Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Catherine Yang and Version 1 by Stavros Spiliopoulos.
Percutaneous deep venous arterialization (DVA) is considered the last-chance treatment for ‘no-option’ patients with chronic limb-threatening ischemia (CLTI). Palliative amputation, which is linked to a high risk of peri-procedural and 1-year mortality, is thereby potentially avoided. Due to the lack of reproducibility and standardization of the procedure, DVA has been attempted in multiple settings using tools and techniques currently available (i.e., ‘off-label’). In studies where alternate techniques of venous arterialization have been used, technical and clinical success varied from 77% to 100% and from 29% to 75%, respectively.
- chronic limb-threatening ischemia
- percutaneous deep venous arterialization
- endovascular treatment
1. Benefits of pDVA
The lack of options for end-stage CLTI patients highlights the importance of pDVA as an option for limb salvage when amputation seems to be the only option remaining. The major advantage of this approach is that it offers a minimally invasive option for patients with adequate endoluminal access. This, in turn, leads to lower periprocedural risks and the absence of wound creation, which is additionally important in an already ischemic leg [1].
2. Adverse Effects and Disadvantages of pDVA
As with any percutaneous catheterization, possible adverse effects of pDVA include vascular complications at the access site (i.e., bleeding events, hematoma), arterial and venous thromboembolic events (myocardial infarction, stroke, pulmonary embolism, etc.), contrast-induced nephropathy, or renal failure [1]. Apart from this, stent placement between the arterial and venous systems may lead to complications, such as thrombosis, circuit stenosis, and vessel spasm.
The advantages and adverse effects of pDVA are summarized in Table 1.
Table 1.
MI: myocardial infraction; PE: pulmonary embolism; DVT: deep vein thrombosis.
| Advantages | Adverse Effects |
|---|---|
| Minimally invasive approach | Allergic reactions |
| Less chance of infection | Vascular complications (bleeding, hematoma) |
| Absence of wounds | Arterial and venous thromboembolic events (angina, stroke, limb ischemia, MI, PE, DVT) |
| Contrast-induced nephropathy and renal failure | |
| Local or systemic infection | |
| Stent thrombosis, in-stent stenosis | |
| Unsuccessful disruption of valves |
3. pDVA Techniques
Different methods are used to perform pDVA, but in each method, there are three important steps: firstly, the formation of an arterio-venous (AV) fistula; secondly, disruption of venous valves so that flow reversal is possible; and thirdly, the prevention of the shunting of arterial blood through the AV fistula and other interconnecting veins. For the creation of an AV fistula, re-entry devices can be used as well as catheters, techniques involving snares (the so-called ‘venous arterialization simplified technique’; VAST), or double punctures (‘AV spear’) [2][3][4]. However, up to 20% of these procedures can fail because of the inability to create an AV communication. For instance, when the vessel wall is heavily calcified, the wire tip or needle cannot penetrate when a penetration wire or reentry device is used [5][6]. The LimFlow system (LimFlow SA, Paris, France) is currently the only registered device with which a pDVA procedure can be performed in its entirety, overcoming the challenge of AV fistula creation. However, it may increase the overall procedure cost, and regulatory approval is still pending in several markets [7].
4. Selection Criteria for pDVA Intervention
DVA is advocated in Rutherford class 5 or 6 patients who lack a distal target vessel for bypass or endovascular intervention and who have at least one patent tibial artery in the proximal segment [8]. As reported by Kum et al., general factors, angiographic factors, and wound-related factors should be the three focus areas in order to select patients suitable for undergoing pDVA [7]. In particular, concerning general factors, patients with poor cardiac function, pre-existing coronary lesions, and high output fistulas for dialysis access should be excluded. The same is true for patients with a history of deep venous thrombosis (DVT). With regard to angiographic factors, it is important to ensure the suitability of the tibial vessel used for crossing as well as the patency of the ipsilateral iliac and femoropopliteal arteries, in order to optimize inflow. Finally, in terms of the wound, patients with acute limb ischemia, systemic sepsis, and wounds around the distal retrograde access site should be considered unsuitable.
5. DVA Techniques
Several described methodologies for DVA have been described in the literature, though there has not been much standardization of procedural steps outside of the LimFlow trials. Techniques include a combination of open surgical, hybrid, and fully percutaneous approaches. Results across all techniques have been promising, demonstrating the feasibility of the arterialization of deep veins for restoring blood flow to the lower limb and potentially preventing major amputation. Open surgical DVA is achieved by pairing a surgical AV bypass with the physical effacement of the valves of the pedal loop vein. This technique has been shown to have comparable patency and limb salvage rates to surgical pedal bypass [9]. Hybrid superficial venous arterialization involves making an in situ open surgical anastomosis from the greater saphenous vein to the popliteal artery, followed by valve lysis and the distal endovascular focalization of flow [10][11]. Additionally, a single-stage approach has been described which involves open bypass with a stent to the venous outflow followed by percutaneous valve lysis [12]. A study conducted by Montelione et al. assessed patients undergoing the surgical arterialization of the deep venous circulation, followed by the endovascular selective embolization of venous escape routes from the foot. They found that this approach was a viable and efficient solution for preserving limbs in patients suffering from CLTI when no other options were available [13].
Finally, the fully percutaneous approach utilizes the tibial arteries as the donor vessel to create an AV crossing into the tibial vein, which is lined with a covered stent to focalize blood flow into the pedal veins, the valves of which are effaced percutaneously. pDVA can be performed using off-the-shelf devices or the proprietary LimFlow System, which is commercially available in Europe and the United States.
6. Benefits of LimFlow System—A Purpose-Built Device
The LimFlow System consists of a series of purpose-built devices, including crossing catheters for the creation of an AV fistula, a pushing valvulotome, and tapered and straight stent grafts. The arterial crossing catheter has a needle with a 10 mm reach that is deployed through the arterial wall into a 6 mm self-expanding nitinol basket deployed in the paired vein. This radiopaque target allows for easy AV alignment and wire capture for the AV crossing. The pushing valvulotome lyses the valves within the veins below the crossover point and distally into the pedal venous loop, allowing forward blood flow. Utilizing the valvulotome reduces the trauma to the vessel wall, avoiding trauma-induced stenosis in the outflow veins that can occur if using a balloon to efface the valves [11]. The tapered crossing stent allows for optimal sizing for both the artery and vein, and the straight extension stents prevent smaller veins from stealing flow back up the leg, focalizing the blood flow into the foot.
7. The pDVA Procedure Using the LimFlow System
Venous access is achieved in the lateral plantar vein (Figure 1), and the venous catheter is advanced into the target vein to the level of the intended crossing point into the tibial artery. Using simultaneous digital subtraction angiography (DSA) and venography, the shortest distance between the vessels is identified. An arterial crossing catheter is inserted via an antegrade femoral arterial approach and advanced distally into the artery to the below-the-knee crossing point. When the two catheters are aligned, artery-to-vein crossing is achieved by advancing the needle from the arterial crossing catheter into the basket of the venous catheter to achieve through-and-through wire access. The AV connection is then dilated using a small angioplasty balloon, allowing the passage of other devices. A 4F over-the-wire push valvulotome is then deployed and serves to render the venous valves insufficient, which allows the reversal of flow in the vein, thus directing arterial oxygenated blood into the foot. Self-expanding stent-grafts are then placed from the level of the ankle to just below the crossing point, creating a conduit for continuous blood flow and covering venous collaterals. Finally, a tapered, self-expanding, covered stent graft is deployed to optimize the transition and flow from the artery into the vein, preventing cardiac overload as well as leakage at the crossover point. Figure 2 illustrates the basic procedural steps. Procedural time ranges from 90 min to 4 h. After the procedure, patients should be maintained on lifelong antiplatelet therapy (oral clopidogrel or aspirin) in combination with warfarin or direct oral anticoagulants for 3 months. A follow-up protocol for monitoring blood flow into the foot is necessary using duplex ultrasound [14]. The LimFlow system earned a CE mark in 2016 and FDA approval in 2023, and is available in Europe and the United States.
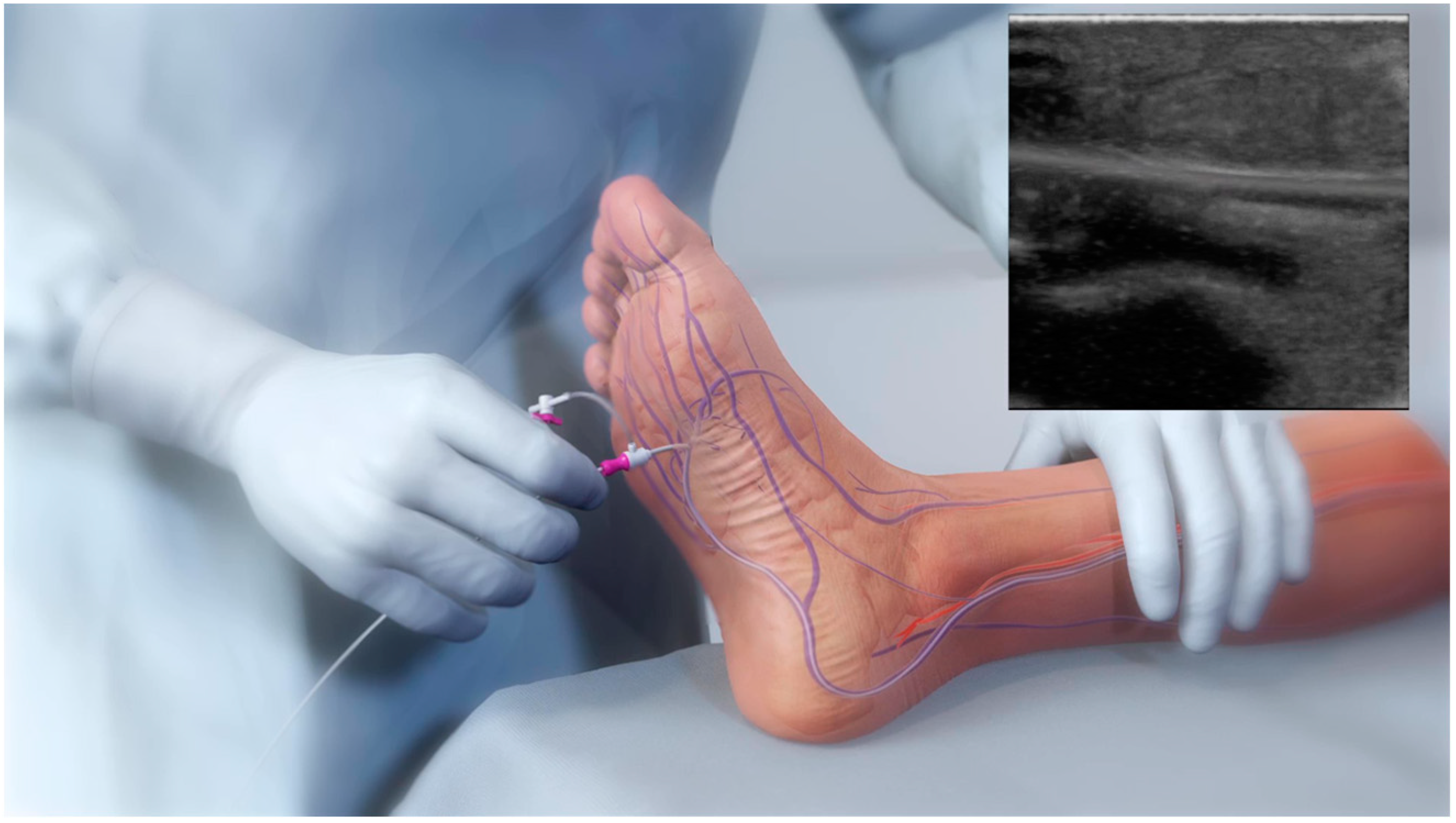
Figure 1.
Retrograde lateral plantar vein access enabling the insertion of the venous catheter during the pDVA technique.

Figure 2. Illustration of pDVA procedural steps. (a) Advancing the needle from the arterial crossing catheter into the basket of the venous catheter to create the fistula. (b) Use of a push valvulotome to allow reversal flow in the vein and (c) final stent graft deployment for arteriovenous bridging.
Notably, an important observation that should be taken into consideration when using the LimFlow device is that prior to the creation of the AV fistula, patients may require an arterial and/or venous intervention (i.e., an arterioplasty, venoplasty) in order to optimize the DVA inflow and outflow vessels [7].
References
- Schreve, M.A.; Lichtenberg, M.; Ünlü, Ç.; Branzan, D.; Schmidt, A.; van den Heuvel, D.A.; Blessing, E.; Brodmann, M.; Cabane, V.; Lin, W.T.Q. PROMISE international; a clinical post marketing trial investigating the percutaneous deep vein arterialization (LimFlow) in the treatment of no-option chronic limb ischemia patient. CVIR Endovasc. 2019, 2, 26.
- Ysa, A.; Lobato, M.; Mikelarena, E.; Arruabarrena, A.; Gómez, R.; Apodaka, A.; Metcalfe, M.; Fonseca, J.L. Homemade Device to Facilitate Percutaneous Venous Arterialization in Patients with No-Option Critical Limb Ischemia. J. Endovasc. Ther. 2019, 26, 213–218.
- Ichihashi, S.; Shimohara, Y.; Bolstad, F.; Iwakoshi, S.; Kichikawa, K. Simplified Endovascular Deep Venous Arterialization for Non-option CLI Patients by Percutaneous Direct Needle Puncture of Tibial Artery and Vein under Ultrasound Guidance (AV Spear Technique). Cardiovasc. Interv. Radiol. 2020, 43, 339–343.
- Migliara, B.; Cappellari, T.F. A Novel Technique to Create an Arteriovenous Fistula during Total Percutaneous Deep Foot Venous Arterialisation Using an IVUS Guided Catheter. Eur. J. Vasc. Endovasc. Surg. 2018, 55, 735.
- Gandini, R.; Merolla, S.; Scaggiante, J.; Meloni, M.; Giurato, L.; Uccioli, L.; Konda, D. Endovascular Distal Plantar Vein Arterialization in Dialysis Patients with No-Option Critical Limb Ischemia and Posterior Tibial Artery Occlusion: A Technique for Limb Salvage in a Challenging Patient Subset. J. Endovasc. Ther. 2018, 25, 127–132.
- Ichihashi, S.; Iwakoshi, S.; Nakai, T.; Yamamoto, Y.; Hirose, T.; Furuichi, K.; Tamura, Y.; Tanaka, T. Role of Percutaneous Deep Venous Arterialization for Patients with Chronic Limb-threatening Ischemia. Interv. Radiol. 2023, 8, 97–104.
- Kum, S.; Tan, Y.K.; Schreve, M.A.; Ferraresi, R.; Varcoe, R.L.; Schmidt, A.; Scheinert, D.; Mustapha, J.A.; Lim, D.A.; Ho, D. Midterm Outcomes from a Pilot Study of Percutaneous Deep Vein Arterialization for the Treatment of No-Option Critical Limb Ischemia. J. Endovasc. Ther. 2017, 24, 619–626.
- Sheil, G.R. Treatment of critical ischaemia of the lower limb by venous arterialization: An interim report. Br. J. Surg. 1977, 64, 197–199.
- Schreve, M.A.; Minnee, R.C.; Bosma, J.; Leijdekkers, V.J.; Idu, M.M.; Vahl, A.C. Comparative study of venous arterialization and pedal bypass in a patient cohort with critical limb ischemia. Ann. Vasc. Surg. 2014, 28, 1123–1127.
- Montero-Baker, M.; Sommerset Rvt, J.; Miranda, J.A. How I Do It: Hybrid Superficial Venous Arterialization and Endovascular Deep Venous Arterialization. J. Vasc. Surg. Cases Innov. Tech. 2023, 9, 101160.
- Ferraresi, R.; Casini, A.; Losurdo, F.; Caminiti, M.; Ucci, A.; Longhi, M.; Schreve, M.; Lichtenberg, M.; Kum, S.; Clerici, G. Hybrid Foot Vein Arterialization in No-Option Patients with Critical Limb Ischemia: A Preliminary Report. J. Endovasc. Ther. 2019, 26, 7–17.
- Alexandrescu, V.; Ngongang, C.; Vincent, G.; Ledent, G.; Hubermont, G. Deep calf veins arterialization for inferior limb preservation in diabetic patients with extended ischaemic wounds, unfit for direct arterial reconstruction: Preliminary results according to an angiosome model of perfusion. Cardiovasc. Revasc. Med. 2011, 12, 10–19.
- Montelione, N.; Catanese, V.; Gabellini, T.; Codispoti, F.A.; Nenna, A.; Spinelli, F.; Stilo, F. Duplex and Angiographic-Assisted Evaluation of Outcomes of Endovascular Embolization after Surgical Deep Vein Arterialization for the Treatment No-Option Critical Limb Ischemia Patients. Diagnostics 2022, 12, 2986.
- Takagi, T.; Miyamoto, A.; Ohura, N.; Yamauchi, Y. Percutaneous deep venous arterialization with balloon angioplasty salvaged a life-threatening critical limb in a hemodialysis patient after repeated pedal angioplasty failed: A case report. Clin. Case Rep. 2023, 11, e7589.
More
