Exosomes are a subtype of extracellular vesicles (EVs) with a diameter of 30~150 nm (averaging ~100 nm) that are primarily produced through the endosomal pathway, and carry various components such as lipids, proteins, RNA, and other small molecular substances. Exosomes can mediate intercellular communication through the bioactive substances they carry, thus participating in different physiological activities. Metabolic syndrome (MS) is a disease caused by disturbances in the body’s metabolism, mainly including insulin resistance (IR), diabetes, obesity, non-alcoholic fatty liver disease (NAFLD), hyperlipidemia, and atherosclerosis (AS). Exosomes are closely related to the occurrence and development of MS. Exosomes can act as messengers to mediate signaling transductions between metabolic cells in the organism and play a bidirectional regulatory role in the MS process.
- exosome
- biogeneration
- function
- metabolic syndrome
1. Introduction
2. Biogenesis of Exosomes
2.1. ILVs Production Involves Two Mechanisms according to Their Reliance on Endosomal Sorting Complex Required for Transport (ESCRT), Hence Referred to as ESCRT-Dependent and -Independent Pathways
For the ESCRT-dependent pathway, the initial stage involves the capture of ubiquitinated proteins by the ESCRT-0 complex on the cell membrane, thereby concentrating cargoes (such as DNA, RNA, and proteins) on the membrane [10][9]. While the cargoes are aggregated, ESCRT-I and ESCRT-II complexes are recruited to the cell membrane, leading to membrane protrusion and the initial formation of intraluminal vesicles (ILVs) [11][10]. Subsequently, the ubiquitinated cargoes interact with the ESCRT-I component TSG101 and the HRS protein in ESCRT-0 to be further concentrated and immobilized on the membrane [12][11]. Finally, the ILVs maturation is promoted by the ESCRT-III complex, VPS4 protein, and auxiliary protein VTA1, which are able to provide energy. Thereafter, ILVs accumulate inside the cells and develop into multivesicular bodies (MVBs) (Figure 1) [13][12].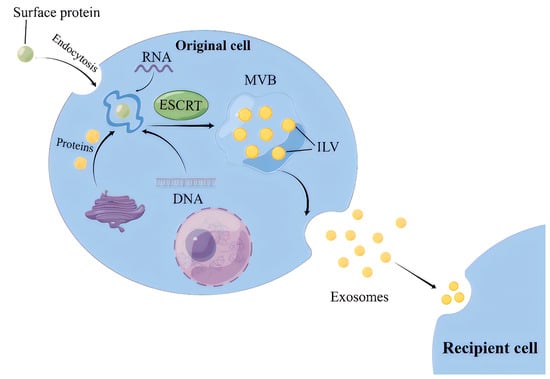
2.2. MVBs Transportation and Exosomes Release
3. Biological Characteristics of Exosomes
The outer membrane of exosomes is rich in cholesterol, sphingolipids, ceramides, glycolipids, and glycerophospholipid chains, which together play crucial roles in the cellular microenvironment. The main roles of the lipid components in the exosome membrane are to provide some stiffness, ensure bioavailability, and prevent the bioactive materials carried from degradation. Moreover, evidence have shown that exosomes lipids are also involved in the transport of lipids in vivo [26,27][15][16]. Different cells produce different exosomes that carry similar conserved proteins on their surface alone, such as the major histocompatibility complex MHC I and MHC II molecules, heat-shock proteins, four transmembrane proteins (CD9, CD63, CD81), integrins, cytoskeletal proteins and some biological enzymes [4,28,29,30][4][17][18][19]. The inclusions of exosomes mainly include proteins, including cytokines, miRNA, mRNA, DNA, and lipids.4. Exosome Separation and Technical Challenges
5. Biological Functions of Exosomes
Exosomes are natural transport carriers, and their inner lumen can be loaded with various biomolecules, such as proteins and nucleic acids. Exosomes also embed and anchor various protein ligands on their surface, which upon recognition and binding to the recipient cells can lead to reactive changes, thus affecting intracellular signaling pathways and the physiological state of the recipient cells. Therefore, exosomes can serve as carriers for biological molecules and signals for communication between cells. Different cells achieve intercellular communication by secreting exosomes carrying different components, and these exosomes are taken up by the recipient cells to exchange molecules or signals through substance exchange and release of endosomes, thus leading to the subsequent influence of recipient cells behavior and phenotype features. The intercellular communications mainly undergo via the following mechanisms. First, exosomes surface proteins can modulate the signaling pathways of target cells via directly binding to its cell receptors; Jodo et al. showed that the membrane of exosomes secreted by T cells contains a signaling molecule, Fas L, which specifically binds to transmembrane protein, Fas, on the receptor cell membrane and induces its trimerization, thus forming an apoptosis-inducing complex to initiate cell death [47][28]. Second, exosomes can fuse with cell membranes and deliver functional proteins, miRNAs, mRNAs, and other biomolecules to target cells.6. Application of Exosomes
Due to their excellent properties, exosomes play an extremely important role in disease diagnosis and treatment. First, given their presence in fluids like blood, urine, and saliva, exosomes can be isolated using liquid biopsy, providing a non-invasive strategy alternative to traditional biopsies [54][29], Second, the compositions of exosomes can be analyzed by mass spectrometry and other analytical methods to obtain a large amount of information about molecular carriers inside and outside the cells, on the basis of which a comprehensive diagnosis can be achieved at different states of disease progress. It was reported that 120 plasma exosome samples collected from the patients were used to screen the specific biomarkers of 16 different types of tumors by proteomic analysis. The results show that they achieved 95% sensitivity and 90% specificity for the classification of various tumors [55][30]. EPI-CE kit (Exosome dx company) and ExoDx Prostate-IntelliScore Diagnostics Product (Bio-Techne company) have been developed and marketed for the diagnosis of prostate cancer by screening RNA biomarkers originating from prostate-specific or urine exosomes [56,57][31][32]. Third, exosomes are natural carriers with the advantages of long half-life and natural non-toxicity. They also have the unique ability to target homing and deliver the substances to the target cells. The biological nature of exosomes allows them to easily cross the blood–brain barrier, making them an ideal cargo for drug delivery into the brain. For instance, exosomes can bind to an anti-CD22 monoclonal antibody fragment (CD22-F (ab’) 2) and wrap doxorubicin (DOX) to form CD22-F (ab’) 2-Exo-DOX, which can penetrate the BBB and accurately deliver DOX to lymphoma cells of primary central nervous system, thus enhancing anti-tumor activity in tumor-bearing mice [58][33].7. Exosomes Involved in MS Progress
7.1. IR
IR is a central factor contributing to MS and a driving force for the development of cardiovascular complications associated with type 2 diabetes mellitus (T2DM) and hypertension [61][34]. It has been found that exosomes can enter the receptor cells by wrapping different inclusions to regulate the transduction of the insulin signaling pathway, which contributes to IR development (Figure 32). Exosomes carrying miR-155 secreted by adipose tissue macrophages into hepatocytes can downregulate glucose transporter-4 (Glut-4) by targeting peroxisome proliferator-activated receptor γ (PPARγ), which ultimately leads to a decrease in insulin sensitivity in the liver and exacerbates the process of diabetes in obese mice [62][35].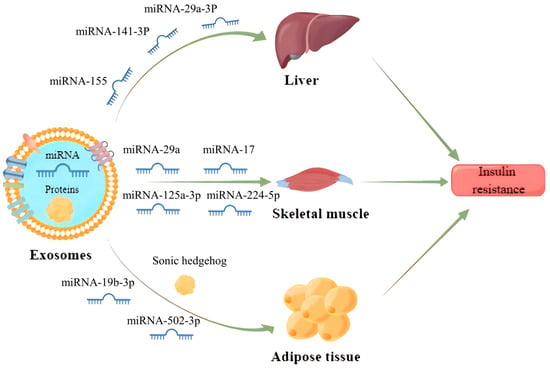
7.2. DM and Its Related Complications
7.3. Obesity
Obesity is one of the most common metabolic diseases, and the prevalence of obesity has greatly increased in past decades in the world [83][42]. Evidence have demonstrated that circulating exosomes miRNAs primarily secreted by adipose tissue exhibit different profiles between obese patients and healthy individuals [84][43], and exosomes play a vital role in the exchange of information between adipose tissue and other tissues, thereby contributing to the pathogenesis of obesity and its related diseases [85][44]. Adipocyte-derived exosomes miR-27a can regulate the hepatic lipid synthesis pathway by inhibiting PPARγ [86][45]. Exosomes also play an important role in lipid transport by regulating the expression of classical lipid transporters (e.g., ABCA1, ABCG1, LDLR, CD36), where plasma exosomes miR-30e and miR-92a can disrupt lipid metabolism and cause inadequate cholesterol efflux by inhibiting ABCA1 and ABCG1 [87][46].7.4. NAFLD
NAFLD, like obesity and diabetes, is a highly prevalent global metabolic disease that affects about a quarter of the global adult population and poses a serious health burden [89][47]. There is growing evidence that exosomes are involved in the development and occurrence of NAFLD [90][48]. The severity of liver inflammation in NAFLD patients positively correlates with the level of toxic hepatocyte-derived exosomes miR-192-5p [91][49], indicating that exosomes miR-192-5p can be used as a molecular marker for NAFLD. The release of miR-223-rich exosomes from macrophages can inhibit the expression of transcriptional activator with PDZ-binding motif (TAZ) in hepatocytes, thereby alleviating the progression of NAFLD to NASH and even liver fibrosis [92][50].7.5. Hyperlipidemia and AS
Hyperlipidemia is caused by higher percentages of fatty droplets and oxidized low-density lipoprotein (Ox-LDL) in blood composition. Such substances can increase the viscosity of the blood, which leads to a decrease in the oxygen-carrying capacity of the blood and promotes damage to the mucous membranes on the walls of blood vessels, resulting in the formation of atheromatous plaques and AS. It has been shown that macrophage-derived exosomes miR-223 promotes macrophage differentiation, inflammatory response, and disturbs lipid metabolism in adipose tissue, thus exacerbating the progression of AS [95,96][51][52].7.6. Hypertension
Hypertension is a complex multifactorial disease, primarily attributable to the interaction between genetic and environmental factors [105][53]. There is emerging evidence showing that exosomes play a key role in the progression of hypertension. A study has revealed that miRNAs and proteins contained in exosomes derived from endothelial cells and immune cells drastically elicit VSMC proliferation and migration, leading to vascular remodeling, a core mechanism in the etiology of hypertension [106][54]. Specific miRNAs, such as miR-155, miR-21, miR-34a, miR-145, and miR-22, present in circulating exosomes have been demonstrated to play a crucial role in the pathogenesis of hypertension. They can modulate vascular dilation and constriction by carrying or altering the expression of endothelial nitric oxide synthase (eNOS), thereby participating in blood pressure regulation [107][55].References
- He, C.; Zheng, S.; Luo, Y.; Wang, B. Exosome theranostics: Biology and translational medicine. Theranostics 2018, 8, 237.
- Xu, J.-Y.; Chen, G.-H.; Yang, Y.-J. Exosomes: A rising star in failing hearts. Front. Physiol. 2017, 8, 494.
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750.
- Pegtel, D.M.; Gould, S.J. Exosomes. Annu. Rev. Biochem. 2019, 88, 487–514.
- Golchin, A.; Hosseinzadeh, S.; Ardeshirylajimi, A. The exosomes released from different cell types and their effects in wound healing. J. Cell. Biochem. 2018, 119, 5043–5052.
- Greening, D.W.; Gopal, S.K.; Xu, R.; Simpson, R.J.; Chen, W. Exosomes and their roles in immune regulation and cancer. In Seminars in Cell & Developmental Biology; Academic Press: Cambridge, MA, USA, 2015; pp. 72–81.
- Gutierrez-Millan, C.; Calvo Díaz, C.; Lanao, J.M.; Colino, C.I. Advances in exosomes-based drug delivery systems. Macromol. Biosci. 2021, 21, 2000269.
- Zhang, Y.; Bi, J.; Huang, J.; Tang, Y.; Du, S.; Li, P. Exosome: A review of its classification, isolation techniques, storage, diagnostic and targeted therapy applications. Int. J. Nanomed. 2020, 15, 6917–6934.
- Skowyra, M.L.; Schlesinger, P.H.; Naismith, T.V.; Hanson, P.I. Triggered recruitment of ESCRT machinery promotes endolysosomal repair. Science 2018, 360, eaar5078.
- Nour, A.M.; Modis, Y. Endosomal vesicles as vehicles for viral genomes. Trends Cell Biol. 2014, 24, 449–454.
- Mosesso, N.; Nagel, M.-K.; Isono, E. Ubiquitin recognition in endocytic trafficking–with or without ESCRT-0. J. Cell Sci. 2019, 132, jcs232868.
- Xu, Y.; Feng, K.; Zhao, H.; Di, L.; Wang, L.; Wang, R. Tumor-derived extracellular vesicles as messengers of natural products in cancer treatment. Theranostics 2022, 12, 1683.
- Fader, C.M.; Sánchez, D.G.; Mestre, M.B.; Colombo, M.I. TI-VAMP/VAMP7 and VAMP3/cellubrevin: Two v-SNARE proteins involved in specific steps of the autophagy/multivesicular body pathways. Biochim. Biophys. Acta 2009, 1793, 1901–1916.
- Matsui, T.; Sakamaki, Y.; Hiragi, S.; Fukuda, M. VAMP5 and distinct sets of cognate Q-SNAREs mediate exosome release. Cell Struct. Funct. 2023, 48, 187–198.
- Mesmin, B.; Maxfield, F.R. Intracellular sterol dynamics. Biochim. Biophys. Acta BBA Mol. Cell Biol. Lipids 2009, 1791, 636–645.
- Vance, J.E.; Voelker, D.R. Cellular lipid transport processes and their role in human disease. Biochim. Biophys. Acta 2009, 1791, 561–562.
- Ludwig, A.-K.; Giebel, B. Exosomes: Small vesicles participating in intercellular communication. Int. J. Biochem. Cell Biol. 2012, 44, 11–15.
- Masyuk, A.I.; Masyuk, T.V.; LaRusso, N.F. Exosomes in the pathogenesis, diagnostics and therapeutics of liver diseases. J. Hepatol. 2013, 59, 621–625.
- Peinado, H.; Alečković, M.; Lavotshkin, S.; Matei, I.; Costa-Silva, B.; Moreno-Bueno, G.; Hergueta-Redondo, M.; Williams, C.; García-Santos, G.; Ghajar, C.M. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 2012, 18, 883–891.
- O’Brien, K.; Breyne, K.; Ughetto, S.; Laurent, L.C.; Breakefield, X.O. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat. Rev. Mol. Cell Biol. 2020, 21, 585–606.
- Abels, E.R.; Breakefield, X.O. Introduction to extracellular vesicles: Biogenesis, RNA cargo selection, content, release, and uptake. Cell. Mol. Neurobiol. 2016, 36, 301–312.
- Hessvik, N.P.; Phuyal, S.; Brech, A.; Sandvig, K.; Llorente, A. Profiling of microRNAs in exosomes released from PC-3 prostate cancer cells. Biochim. Biophys. Acta BBA Gene Regul. Mech. 2012, 1819, 1154–1163.
- Jeppesen, D.K.; Hvam, M.L.; Primdahl-Bengtson, B.; Boysen, A.T.; Whitehead, B.; Dyrskjøt, L.; Orntoft, T.F.; Howard, K.A.; Ostenfeld, M.S. Comparative analysis of discrete exosome fractions obtained by differential centrifugation. J. Extracell. Vesicles 2014, 3, 25011.
- Li, P.; Kaslan, M.; Lee, S.H.; Yao, J.; Gao, Z. Progress in Exosome Isolation Techniques. Theranostics 2017, 7, 789–804.
- Gardiner, C.; Di Vizio, D.; Sahoo, S.; Théry, C.; Witwer, K.W.; Wauben, M.; Hill, A.F. Techniques used for the isolation and characterization of extracellular vesicles: Results of a worldwide survey. J. Extracell. Vesicles 2016, 5, 32945.
- Niamtu, J., 3rd. Lip reduction surgery (reduction cheiloplasty). Facial Plast. Surg. Clin. N. Am. 2010, 18, 79–97.
- Gholizadeh, S.; Shehata Draz, M.; Zarghooni, M.; Sanati-Nezhad, A.; Ghavami, S.; Shafiee, H.; Akbari, M. Microfluidic approaches for isolation, detection, and characterization of extracellular vesicles: Current status and future directions. Biosens. Bioelectron. 2017, 91, 588–605.
- Jodo, S.; Xiao, S.; Hohlbaum, A.; Strehlow, D.; Marshak-Rothstein, A.; Ju, S.T. Apoptosis-inducing membrane vesicles. A novel agent with unique properties. J. Biol. Chem. 2001, 276, 39938–39944.
- Pantel, K.; Alix-Panabières, C. Liquid biopsy and minimal residual disease—Latest advances and implications for cure. Nat. Rev. Clin. Oncol. 2019, 16, 409–424.
- Hoshino, A.; Kim, H.S.; Bojmar, L.; Gyan, K.E.; Cioffi, M.; Hernandez, J.; Zambirinis, C.P.; Rodrigues, G.; Molina, H.; Heissel, S. Extracellular vesicle and particle biomarkers define multiple human cancers. Cell 2020, 182, 1044–1061.e18.
- Heiss, J.A.; Brennan, K.J.; Baccarelli, A.A.; Téllez-Rojo, M.M.; Estrada-Gutiérrez, G.; Wright, R.O.; Just, A.C. Battle of epigenetic proportions: Comparing Illumina’s EPIC methylation microarrays and TruSeq targeted bisulfite sequencing. Epigenetics 2020, 15, 174–182.
- Tutrone, R.; Donovan, M.J.; Torkler, P.; Tadigotla, V.; McLain, T.; Noerholm, M.; Skog, J.; McKiernan, J. Clinical utility of the exosome based ExoDx Prostate(IntelliScore) EPI test in men presenting for initial Biopsy with a PSA 2–10 ng/mL. Prostate Cancer Prostatic Dis. 2020, 23, 607–614.
- Liu, X.; Xia, T.; Fang, Y.; Zuo, H.; Dong, X.; Xu, P.; Ouyang, J. Overcoming the blood–brain barrier by using a multistage exosome delivery system to inhibit central nervous system lymphoma. Nanomed. Nanotechnol. Biol. Med. 2022, 41, 102523.
- O’Neill, S.; O’Driscoll, L. Metabolic syndrome: A closer look at the growing epidemic and its associated pathologies. Obes. Rev. 2015, 16, 1–12.
- Ying, W.; Riopel, M.; Bandyopadhyay, G.; Dong, Y.; Birmingham, A.; Seo, J.B.; Ofrecio, J.M.; Wollam, J.; Hernandez-Carretero, A.; Fu, W. Adipose tissue macrophage-derived exosomal miRNAs can modulate in vivo and in vitro insulin sensitivity. Cell 2017, 171, 372–384.e12.
- Nair, S.; Jayabalan, N.; Guanzon, D.; Palma, C.; Scholz-Romero, K.; Elfeky, O.; Zuñiga, F.; Ormazabal, V.; Diaz, E.; Rice, G.E.; et al. Human placental exosomes in gestational diabetes mellitus carry a specific set of miRNAs associated with skeletal muscle insulin sensitivity. Clin. Sci. 2018, 132, 2451–2467.
- Al-Κafaji, G.; Al-Muhtaresh, H.A.; Salem, A.H. Expression and clinical significance of miR-1 and miR-133 in pre-diabetes. Biomed. Rep. 2021, 14, 33.
- Tsukita, S.; Yamada, T.; Takahashi, K.; Munakata, Y.; Hosaka, S.; Takahashi, H.; Gao, J.; Shirai, Y.; Kodama, S.; Asai, Y. MicroRNAs 106b and 222 improve hyperglycemia in a mouse model of insulin-deficient diabetes via pancreatic β-cell proliferation. eBioMedicine 2017, 15, 163–172.
- Sun, Y.; Zhou, Y.; Shi, Y.; Zhang, Y.; Liu, K.; Liang, R.; Sun, P.; Chang, X.; Tang, W.; Zhang, Y. Expression of miRNA-29 in pancreatic β cells promotes inflammation and diabetes via TRAF3. Cell Rep. 2021, 34, 108576.
- Liu, Y.-P.; Tian, M.-Y.; Yang, Y.-D.; Li, H.; Zhao, T.-T.; Zhu, J.; Mou, F.-F.; Cui, G.-H.; Guo, H.-D.; Shao, S.-J. Schwann cells-derived exosomal miR-21 participates in high glucose regulation of neurite outgrowth. iScience 2022, 25, 105141.
- Jia, Y.; Chen, J.; Zheng, Z.; Tao, Y.; Zhang, S.; Zou, M.; Yang, Y.; Xue, M.; Hu, F.; Li, Y. Tubular epithelial cell-derived extracellular vesicles induce macrophage glycolysis by stabilizing HIF-1α in diabetic kidney disease. Mol. Med. 2022, 28, 95.
- Rulkiewicz, A.; Pilchowska, I.; Lisik, W.; Pruszczyk, P.; Ciurzynski, M.; Domienik-Karlowicz, J. Prevalence of Obesity and Severe Obesity among Professionally Active Adult Population in Poland and Its Strong Relationship with Cardiovascular Co-Morbidities-POL-O-CARIA 2016–2020 Study. J. Clin. Med. 2022, 11, 3720.
- Ji, C.; Guo, X. The clinical potential of circulating microRNAs in obesity. Nat. Rev. Endocrinol. 2019, 15, 731–743.
- Kumar, A.; Sundaram, K.; Mu, J.; Dryden, G.W.; Sriwastva, M.K.; Lei, C.; Zhang, L.; Qiu, X.; Xu, F.; Yan, J. High-fat diet-induced upregulation of exosomal phosphatidylcholine contributes to insulin resistance. Nat. Commun. 2021, 12, 213.
- Yu, Y.; Du, H.; Wei, S.; Feng, L.; Li, J.; Yao, F.; Zhang, M.; Hatch, G.M.; Chen, L. Adipocyte-derived exosomal MiR-27a induces insulin resistance in skeletal muscle through repression of PPARγ. Theranostics 2018, 8, 2171.
- Wang, Z.; Zhang, J.; Zhang, S.; Yan, S.; Wang, Z.; Wang, C.; Zhang, X. MiR-30e and miR-92a are related to atherosclerosis by targeting ABCA1. Mol. Med. Rep. 2019, 19, 3298–3304.
- Lazarus, J.V.; Mark, H.E.; Anstee, Q.M.; Arab, J.P.; Batterham, R.L.; Castera, L.; Cortez-Pinto, H.; Crespo, J.; Cusi, K.; Dirac, M.A. Advancing the global public health agenda for NAFLD: A consensus statement. Nat. Rev. Gastroenterol. Hepatol. 2022, 19, 60–78.
- Wu, D.; Zhu, H.; Wang, H. Extracellular vesicles in non-alcoholic fatty liver disease and alcoholic liver disease. Front. Physiol. 2021, 12, 707429.
- Liu, X.L.; Pan, Q.; Cao, H.X.; Xin, F.Z.; Zhao, Z.H.; Yang, R.X.; Zeng, J.; Zhou, H.; Fan, J.G. Lipotoxic hepatocyte-derived exosomal microRNA 192-5p activates macrophages through rictor/Akt/forkhead box transcription factor O1 signaling in nonalcoholic fatty liver disease. Hepatology 2020, 72, 454–469.
- Hou, X.; Yin, S.; Ren, R.; Liu, S.; Yong, L.; Liu, Y.; Li, Y.; Zheng, M.H.; Kunos, G.; Gao, B. Myeloid-Cell–Specific IL-6 Signaling Promotes MicroRNA-223-Enriched Exosome Production to Attenuate NAFLD-Associated Fibrosis. Hepatology 2021, 74, 116–132.
- Ismail, N.; Wang, Y.; Dakhlallah, D.; Moldovan, L.; Agarwal, K.; Batte, K.; Shah, P.; Wisler, J.; Eubank, T.D.; Tridandapani, S.; et al. Macrophage microvesicles induce macrophage differentiation and miR-223 transfer. Blood 2013, 121, 984–995.
- Zhuang, G.; Meng, C.; Guo, X.; Cheruku, P.S.; Shi, L.; Xu, H.; Li, H.; Wang, G.; Evans, A.R.; Safe, S.; et al. A novel regulator of macrophage activation: miR-223 in obesity-associated adipose tissue inflammation. Circulation 2012, 125, 2892–2903.
- Taylor, D.A. Hypertensive Crisis: A Review of Pathophysiology and Treatment. Crit. Care Nurs. Clin. 2015, 27, 439–447.
- Jansen, F.; Yang, X.; Proebsting, S.; Hoelscher, M.; Przybilla, D.; Baumann, K.; Schmitz, T.; Dolf, A.; Endl, E.; Franklin, B.S.; et al. MicroRNA expression in circulating microvesicles predicts cardiovascular events in patients with coronary artery disease. J. Am. Heart Assoc. 2014, 3, e001249.
- Ong, S.G.; Lee, W.H.; Huang, M.; Dey, D.; Kodo, K.; Sanchez-Freire, V.; Gold, J.D.; Wu, J.C. Cross talk of combined gene and cell therapy in ischemic heart disease: Role of exosomal microRNA transfer. Circulation 2014, 130, S60–S69.
