As a novel type of green energy storage device, supercapacitors exhibit several orders of magnitude higher capacities than the traditional dielectric capacitors and significantly higher power density than the traditional secondary batteries. Supercapacitors have been widely applied in energy storage fields. Electrode materials, as pivotal components of supercapacitors, play an important role in electrochemical performance. Molybdenum-based materials have attracted widespread attention for their high theoretical capacitance, abundant resources, and facile synthesis tactics. Therefore, it is necessary to systematically summarize the application of Mo-based electrode materials in high-performance supercapacitors and unveil their developmental direction and trends. In this paper, we provide a review of binary Mo-based materials, ternary Mo-based materials, nanocomposites of Mo-based materials, and Mo-based MOFs and derivative materials. In addition, we further point out the key issues on the development of Mo-based materials in supercapacitors. This review may inspire more insightful works and enlighten other electrochemical areas concerning Mo-based materials.
- supercapacitors
- Mo-based electrode materials
- binary materials
1. Introduction
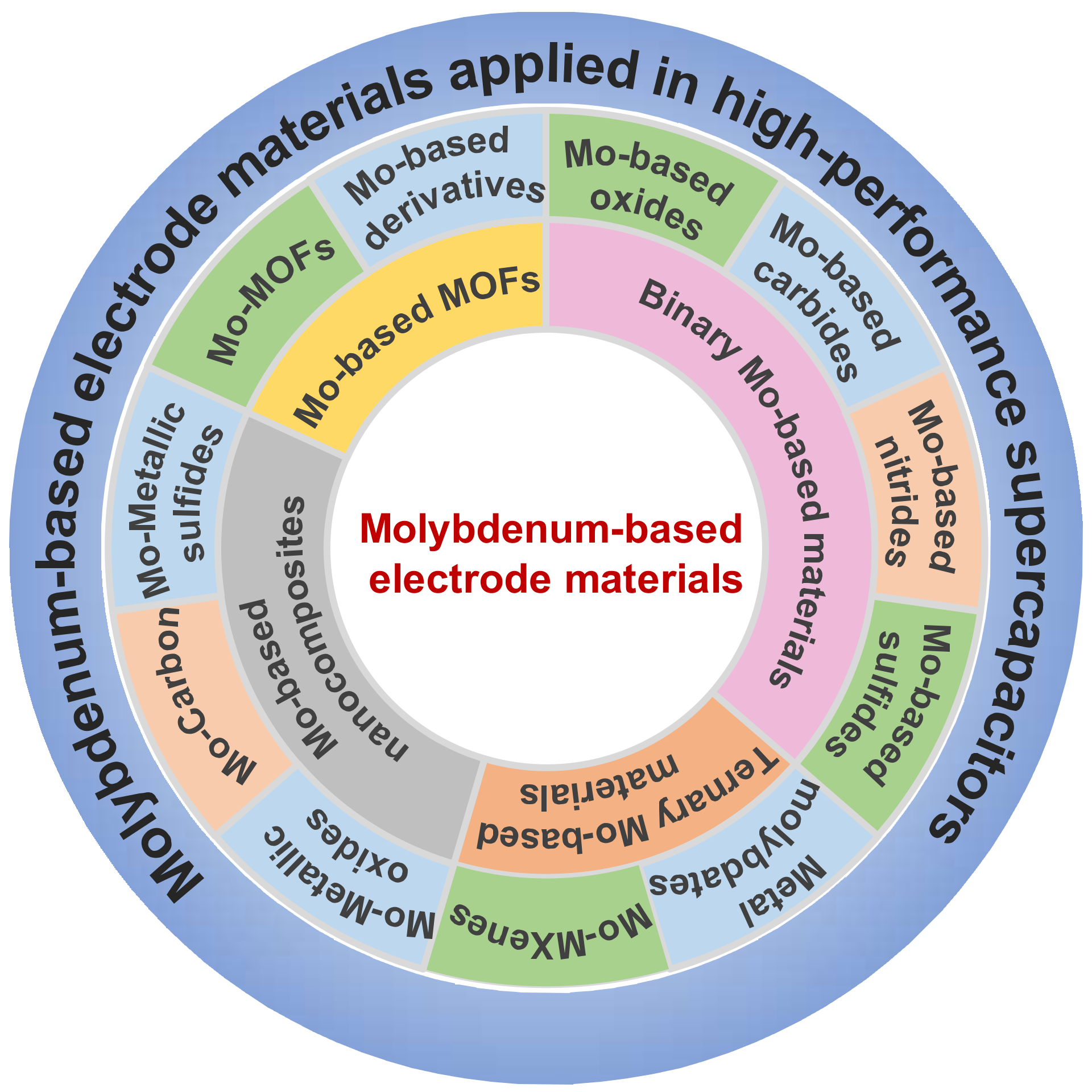
2. Mo-Based Electrode Materials for Supercapacitors
2.1. Binary Mo-Based Materials
2.1.1. Molybdenum Oxides

2.1.2. Molybdenum Carbides
2.1.3. Molybdenum Nitrides

2.1.4. Molybdenum Sulfides
2.2. Ternary Mo-Based Materials
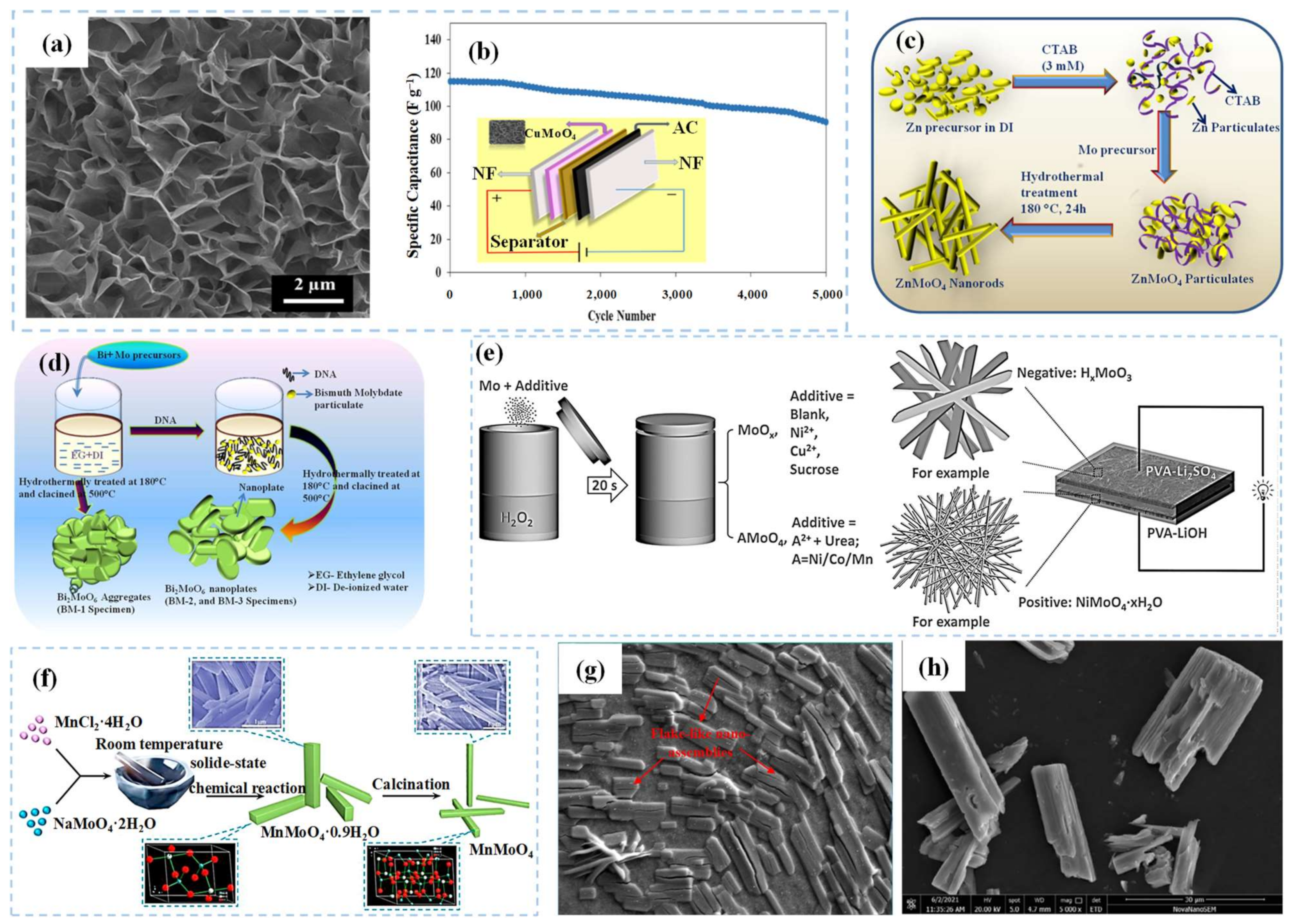
2.2.1. Metal Molybdates
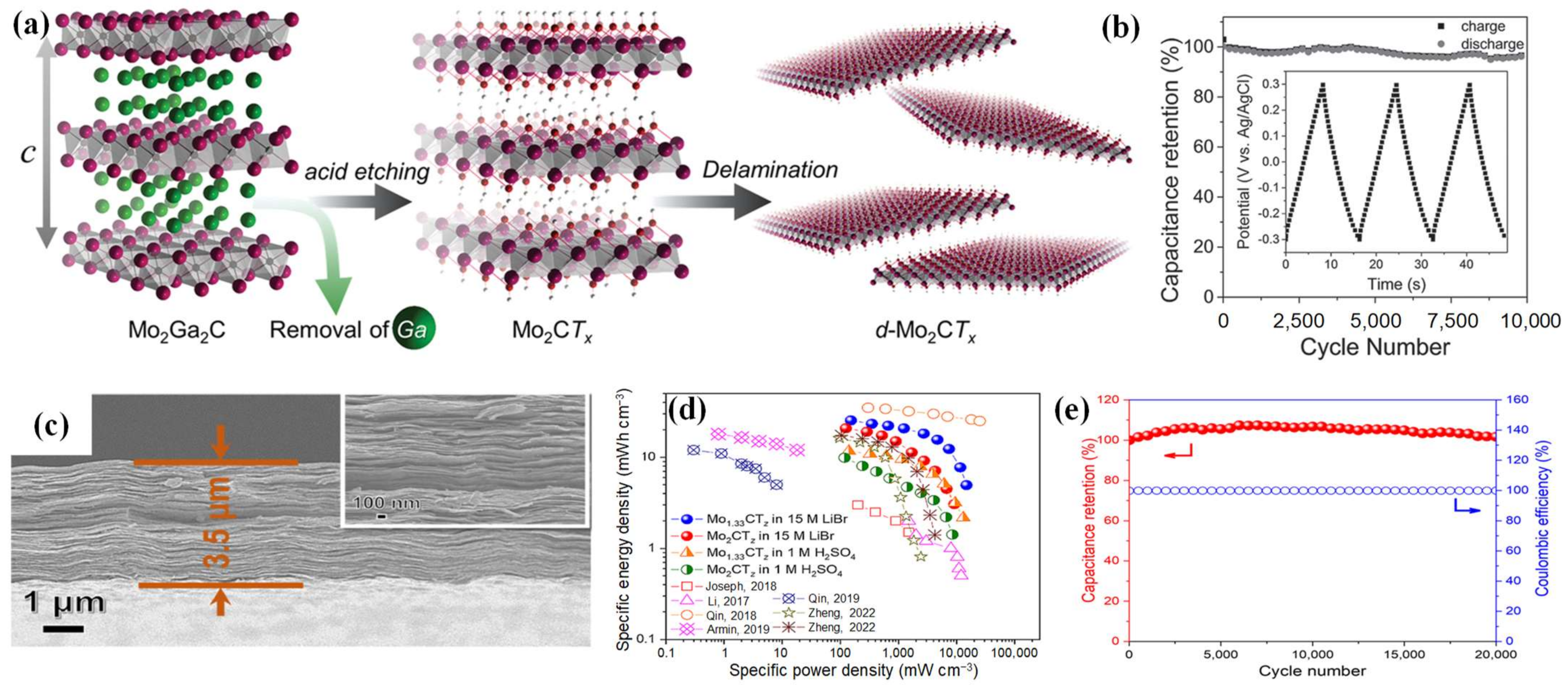
2.2.2. Mo-MXenes
References
- Song, D.; Liu, Y.; Qin, T.; Gu, H.; Cao, Y.; Shi, H. Overview of the policy instruments for renewable energy development in China. Energies 2022, 15, 6513.
- Pan, Y.; Dong, F. Dynamic evolution and driving factors of new energy development: Fresh evidence from China. Technol. Forecast. Soc. Chang. 2022, 176, 14.
- Guo, L.; Hu, P.; Wei, H. Development of supercapacitor hybrid electric vehicle. J. Energy Storage 2023, 65, 8.
- Lamba, P.; Singh, P.; Singh, P.; Singh, P.; Bharti; Kumar, A.; Gupta, M.; Kumar, Y. Recent advancements in supercapacitors based on different electrode materials: Classifications, synthesis methods and comparative performance. J. Energy Storage 2022, 48, 103871.
- Frackowiak, E.; Abbas, Q.; Béguin, F. Carbon/carbon supercapacitors. J. Energy Chem. 2013, 22, 226–240.
- Conway, B.E. Transition from “supercapacitor” to “battery” behavior in electrochemical energy storage. J. Electrochem. Soc. 1991, 138, 1539–1548.
- Ji, H.; Zhao, X.; Qiao, Z.; Jung, J.; Zhu, Y.; Lu, Y.; Zhang, L.L.; MacDonald, A.H.; Ruoff, R.S. Capacitance of carbon-based electrical double-layer capacitors. Nat. Commun. 2014, 5, 3317.
- Lu, X.F.; Wang, A.L.; Xu, H.; He, X.J.; Tong, Y.X.; Li, G.R. High-performance supercapacitors based on MnO2 tube-in-tube arrays. J. Mater. Chem. A 2015, 3, 16560–16566.
- Sharma, S.; Chand, P. Supercapacitor and electrochemical techniques: A brief review. Results Chem. 2023, 5, 100885.
- Kumar, S.; Saeed, G.; Zhu, L.; Hui, K.N.; Kim, N.H.; Lee, J.H. 0D to 3D carbon-based networks combined with pseudocapacitive electrode material for high energy density supercapacitor: A review. Chem. Eng. J. 2021, 403, 126352.
- Wang, D.; Tang, K.; Xiao, J.; Li, X.; Long, M.; Chen, J.; Gao, H.; Chen, W.; Liu, C.; Liu, H. Advances of electrospun Mo-based nanocomposite fibers as anode materials for supercapacitors. Sustain. Mater. Technol. 2021, 29, e00302.
- Xiong, C.; Zhang, Y.; Xu, J.; Dang, W.; Sun, X.; An, M.; Ni, Y.; Mao, J. Kinetics process for structure-engineered integrated gradient porous paper-based supercapacitors with boosted electrochemical performance. Nano Res. 2023, 16, 9471–9479.
- Xiong, C.; Zheng, C.; Jiang, X.; Xiao, X.; Wei, H.; Zhou, Q.; Ni, Y. Recent progress of green biomass based composite materials applied in supercapacitors, sensors, and electrocatalysis. J. Energy Storage 2023, 72, 108633.
- Xiong, C.; Wang, T.; Han, J.; Zhang, Z.; Ni, Y. Recent research progress of paper-based supercapacitors based on cellulose. Energy Environ. Mater. 2023, e12651.
- Mendoza-Sánchez, B.; Brousse, T.; Ramirez Castro, C.; Nicolosi, V.; Grant, P.S. An investigation of nanostructured thin film α-MoO3 based supercapacitor electrodes in an aqueous electrolyte. Electrochim. Acta 2013, 91, 253–260.
- Ma, H.; Liang, J.; Qiu, J.; Jiang, L.; Ma, L.; Sheng, H.; Shao, M.; Wang, Q.; Li, F.; Fu, Y.; et al. A biocompatible supercapacitor diode with enhanced rectification capability toward ion/electron-coupling logic operations. Adv. Mater. 2023, 35, e2301218.
- Vikraman, D.; Hussain, S.; Karuppasamy, K.; Santhoshkumar, P.; Kathalingam, A.; Jung, J.; Kim, H.S. Fabrication of asymmetric supercapacitors using molybdenum dichalcogenide nanoarray structures. Int. J. Energy Res. 2022, 46, 18410–18425.
- Yuksel, R.; Coskun, S.; Unalan, H.E. Coaxial silver nanowire network core molybdenum oxide shell supercapacitor electrodes. Electrochim. Acta 2016, 193, 39–44.
- Saji, V.S.; Lee, C.W. Molybdenum, molybdenum oxides, and their electrochemistry. ChemSusChem 2012, 5, 1146–1161.
- Mai, L.Q.; Hu, B.; Chen, W.; Qi, Y.Y.; Lao, C.S.; Yang, R.S.; Dai, Y.; Wang, Z.L. Lithiated MoO3 nanobelts with greatly improved performance for lithium batteries. Adv. Mater. 2007, 19, 3712–3716.
- De Castro, I.A.; Datta, R.S.; Ou, J.Z.; Castellanos Gomez, A.; Sriram, S.; Daeneke, T.; Kalantar-Zadeh, K. Molybdenum oxides—From fundamentals to functionality. Adv. Mater. 2017, 29, 31.
- Huang, C.; Zhang, W.; Zheng, W. The debut and spreading the landscape for excellent vacancies-promoted electrochemical energy storage of nano-architected molybdenum oxides. Mater. Today Energy 2022, 30, 101154.
- Pan, W.; Tian, R.; Jin, H.; Guo, Y.; Zhang, L.; Wu, X.; Zhang, L.; Han, Z.; Liu, G.; Li, J.; et al. Structure, optical, and catalytic properties of novel hexagonal metastable h-MoO3 nano- and microrods synthesized with modified liquid-phase processes. Chem. Mater. 2010, 22, 6202–6208.
- Prakash, N.G.; Dhananjaya, M.; Narayana, A.L.; Shaik, D.P.M.D.; Rosaiah, P.; Hussain, O.M. High performance one dimensional α-MoO3 nanorods for supercapacitor applications. Ceram. Int. 2018, 44, 9967–9975.
- Niu, Y.; Li, X.; Su, H.; Li, J.; Qi, Y. Formation of three dimensional porous h-MoO3 architecture and its application in supercapacitors. Mater. Lett. 2022, 316, 132062.
- Zhu, Y.; Tan, Y.; Li, H. MoO3 nanoplates preparation via self-sacrifice C3N4 for supercapacitors in an acid electrolyte. J. Energy Storage 2023, 60, 106657.
- Hao, J.; Zhang, J.; Xia, G.; Liu, Y.; Zheng, Y.; Zhang, W.; Tang, Y.; Pang, W.K.; Guo, Z. Heterostructure manipulation via in situ localized phase transformation for high-rate and highly durable lithium ion storage. ACS Nano 2018, 12, 10430–10438.
- Ma, B.; Hao, W.; Ruan, W.; Yuan, C.; Wang, Q.; Teng, F. Unveiling capacitive behaviors of MoO2 in different electrolytes and flexible MoO2-based asymmetric micro-supercapacitor. J. Energy Storage 2022, 52, 104833.
- Wu, K.; Zhao, J.; Zhang, X.; Zhou, H.; Wu, M. Hierarchical mesoporous MoO2 sphere as highly effective supercapacitor electrode. J. Taiwan Inst. Chem. Eng. 2019, 102, 212–217.
- Zhao, X.; Wang, H.E.; Cao, J.; Cai, W.; Sui, J. Amorphous/crystalline hybrid MoO2 nanosheets for high-energy lithium-ion capacitors. Chem. Commun. 2017, 53, 10723–10726.
- Luo, Z.; Miao, R.; Huan, T.D.; Mosa, I.M.; Poyraz, A.S.; Zhong, W.; Cloud, J.E.; Kriz, D.A.; Thanneeru, S.; He, J.; et al. Mesoporous MoO3–x material as an mfficient mlectrocatalyst for hydrogen evolution reactions. Adv. Energy Mater. 2016, 6, 11.
- Gurusamy, L.; Karuppasamy, L.; Anandan, S.; Liu, N.; Lee, G.J.; Liu, C.H.; Wu, J.J. Enhanced performance of charge storage supercapattery by dominant oxygen deficiency in crystal defects of 2-D MoO3-x nanoplates. Appl. Surf. Sci. 2021, 541, 148676.
- Kuwahara, Y.; Furuichi, N.; Seki, H.; Yamashita, H. One-pot synthesis of molybdenum oxide nanoparticles encapsulated in hollow silica spheres: An efficient and reusable catalyst for epoxidation of olefins. J. Mater. Chem. A 2017, 5, 18518–18526.
- Li, X.; Wang, D.; Zhang, Y.; Liu, L.; Wang, W. Surface-ligand protected reduction on plasmonic tuning of one-dimensional MoO3−x nanobelts for solar steam generation. Nano Res. 2020, 13, 3025–3032.
- Wu, Q.L.; Zhao, S.X.; Yu, L.; Zheng, X.X.; Wang, Y.F.; Yu, L.Q.; Nan, C.W.; Cao, G.Z. Oxygen vacancy-enriched MoO3-x nanobelts for asymmetric supercapacitors with excellent room/low temperature performance. J. Mater. Chem. A 2019, 7, 13205–13214.
- Cong, S.; Hadipour, A.; Sugahara, T.; Wei, T.; Jiu, J.; Ranjbar, S.; Hirose, Y.; Karakawa, M.; Nagao, S.; Aernouts, T.; et al. Modifying the valence state of molybdenum in the efficient oxide buffer layer of organic solar cells via a mild hydrogen peroxide treatment. J. Mater. Chem. C 2017, 5, 889–895.
- Bai, Y.; Ma, Y.; Zheng, S.; Zhang, C.; Hu, C.; Liang, B.; Xu, Y.; Huang, G.; Yang, R. Oxygen deficiency and single-crystalline MoO3−x nanobelt as advanced supercapacitor negative electrode and dye adsorbent. Colloids Surf. A 2022, 647, 129064.
- Salkar, A.V.; Naik, A.P.; Peña, G.D.J.G.; Bharath, G.; Haija, M.A.; Banat, F.; Morajkar, P.P. 2D α-MoO3-x truncated microplates and microdisks as electroactive materials for highly efficient asymmetric supercapacitors. J. Energy Storage 2022, 48, 103958.
- Cao, Y.B.; Zhi, S.X.; Qi, H.B.; Zhang, Y.; Qin, C.; Yang, S.P. Evolution behavior of ex-situ NbC and properties of Fe-based laser clad coating. Opt. Laser Technol. 2020, 124, 8.
- Gao, J.; Xu, C.; Tian, X.; Sun, M.; Zhao, J.; Ma, J.Y.; Zhou, H.; Xiao, J.; Wu, M. Design bifunctional vanadium carbide embedded in mesoporous carbon electrode for supercapacitor and dye-sensitized solar cell. Sol. Energy 2020, 206, 848–854.
- Wan, C.; Zhang, R.; Wang, S.; Liu, X. Molten salt electrolytic fabrication of TiC-CDC and its applications for supercapacitor. J. Mater. Sci. Technol. 2017, 33, 788–792.
- Xu, Y.; Yang, Q.Q.; Zhang, W.B.; Liu, M.C.; Kong, L.B. Enhanced performance for a high electrical conductive Mo2C electrode based proton ionic liquid electrolytes in supercapacitors. Mater. Res. Express 2018, 5, 75508.
- Yu, Y.; Guo, Z.; Peng, Q.; Zhou, J.; Sun, Z. Novel two-dimensional molybdenum carbides as high capacity anodes for lithium/sodium-ion batteries. J. Mater. Chem. A 2019, 7, 12145–12153.
- Luo, Q.; Lu, C.; Liu, L.; Zhu, M. A review on the synthesis of transition metal nitride nanostructures and their energy related applications. Green Energy Environ. 2023, 8, 406–437.
- Xiao, X.; Yu, H.; Jin, H.; Wu, M.; Fang, Y.; Sun, J.; Hu, Z.; Li, T.; Wu, J.; Huang, L.; et al. Salt-templated synthesis of 2D metallic MoN and other nitrides. ACS Nano 2017, 11, 2180–2186.
- Gao, Z.; Zhang, T.; Wang, Q.; Heinz Mayrhofer, P. Nanostructured zig-zag γ-Mo2N thin films produced by glancing angle deposition for flexible symmetrical solid-state supercapacitors. Mater. Des. 2023, 225, 111432.
- Djire, A.; Siegel, J.B.; Ajenifujah, O.; He, L.; Thompson, L.T. Pseudocapacitive storage via micropores in high-surface area molybdenum nitrides. Nano Energy 2018, 51, 122–127.
- Wang, L.; Liu, X.; Luo, J.; Duan, X.; Crittenden, J.; Liu, C.; Zhang, S.; Pei, Y.; Zeng, Y.; Duan, X. Self-Optimization of the Active Site of Molybdenum Disulfide by an Irreversible Phase Transition during Photocatalytic Hydrogen Evolution. Angew. Chem. Int. Ed. 2017, 56, 7610–7614.
- Li, R.; Liang, S.; Aihemaiti, A.; Li, S.; Zhang, Z. Effectively enhanced piezocatalytic activity in flower-like 2H-MoS2 with tunable S vacancy towards organic pollutant degradation. Appl. Surf. Sci. 2023, 631, 157461.
- Marinov, A.D.; Bravo Priegue, L.; Shah, A.R.; Miller, T.S.; Howard, C.A.; Hinds, G.; Shearing, P.R.; Cullen, P.L.; Brett, D.J.L. Ex situ characterization of 1T/2H MoS2 and their carbon composites for energy applications, a review. ACS Nano 2023, 17, 5163–5186.
- Li, H.; Han, X.; Jiang, S.; Zhang, L.; Ma, W.; Ma, R.; Zhou, Z. Controllable fabrication and structure evolution of hierarchical 1T-MoS2 nanospheres for efficient hydrogen evolution. Green Energy Environ. 2022, 7, 314–323.
- Liu, X.; Liu, L.; Wu, Y.; Wang, Y.; Yang, J.; Wang, Z. Rosette-like MoS2 nanoflowers as highly active and stable electrodes for hydrogen evolution reactions and supercapacitors. RSC Adv. 2019, 9, 13820–13828.
- Teli, A.M.; Beknalkar, S.A.; Mane, S.M.; Bhat, T.S.; Kamble, B.B.; Patil, S.B.; Sadale, S.B.; Shin, J.C. Electrodeposited crumpled MoS2 nanoflakes for asymmetric supercapacitor. Ceram. Int. 2022, 48, 29002–29010.
- Joseph, N.; Muhammed Shafi, P.; Chandra Bose, A. Metallic 1T-MoS2 with defect induced additional active edges for high performance supercapacitor application. N. J. Chem. 2018, 42, 12082–12090.
- Gupta, H.; Chakrabarti, S.; Mothkuri, S.; Padya, B.; Rao, T.N.; Jain, P.K. High performance supercapacitor based on 2D-MoS2 nanostructures. Mater. Today Proc. 2020, 26, 20–24.
- Sha, R.; Maity, P.C.; Rajaji, U.; Liu, T.-Y.; Bhattacharyya, T.K. Review—MoSe2 nanostructures and related electrodes for advanced supercapacitor developments. J. Electrochem. Soc. 2022, 169, 13503.
- Vattikuti, S.V.P.; Devarayapalli, K.C.; Nagajyothi, P.C.; Shim, J. Microwave synthesized dry leaf-like mesoporous MoSe2 nanostructure as an efficient catalyst for enhanced hydrogen evolution and supercapacitor applications. Microchem. J. 2020, 153, 104446.
- Upadhyay, S.; Pandey, O.P. Synthesis of layered 2H-MoSe2 nanosheets for the high-performance supercapacitor electrode material. J. Alloys Compd. 2021, 857, 157522.
- Zhang, H.-J.; Wang, Y.-K.; Kong, L.-B. A facile strategy for the synthesis of three-dimensional heterostructure self-assembled MoSe2 nanosheets and their application as an anode for high-energy lithium-ion hybrid capacitors. Nanoscale 2019, 11, 7263–7276.
- Zhang, L.; Zheng, S.; Wang, L.; Tang, H.; Xue, H.; Wang, G.; Pang, H. Fabrication of metal molybdate micro/nanomaterials for electrochemical energy storage. Small 2017, 13, 19.
- Yang, J.; Yao, G.; Sun, S.; Chen, Z.; Yuan, S.; Wu, K.; Fu, X.; Wang, Q.; Cui, W. Structural, magnetic properties of in-plane chemically ordered (Mo2/3R)2AlC (R=Gd, Tb, Dy, Ho, Er and Y) MAX phase and enhanced capacitance of Mo1.33C MXene derivatives. Carbon 2021, 179, 104–110.
- Farahpour, M.; Arvand, M. Single-pot hydrothermal synthesis of copper molybdate nanosheet arrays as electrode materials for high areal-capacitance supercapacitor. J. Energy Storage 2021, 40, 102742.
- Gurusamy, H.; Sivasubramanian, R.; Johnbosco, Y.; Bhagavathiachari, M. Morphology-controlled synthesis of one-dimensional zinc molybdate nanorods for high-performance pseudocapacitor electrode application. Chem. Pap. 2020, 75, 1715–1726.
- Yesuraj, J.; Austin Suthanthiraraj, S.; Padmaraj, O. Synthesis, characterization and electrochemical performance of DNA-templated Bi2MoO6 nanoplates for supercapacitor applications. Mater. Sci. Semicond. Process. 2019, 90, 225–235.
- Qu, G.; Li, T.; Jia, S.; Zheng, H.; Li, L.; Cao, F.; Wang, H.; Ma, W.; Tang, Y.; Wang, J. Rapid and scalable synthesis of Mo-based binary and ternary oxides for electrochemical applications. Adv. Funct. Mater. 2017, 27, 1700928.
- Sheng, R.; Hu, J.; Lu, X.; Jia, W.; Xie, J.; Cao, Y. Solid-state synthesis and superior electrochemical performance of MnMoO4 nanorods for asymmetric supercapacitor. Ceram. Int. 2021, 47, 16316–16323.
- Sakthikumar, K.; Ede, S.R.; Mishra, S.; Kundu, S. Shape-selective synthesis of Sn(MoO4)2 nanomaterials for catalysis and supercapacitor applications. Dalton Trans. 2016, 45, 8897–8915.
- Li, L.; Zhou, J.; Zhang, Y.; Pei, X. Synthesis process optimization and electrochemical properties of CoMoO4 supercapacitor prepared by in situ growth method. J. Mater. Sci. Mater. Electron. 2022, 33, 23851–23866.
- Liang, C.; Meng, Y.; Zhang, Y.; Zhang, H.; Wang, W.; Lu, M.; Wang, G. Insights into the impact of interlayer spacing on MXene-based electrodes for supercapacitors: A review. J. Energy Storage 2023, 65, 20.
- Garg, R.; Agarwal, A.; Agarwal, M. A review on MXene for energy storage application: Effect of interlayer distance. Mater. Res. Express 2020, 7, 21.
- Halim, J.; Kota, S.; Lukatskaya, M.R.; Naguib, M.; Zhao, M.Q.; Moon, E.J.; Pitock, J.; Nanda, J.; May, S.J.; Gogotsi, Y.; et al. Synthesis and characterization of 2D molybdenum carbide (MXene). Adv. Funct. Mater. 2016, 26, 3118–3127.
- Das, M.; Ghosh, S. Theoretical investigation of capacitances in functionalised MXene supercapacitors Mn+1CnO2, M. = Ti, V., Nb, Mo. J. Phys. D Appl. Phys. 2021, 55, 85502.
- Zheng, W.; Halim, J.; Persson, P.O.Å.; Rosen, J.; Barsoum, M.W. Effect of vacancies on the electrochemical behavior of Mo-based MXenes in aqueous supercapacitors. J. Power Sources 2022, 525, 231064.
