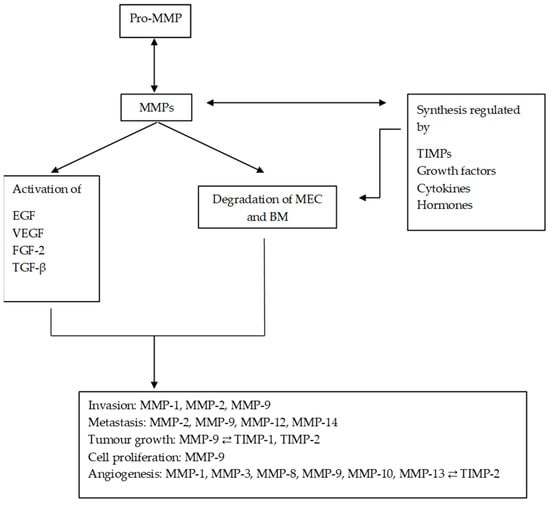
Figure 1. Diagram of the main processes in which MMPs are involved and their regulation by TIMPs. MMPs regulate the synthesis and activation of other substances, promoting and altering the tumour microenvironment. MMPs: matrix metalloproteinases; ECM: extracellular matrix; EGF: epithelial growth factor; VEGF: vascular endothelial growth factor; TGF-β: transforming growth factor beta; FGF-2: fibroblast growth factor 2.
