Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Rita Xu and Version 1 by Jelica Grujic-Milanovic.
Cardiovascular diseases (CVDs) are a group of diseases with a very high rate of morbidity and mortality. The clinical presentation of CVDs can vary from asymptomatic to classic symptoms such as chest pain in patients with myocardial infarction. Current therapeutics for CVDs mainly target disease symptoms. The most common CVDs are coronary artery disease, acute myocardial infarction, atrial fibrillation, chronic heart failure, arterial hypertension, and valvular heart disease.
- cardiovascular diseases
- oxidative stress
- natural products
- atioxidants
1. Introduction
Cardiovascular diseases (CVDs) are a group of diseases of the heart and blood vessels that contribute most to morbidity and mortality in the human population [1,2][1][2]. Atherosclerosis and arterial thrombosis lead to ischemic damage of different organs such as the heart, brain, kidneys, and eyes, which can induce different failures of these organs [3]. The incidence of CVDs doubled in the last three decades, from 271 million in 1990 to 523 million in 2020, with an extremely high mortality rate of over 32% [4]. Over the past 30 years, mortality from CVDs has steadily increased. Today, one person dies every half a minute from CVDs indicating the devastating fact that one-third of all deaths in the world are due to CVDs [5]. Among the main modifiable risk factors that contribute to the development and prognosis of CVDs are a combination of different psychosocial factors: socioeconomic, behavioural, unhealthy diet, physical inactivity, illicit substance use, smoking, and environmental risk factors are of most importance.
Other nonmodifiable factors may also affect the risk of CVDs, such as genetic predisposition, ethnicity, gender, and age [6].
The clinical presentation of CVDs can vary from asymptomatic in patients with atherosclerosis [7], or often with arterial hypertension [8,9][8][9], or manifest as unspecified symptoms such as weakness, light-headedness, and nausea, or classic symptoms such as chest pain in patients with coronary artery disease (CAD) [10] or acute coronary syndromes (i.e., acute myocardial infarction) [11,12][11][12]. Different etiologic and clinical symptoms of CVDs share some common features at the cellular and molecular levels: chronic inflammation [13], mitochondrial dysfunction [14[14][15][16],15,16], and oxidative damage [17] to biomolecules including proteins, lipids, and nucleic acids. These factors are believed to be a progressive process that may occur as early as childhood [18].
Numerous studies in the past decades have been performed to develop better therapeutic strategies, but current medications for CVDs mainly target disease symptoms like therapeutics for CAD disease [19,20[19][20][21][22],21,22], acute myocardial infarction [23], atrial fibrillation [24[24][25][26],25,26], chronic heart failure [27[27][28],28], and arterial hypertension [29[29][30][31][32],30,31,32], respectively. Physicians should be careful in choosing the right kind of treatment depending on the type of disease that a patient has. Especially since certain therapeutics are not effective enough in the treatment of certain CVDs or show intolerance or side effects. Therefore, it is important to improve prevention and early diagnosis and develop therapeutic options to reduce the currently very high risk of CVDs. In recent years, the search for active ingredients from natural products and plant sources for the treatment, prevention and/or supportive therapy of various types of cardiovascular disease has become a hotspot.
The World Health Organization (WHO) estimates that approximately 75% of the world medical market consists of phytomedicine [2]. Numerous therapeutics approved by the Food and Drug Administration (FDA) used today to treat the most common CVDs have been extensively studied in preclinical and clinical studies. The efficacy of herbal medicine has been carefully reviewed in the preclinical field; no comparative studies have been found to confirm the efficacy of natural products compared to FDA-approved therapeutics for the treatment of CVDs.
2. Most Frequent Cardiovascular Diseases
CVDs is an umbrella term for all diseases of the heart and circulation [1]. The pathophysiology of the occurrence of CVDs depends on a whole range of different factors (Figure 1). Numerous studies have shown that several potential mechanisms, including endothelial dysfunction, inflammation, oxidative stress, atherosclerosis, dysregulated haemostasis, cardiac stress, and epigenetics, play a role in the development of vascular and cardiac damage [33]. The most common types of heart diseases are CAD including acute coronary syndromes, atrial fibrillation, chronic heart failure, valvular heart disease, arterial hypertension, and congenital heart disease [34].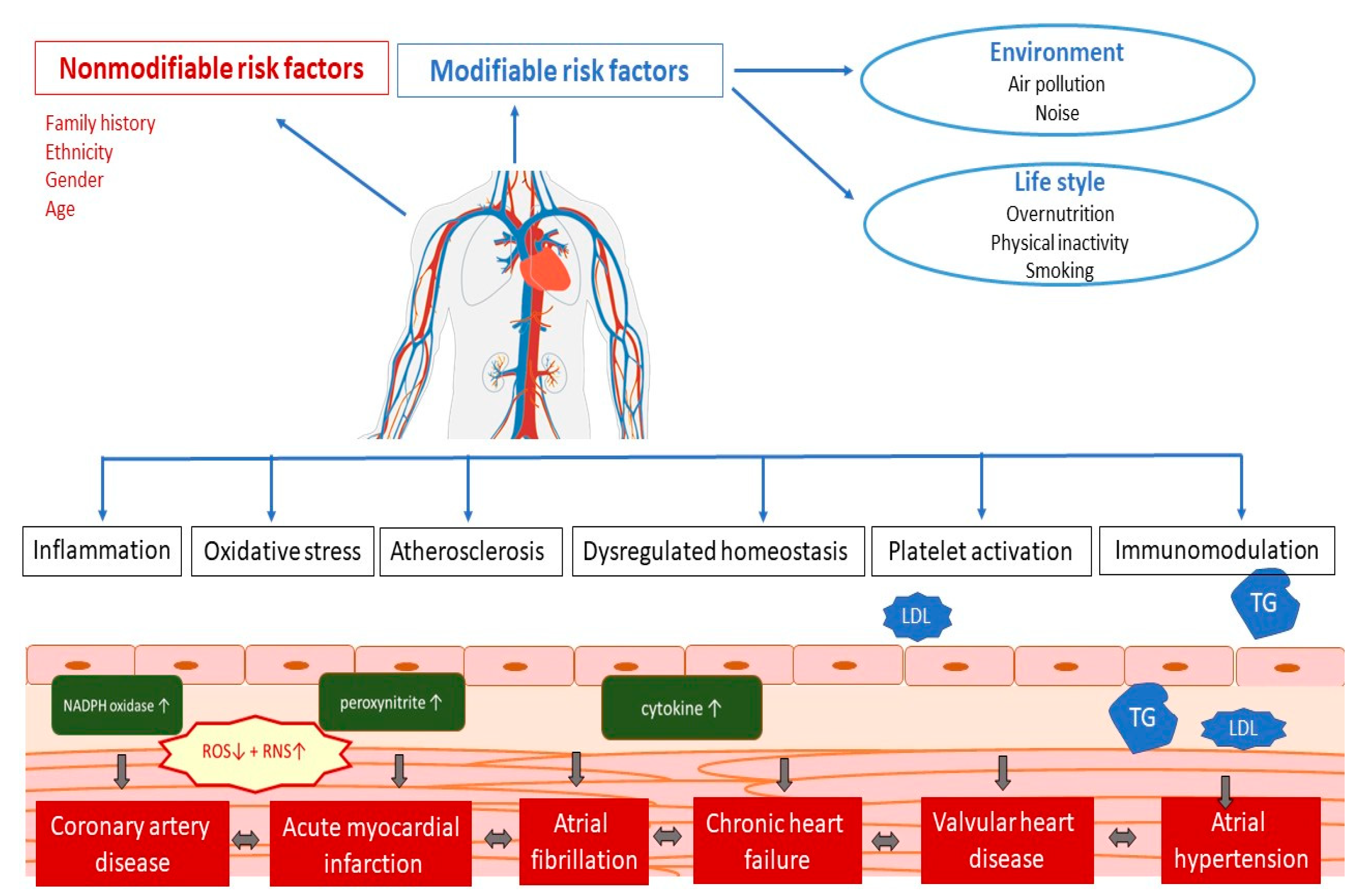
Figure 1. Pathophysiology of cardiovascular disease. LDL—low-density cholesterol; TG—triglycerides; ROS—reactive oxygen species; RNS—reactive nitrogen species.
2.1. Coronary Artery Disease
Coronary artery disease (CAD) is the most common CVD. Coronary atherosclerosis is a slow process that leads to the gradual intima thickening of the coronary arteries and subsequent development of atherosclerotic plaques that might be stable or prone to rupture due to inflammation. Atherosclerosis is the main factor that affects artery blood flow and leads to myocardial ischemia [7]. Coronary stenosis or occlusion may occur as a result of the formation of an intraluminal coronary thrombus [35]. Worldwide, an estimated 200 million people have CAD, and one in six deaths are caused by this disease [36]. In people with suspected CAD, the first option in a diagnosis is clinical diagnosis along with laboratory tests, electrocardiogram, exercise stress test, echocardiogram, and cardiac CT angiography [37].2.1.1. Treatment of Coronary Artery Disease Using Approved Drugs
Clinical guidelines for CAD treatments recommend a combination of lifestyle changes, pharmacological treatment, and, in some cases, cardiac interventions [21,38,39][21][38][39]. Lifestyle modification includes a healthy diet, smoking cessation, optimal physical activity, and stress management (Figure 2). As the development of CAD includes several risk factors such as hyperlipidaemia, obesity, diabetes mellitus, arterial hypertension, and smoking [16], pharmacological treatment includes target antiplatelet agents such as acetylsalicylic acid, clopidogrel, and blockers of adrenergic β receptors (beta blockers), hypolipemic drugs such as statins, fibrates or proprotein convertase subtilisin/kexin type 9 (PCSK-9) inhibitors, calcium channel blockers, organic nitrates, and various antihypertensive drugs (Figure 2) [21].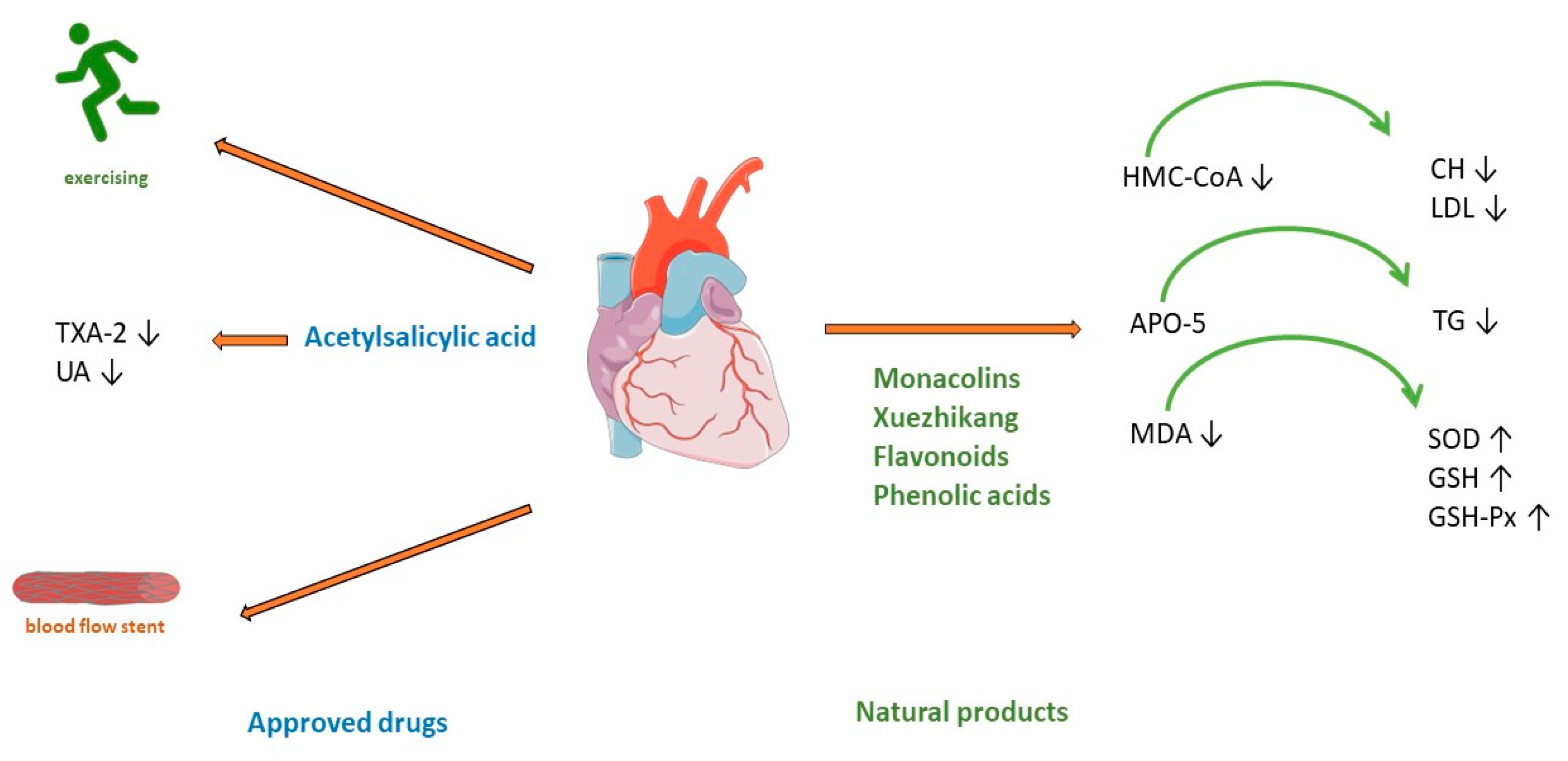
Figure 2. Treatment of coronary artery disease. TXA-2—Thromboxane A2; UA—uric acid; HMG-CoA—(3-hydroxy-3-methylglutaryl-coenzyme A) reductase; APO5—apolipoprotein A5; MDA—malondialdehyde; CH—cholesterol; LDL—low-density cholesterol; TG—triglyceride; SOD—superoxide dismutase; GSH—glutathione; GSH—glutathione peroxidase.
2.1.2. Treatment of Coronary Heart Disease Using Natural Products
Red yeast rice has been used as a herbal supplement for lowering cholesterol and lipoprotein in human blood. It is made by fermenting white rice with the yeast Monascus purpureus. Monacolin K is chemically like the cholesterol-lowering drug lovastatin. It acts by competitively inhibiting HMG-CoA (3-hydroxy-3-methylglutaryl-coenzyme A) reductase, the rate-limiting enzyme of the pathway of cholesterol synthesis (Figure 2). A meta-analysis of 6663 patients (from 20 randomized clinical trials) treated with red yeast rice extract showed a reduction in low-density cholesterol (LDL) [44][43]. The applied dose varies from 4.8 to 24 mg of monacolin K (1200–2400 mg of red yeast rice). The advantage of this treatment shows a significant reduction in the incidence of kidney injury and liver abnormalities compared with standard statin therapy [44][43]. However, research stated the limitation that reporting of adverse events was insufficient in most of studies. Thus, red yeast rice may be an effective treatment for reducing cardiovascular risk in statin-tolerant patients only when a mild profile of adverse reaction is confirmed [45][44]. Another meta-analysis of 15 high-quality randomized clinical trials with red yeast rice applied in doses of 200–4800 mg daily showed its efficacy and safety in the treatment of hyperlipidaemia. Hypertriglyceridemia represents an independent risk of coronary heart disease [46][45], but in most patients with this disease, high-intensity statin therapy is not useful because of the high incidence of statin intolerance [47][46], so treatment with Xuezhikang, may be a better alternative (Figure 2). Xuezhikang, an extract of Monascus purpureus, contains monacolins, PUFAs, flavonoids, and ergosterol. Xuezhikang is a supplementary product approved by the US Food and Drug Administration and has an excellent lowering performance on triglyceride and LDL-C levels (Figure 2). In coronary heart disease patients, 6 weeks of treatment with Xuezhikang extract (1200 mg/daily) resulted in a significant reduction in cholesterol, LDL-C, and triglycerides levels [48][47]. A review of 22 clinical randomized trials (most of them published in Chinese) showed that Xuezhikang is safe and effective in reducing cardiovascular events in coronary heart disease complicated by dyslipidaemia [49][48]. In rat models of high-fructose-diet-induced hypertriglyceridemia, Xuezhikang (XZK) was compared with simvastatin. Xuezhikang had a similar effect to simvastatin in lowering LDL-C, but a significantly higher hypotriglyceridaemic performance was attributed to the upregulation of apolipoprotein A5 (apoA5) via the peroxisome proliferator-activated receptor α (PPARα) signalling pathway [50][49]. Xuezhikang contributes to greater triglyceride reduction than simvastatin in hypertriglyceridemia rats by apoA5 elevation in hepatocytes [50][49]. Apo A5 is a target gene of PPARa and an important regulator of triglyceride metabolism [51][50]. Numerous studies have demonstrated the antioxidant effects of flavonoids. In a rat model of hyperlipidaemia, the administration of flavonoids from the seed of Amygdalus mongolica significantly lowered total cholesterol (TC), LDL-C, and the atherosclerosis index (Figure 2) [52][51]. The hypocholesterolaemic activity of the extract could be attributed to the fact it reduced malondialdehyde (MDA) and significantly increased activities of the antioxidant enzymes superoxide dismutase (SOD), glutathione (GSH), and glutathione peroxidase (GSH-Px) (Figure 2) [52][51]. In a meta-analysis of 39 prospective cohort studies (23,664 individuals with CHD), the intake of quercetin and kaempferol was linearly associated with a lower risk of CHD [53][52]. The lowest risk was observed in individuals whose intake was up to 12–14 mg/day of quercetin. Four phenolic acids are major compounds present in the methanolic extract of Quercus acutissima fruit (QF): caffeic acid, ellagic acid, gallic acid, and protocatechuic acid [54][53]. A recent investigation confirmed the important role of QF in cellular functions, such as gene regulation, cytoskeleton dynamics, receptor signalling, and cellular metabolism [55][54]. The anti-obesity, anti-hyperlipidaemic, anti-cholesterol, and anti-oxidative effects of QF are associated with the inhibition of acetylation, an important factor included in metabolic regulation (Figure 2) [56][55]. Saponin shows antiatherosclerosis activity by regulating lipid metabolism. A randomized controlled trial with Panax notoginseng saponins on 84 patients with CAD showed anti-lipidemic and anti-inflammatory effects. After 30 days of treatment with this saponin, high-density lipoprotein significantly increased, and white blood cell count decreased significantly [57][56]. An important mechanism of Panax notoginseng in vitro activity changes the methylation of miR-194, its promoter, and MAPK, FAS, RAS, and FOS, and significantly decreases the apoptosis rate of HUVECs cells [57][56]. The compound of Panax notoginseng saponin is available on drug markets as an over-the-counter drug in China and around the world [58][57]. Hydroxysaf flower yellow A is a c-glycosyl compound, a member of phenols, extracted from safflower (Carthamus tinctorius L.) which shows excellent therapeutic effects on CVDs by different mechanisms, is antioxidative, and has free radical scavenging abilities and anti-inflammatory activity. In models of atherosclerosis, it can suppress foam cell formation, vascular endothelial cell dysfunction, vascular smooth muscle cell proliferation and migration, and platelet activation by regulation of the reverse cholesterol process, fatty acids synthesis, and regulation of oxidative stress parameters [59][58]. Hydroxysaf flower yellow A reduces vascular inflammation by regulating the expression of NF-kappaB, Bax/Bcl-2, and TLR4/Rac1/Akt, PI3K/Akt/mTOR signalling pathways [59][58].2.2. Acute Myocardial Infarction
Acute myocardial infarction occurs when the blood supply to the heart is interrupted. In this situation, the heart is no longer supplied with sufficient oxygen and nutrients, so the muscle begins to die. In many cases, myocardial infarction is not fatal, especially if patients receive early treatment [11]. Myocardial infarction is the leading cause of death worldwide, with a prevalence approaching 3 million people [12].2.2.1. Treatment of Acute Myocardial Infarction Using Approved Drugs
The type of acute myocardial infarction (AMI) depends on the degree of coronary artery occlusion (Figure 3). The traditional recommendation for patients is to take one nitro-glycerine dose sublingually, 5 min apart, for up to three doses before admission to the emergency department [64][59]. After AMI, it is crucial to improve cardiac function and prevent postinfarction pathophysiologic remodelling [11]. Timely revascularization of the heart after AMI depends on the infarct size; therefore, an adequate reaction of physicians is very important. Standard treatment includes the use of antiplatelets and/or anticoagulants, beta-blockers, antiarrhythmics, opiate analgesics, antihypertensives such as angiotensin-converting enzyme (ACE) inhibitors, diuretics or calcium channel blockers, and oxygen therapy. Even prognosis most often depends on the type of AMI and administration of thrombolytic treatment or PCI [23]. Consequently, many patients in which this approach is used still progress to cardiac hypertrophy and heart failure.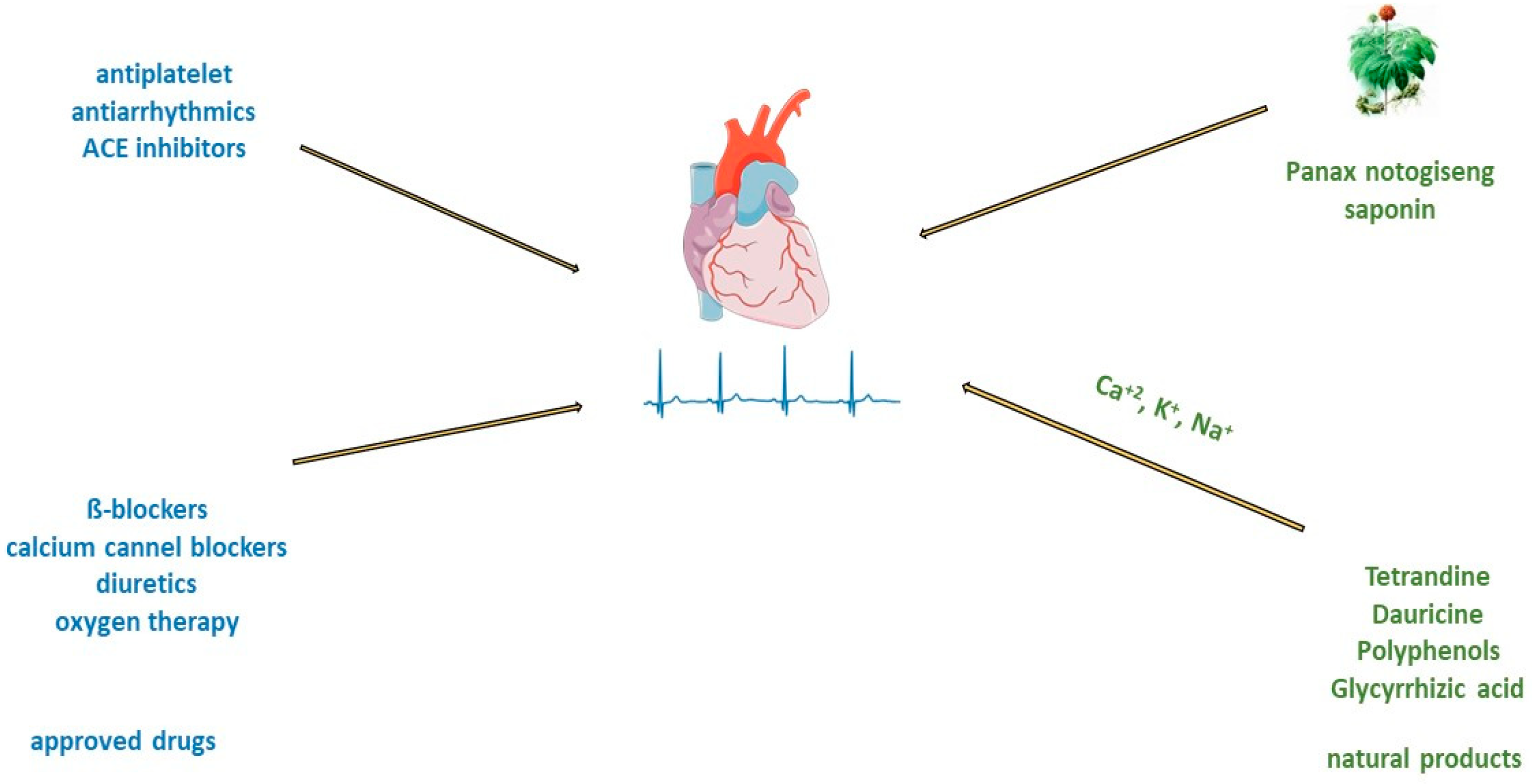
Figure 3. Treatment of acute myocardial infarction and atrial fibrillation with approved drugs or natural products.
2.2.2. Treatment of Acute Myocardial Infarction Using Natural Products
Saponin from Panax notoginseng exerts a cardioprotective effect in acute myocardial infarction [65][60]. In traditional medicine, the freeze-dried extract of Panax notoginseng for intravenous administration is used in the clinic for the prevention and treatment of cerebral ischemic injuries [66][61]. In addition, preclinical studies have shown the antioxidant and anti-inflammatory properties of this saponin [67,68][62][63]. Administration of Panax notoginseng injection to patients with myocardial infarction improved survival and cardiac function and decreased infarct size by direct inhibition of platelet aggregation and improved endothelial cell migration and angiogenesis (Figure 3). Panax notoginseng treatment significantly lowers lactate dehydrogenase and cardiac troponin I concentrations in the plasma of mice with MI. The mechanism of Panax notoginseng is manifested through the phosphorylation of AMPK and CaMKII in cardiomyocytes which induces autophagy [65][60]. Salvianolic acid B extracted from Salvia miltiorrhiza Bunge, promote angiogenesis in the marginal zone of MI by increasing the expression of VEGF [69][64]. In large myocardial infarction of rats, pretreatment with salvianolic acid B promotes the differentiation of mesenchymal stem cells into endothelial cells and has greater effects than the angiotensin-converting enzyme inhibitor benazepril [70][65]. In hyperlipidaemic animals with myocardial ischemia/reperfusion, hydroxysafflower yellow A inhibited the NF-κB signalling pathway, TLR4 signalling pathway, and phosphorylation of p38 [71][66]. Experimental acute myocardial ischemic models reduced serum levels of inflammatory factors such as TNF-alpha, IL-1β, and IL-18, reduced NLRP3 inflammasome expression, and induced autophagy [72][67].2.3. Atrial Fibrillation
Atrial fibrillation is a disorder of myocardial electrical conductivity that causes arrhythmia with various heart rhythms and rates [24]. As a result, too little blood is transported into the heart chambers (ventricles). This increases the risk of lung congestion and atrial thrombosis, as well as systemic thrombosis that causes a stroke. Uncontrolled atrial fibrillation can lead to chronic and acute heart failure [77][68]. The prevalence of atrial fibrillation ranged from 0.5% to 9% for people aged 50 to 90 years, respectively [42,78][42][69]. Causes of atrial fibrillation include sinus node dysfunction, coronary artery disease, rheumatic heart disease, arterial hypertension, hyperthyroidism, and alcohol [42]. Pathophysiological changes in atrial fibrillation include electrical remodelling, impaired atrial structure, autonomic nerve dysfunction, metabolic abnormalities, oxidative stress, etc. [78][69].2.3.1. Treatment of Atrial Fibrillation Using Approved Drugs
Treatment of atrial fibrillation usually includes rate and rhythm control, anticoagulation, and left atrial appendage closure. There is consensus that in patients with acute atrial fibrillation, parenteral anticoagulants such as heparin must be administered before cardioversion to reduce the risk of embolism [79][70]. Guidelines from various professional societies (The European Association of Cardio-Thoracic Surgery, American Heart Association (AHA)/American College of Cardiology (ACC)/Heart Rhythm Society (HRS)) recommend catheter ablation to restore sinus rhythm in patients with atrial fibrillation [80][71]. Patients with atrial fibrillation have an impaired quality of life and an increased risk of stroke, heart failure, cardiomyopathy, and acute coronary syndrome [79][70]. Administration of beta-blockers and calcium channel blockers is recommended as a first-line treatment for rate control of atrial fibrillation (Figure 3) [24]. Oral or intravenous application of different antiarrhythmic drugs, amiodarone, digoxin, flecainide, and ibutilide, increase the likelihood of reversion to sinus rhythm and can cause ventricular arrhythmias [25,26,81,82][25][26][72][73]. In addition, there are limitations such as that flecainide and propafenone should not be used in people with ischemic heart disease [25,26][25][26]. Treatment with verapamil, diltiazem, and digoxin may control heart rate, but they are unlikely to restore sinus rhythm [82][73]. On the other side, the long-time application of amiodarone can cause hepatotoxicity, interstitial lung disease, and thyroid dysfunction [83][74]. Therefore, the search for antiarrhythmic drugs from natural sources has been one of the priorities of scientists in recent years.2.3.2. Treatment of Atrial Fibrillation and Natural Compounds
There are many electrolytes in the human body; however, some of them, such as potassium, calcium, and sodium, play an important role in regulating signal transduction and ion transport across cell membranes. In patients with atrial fibrillation, due to electrolyte imbalance, the expression of ion channel proteins as well as gene transcription is altered, and fibrosis develops [84][75]. In an animal model of middle cerebral artery occlusion, saponin extracted from the roots of Panax notoginseng has significant antiarrhythmic and antiplatelet effects, regulates glycoprotein Ib-α, and reduces von Willebrand factor (VWF)-mediated platelet adhesion [85][76]. Myocardial tissue from the right and left atria of patients with atrial fibrillation after treatment with saponin increases in mitochondrial respiration rate [86][77]. The other group of saponins, ginsenosides, exert antiarrhythmic effects by modulating intracellular Ca2+ signalling through the inhibition of Ca2+ channels [87][78], or by regulating sodium, potassium, and calcium channels [88][79], or inhibiting collagen deposition in cardiomyocyte (Figure 3) [89][80]. Alkaloids are widely distributed in advanced plants and contain at least one nitrogen group. One of them, berberine, inhibits the occurrence of atrioventricular re-entrant tachycardia by regulating potassium and calcium ion channels and cyclic nucleotide-gated cation channels activated by hyperpolarization [90][81], or prolongs action potential duration and the effective refractory period in cardiac myocytes of rabbits [91][82]. Another alkaloid, tetrandrine, is antiarrhythmic by the inhibition of calcium, potassium, and sodium channels. An in vitro study of tetrandrine at a dosage of 100 µmol/L in rat cardiomyocytes, reduced Ca2+ influx into the sarcolemma and inhibited Ca2+ uptake into the sarcoplasmic reticulum by inhibiting ATP [92][83]. The significantly low dosage of tetrandrine, 15 µmol/L, increased the opening frequency and prolonged the opening time of calcium-activated potassium channels [93][84]. In a concentration-dependent manner (25, 125, 250, 400, 1000, and 2500 μmol/L) guanfacine blocked the L-type calcium channel and inhibited potassium currents in rat ventricular myocytes [94][85]. Dauricine reduced intracellular Ca2+ concentration by Na+-K+-ATPase and Ca2+-Mg2+-ATPase activation [95][86]. Matrine at a high concentration of 100 μM inhibited the expression of the human ether-a-go-go-related gene (hERG), encoded the rapidly activating, delayed rectifier potassium channel (IKr) important for cardiac repolarization, and at a low concentration of 1 μM, martine promoted hERG expression in rat cardiomyocytes. Indeed, matrine prolonged the action potential duration and the effective refractory period of cardiomyocytes [96][87]. Polyphenols are secondary metabolites widely distributed in the skin, roots, and leaves of fruits and medicinal plants. In vitro, cardiac arrhythmias caused by oxidative stress and calcium overload were significantly reduced in guinea pigs’ ventricular myocytes after treatment with resveratrol. Resveratrol reduced oxygen-free radical production, prevented the activation of calmodulin-activated protein kinase II, and inhibited L-type calcium channels [97][88]. Hydrogen-peroxide-induced ischemic arrhythmias in ventricular myocytes were reduced after resveratrol treatment by decreasing sodium concentration and reversing the sodium–calcium exchange current [98][89]. Puerarin protected rats’ ventricular myocytes against ischemia and reperfusion injury by regulating the calcium-activated potassium channel and activating protein kinase C [99][90].2.4. Chronic Heart Failure
Heart failure is a chronic, long-term condition in which the heart can no longer provide sufficient minute volume. This leads to circulus viciousness in terms of fluid retention starting from the legs, abdomen, and lungs to general edema (anasarca) in association with other symptoms of chronic heart failure. Chronic heart failure has increased to an estimated 37.7 million people, and almost 50% of these patients die within 5 years after diagnosis [101][91]. The risk increases with age, obesity, diabetes, smoking, alcohol abuse, or cocaine use. The guidelines of the American College of Cardiology Foundation (ACCF)/American Heart Association (AHA) defined chronic heart failure based on ejection fraction as preserved, intermediate, and heart failure reduced ejection fraction [101][91]. Additionally, in pathogenesis, myocardial interstitial fibrosis contributes to left ventricular dysfunction defined by the diffuse, disproportionate accumulation of collagen in the myocardial interstitium and activation of multiple molecular signalling pathways, such as endothelial dysfunction, hypertrophy of cardiomyocytes, and cardiac inflammation [10].2.4.1. Treatment of Chronic Heart Failure Using Approved Drugs
According to the guidelines for the diagnosis and treatment of acute and chronic heart failure, the following pharmacotherapeutic groups are recommended: drugs for the modulation of the renin-angiotensin-aldosterone (RAAS) and sympathetic nervous systems with ACE inhibitors or an angiotensin receptor-neprilysin inhibitor (ARNI), beta-blockers, mineralocorticoid receptor antagonists (MRA), loop and thiazide diuretics, and newly introduced gliflozins (inhibitors of sodium-glucose transport proteins 2) and ivabradine [101][91]. The side effects of high-dose diuretics can lead to low blood pressure, electrolyte disorders, and worsening of heart failure symptoms. Aldosterone antagonists can induce hyperkalaemia (Figure 4).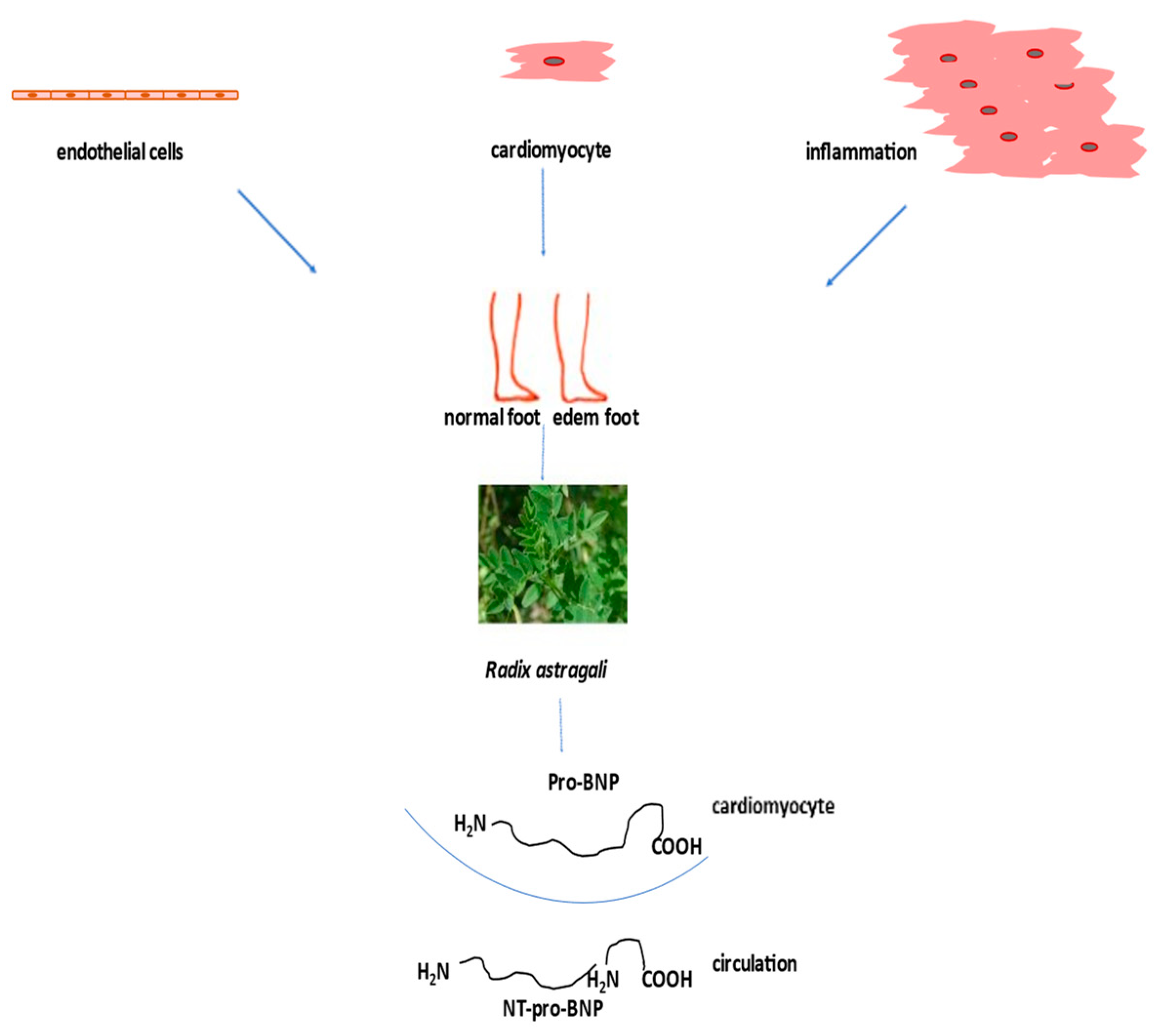
Figure 4. Treatment of heart failure with naturally derived astragaloside IV.
2.4.2. Treatment of Chronic Heart Failure and Natural Compounds
Heart failure is usually associated with different risk factors such as chronic inflammation, hypertension, type 2 diabetes mellitus (T2DM), obesity, coronary artery disease, and sarcopenia. Since the incidence of heart failure has increased in recent decades, and there are no adequate pharmacological therapies, there is an urgent need to test nonpharmacological strategies, such as the use of natural products, to improve clinical outcomes in these patients. One of the most commonly used natural products to treat heart failure when other medications do not help is digoxin, a secondary glycoside. Its side effects include digestive problems, confusion, and visual disturbances [102][92]. Saponin astragaloside IV (AS-IV) is one of the main active ingredients of the aqueous extract Radix Astragali Huangqi (The Root of Astragalus membranaceus var. mongholicus) injected into heart failure patients. This injection improves cardiac function by increasing left ventricular ejection fraction and decreasing stroke volume [103][93]. A meta-analysis of seven randomized clinical trial with 550 patients in total has shown that Di’ao Xinxuekang capsules, steroidal saponins, extracted from Dioscorea panthaica, have a better protective effect on heart failure patients than isosorbide dinitrate [104][94]. Also, the incidence of adverse events was lower in Di’ao Xinxuekang capsule-treated patients. In a clinical trial, a total of 512 patients with chronic heart failure were divided into a control/placebo group and a treated group. The treated group took capsules of qili qiangxin and saponin for 12 weeks. Treatment significantly decreased plasma N-terminal pro-B-type natriuretic peptide and improved left ventricular ejection fraction (Figure 4) and quality of life [105][95]. In chronic heart failure patients, Shenmai injection, ginsenoside saponin, improved left ventricular diastolic function [106][96].References
- Olvera Lopez, E.; Ballard, B.D.; Jan, A. Cardiovascular Disease. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2023.
- Organization., W.H. Available online: https://www.who.int/news-room/fact-sheets/detail/hypertension (accessed on 8 October 2023).
- Willeit, J.; Kiechl, S. Biology of arterial atheroma. Cerebrovasc. Dis. 2000, 10 (Suppl. S5), 1–8.
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021.
- Centers for Disease Control and Prevention. CDC Protects and Prepares Communities; Department of Health & Human Services, CDC: Atlanta, GA, USA, 2020.
- Bowman, L.; Weidinger, F.; Albert, M.A.; Fry, E.T.A.; Pinto, F.J.; Clinical Trial Expert Group and ESC Patient Forum. Randomized Trials Fit for the 21st Century: A Joint Opinion From the European Society of Cardiology, American Heart Association, American College of Cardiology, and the World Heart Federation. Circulation 2023, 147, 925–929.
- Frak, W.; Wojtasinska, A.; Lisinska, W.; Mlynarska, E.; Franczyk, B.; Rysz, J. Pathophysiology of Cardiovascular Diseases: New Insights into Molecular Mechanisms of Atherosclerosis, Arterial Hypertension, and Coronary Artery Disease. Biomedicines 2022, 10, 1938.
- Verma, N.; Rastogi, S.; Chia, Y.C.; Siddique, S.; Turana, Y.; Cheng, H.M.; Sogunuru, G.P.; Tay, J.C.; Teo, B.W.; Wang, T.D.; et al. Non-pharmacological management of hypertension. J. Clin. Hypertens. 2021, 23, 1275–1283.
- Vidal-Petiot, E. Thresholds for Hypertension Definition, Treatment Initiation, and Treatment Targets: Recent Guidelines at a Glance. Circulation 2022, 146, 805–807.
- Schirone, L.; Forte, M.; Palmerio, S.; Yee, D.; Nocella, C.; Angelini, F.; Pagano, F.; Schiavon, S.; Bordin, A.; Carrizzo, A.; et al. A Review of the Molecular Mechanisms Underlying the Development and Progression of Cardiac Remodeling. Oxid. Med. Cell. Longev. 2017, 2017, 3920195.
- Lu, L.; Liu, M.; Sun, R.; Zheng, Y.; Zhang, P. Myocardial Infarction: Symptoms and Treatments. Cell Biochem. Biophys. 2015, 72, 865–867.
- Salari, N.; Morddarvanjoghi, F.; Abdolmaleki, A.; Rasoulpoor, S.; Khaleghi, A.A.; Hezarkhani, L.A.; Shohaimi, S.; Mohammadi, M. The global prevalence of myocardial infarction: A systematic review and meta-analysis. BMC Cardiovasc. Disord. 2023, 23, 206.
- Ferrucci, L.; Fabbri, E. Inflammageing: Chronic inflammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 2018, 15, 505–522.
- Dikalova, A.; Dikalov, S. Response by Dikalova and Dikalov to Letter Regarding Article, “Mitochondrial Deacetylase Sirt3 Reduces Vascular Dysfunction and Hypertension While Sirt3 Depletion in Essential Hypertension Is Linked to Vascular Inflammation and Oxidative Stress”. Circ. Res. 2020, 126, e33–e34.
- Zhao, M.; Wang, Y.; Li, L.; Liu, S.; Wang, C.; Yuan, Y.; Yang, G.; Chen, Y.; Cheng, J.; Lu, Y.; et al. Mitochondrial ROS promote mitochondrial dysfunction and inflammation in ischemic acute kidney injury by disrupting TFAM-mediated mtDNA maintenance. Theranostics 2021, 11, 1845–1863.
- Sauer, F.; Riou, M.; Charles, A.L.; Meyer, A.; Andres, E.; Geny, B.; Talha, S. Pathophysiology of Heart Failure: A Role for Peripheral Blood Mononuclear Cells Mitochondrial Dysfunction? J. Clin. Med. 2022, 11, 741.
- Shaito, A.; Aramouni, K.; Assaf, R.; Parenti, A.; Orekhov, A.; Yazbi, A.E.; Pintus, G.; Eid, A.H. Oxidative Stress-Induced Endothelial Dysfunction in Cardiovascular Diseases. Front. Biosci. 2022, 27, 105.
- McGill, H.C., Jr.; McMahan, C.A.; Zieske, A.W.; Tracy, R.E.; Malcom, G.T.; Herderick, E.E.; Strong, J.P. Association of Coronary Heart Disease Risk Factors with microscopic qualities of coronary atherosclerosis in youth. Circulation 2000, 102, 374–379.
- Alderman, M.; Aiyer, K.J. Uric acid: Role in cardiovascular disease and effects of losartan. Curr. Med. Res. Opin. 2004, 20, 369–379.
- Antithrombotic Trialists, C.; Baigent, C.; Blackwell, L.; Collins, R.; Emberson, J.; Godwin, J.; Peto, R.; Buring, J.; Hennekens, C.; Kearney, P.; et al. Aspirin in the primary and secondary prevention of vascular disease: Collaborative meta-analysis of individual participant data from randomised trials. Lancet 2009, 373, 1849–1860.
- Qian, X.; Deng, H.; Yuan, J.; Hu, J.; Dai, L.; Jiang, T. Evaluating the efficacy and safety of percutaneous coronary intervention (PCI) versus the optimal drug therapy (ODT) for stable coronary heart disease: A systematic review and meta-analysis. J. Thorac. Dis. 2022, 14, 1183–1192.
- Cheng, A.; Malkin, C.; Briffa, N.P. Antithrombotic therapy after heart valve intervention: Review of mechanisms, evidence and current guidance. Heart 2023.
- Huang, S.; Frangogiannis, N.G. Anti-inflammatory therapies in myocardial infarction: Failures, hopes and challenges. Br. J. Pharmacol. 2018, 175, 1377–1400.
- Lip, G.Y.; Fauchier, L.; Freedman, S.B.; Van Gelder, I.; Natale, A.; Gianni, C.; Nattel, S.; Potpara, T.; Rienstra, M.; Tse, H.F.; et al. Atrial fibrillation. Nat. Rev. Dis. Primers 2016, 2, 16016.
- Holmes, A.P.; Saxena, P.; Kabir, S.N.; O’Shea, C.; Kuhlmann, S.M.; Gupta, S.; Fobian, D.; Apicella, C.; O’Reilly, M.; Syeda, F.; et al. Atrial resting membrane potential confers sodium current sensitivity to propafenone, flecainide and dronedarone. Heart Rhythm 2021, 18, 1212–1220.
- Cay, S.; Kara, M.; Ozcan, F.; Ozeke, O.; Aksu, T.; Aras, D.; Topaloglu, S. Propafenone use in coronary artery disease patients undergoing atrial fibrillation ablation. J. Interv. Card. Electrophysiol. 2022, 65, 381–389.
- Faragli, A.; Tano, G.D.; Carlini, C.; Nassiacos, D.; Gori, M.; Confortola, G.; Lo Muzio, F.P.; Rapis, K.; Abawi, D.; Post, H.; et al. In-hospital Heart Rate Reduction With Beta Blockers and Ivabradine Early After Recovery in Patients With Acute Decompensated Heart Failure Reduces Short-Term Mortality and Rehospitalization. Front. Cardiovasc. Med. 2021, 8, 665202.
- Kim, R.; Suresh, K.; Rosenberg, M.A.; Tan, M.S.; Malone, D.C.; Allen, L.A.; Kao, D.P.; Anderson, H.D.; Tiwari, P.; Trinkley, K.E. A machine learning evaluation of patient characteristics associated with prescribing of guideline-directed medical therapy for heart failure. Front. Cardiovasc. Med. 2023, 10, 1169574.
- Bertoluci, C.; Foppa, M.; Santos, A.B.S.; Fuchs, S.C.; Fuchs, F.D. Diuretics are Similar to Losartan on Echocardiographic Target-Organ Damage in Stage I Hypertension. PREVER-Treatment Study. Arq. Bras. Cardiol. 2019, 112, 87–90.
- Benard, B.; Durand, M.; Berthoumieux, S.; Gauthier, M.; L’Archeveque, H.; Lamarre-Cliche, M.; Laskine, M. The impact of beta-blockers on the central and delta systolic pressures in a real-world population with treated hypertension: A cross-sectional study. Health Sci. Rep. 2022, 5, e948.
- Faucon, A.L.; Fu, E.L.; Stengel, B.; Mazhar, F.; Evans, M.; Carrero, J.J. A nationwide cohort study comparing the effectiveness of diuretics and calcium channel blockers on top of renin-angiotensin system inhibitors on chronic kidney disease progression and mortality. Kidney Int. 2023, 104, 542–551.
- Nachawati, D.; Patel, J.B. Alpha-Blockers. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2023.
- Lee, S.N.; Yun, J.S.; Ko, S.H.; Ahn, Y.B.; Yoo, K.D.; Her, S.H.; Moon, D.; Jung, S.H.; Won, H.H.; Kim, D. Impacts of gender and lifestyle on the association between depressive symptoms and cardiovascular disease risk in the UK Biobank. Sci. Rep. 2023, 13, 10758.
- Sacco, R.L.; Roth, G.A.; Reddy, K.S.; Arnett, D.K.; Bonita, R.; Gaziano, T.A.; Heidenreich, P.A.; Huffman, M.D.; Mayosi, B.M.; Mendis, S.; et al. The Heart of 25 by 25: Achieving the Goal of Reducing Global and Regional Premature Deaths From Cardiovascular Diseases and Stroke: A Modeling Study From the American Heart Association and World Heart Federation. Glob. Heart 2016, 11, 251–264.
- Ambrose, J.A.; Singh, M. Pathophysiology of coronary artery disease leading to acute coronary syndromes. F1000Prime Rep. 2015, 7, 8.
- Disease, G.B.D.; Injury, I.; Prevalence, C. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858.
- Li, J.L.; Zhou, J.R.; Tan, P.; Chen, J. Dynamic assessment of coronary artery during different cardiac cycle in patients with coronary artery disease using coronary CT angiography. Perfusion 2023, 38, 1453–1460.
- Knuuti, J.; Wijns, W.; Saraste, A.; Capodanno, D.; Barbato, E.; Funck-Brentano, C.; Prescott, E.; Storey, R.F.; Deaton, C.; Cuisset, T.; et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur. Heart J. 2020, 41, 407–477.
- Byrne, R.A.; Rossello, X.; Coughlan, J.J.; Barbato, E.; Berry, C.; Chieffo, A.; Claeys, M.J.; Dan, G.A.; Dweck, M.R.; Galbraith, M.; et al. 2023 ESC Guidelines for the management of acute coronary syndromes. Eur. Heart J. 2023, 44, 3720–3826.
- Park, J.; Kim, S.H.; Kim, M.; Lee, J.; Choi, Y.; Kim, H.; Kim, T.O.; Kang, D.Y.; Ahn, J.M.; Yoo, J.S.; et al. Impact of Optimal Medical Therapy on Long-Term Outcomes After Myocardial Revascularization for Multivessel Coronary Disease. Am. J. Cardiol. 2023, 203, 81–91.
- Yoon, G.S.; Choi, S.H.; Kwon, S.W.; Park, S.D.; Woo, S.I. A prospective double-blinded randomized study on drug-eluting stent implantation into nitrate-induced maximally dilated vessels in patients with coronary artery disease. Trials 2023, 24, 460.
- Lippi, G.; Sanchis-Gomar, F.; Cervellin, G. Global epidemiology of atrial fibrillation: An increasing epidemic and public health challenge. Int. J. Stroke 2021, 16, 217–221.
- Gerards, M.C.; Terlou, R.J.; Yu, H.; Koks, C.H.; Gerdes, V.E. Traditional Chinese lipid-lowering agent red yeast rice results in significant LDL reduction but safety is uncertain—A systematic review and meta-analysis. Atherosclerosis 2015, 240, 415–423.
- Li, P.; Wang, Q.; Chen, K.; Zou, S.; Shu, S.; Lu, C.; Wang, S.; Jiang, Y.; Fan, C.; Luo, Y. Red Yeast Rice for Hyperlipidemia: A Meta-Analysis of 15 High-Quality Randomized Controlled Trials. Front. Pharmacol. 2021, 12, 819482.
- Liu, J.; Zeng, F.F.; Liu, Z.M.; Zhang, C.X.; Ling, W.H.; Chen, Y.M. Effects of blood triglycerides on cardiovascular and all-cause mortality: A systematic review and meta-analysis of 61 prospective studies. Lipids Health Dis. 2013, 12, 159.
- Zhao, S.; Wang, Y.; Mu, Y.; Yu, B.; Ye, P.; Yan, X.; Li, Z.; Wei, Y.; Ambegaonakr, B.M.; Hu, D.; et al. Prevalence of dyslipidaemia in patients treated with lipid-lowering agents in China: Results of the DYSlipidemia International Study (DYSIS). Atherosclerosis 2014, 235, 463–469.
- Zhu, L.Y.; Wen, X.Y.; Xiang, Q.Y.; Guo, L.L.; Xu, J.; Zhao, S.P.; Liu, L. Comparison of the Reductions in LDL-C and Non-HDL-C Induced by the Red Yeast Rice Extract Xuezhikang between Fasting and Non-fasting States in Patients with Coronary Heart Disease. Front. Cardiovasc. Med. 2021, 8, 674446.
- Shang, Q.; Liu, Z.; Chen, K.; Xu, H.; Liu, J. A systematic review of xuezhikang, an extract from red yeast rice, for coronary heart disease complicated by dyslipidemia. Evid. Based Complement. Altern. Med. 2012, 2012, 636547.
- Zhao, S.P.; Li, R.; Dai, W.; Yu, B.L.; Chen, L.Z.; Huang, X.S. Xuezhikang contributes to greater triglyceride reduction than simvastatin in hypertriglyceridemia rats by up-regulating apolipoprotein A5 via the PPARalpha signaling pathway. PLoS ONE 2017, 12, e0184949.
- Lien, C.F.; Lin, C.S.; Shyue, S.K.; Hsieh, P.S.; Chen, S.J.; Lin, Y.T.; Chien, S.; Tsai, M.C. Peroxisome proliferator-activated receptor delta improves the features of atherosclerotic plaque vulnerability by regulating smooth muscle cell phenotypic switching. Br. J. Pharmacol. 2023, 180, 2085–2101.
- Zheng, Q.N.; Wang, J.; Zhou, H.B.; Niu, S.F.; Liu, Q.L.; Yang, Z.J.; Wang, H.; Zhao, Y.S.; Shi, S.L. Effectiveness of Amygdalus mongolica oil in hyperlipidemic rats and underlying antioxidant processes. J. Toxicol. Environ. Health A 2017, 80, 1193–1198.
- Micek, A.; Godos, J.; Del Rio, D.; Galvano, F.; Grosso, G. Dietary Flavonoids and Cardiovascular Disease: A Comprehensive Dose-Response Meta-Analysis. Mol. Nutr. Food Res. 2021, 65, e2001019.
- Elansary, H.O.; Szopa, A.; Kubica, P.; Ekiert, H.; Mattar, M.A.; Al-Yafrasi, M.A.; El-Ansary, D.O.; El-Abedin, T.K.Z.; Yessoufou, K. Polyphenol Profile and Pharmaceutical Potential of Quercus spp. Bark Extracts. Plants 2019, 8, 486.
- Verdin, E.; Ott, M. 50 years of protein acetylation: From gene regulation to epigenetics, metabolism and beyond. Nat. Rev. Mol. Cell Biol. 2015, 16, 258–264.
- Hwang, J.T.; Choi, H.K.; Kim, S.H.; Chung, S.; Hur, H.J.; Park, J.H.; Chung, M.Y. Hypolipidemic Activity of Quercus acutissima Fruit Ethanol Extract is Mediated by Inhibition of Acetylation. J. Med. Food 2017, 20, 542–549.
- Duan, L.; Liu, Y.; Li, J.; Zhang, Y.; Dong, Y.; Liu, C.; Wang, J. Panax notoginseng Saponins Alleviate Coronary Artery Disease through Hypermethylation of the miR-194-MAPK Pathway. Front. Pharmacol. 2022, 13, 829416.
- Duan, L.; Xiong, X.; Hu, J.; Liu, Y.; Li, J.; Wang, J. Panax notoginseng Saponins for Treating Coronary Artery Disease: A Functional and Mechanistic Overview. Front. Pharmacol. 2017, 8, 702.
- Xue, X.; Deng, Y.; Wang, J.; Zhou, M.; Liao, L.; Wang, C.; Peng, C.; Li, Y. Hydroxysafflor yellow A, a natural compound from Carthamus tinctorius L with good effect of alleviating atherosclerosis. Phytomedicine 2021, 91, 153694.
- Antman, E.M.; Anbe, D.T.; Armstrong, P.W.; Bates, E.R.; Green, L.A.; Hand, M.; Hochman, J.S.; Krumholz, H.M.; Kushner, F.G.; Lamas, G.A.; et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction--executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1999 Guidelines for the Management of Patients With Acute Myocardial Infarction). Circulation 2004, 110, 588–636.
- Wang, D.; Lv, L.; Xu, Y.; Jiang, K.; Chen, F.; Qian, J.; Chen, M.; Liu, G.; Xiang, Y. Cardioprotection of Panax Notoginseng saponins against acute myocardial infarction and heart failure through inducing autophagy. Biomed. Pharmacother. 2021, 136, 111287.
- Guo, H.; Adah, D.; James, P.B.; Liu, Q.; Li, G.; Ahmadu, P.; Chai, L.; Wang, S.; Liu, Y.; Hu, L. Xueshuantong Injection (Lyophilized) Attenuates Cerebral Ischemia/Reperfusion Injury by the Activation of Nrf2-VEGF Pathway. Neurochem. Res. 2018, 43, 1096–1103.
- Zheng, Q.; Bao, X.Y.; Zhu, P.C.; Tong, Q.; Zheng, G.Q.; Wang, Y. Ginsenoside Rb1 for Myocardial Ischemia/Reperfusion Injury: Preclinical Evidence and Possible Mechanisms. Oxid. Med. Cell Longev. 2017, 2017, 6313625.
- Kim, J.H. Pharmacological and medical applications of Panax ginseng and ginsenosides: A review for use in cardiovascular diseases. J. Ginseng Res. 2018, 42, 264–269.
- Lin, C.; Liu, Z.; Lu, Y.; Yao, Y.; Zhang, Y.; Ma, Z.; Kuai, M.; Sun, X.; Sun, S.; Jing, Y.; et al. Cardioprotective effect of Salvianolic acid B on acute myocardial infarction by promoting autophagy and neovascularization and inhibiting apoptosis. J. Pharm. Pharmacol. 2016, 68, 941–952.
- He, H.B.; Yang, X.Z.; Shi, M.Q.; Zeng, X.W.; Wu, L.M.; Li, L.D. Comparison of cardioprotective effects of salvianolic acid B and benazepril on large myocardial infarction in rats. Pharmacol. Rep. 2008, 60, 369–381.
- Han, D.; Wei, J.; Zhang, R.; Ma, W.; Shen, C.; Feng, Y.; Xia, N.; Xu, D.; Cai, D.; Li, Y.; et al. Hydroxysafflor yellow A alleviates myocardial ischemia/reperfusion in hyperlipidemic animals through the suppression of TLR4 signaling. Sci. Rep. 2016, 6, 35319.
- Ye, J.; Lu, S.; Wang, M.; Ge, W.; Liu, H.; Qi, Y.; Fu, J.; Zhang, Q.; Zhang, B.; Sun, G.; et al. Hydroxysafflor Yellow A Protects Against Myocardial Ischemia/Reperfusion Injury via Suppressing NLRP3 Inflammasome and Activating Autophagy. Front. Pharmacol. 2020, 11, 1170.
- He, J.; Li, S.; Ding, Y.; Tong, Y.; Li, X. Research Progress on Natural Products’ Therapeutic Effects on Atrial Fibrillation by Regulating Ion Channels. Cardiovasc. Ther. 2022, 2022, 4559809.
- Pistoia, F.; Sacco, S.; Tiseo, C.; Degan, D.; Ornello, R.; Carolei, A. The Epidemiology of Atrial Fibrillation and Stroke. Cardiol. Clin. 2016, 34, 255–268.
- Isakadze, N.; Kazzi, Z.; Bantsadze, T.; Gotsadze, G.; Butkhikridze, N.; El Chami, M.; Papiashvili, G. Updated Atrial Fibrillation Management Recommendations for Georgian Hospitals Based on the 2020 European Society of Cardiology Atrial Fibrillation Guidelines. Georgian Med. News 2022, 333, 13–16.
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomstrom-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur. Heart J. 2021, 42, 373–498.
- Camm, A.J.; Savelieva, I. Some patients with paroxysmal atrial fibrillation should carry flecainide or propafenone to self treat. BMJ 2007, 334, 637.
- Levy, S. Cardioversion of recent-onset atrial fibrillation using intravenous antiarrhythmics: A European perspective. J. Cardiovasc. Electrophysiol. 2021, 32, 3259–3269.
- Siemers, L.A.; MacGillivray, J.; Andrade, J.G.; Turgeon, R.D. Chronic Amiodarone Use and the Risk of Cancer: A Systematic Review and Meta-analysis. CJC Open 2021, 3, 109–114.
- Lu, Y.Y.; Cheng, C.C.; Chen, Y.C.; Lin, Y.K.; Chen, S.A.; Chen, Y.J. Electrolyte disturbances differentially regulate sinoatrial node and pulmonary vein electrical activity: A contribution to hypokalemia- or hyponatremia-induced atrial fibrillation. Heart Rhythm 2016, 13, 781–788.
- Xu, Z.Y.; Xu, Y.; Xie, X.F.; Tian, Y.; Sui, J.H.; Sun, Y.; Lin, D.S.; Gao, X.; Peng, C.; Fan, Y.J. Anti-platelet aggregation of Panax notoginseng triol saponins by regulating GP1BA for ischemic stroke therapy. Chin. Med. 2021, 16, 12.
- Slagsvold, K.H.; Johnsen, A.B.; Rognmo, O.; Hoydal, M.A.; Wisloff, U.; Wahba, A. Mitochondrial respiration and microRNA expression in right and left atrium of patients with atrial fibrillation. Physiol. Genom. 2014, 46, 505–511.
- Wang, Y.G.; Zima, A.V.; Ji, X.; Pabbidi, R.; Blatter, L.A.; Lipsius, S.L. Ginsenoside Re suppresses electromechanical alternans in cat and human cardiomyocytes. Am. J. Physiol. Heart Circ. Physiol. 2008, 295, H851–H859.
- Jiang, L.; Yin, X.; Chen, Y.H.; Chen, Y.; Jiang, W.; Zheng, H.; Huang, F.Q.; Liu, B.; Zhou, W.; Qi, L.W.; et al. Proteomic analysis reveals ginsenoside Rb1 attenuates myocardial ischemia/reperfusion injury through inhibiting ROS production from mitochondrial complex I. Theranostics 2021, 11, 1703–1720.
- Li, X.; Xiang, N.; Wang, Z. Ginsenoside Rg2 attenuates myocardial fibrosis and improves cardiac function after myocardial infarction via AKT signaling pathway. Biosci. Biotechnol. Biochem. 2020, 84, 2199–2206.
- Mirhadi, E.; Rezaee, M.; Malaekeh-Nikouei, B. Nano strategies for berberine delivery, a natural alkaloid of Berberis. Biomed. Pharmacother. 2018, 104, 465–473.
- Zhou, Z.W.; Zheng, H.C.; Zhao, L.F.; Li, W.; Hou, J.W.; Yu, Y.; Miao, P.Z.; Zhu, J.M. Effect of berberine on acetylcholine-induced atrial fibrillation in rabbit. Am. J. Transl. Res. 2015, 7, 1450–1457.
- Wang, H.X.; Kwan, C.Y.; Wong, T.M. Tetrandrine inhibits electrically induced i transient in the isolated single rat cardiomyocyte. Eur. J. Pharmacol. 1997, 319, 115–122.
- Wu, S.N.; Li, H.F.; Lo, Y.C. Characterization of tetrandrine-induced inhibition of large-conductance calcium-activated potassium channels in a human endothelial cell line (HUV-EC-C). J. Pharmacol. Exp. Ther. 2000, 292, 188–195.
- Huang, B.; Qin, D.; El-Sherif, N. Spatial alterations of Kv channels expression and K+ currents in post-MI remodeled rat heart. Cardiovasc. Res. 2001, 52, 246–254.
- Liu, Q.N.; Zhang, L.; Gong, P.L.; Yang, X.Y.; Zeng, F.D. Inhibitory effects of dauricine on early afterdepolarizations and L-type calcium current. Can. J. Physiol. Pharmacol. 2009, 87, 954–962.
- Zhou, J.; Ma, W.; Wang, X.; Liu, H.; Miao, Y.; Wang, J.; Du, P.; Chen, Y.; Zhang, Y.; Liu, Z. Matrine Suppresses Reactive Oxygen Species (ROS)-Mediated MKKs/p38-Induced Inflammation in Oxidized Low-Density Lipoprotein (ox-LDL)-Stimulated Macrophages. Med. Sci. Monit. 2019, 25, 4130–4136.
- Hernandez-Cascales, J. Resveratrol enhances the inotropic effect but inhibits the proarrhythmic effect of sympathomimetic agents in rat myocardium. PeerJ 2017, 5, e3113.
- Qian, C.; Ma, J.; Zhang, P.; Luo, A.; Wang, C.; Ren, Z.; Kong, L.; Zhang, S.; Wang, X.; Wu, Y. Resveratrol attenuates the Na+-dependent intracellular Ca2+ overload by inhibiting H2O2-induced increase in late sodium current in ventricular myocytes. PLoS ONE 2012, 7, e51358.
- Gao, Q.; Yang, B.; Ye, Z.G.; Wang, J.; Bruce, I.C.; Xia, Q. Opening the calcium-activated potassium channel participates in the cardioprotective effect of puerarin. Eur. J. Pharmacol. 2007, 574, 179–184.
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Bohm, M.; Burri, H.; Butler, J.; Celutkiene, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2021, 42, 3599–3726.
- Chang, K.Y.; Giorgio, K.; Schmitz, K.; Walker, R.F.; Prins, K.W.; Pritzker, M.R.; Archer, S.L.; Lutsey, P.L.; Thenappan, T. Effect of Chronic Digoxin Use on Mortality and Heart Failure Hospitalization in Pulmonary Arterial Hypertension. J. Am. Heart Assoc. 2023, 12, e027559.
- Zhou, Z.L.; Yu, P.; Lin, D. Study on effect of Astragalus injection in treating congestive heart failure. Zhongguo Zhong Xi Yi Jie He Za Zhi 2001, 21, 747–749.
- Jia, Y.; Chen, C.; Ng, C.S.; Leung, S.W. Meta-Analysis of Randomized Controlled Trials on the Efficacy of Di’ao Xinxuekang Capsule and Isosorbide Dinitrate in Treating Angina Pectoris. Evid. Based Complement. Altern. Med. 2012, 2012, 904147.
- Li, X.; Zhang, J.; Huang, J.; Ma, A.; Yang, J.; Li, W.; Wu, Z.; Yao, C.; Zhang, Y.; Yao, W.; et al. A multicenter, randomized, double-blind, parallel-group, placebo-controlled study of the effects of qili qiangxin capsules in patients with chronic heart failure. J. Am. Coll. Cardiol. 2013, 62, 1065–1072.
- Ma, R.G.; Wang, C.X.; Shen, Y.H.; Wang, Z.Q.; Ma, J.H.; Huang, L.S. Effect of Shenmai Injection on ventricular diastolic function in patients with chronic heart failure: An assessment by tissue Doppler imaging. Chin. J. Integr. Med. 2010, 16, 173–175.
More
