In this regard, two-dimensional (2D) MXenes have gained more attention than other semiconductors for the realization of RT gas sensors because of their good conductivity, high surface area, ease of synthesis, tunable band gaps, ease of operation, and unique electrical characteristics. Furthermore, they have plenty of functional groups on their surfaces which are favorable sites for adsorption of gas molecules, which makes them very sensitive to changes in the gas concentrations in the surrounding atmosphere
[5]. By controlling the type and amount of functional groups, it is possible to increase the sensitivity of these sensing materials. Owing to working at RT, they can be used for detection of explosive gases with high safety. In addition, they can be used in electronic devices and smartphones because of their low power consumption. Furthermore, they can be used for realization of flexible and wearable gas sensors which have applications in wearable electronic devices
[6][7][6,7].
2. MXenes
Since the discovery of 2D MXenes with nanosheet (NS) morphology by Gogotsi et al.
[8][34] in 2011, MXenes have attracted much interest because of their features such as 2D morphology, high conductance, tunable bandgap, high mechanical flexibility, and hydrophilicity
[9][10][35,36]. Their general formula is
Mn+1XnT
x, in which
M represents the transition metal (Mo, Ti, Zr, Cr, etc.),
X stands for “C” or “N” sites,
n = 1 to 4, and T
x (
x is variable) indicates surface termination groups such as -H, -O, -OH, -F
[11][37].
In these materials, layers of
M atoms are arranged in a honeycomb-like 2D lattice intervened by X ions in the octahedral sites between adjacent metal layers. The precursors of MXenes are from MAX phases, with the formula
Mn+1AXn (MAX), where “
n” = 1 to 3, M is a transition metal, and
A is a group 13–16 element. Various routes are available for synthesizing MXenes
[12][13][39,40]. However, they are generally produced by etching the MAX phase, in which the A-layer atoms are selectively etched to generate loosely stacked MX layers
[14][15][41,42]. Mostly, the HF etchant with varying chemical composition is used to synthesis MXenes by the selective etching of Al atoms from Al-MAX phases. However, to achieve successful conversion of the MAX phase into the MXene, the control of the
HF concentration, the reaction temperature and time are essential, since the M-Al bond strength depends on the type of
M elements. It should be noted that intense etching lead to formation of defects, affecting the MXene quality. In addition, HF etching converts the MAX phase into accordion-like multilayered MXenes, in which individual NSs are held together by van der Waals forces and hydrogen bonds. During HF etching, the following reactions cause the removal of “
A” atoms and generation of MXene.
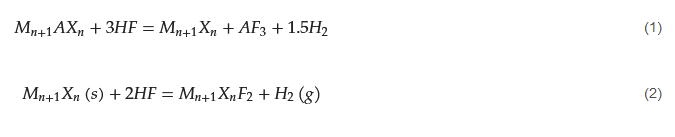
Due to the high toxicity of
HF, MXenes are also synthesized by using fluoride salt-derived in situ
HF etching, fluoride-free etching, molten salt etching, and electrochemical-derived etching of MAX phases
[16][17][18][43,44,45]. MXenes have also been used as templates for the synthesis of other materials
[19][46]. Owing to their large surface area, controllable interlayer spacing, abundant functional groups, and unique electrical properties, MXenes are considered promising materials for realizing RT gas sensors
[20][21][47,48]. Among MXenes, the most widely used for gas sensors is Ti
3C
2T
x MXene due to the following facts: (i) large specific area and numerous terminal functional groups (-OH, -O, and -F) on Ti
3C
2T
x MXene can lead to the strong interface chemical connection with semiconductors and form a Schottky junction. (ii) The high metallic conductivity of Ti
3C
2T
x assures rapid carrier migration. (iii) The exposed terminal metal sites on MXenes may result in more active reactivity than that of carbon materials
[22][49].
One of the major limitations for the practical application of MXenes is their poor oxidation stability under ambient conditions. In fact, exposure to water, air, heat, and light degrades the 2D MXenes into a composite of metal oxide nanoparticles and amorphous carbon. The oxidation of the MXene NSs is generally initiated at surface defects/edges and then propagates to other parts of the nanosheet
[16][43]. The oxidation and/or the state of MXene oxidation can be investigated by Raman spectroscopy
[23][50]. For sensing applications, generally at RT there is no notable oxidation, as the sensing temperature is low. However, at high temperatures, MXene composition changes due to partial oxidation, which leads to changes in the response to the target gas. Therefore, MXenes are mostly used at low temperature due to their relatively low oxidation resistance. Also, for RT MXene-based gas sensors, the long-term stability is acceptable due to working at low temperatures.
2.1. Pristine MXene Gas Sensors
For pristine MXene gas sensors, the sensing mechanism relies on three factors. First, the oxygen molecules can be directly adsorbed on the sensor surface and change the amount of charge carriers on the outer surfaces of MXene, leading to formation of a hole accumulation layer (HAL) or an electron depletion layer (EDL) on p- and n-type MXenes, respectively. Upon exposure to target gases, the amount of charge carriers significantly changes, leading to significant modulation of the sensor resistance. Second, since MXenes are generally synthesized by selective etching, some defects will be present on their surfaces which act as favorable sites for adsorption of gas molecules. Third, the surface functional groups on the sensor surface also are considered as potential sites for adoption of target gas. In addition, the high surface area of MXene NSs provides plenty of adsorption sites for incoming gas molecules.
2.1.1. Delaminated MXene Gas Sensors
Two-dimensional Ti
3C
2T
x MXene NSs were fabricated by etching Ti
3AlC
2 in a mixture of 9 M HCl and LiF (~1 g) at 35 °C for 24 h, and they were delaminated by sonication. Then, they were deposited on a flexible polyimide (PI) substrate for NH
3 gas sensing at RT
[24][51]. Polymeric substrates are generally employed to realize flexible/wearable sensors owing to their low cost, flexibility, and stretchability. Flexible PI is widely used as a flexible substrate because of its excellent bendability, high thermal stability, and high chemical stability
[25][26][52,53]. One fabricated sensor exhibited a response of 21% ([(|R
g − R
a|)/R
a] × 100) to 100 NH
3 at RT. The p-type semiconducting behavior of Ti
3C
2T
x originated from the presence of H
2O and oxygen on the surface of MXene, which were added during etching. NH
3 gas was adsorbed by both defects and functional groups, such as -O and -OH, on Ti
3C
2T
x MXene. The bonding of NH
3 was stronger via hydrogen bonds, leading to the transfer of electrons from NH
3 to Ti
3C
2T
x, which combined with the holes inside the Ti
3C
2T
x sensor, leading to an increase in the resistance and generation of the sensing signal
[24][51].
High-performance gas sensors should have low electrical noise, owing to their high conductivity, and strong signals owing to their strong and abundant adsorption sites
[27][54]. However, it is difficult to satisfy these conditions. In fact, metal oxide gas sensors only show both a high signal and low noise at high temperatures due to the presence of activation energy. On the other hand, highly conductive channel materials are likely to yield low noise but lack the gas adsorption sites required for a high signal. In this regard, MXenes with both high conductivity and a high amount of adsorption sites are promising candidates. In an interesting study, Ti
3C
2T
x MXenes were synthesized by etching using a mixture of lithium fluoride (LiF) and 9 M hydrochloric acid (HCl) while stirring. Then, they were delaminated by sonication. The Ti
3C
2T
x sensor exhibited high selectivity to hydrogen-bonded gases over acidic gases and showed an empirical limit of detection (LOD) of 50 ppb. The Ti
3C
2T
x sensor displayed significantly high signal-to-noise ratios (SNR) over the entire ppb range, and an SNR of 25.6 was obtained for acetone detection at 50 ppb. Both the metallic-like conductivity and presence of many adsorption sites are responsible for the high SNR of the sensor
[27][54].
Because the-O and -OH intrinsic groups on the MXene surface act as favorable adsorption sites, their amounts can be increased to enhance the overall gas-sensing performance. In this context, plasma exposure is a highly promising technique with good control over parameters such as power, exposure duration, and atmosphere. In addition, the plasma exposure does not damage the structure of the MXenes. Exposure is particularly advantageous because the delicate MXene layers will not be damaged. In this context, a large number of oxygen functional groups were grafted onto delaminated Ti
3C
2T
x MXenes via in situ plasma exposure. Air was first introduced at a flow rate of 400 sccm in a chamber at a vacuum level of 101 mbar. Next, the sensors were irradiated by plasma. The optimal sensor displayed a response of 13.8% ([(|R
g − R
a|)/R
a] × 100) to 10 ppm NO
2 at RT. DFT calculations revealed that the oxygen functional groups were associated with increased NO
2 adsorption energy, thereby enhancing the gas response
[28][57].
Surface treatment is a feasible strategy to enhance the stability of MXene-based sensors at RT. 3-Aminopropyl triethoxysilane (APTES), as a silane coupling reagent, can not only decrease the oxidation of MXenes by the addition of a protective layer but can also add additional reactive groups such as -NH
2 to MXene, which are promising for the detection of acidic gases. In this regard, delaminated Nb
2CT
x MXene was dispersed in a water and ethanol mixture of 1:9 ratio to provide enough water for a hydrolysis reaction. Subsequently, three different concentrations of APTES (0.1 mL, 0.2 mL, and 0.3 mL) were added to the above mixture. The hydrophilic -NH
2 group with an electron-donating nature was useful for NO
2 adsorption. The responses of 0.1, 0.2, and 0.3 mL APTES-functionalized Nb
2CT
x MXene sensors to 25 ppm NO
2 gas were 22.5, 31.52, and 26.8% ([(|R
g − R
a|)/R
a] × 100), respectively, while that of the pristine sensor was only 12.5%. Hence, the addition of APTES to Nb
2CT
x MXenes enhanced the sensing response via amine functionalization. However, upon 0.3 mL addition of APTES, due to the increase in the number of Si-O-Si groups, the resistance of MXene increased, which limited the charge transfer and decreased the sensor response
[29][60].
2.1.2. Multilayered MXene Gas Sensors
Generally, van der Waals attractions between MXene NSs lead to self-stacking and agglomeration, which limits the adsorption sites on the sensor surface. Accordion-like Ti
3C
2T
x MXenes were synthesized using an HF etching method for acetone sensing. The response of the sensor to 100 ppm acetone was 100% ([(|R
g − R
a|)/R
a] × 100) at RT, and it was able to detect 250 ppb acetone with a fast response time (t
res) of 53 s at RT. The high surface area is due to the accordion-like morphology of Ti
3C
2T
x as well as the presence of a large number of hydrogen bonds between the functional groups on the MXene surface and acetone vapor attributed to the sensing enhancement. However, the response of the gas sensor in the presence of 60% or higher RH was significantly decreased
[30][68].
Mo-based MXenes, such as Mo
2CT
x, offer more conductance and higher reactivity than Ti-based MXenes; however, less attention has been paid to them. Three gas sensors based on multilayered Mo
2CT
x MXenes on glass, crystalline Si (cSi), and porous Si (pSi) substrates were used for CO
2 sensing. The sensor deposited on glass Si substrate displayed the best response to CO
2 gas, with a good response of 2.3% ([(|R
g − R
a|)/R
a] × 100) at RT to 50 ppm CO
2 and fast t
res and recovery times (t
rec) of 28 and 40 s, respectively
[31][69]. However, at higher temperatures, the sensor deposited on pSi exhibited an enhanced response to CO
2 gas. The enhanced gas response was justified by the lack of charge transfer from either the cSi or pSi substrates to MXene at RT. However, at higher temperatures, the charge transfer from these substrates to MXene leads to a decrease in resistance, which ultimately contributes to the sensing response.
2.2. Composite MXene Gas Sensors
2.2.1. MXene–Metal Oxide Composites
The combination of MXenes with metal oxides is a promising strategy for enhancing the RT-sensing properties of the resultant composite, which generally leads to high-performance gas sensors at RT. Therefore, a SnO
2/Ti
3C
2T
x composite was synthesized hydrothermally. A mixture of MXene powder and stannic chloride pentahydrate (SnCl
4·5H
2O) was prepared. Then, it was put into a 50 mL Teflon-lined autoclave and heated at 180 °C for 12 h. The fabricated sensor offered a response of 40% ([(|R
g − R
a|)/R
a] × 100) to 40 ppm NH
3 at RT, which was higher than a pristine sensor. In the composite, the 2D MXene provided a matrix with high conductivity, which enabled RT sensing. NH
3 absorption at the defect sites on the MXene surface, as well as the interaction with functional groups, resulted in an enhanced gas response. Furthermore, the formation of heterojunctions between MXene and SnO
2 NPs, which acted as resistance modulation sources, contributed to the sensing enhancement
[32][75].
The influence of the amount of MXene in the Ti
3C
2T
x-SnO
2 composite was also investigated. Ti
3C
2T
X MXene (10–40 wt%)-SnO
2 composites were prepared using a hydrothermal route for NO
2 sensing at RT. SnO
2 and Ti
3C
2T
X (10, 20, 30, and 40 wt%) were sonicated, and 0.32 g of urea was added into it along with the dropwise addition of 40-µL HCl. The obtained solution was put inside an autoclave and heated at 120 °C for 8 h. All the SnO
2/MXene composite sensors exhibited superior performance to NO
2 gas relative to pristine MXene. The composite sensor not only had a higher surface area (~25–30 m
2/g) than pristine MXene (8 m
2/g) due to combination of NS morphology of MXene with SnO
2 NPs, but also a higher conductivity relative to pristine SnO
2 NPs owing to the presence of MXene. The presence of MXene facilitated charge-carrier transport during gas sensing, resulting in faster t
res and t
rec. In addition, the functional groups of the Ti
3C
2T
X MXenes are favorable sites for gas adsorption. The sensor with the lowest amount of MXene (10 wt%) showed some agglomeration between the SnO
2 NPs, while the sensor with 20 wt% Ti
3C
2T
X showed the maximum performance. The response decreased with a further increase in the MXene amount, which was related to the presence of enormous -OH termination groups, hindering the number of active sites for NO
2 gas.
SnO
2 NPs (5–10 nm) were dispersed on the Ti
3C
2T
x MXene surface during the hydrothermal synthesis. SnCl
4·5 H
2O and different volumes (10, 15, 20 and 25 mL) of the few-layered Ti
3C
2T
x colloidal solution were poured to form a homogeneous mixture by means of electrostatic self-assembly; the obtained samples were named as 4.8%, 9.2%, 13.1% and 16.8%Ti
3C
2T
x-SnO
2 composites, respectively. Then, CO(NH
2)
2 dissolved in various deionized water (30, 25, 20 and 15 mL) was dripped slowly into the mixture and stirred for 0.5 h. The resulting mixed solution was subsequently transferred into a 100 mL Teflon-lined autoclave and kept at 180 °C for 12 h.
An ultrasonic method was used to prepare NiO/Ti
3C
2T
x MXene nanocomposites. NiO and MXene powders were dissolved in deionized water and then subjected to ultrasonic treatment for 5 h. Then, it was centrifuged to separate the precipitate, and the NiO/Ti
3C
2T
x MXene composite was obtained. The sensor exhibited a high response of 6.13% ([(|R
g − R
a|)/R
a] × 100) to 50 ppm NH
3 at RT, which was ~9 times more than that of the pristine MXene sensor. Well-dispersed NiO particles in the interlayers of the accordion-like MXene not only prevented agglomeration but also increased the surface area. Hence, it improves the absorption and diffusion of NH
3 molecules. Furthermore, many functional groups on MXene can easily form strong hydrogen bonds with NH
3 gas. In addition, the high conductivity of MXenes accelerates charge transfer, thereby enhancing the gas response. The formation of heterojunctions also accounted for the sensing enhancement.
A MXene/NiO composite was synthesized via an in situ precipitation method. A NiSO
4·6H
2O and MXene solution was prepared, and then a NaOH aqueous solution was dropped into the solution. After being stirred for 2 h, the precipitates were collected and washed three times with deionized water and three times with ethanol. Then, the samples were dried at 60 °C for 24 h. Finally, the MXene/NiO composite materials were obtained after being calcined at 350 °C for 2 h under N
2 atmosphere. The sensor response to 50 ppm HCHO gas was 8.8 at RT. Based on FTIR analysis, numerous hydroxyl and other oxygen-containing functional groups were present on the sensor surface, which are important for NH
3 gas sensing.
2.2.2. MXene-TMD Composites
Two-dimensional transition metal dichalcogenides (TMDs) have high surface areas, abundant adsorption sites, and high surface reactivities; therefore, their composites with MXenes are promising for sensing studies
[33][34][99,100]. In a relevant study, a Ti
3C
2T
x-WSe
2 composite was chemically prepared and the fabricated sensor displayed a response of 9% ([(|R
g − R
a|)/R
a] × 100) to 40 ppm ethanol at RT. In addition, fast t
res (9.7 s) and t
rec (6.6 s) were recorded. The enhanced response to ethanol gas is related to the numerous heterojunctions generated between Ti
3C
2T
x and WSe
2. The enhanced response to ethanol gas is related to the numerous heterojunctions generated between Ti
3C
2T
x and WSe
2. In heterojunctions, band bending occurs, and as a result, potential barriers will be formed between two materials, leading to difficulty of flow of the charge carriers. Upon injection of the target gas, the height of potential barriers changes, contributing to significant resistance changes in heterojunctions. More heterojunctions result in higher modulation of the sensor resistance. In addition, after 1000 bending cycles, the performance not only did not decrease, but also slightly increased owing to the creation of microcracks and wrinkles by the strain forces, which acted as adsorption sites
[35][101].
The influence of the electrode type on sensing performance was explored. A flexible paper-based sensor using a Ti
3C
2T
x/WS
2 composite was fabricated using either a Ti
3C
2T
x-MXene electrode (ME) or a Au electrode (AE). The ME + Ti
3C
2T
x/WS
2 gas sensor exhibited the highest response of 15.2% ([(|R
g − R
a|)/R
a] × 100) to 1 ppm NO
2 gas at RT, owing to the formation of Ohmic contact between the sensing layer and ME, in contrast to the Schottky contact formed between the sensor and AE. When Au and Ti
3C
2T
x/WS
2 were in contact, the formation of Schottky potential barriers prevented the transport of charges between the two materials, and only a small number of carriers were able to cross the junction. In contrast, when Ti
3C
2T
x/WS
2 contacted the ME, the height of the barrier between the nonmetal ME and the Ti
3C
2T
x/WS
2 sensor was much lower, which allowed the easy transport of charge carriers across the junction. Furthermore, the flexible 2D ME has a large specific surface area and offers adequate adsorption and reaction sites for oxygen and the target gas. In addition, numerous surface groups are present on the ME surface, which affect the sensing performance. Finally, the excellent conductivity of Ti
3C
2T
x accelerated the electron flow during the sensing process and shortened t
rec. The optimized sensor showed good flexibility by maintaining its performance even after bending 500 times by 60°
[36][102].
2.2.3. MXene-Conducting Polymers Composites
Conducting polymers (CPs) are promising materials for gas sensors because of their high conductivity, possibility of working at RT, tunable chemical composition, easy doping, and low price
[37][38][39][107,108,109]; therefore, they can be used with MXene to boost the RT gas-sensing properties of the resultant composite. A sensor was fabricated for RT ammonia sensing by the in situ polymerization of PEDOT and PSS on Ti
3C
2T
x MXene. The sensor showed a high response of 36.6% ([(|R
g − R
a|)/R
a] × 100) to 100 ppm of NH
3 with t
res and t
rec of 2 min and 40 s, respectively. In addition, the sensor on the flexible PI substrate exhibited good mechanical flexibility by maintaining its performance at different bending angles. Charge flow occurred between the NH
3 molecules and the sensor surface, leading to a change in the electrical conductivity. Furthermore, the high specific surface area of the composite, along with π = π interactions, increased the concentration of charge carriers
[40][110].
A Ti
3C
2T
x MXene/urchin-like polyaniline (PANI) composite was produced using a template method by employing sulfonated PS nanosphere templates and in situ polymerization on flexible polyethylene terephthalate (PET). The sensor disclosed a high response of 3.70 to 10 ppm NH
3 at RT, which was higher than that of the pristine sensor. The enhanced sensing was related to the hollow urchin-like morphology of PANI and the NS morphology of Ti
3C
2T
x, both of which were beneficial for providing more adsorption sites for NH
3 gas. Second, Schottky heterojunctions were generated by the intimate contact between PANI and the Ti
3C
2T
x NS, which shortened the diffusion length for charges and led to fast charge flow. Furthermore, the degree of protonation of PANI increased through its connection with the Ti
3C
2T
x NS. The increased -NH
2+ and = NH
+ groups in the composite led to an enhanced response to NH
3 gas. As NH
3 is an indicator of meat freshness, the fabricated sensor was successfully used to evaluate pork meat freshness. After 36 h, the sensor was able to indicate an increase in NH
3 concentration in the meat, confirming spoilage
[41][111]. PANI NPs were decorated with Ti
3C
2T
x NSs via in situ polymerization. The sensor displayed a response (ΔI/I
0) of 40 to 200 ppm ethanol gas at RT. In addition, it exhibited good mechanical flexibility; under bending from 0° to 120°, it exhibited almost the same performance, demonstrating good flexibility. In particular, under bending to ~120° it showed a high response of 27.4% ([(|R
g − R
a|)/R
a] × 100) to 150 ppm ethanol. In addition, the t
res and t
rec were 0.6 and 0.8 s, respectively, after bending. Based on DFT calculations, the adsorption energies of −0.985, −0.689, and −0.544 were calculated for OH-terminated Ti
3C
2, O-terminated Ti
3C
2, and F-terminated Ti
3C
2, respectively. This demonstrates that the OH-terminated Ti
3C
2 had the strongest binding energy for ethanol
[42][112].
2.2.4. Ternary Composites
MXene-based ternary composites have been studied less for gas-sensing applications than binary composites because of the complexity of the synthesis procedure and the need for optimization of the three components. However, they exhibit superior sensing properties because there are more resistance-modulation sources inside the sensing materials.
A hamburger-like SnO-SnO
2/Ti
3C
2T
x MXene nanocomposite was hydrothermally prepared at 120 °C for 8 h. It revealed a high response of 12.1 to 100 ppm acetone at RT, which was higher than that of the pristine sensors. Moreover, it revealed a t
rec of 9 s. The improved response was related to the higher surface area of the SnO-SnO
2-Ti
3C
2T
x MXene composite (46.7 m
2/g), relative to pristine Ti
3C
2T
x, (13.6 m
2/g), and SnO-SnO
2 (38.6 m
2/g) gas sensors. Furthermore, there are more resistance modulation sources in the nanocomposite than in the other sensors.
Ternary 2D Ti
3C
2T
x MXene@TiO
2/MoS
2 composites were prepared using the hydrothermal method for NH
3 sensing at RT. It showed a response of 164% ([(|R
g − R
a|)/R
a] × 100) to 100 ppm NH
3 gas at RT, which was higher than that of the pristine sensor counterparts. The improved sensing of NH
3 was attributed to the layered nanostructure with a unique morphology and p-n heterojunctions. Furthermore, DFT studies indicated that NH
3 was able to transfer more charge to the composite surface than to pristine Ti
3C
2T
x MXene and MoS
2, resulting in a higher modulation of the resistance
[43][117].
A 2D Ti
3C
2T
x MXene-MoO
2/MoO
3 NSs composite was fabricated using the hydrothermal method at 180 °C/10 h for ethanol detection at RT. It revealed a high response of 19.77 to 200 ppm to ethanol, and fast t
res and t
rec (46 s/276 s). The high surface area (13.54 m
2/g) and abundant surface groups on MXene provided more active sites for the adsorption of oxygen and ethanol molecules.
Various nanocomposites such as MXenes with GO, ZnO, CuO, GO/ZnO, GO/CuO, ZnO/CuO, and GO/ZnO/CuO have been hydrothermally synthesized for NH
3 sensing at RT. Among them, Ti
3C
2T
x MXene/GO/CuO/ZnO with an optimal ratio of 9:1:5:5 exhibited the best NH
3 gas sensing without resistance drift. The response to 200 ppm was 96% ([(|R
g − R
a|)/R
a] × 100) along with good humidity independence. The improved sensing response was related to the generation of multiple p-n and p-p heterojunctions, as well as the presence of many functional groups on the surfaces of MXene and GO
[44][119].
A 3D Ti
3C
2T
x MXene/rGO/SnO
2 aerogel was fabricated using a facile solvothermal approach at 140 °C for 24 h. It exhibited a response of 54.97% ([(|R
g − R
a|)/R
a] × 100) to 10 ppm formaldehyde at RT. In addition, it indicated short t
res and t
rec (2.9 and 2.2 s) along with high stability. The high surface area of 103 m
2/g and the generation of p-n junctions between rGO and SnO
2 and p-p junctions between MXene and rGO contributed to the sensing mechanism. Based on DFT calculations, the adsorption energy of HCHO on Ti
3C
2T
x MXene/rGO/SnO
2 was −5.7 eV, which was larger than that for other sensors
[45][120].
2.2.5. Other MXene-Based Composites
A hollow nanofiber GaN/Ti
3C
2T
x composite was synthesized by hydrothermal nitridation at 120 °C/12 h. Ti
3C
2T
x, which has metallic properties, acts as a conductive channel and decreases the overall resistance at RT. In addition, Ti
3C
2T
x accelerated charge flow during the sensing reactions, resulting in fast sensor dynamics at RT. Accordingly, the response of the composite sensor to 50 ppm NH
3 was 3.5 times higher than that of the bare Ti
3C
2T
x. The large specific surface area and unique hollow porous morphology of the GaN NFs provide sufficient adsorption sites for NH
3 gas. The formation of p-n Ti
3C
2T
x-GaN heterojunctions is beneficial for resistance modulation. The responses to NH
3 were not affected by 20–80%RH. At high humidity, the sensor was covered with multilayered physisorbed water, leading to the inhibition of the direct reaction between the adsorbed oxygen and NH
3 [46][121].
Ni(OH)
2 has features such as non-toxicity, low cost, ease of synthesis, and semiconducting properties. Ni(OH)
2/Ti
3C
2T
x composites were synthesized via in situ electrostatic self-assembly. The sensor with ~7.8 wt% Ni(OH)
2 revealed the highest response, of 13% ([(|R
g − R
a|)/R
a] × 100) to 50 ppm NH
3 gas at RT. A further increase in Ni(OH)
2 resulted in the partial aggregation of Ni(OH)
2, causing a decrease in the number of adsorption sites and the sensing response. The formation of interfacial Schottky junctions between the two components and the increase in adsorption sites owing to the high surface area (54 m
2/g) are attributed to the sensing mechanism
[47][122].
A BiOCl-Ti
3C
2T
x MXene composite with an NS morphology, excellent homogeneity, and good electronic characteristics was synthesized for sensing studies. It revealed a high response to 34.58 to 100 ppm NO
2 gas at 80%RH. The high response of the gas sensor was attributed to the formation of p-p heterojunctions between BiOCl and MXene
[48][123].
The SnS
2/Ti
3C
2 MXene composites were produced via electrostatic interactions. The response to 50 ppm acetone was 29.8% ([(|R
g − R
a|)/R
a] × 100) at RT, and the t
res and t
rec were ~90 and 355 s, respectively. The oxygen-containing functional groups on Ti
3C
2 formed hydrogen bonds with acetone. Electrons flowed from Ti
3C
2T
x to SnS
2 to form heterojunctions with potential barriers, the heights of which were changed upon exposure to target gas. The sensor could detect acetone in both the optical and electrical modes. To demonstrate the optical mode of the sensor, the sensor signal was connected to an LED, and the blue light evolution images of the LED at various acetone concentrations were processed.
2.2.6. Doped/Decorated MXenes
Doping is a popular method for enhancing the gas-sensing properties of metal oxides
[49][125]. Few studies on the doping of MXenes for gas-sensing applications have been reported. Generally, noble metals with catalytic activity are used for decoration on the sensing materials, and since they are much more expensive than other materials, fewer studies have been conducted using noble metal decoration on MXenes. Also, doped MXenes are less studied relative to composite-based MXenes for gas-sensing application due to the lower impact of doping on the gas response relative to heterojunctions. However, in future studies much more attention should be paid to doped and decorated MXenes for gas-sensing studies. Heteroatom additions to MXenes can go to lattice sites, functional group sites or become adsorbed on surfaces. In general, element doping effects are as follows: (i) generation of active species and increase in conductance; (ii) adjustment of the electronic structure by introduction of defects; (iii) changing of the surface nature and chemical bonds in MXene; (iv) adjustment of the surface chemical properties to increase catalytic performance
[50][126]. In this regard, S atoms with high electronegativity can decrease the electron density of the Ti atom, leading to a higher binding energy than that of Ti-C bonds. In a relevant study, it was demonstrated that the S doping of Ti
3C
2T
x MXene led to a higher gas-sensing response to toluene than that of the pristine sensor. An enhanced response of 214% ([(|R
g − R
a|)/R
a] × 100) to 1 ppm toluene was obtained after sulfur doping. Expansion of the interlayer spacing after sulfur doping has been reported; therefore, a larger surface area resulted in effective gas diffusion and provided more sites for toluene gas. Furthermore, the S at the surface of the MXenes acted like oxygen ions, leading to the expansion of the electron depletion layer (EDL) on the MXene. Upon interaction of toluene gas with these adsorbed sulfur species, they react with sulfur ions, and the liberated electrons increase the concentration of electrons, leading to the appearance of a sensor signal. Moreover, owing to the donating effect of the ethyl group, a remarkable enhancement in the activity of the H
2 atoms on the benzene ring was observed, leading to enhanced selectivity for toluene gas. In addition, DFT calculations revealed an increase in the binding energy of toluene to the S-doped MXenes
[51][127].
3. Conclusions
Pristine MXene gas sensors without any modification often exhibit poor performance; hence, their surfaces can be modified to increase the number of surface functional groups or add new functional groups. Therefore, the response and selectivity can be increased. Composite fabrication with other materials, such as metal oxides, TMDS, and CPs, is a very popular and promising strategy for enhancing the RT performance of gas sensors. In particular, because of the highly intrinsic sensing properties of metal oxides, their composites with MXenes have led to the realization of high-performance gas sensors that can work at RT. Composites based on MXenes-TMDs have high surface areas and abundant surface groups, both of which are beneficial for gas sensing. Furthermore, composites with CPs are highly sensitive to NH
3 because of the high intrinsic sensitivities of both MXenes and CPs to this gas. Compared to MXene composites, less attention has been paid to doped MXenes, and more studies are needed in the future. Ternary composites are also promising for sensing applications; however, the optimization of all components is often difficult, and in this regard, more detailed studies are needed. In addition, the use of UV light to promote surface reactions and increase the number of active surface sites is a promising technique for enhancing the RT-sensing properties of MXene-based gas sensors.
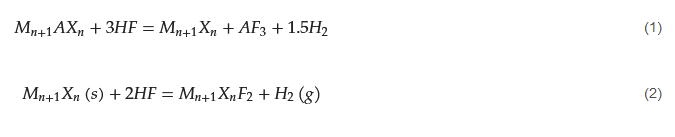 Due to the high toxicity of HF, MXenes are also synthesized by using fluoride salt-derived in situ HF etching, fluoride-free etching, molten salt etching, and electrochemical-derived etching of MAX phases [16][17][18][43,44,45]. MXenes have also been used as templates for the synthesis of other materials [19][46]. Owing to their large surface area, controllable interlayer spacing, abundant functional groups, and unique electrical properties, MXenes are considered promising materials for realizing RT gas sensors [20][21][47,48]. Among MXenes, the most widely used for gas sensors is Ti3C2Tx MXene due to the following facts: (i) large specific area and numerous terminal functional groups (-OH, -O, and -F) on Ti3C2Tx MXene can lead to the strong interface chemical connection with semiconductors and form a Schottky junction. (ii) The high metallic conductivity of Ti3C2Tx assures rapid carrier migration. (iii) The exposed terminal metal sites on MXenes may result in more active reactivity than that of carbon materials [22][49].
One of the major limitations for the practical application of MXenes is their poor oxidation stability under ambient conditions. In fact, exposure to water, air, heat, and light degrades the 2D MXenes into a composite of metal oxide nanoparticles and amorphous carbon. The oxidation of the MXene NSs is generally initiated at surface defects/edges and then propagates to other parts of the nanosheet [16][43]. The oxidation and/or the state of MXene oxidation can be investigated by Raman spectroscopy [23][50]. For sensing applications, generally at RT there is no notable oxidation, as the sensing temperature is low. However, at high temperatures, MXene composition changes due to partial oxidation, which leads to changes in the response to the target gas. Therefore, MXenes are mostly used at low temperature due to their relatively low oxidation resistance. Also, for RT MXene-based gas sensors, the long-term stability is acceptable due to working at low temperatures.
Due to the high toxicity of HF, MXenes are also synthesized by using fluoride salt-derived in situ HF etching, fluoride-free etching, molten salt etching, and electrochemical-derived etching of MAX phases [16][17][18][43,44,45]. MXenes have also been used as templates for the synthesis of other materials [19][46]. Owing to their large surface area, controllable interlayer spacing, abundant functional groups, and unique electrical properties, MXenes are considered promising materials for realizing RT gas sensors [20][21][47,48]. Among MXenes, the most widely used for gas sensors is Ti3C2Tx MXene due to the following facts: (i) large specific area and numerous terminal functional groups (-OH, -O, and -F) on Ti3C2Tx MXene can lead to the strong interface chemical connection with semiconductors and form a Schottky junction. (ii) The high metallic conductivity of Ti3C2Tx assures rapid carrier migration. (iii) The exposed terminal metal sites on MXenes may result in more active reactivity than that of carbon materials [22][49].
One of the major limitations for the practical application of MXenes is their poor oxidation stability under ambient conditions. In fact, exposure to water, air, heat, and light degrades the 2D MXenes into a composite of metal oxide nanoparticles and amorphous carbon. The oxidation of the MXene NSs is generally initiated at surface defects/edges and then propagates to other parts of the nanosheet [16][43]. The oxidation and/or the state of MXene oxidation can be investigated by Raman spectroscopy [23][50]. For sensing applications, generally at RT there is no notable oxidation, as the sensing temperature is low. However, at high temperatures, MXene composition changes due to partial oxidation, which leads to changes in the response to the target gas. Therefore, MXenes are mostly used at low temperature due to their relatively low oxidation resistance. Also, for RT MXene-based gas sensors, the long-term stability is acceptable due to working at low temperatures.
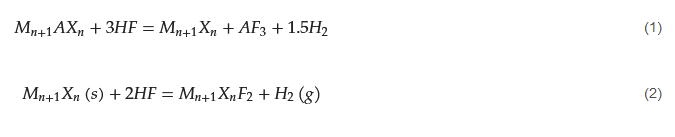 Due to the high toxicity of HF, MXenes are also synthesized by using fluoride salt-derived in situ HF etching, fluoride-free etching, molten salt etching, and electrochemical-derived etching of MAX phases [16][17][18][43,44,45]. MXenes have also been used as templates for the synthesis of other materials [19][46]. Owing to their large surface area, controllable interlayer spacing, abundant functional groups, and unique electrical properties, MXenes are considered promising materials for realizing RT gas sensors [20][21][47,48]. Among MXenes, the most widely used for gas sensors is Ti3C2Tx MXene due to the following facts: (i) large specific area and numerous terminal functional groups (-OH, -O, and -F) on Ti3C2Tx MXene can lead to the strong interface chemical connection with semiconductors and form a Schottky junction. (ii) The high metallic conductivity of Ti3C2Tx assures rapid carrier migration. (iii) The exposed terminal metal sites on MXenes may result in more active reactivity than that of carbon materials [22][49].
One of the major limitations for the practical application of MXenes is their poor oxidation stability under ambient conditions. In fact, exposure to water, air, heat, and light degrades the 2D MXenes into a composite of metal oxide nanoparticles and amorphous carbon. The oxidation of the MXene NSs is generally initiated at surface defects/edges and then propagates to other parts of the nanosheet [16][43]. The oxidation and/or the state of MXene oxidation can be investigated by Raman spectroscopy [23][50]. For sensing applications, generally at RT there is no notable oxidation, as the sensing temperature is low. However, at high temperatures, MXene composition changes due to partial oxidation, which leads to changes in the response to the target gas. Therefore, MXenes are mostly used at low temperature due to their relatively low oxidation resistance. Also, for RT MXene-based gas sensors, the long-term stability is acceptable due to working at low temperatures.
Due to the high toxicity of HF, MXenes are also synthesized by using fluoride salt-derived in situ HF etching, fluoride-free etching, molten salt etching, and electrochemical-derived etching of MAX phases [16][17][18][43,44,45]. MXenes have also been used as templates for the synthesis of other materials [19][46]. Owing to their large surface area, controllable interlayer spacing, abundant functional groups, and unique electrical properties, MXenes are considered promising materials for realizing RT gas sensors [20][21][47,48]. Among MXenes, the most widely used for gas sensors is Ti3C2Tx MXene due to the following facts: (i) large specific area and numerous terminal functional groups (-OH, -O, and -F) on Ti3C2Tx MXene can lead to the strong interface chemical connection with semiconductors and form a Schottky junction. (ii) The high metallic conductivity of Ti3C2Tx assures rapid carrier migration. (iii) The exposed terminal metal sites on MXenes may result in more active reactivity than that of carbon materials [22][49].
One of the major limitations for the practical application of MXenes is their poor oxidation stability under ambient conditions. In fact, exposure to water, air, heat, and light degrades the 2D MXenes into a composite of metal oxide nanoparticles and amorphous carbon. The oxidation of the MXene NSs is generally initiated at surface defects/edges and then propagates to other parts of the nanosheet [16][43]. The oxidation and/or the state of MXene oxidation can be investigated by Raman spectroscopy [23][50]. For sensing applications, generally at RT there is no notable oxidation, as the sensing temperature is low. However, at high temperatures, MXene composition changes due to partial oxidation, which leads to changes in the response to the target gas. Therefore, MXenes are mostly used at low temperature due to their relatively low oxidation resistance. Also, for RT MXene-based gas sensors, the long-term stability is acceptable due to working at low temperatures.
