Flame spray pyrolysis (FSP) is an industrially scalable technology that enables the engineering of a wide range of metal-based nanomaterials with tailored properties nanoparticles. In the present review, we discuss the recent state-of-the-art advances in FSP technology with regard to nanostructure engineering as well as the FSP reactor setup designs. The challenges of in situ incorporation of nanoparticles into complex functional arrays are reviewed, underscoring FSP's transformative potential in next-generation nanodevice fabrication. Key areas of focus include the integration of FSP into the technology readiness level (TRL) for nanomaterials production, the FSP process design, and recent advancements in nanodevice development. With a comprehensive overview of engineering methodologies such as the oxygen-deficient process, double-nozzle configuration, and in situ coatings deposition, this review charts the trajectory of FSP from its foundational roots to its contemporary applications in intricate nanostructure and nanodevice synthesis.
Flame spray pyrolysis (FSP) is an industrially scalable technology that enables the engineering of a wide range of metal-based nanomaterials with tailored properties nanoparticles.
- flame spray pyrolysis
- complex assemblies
- multifunctional nanomaterials/nanodevices
1. Introduction
1.1. Integration of Flame Spray Pyrolysis into the Technology Readiness Level (TRL) Scale for Nanomaterial Production
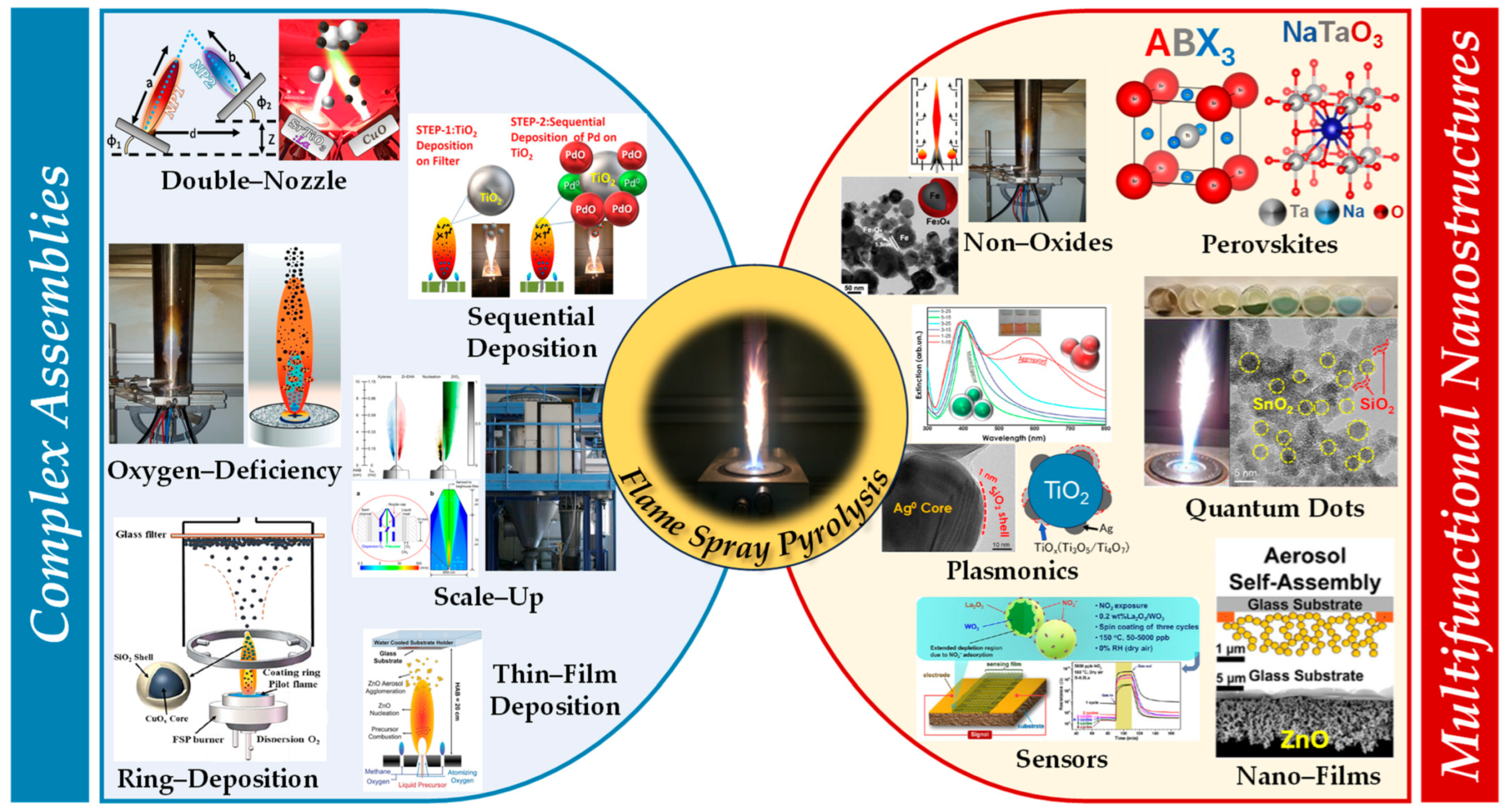
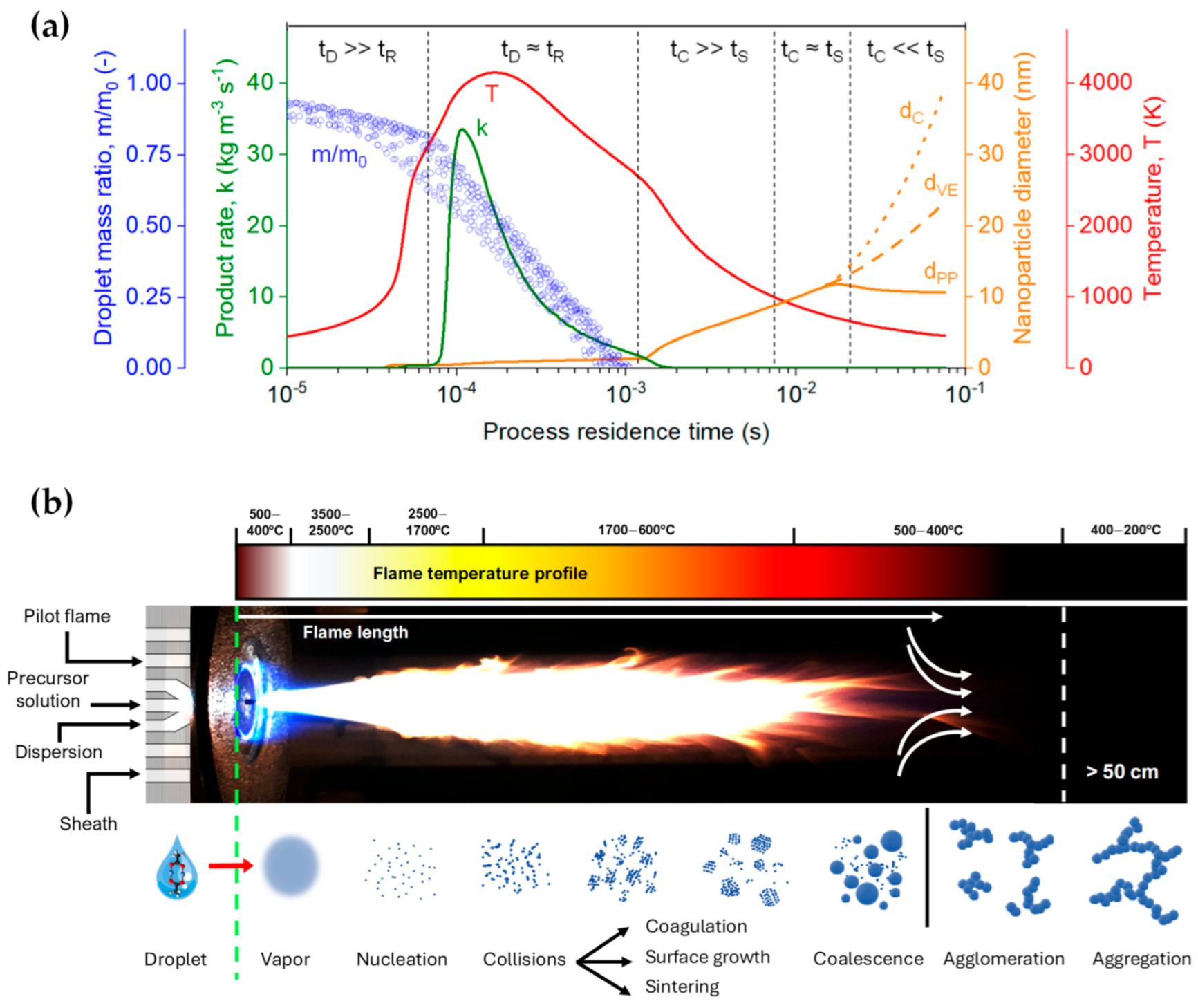
1.2. Process Design
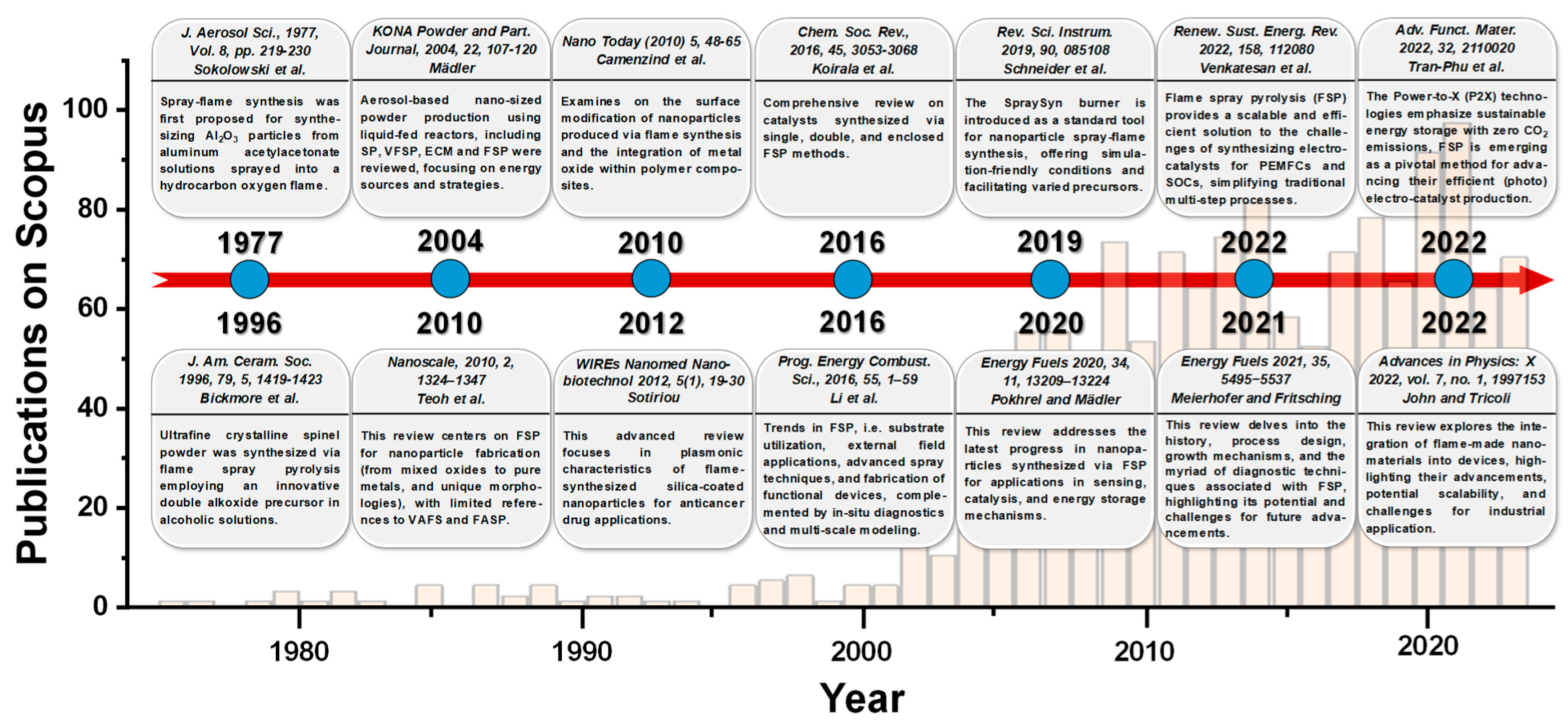
1.3. Recent Advancements in Product/Nanodevice Development
2. Engineering of Complex Nanoassemblies by Flame Spray Pyrolysis
2.1. Oxygen-Deficient FSP Process
The concept of oxygen-deficient synthesis can pertain to anoxic or reduced metal oxides. In the literature, these are referenced as MKOL−x where K and L are the stoichiometry coefficients that determine the stable crystal phase MKOL. In this terminology, x signifies the O-deficiency coefficient. Here, for the sake of the discussion, wresearchers classify these materials in three cases:- [i]
-
O-vacancies generation with no change in the crystal phase: lack of O atoms from the lattice, compared to the formal stoichiometry of the nominal crystal phase, with no modification of the crystal phase.
- [ii]
-
Generation or reduced metal atoms with no change in the crystal phase: lack of O atoms from the lattice can stabilize lower-oxidation states of the metal atoms.
- [iii]
-
Stabilization of a reduced crystal phase via lack of O atoms: certain O-deficient metal oxides can stabilize reduced phases. This occurs when a significant fraction part of the metal atoms is reduced. For example, magnetite Fe3O4, which contains one Fe2+ and two Fe3+, can be formed from Fe2O3 (two Fe3+) when 1/3 of the Fe3+-atoms is reduced to Fe2+. Further reduction in all Fe atoms to Fe2+ forms the FeO phase, while further reduction to Fe0-atoms forms the metallic, zero-valent-iron material. Similarly, Cu2O (SnO) is formed when all Cu2+ (Sn4+) atoms in CuO (SnO2) are reduced to the Cu1+ (Sn2+) state.Strobel and Pratsinis used an oxygen-deficiency FSP process [51][33] in order to synthesize Fe2O3, Fe3O4, and FeO nanoparticles. Their setup featured an FSP nozzle with a metal tube (4 cm in diameter and 40 cm in length) positioned directly above it (as shown in Figure 4b). Situated 20 cm above the FSP nozzle and angled at 45°, an internal mix spray nozzle was directed downward. This nozzle delivered deionized water at a rate of 10 mL/min, dispersed using 5 L/min of N2. A different oxygen-deficiency FSP setup for the production of Fe3O4 nanoparticles may be the utilization of a laminar, inverse diffusion flame [52][34]. This method takes advantage of the properties of the inverse flame, created when an oxidizer is injected into a flow of surrounding fuel [53][35]. Contrary to conventional flame approaches, this setup ensures that the iron particle formation occurs in a predominantly reducing atmosphere. As illustrated in Figure 5a, the burner features two concentric brass tubes with specific outer diameters, enclosed within an 11.4 cm diameter acrylic chamber. This chamber is crucial for protecting the flame from ambient air, preventing additional particle oxidation and potential secondary diffusion flame formation due to excess fuel reacting with room air. The oxidizer, either pure O2 or an O2-Ar mixture, is released from the innermost tube and is encircled by a blend of fuel (methane or ethylene), argon, and iron precursor vapor. A N2 flow enveloped the resulting inverse flame.
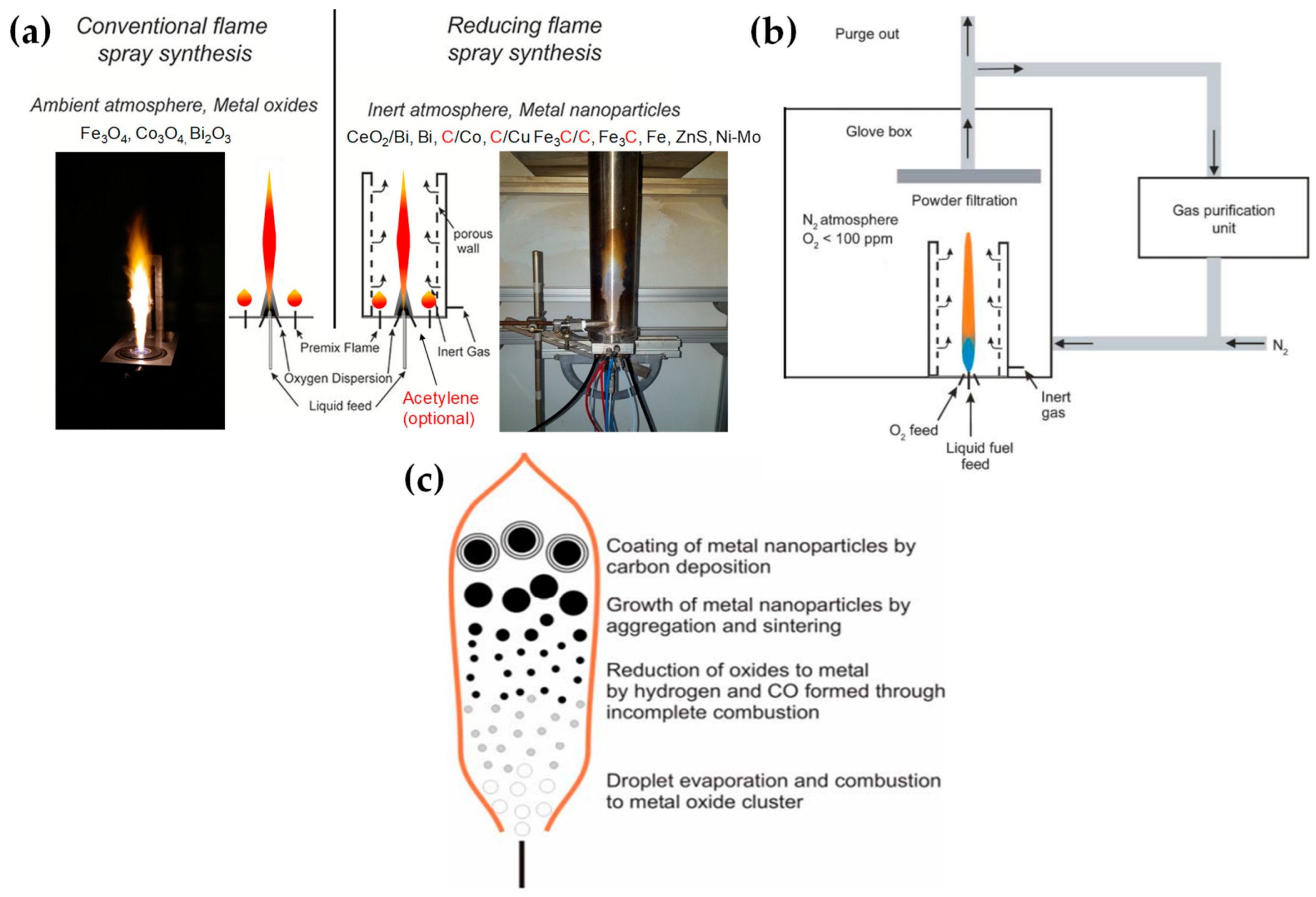 Figure 4. (a) Conventional FSP (left) and our reducing FSP (right), where the anoxic flame is produced by in situ introduction of reducing dispersion gas, e.g., CH4. (b) An anoxic FSP reactor, used by Stark, with the whole reactor enclosed in a glove box filled with an inert atmosphere. By adjusting the gas flow rates, it is possible to achieve highly reduced conditions (O2 < 100 ppm). Used with permission of Royal Society of Chemistry from [45][27]; permission conveyed through Copyright Clearance Center, Inc. (c) Schematic depiction of the step-by-step transformation from precursor to oxide, metal, and carbon-coated metal nanoparticles during the reducing flame synthesis process: Initially, the precursor undergoes evaporation and combustion, resulting in oxide nanoparticles. These particles can then be further reduced to their metallic form by H2 and CO. Throughout this procedure, the nanoparticles increase in size due to aggregation and sintering. By introducing acetylene, these metal nanoparticles can acquire a carbon coating layer.Recently, wresearchers have exemplified a novel anoxic FSP process, to engineer ZrO2–x (see Figure 5b) [54][36] and C@Cu2O/Cu0 (see Figure 5c) [55][37] nanoparticles. OurThe anoxic FSP concept relies on the combustion of CH4 in the dispersion gas. This introduces reducing agents that can modify the primary Zr particle by creating oxygen vacancies (VO). XPS and EPR confirm that the increased dispersion of the CH4 promotes the formation of oxygen vacancies [54][36]. A more complicated oxygen-deficiency FSP setup, which includes a dispersion feed consisting of {oxygen (O2)–methane (CH4)} mixture, in tandem with enclosed FSP flame with radial N2, is necessary for the synthesis of non-graphitized carbon/Cu2O/Cu0 heterojunction (see Figure 5c) [55][37]. The modification in the dispersion gas mixture leads to increased temperatures and generates reducing agents for the controlled phase transformation from CuO to Cu2O and Cu0 (see Figure 5c).
Figure 4. (a) Conventional FSP (left) and our reducing FSP (right), where the anoxic flame is produced by in situ introduction of reducing dispersion gas, e.g., CH4. (b) An anoxic FSP reactor, used by Stark, with the whole reactor enclosed in a glove box filled with an inert atmosphere. By adjusting the gas flow rates, it is possible to achieve highly reduced conditions (O2 < 100 ppm). Used with permission of Royal Society of Chemistry from [45][27]; permission conveyed through Copyright Clearance Center, Inc. (c) Schematic depiction of the step-by-step transformation from precursor to oxide, metal, and carbon-coated metal nanoparticles during the reducing flame synthesis process: Initially, the precursor undergoes evaporation and combustion, resulting in oxide nanoparticles. These particles can then be further reduced to their metallic form by H2 and CO. Throughout this procedure, the nanoparticles increase in size due to aggregation and sintering. By introducing acetylene, these metal nanoparticles can acquire a carbon coating layer.Recently, wresearchers have exemplified a novel anoxic FSP process, to engineer ZrO2–x (see Figure 5b) [54][36] and C@Cu2O/Cu0 (see Figure 5c) [55][37] nanoparticles. OurThe anoxic FSP concept relies on the combustion of CH4 in the dispersion gas. This introduces reducing agents that can modify the primary Zr particle by creating oxygen vacancies (VO). XPS and EPR confirm that the increased dispersion of the CH4 promotes the formation of oxygen vacancies [54][36]. A more complicated oxygen-deficiency FSP setup, which includes a dispersion feed consisting of {oxygen (O2)–methane (CH4)} mixture, in tandem with enclosed FSP flame with radial N2, is necessary for the synthesis of non-graphitized carbon/Cu2O/Cu0 heterojunction (see Figure 5c) [55][37]. The modification in the dispersion gas mixture leads to increased temperatures and generates reducing agents for the controlled phase transformation from CuO to Cu2O and Cu0 (see Figure 5c).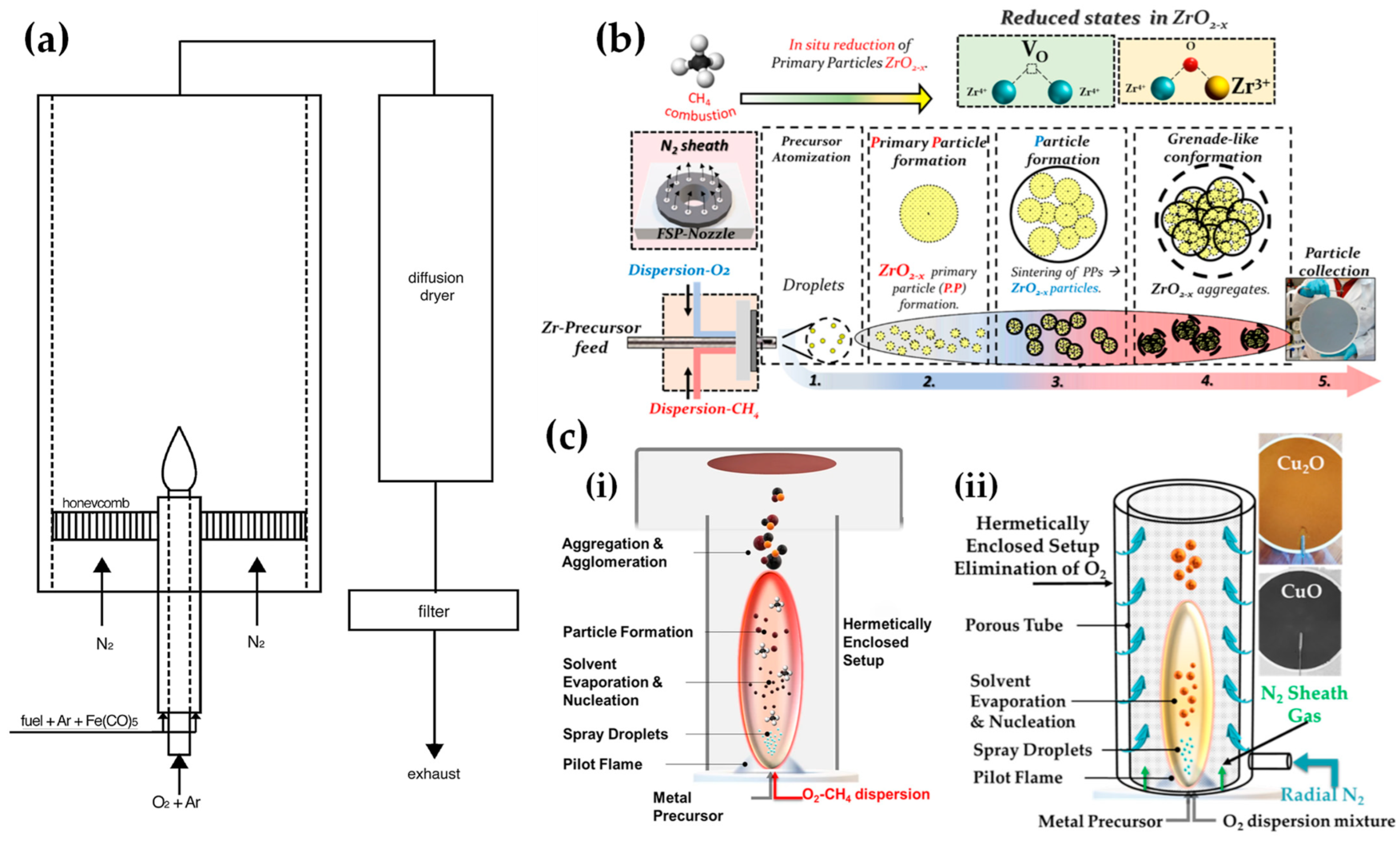 Figure 5. (a) Experimental setup of laminar, inverse diffusion flame stabilized on a burner for the synthesis of magnetic iron oxide nanoparticles with reduced oxidation state. (b) The concept of the novel anoxic FSP, as developed by our lab, for ZrO2−x production. (c) (i) Schematic representation of anoxic FSP reactor used for the synthesis of C@Cu2O/Cu0 nanoparticles. (ii) Anoxic FSP reactor configuration utilized for creating CuO and Cu2O nanomaterials.
Figure 5. (a) Experimental setup of laminar, inverse diffusion flame stabilized on a burner for the synthesis of magnetic iron oxide nanoparticles with reduced oxidation state. (b) The concept of the novel anoxic FSP, as developed by our lab, for ZrO2−x production. (c) (i) Schematic representation of anoxic FSP reactor used for the synthesis of C@Cu2O/Cu0 nanoparticles. (ii) Anoxic FSP reactor configuration utilized for creating CuO and Cu2O nanomaterials.2.2. Double-Nozzle FSP Configuration
In the case of mixed structures, e.g., heterojunctions, core-shell compositions, etc., the application of two FSP nozzles that operate in tandem offers advantages. Typical examples include the cases where a nanomaterial (NP1) and a cocatalytic nanomaterial (NP2) are combined. In the conventional single-nozzle FSP, a single precursor contains both the elements of nanomaterial (NP1) and nanomaterial (NP2) and produces the combined material in a single flame (see Figure 6a).Double-nozzle FSP entails two independent spray flames, with the precursor of NP1 inserted in a different flame than NP2 (see Figure 6). This method unlocks several options for independent size control, mixing, and specific deposition for the two nanomaterials by altering the primary geometrical parameters of distance and intersection of the flames. As shown in Figure 6: (i) At a small flame-intersection distance, where the centers of the flames are in contact, the atoms are in the preliminary stages of crystallization, producing well-mixed particles, tending to be similar to the single-nozzle FSP. In this case, the second flame substantially increases the synthesis overall temperature. (ii) When the intersection occurs after the endpoints of the flames, the materials are well crystallized, resulting in well-mixed primary particles of NP1 and NP2. (iii) At increased intersection distance, the two materials mix at their sintering stage or bigger distances at the agglomeration stage. Thus, by changing the geometrical disposition of the two flames via the parameters a, b, d, Φ1, Φ2, and Z (see Figure 7b), the symmetrical/asymmetrical DN-FSP configuration offers a versatile technology that allows for the control of composite configurations at different synthesis stages, i.e., at the atomic scale, at the particle scale, or the aggregate’s scale (see Figure 6).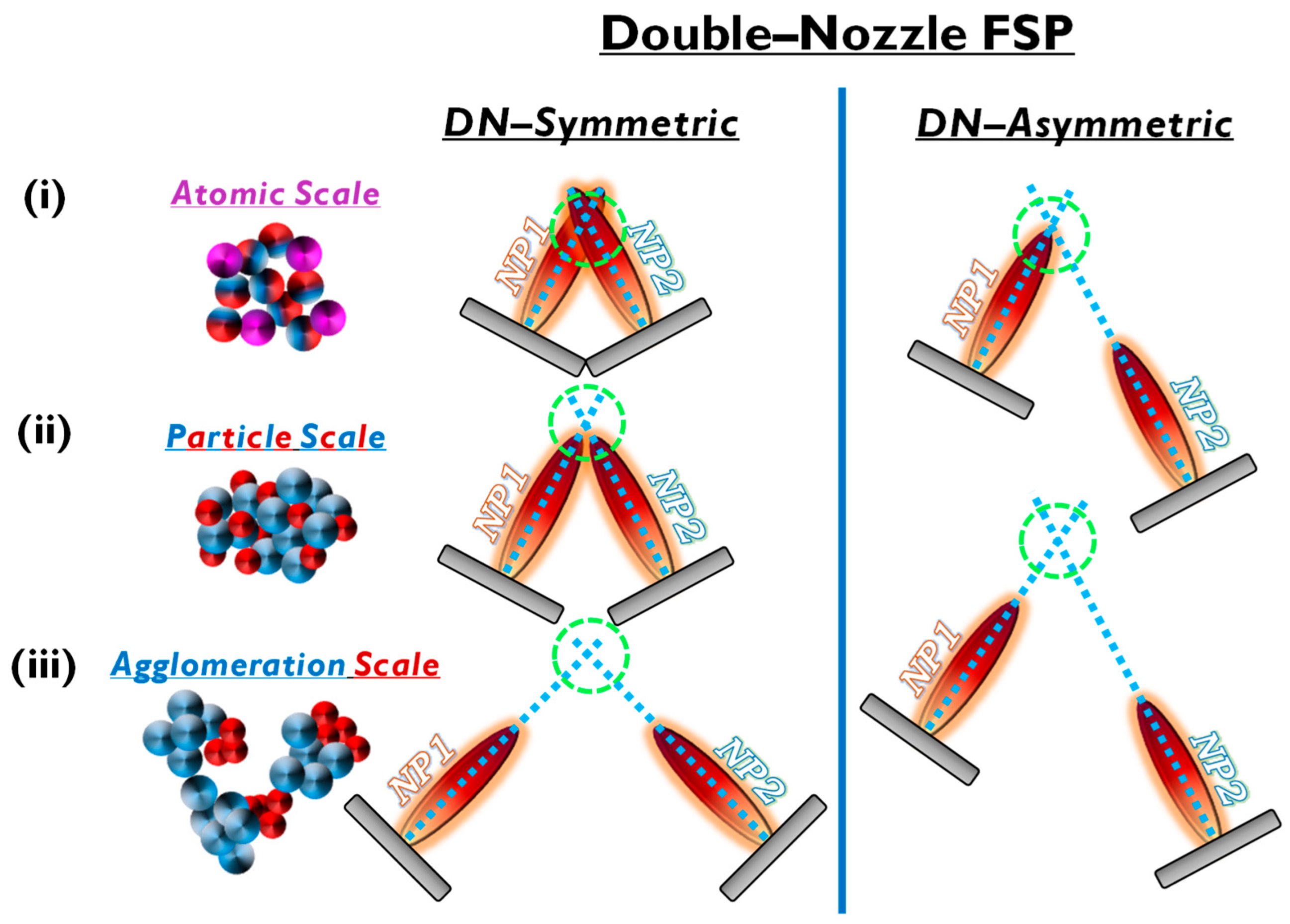 Figure 6.Symmetric and asymmetric DN-FSP configuration for two particle formation regarding the (i) atomic, (ii) particle, or (iii) agglomeration scale.Al2O3: DN-FSP was first implemented by Strobel et al. [59][38], producing in one nozzle Al2O3 and in the second nozzle Pt/BaCO3, thus forming individual Al2O3 and monoclinic BaCO3 nanoparticles. Increasing the internozzle distance delayed flame product mixing, increasing the crystallinity of BaCO3. In contrast, the single-nozzle process yielded Al2O3 particles with amorphous Ba species. The two-nozzle process enhanced NOx storage behavior, while the single-nozzle approach showed negligible NOx retention [59][38]. Following this successful novelty method, a series of Al2O3-based articles were published, herein chronologically presented: Minnermann et al. [60][39] produced in one nozzle Al2O3 and in the other pure oxide or mixed CoOx. Single flame synthesis is inadequate for producing an effective Al2O3/Co FT catalyst due to inadequate reducible cobalt oxide support particle size. The DN-FSP geometry significantly influences the resulting catalyst, yielding smaller alumina particles as the intersection distance increases, resulting in good adhesion of the two oxides and good stabilization. Høj et al. [61][40] produced Al2O3/CoMo by DN-FSP, and varying flame mixing distances (81–175 mm) minimized the formation of CoAl2O4, detectable only at short flame distances. Notably, employing DN-FSP synthesis achieved superior promotion of the active molybdenum sulfide phase, potentially attributed to reduced CoAl2O4 formation, consequently enhancing Co availability for promotion. Schubert et al. [62][41], through DN-FSP, produced Al2O3/Co enhanced with Pt (0.03, 0.43 wt%) deposition in the first nozzle and other materials in the second nozzle. Noble metals enhance catalyst reducibility, yielding abundant metallic Co sites. Due to their high cost, optimizing synthetic strategies for low concentrations is essential. Regardless of the preparation approach, adding 0.03 wt% Pt significantly improves catalytic activity in CO2 methanation, and 0.43 wt% Pt marginally increases the catalyst reduction. Using DN-FSP, Horlyck et al. [63][42] produced Al2O3/Co with Lanthanum doping (0–15 wt%). Increased La content and wider nozzle distance suppressed undesirable CoAl2O4 spinel phase, promoting easily reducible Co species. La addition enhanced carbon resistance, ensuring maximum methane conversions at 15 wt% La without catalyst deactivation or carbon formation. Stahl et al. [64][43] used DN-FSP to produce Co/Al2O3; in the nozzle of Al2O3, one additional particle—SmOx, ZrOx, or Pt—was formed contributing different cocatalytic effects, enhancing surface hydrogen or carbon oxide concentrations (see Figure 8a,b). All catalysts had consistent morphology with interconnected 12 nm alumina oxides and ~ 8 nm cobalt oxides. For CO2 methanation, Pt and zirconia proved optimal, aligning with Pt-enhanced H2 adsorption and zirconia’s higher CO2 adsorption due to oxide sites with medium basicity.
Figure 6.Symmetric and asymmetric DN-FSP configuration for two particle formation regarding the (i) atomic, (ii) particle, or (iii) agglomeration scale.Al2O3: DN-FSP was first implemented by Strobel et al. [59][38], producing in one nozzle Al2O3 and in the second nozzle Pt/BaCO3, thus forming individual Al2O3 and monoclinic BaCO3 nanoparticles. Increasing the internozzle distance delayed flame product mixing, increasing the crystallinity of BaCO3. In contrast, the single-nozzle process yielded Al2O3 particles with amorphous Ba species. The two-nozzle process enhanced NOx storage behavior, while the single-nozzle approach showed negligible NOx retention [59][38]. Following this successful novelty method, a series of Al2O3-based articles were published, herein chronologically presented: Minnermann et al. [60][39] produced in one nozzle Al2O3 and in the other pure oxide or mixed CoOx. Single flame synthesis is inadequate for producing an effective Al2O3/Co FT catalyst due to inadequate reducible cobalt oxide support particle size. The DN-FSP geometry significantly influences the resulting catalyst, yielding smaller alumina particles as the intersection distance increases, resulting in good adhesion of the two oxides and good stabilization. Høj et al. [61][40] produced Al2O3/CoMo by DN-FSP, and varying flame mixing distances (81–175 mm) minimized the formation of CoAl2O4, detectable only at short flame distances. Notably, employing DN-FSP synthesis achieved superior promotion of the active molybdenum sulfide phase, potentially attributed to reduced CoAl2O4 formation, consequently enhancing Co availability for promotion. Schubert et al. [62][41], through DN-FSP, produced Al2O3/Co enhanced with Pt (0.03, 0.43 wt%) deposition in the first nozzle and other materials in the second nozzle. Noble metals enhance catalyst reducibility, yielding abundant metallic Co sites. Due to their high cost, optimizing synthetic strategies for low concentrations is essential. Regardless of the preparation approach, adding 0.03 wt% Pt significantly improves catalytic activity in CO2 methanation, and 0.43 wt% Pt marginally increases the catalyst reduction. Using DN-FSP, Horlyck et al. [63][42] produced Al2O3/Co with Lanthanum doping (0–15 wt%). Increased La content and wider nozzle distance suppressed undesirable CoAl2O4 spinel phase, promoting easily reducible Co species. La addition enhanced carbon resistance, ensuring maximum methane conversions at 15 wt% La without catalyst deactivation or carbon formation. Stahl et al. [64][43] used DN-FSP to produce Co/Al2O3; in the nozzle of Al2O3, one additional particle—SmOx, ZrOx, or Pt—was formed contributing different cocatalytic effects, enhancing surface hydrogen or carbon oxide concentrations (see Figure 8a,b). All catalysts had consistent morphology with interconnected 12 nm alumina oxides and ~ 8 nm cobalt oxides. For CO2 methanation, Pt and zirconia proved optimal, aligning with Pt-enhanced H2 adsorption and zirconia’s higher CO2 adsorption due to oxide sites with medium basicity.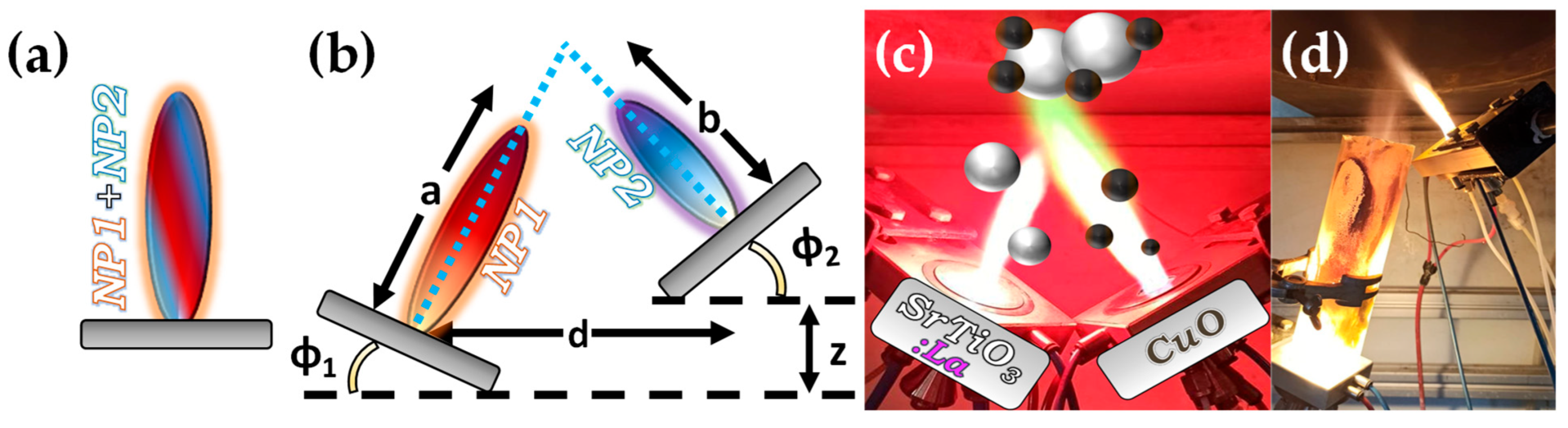 Figure 7. (a) Schematic example of SN-FSP where two precursors are mixed before being fed to the flame. (b) Geometry parameters of DN-FSP. (c) Example of a symmetrical DN-FSP, used for engineering of La-doped SrTiO3, with surface deposition of CuO. (d) Example of asymmetrical DN-FSP.TiO2: Grossmann et al., through the utilization of DN-FSP, produced TiO2 with deposited Pt particles [67][44]. Geometric configurations in DN-FSP strongly influenced Pt particle size and distribution on TiO2. Larger intersection distances and smaller angles result in nonuniform large and broadly distributed Pt clusters on TiO2. Conversely, smaller distances and larger angles enhance Pt dispersion and a uniform mixing, akin to single flame; however, DN-FSP allows for individual tuning of compound particle sizes. Solakidou et al. produced {TiO2-Noble metal} nanohybrids, with deposition of Pt0, Pd0, Au0, or Ag0 [68][45]. As shown, DN-FSP is superior vs. single-nozzle-FSP for finely dispersing noble metals on TiO2 support, achieving a narrower size distribution [50][32]. DN-FSP promoted intraband states in TiO2/noble metal, reducing the band gap. Efficient H2 generation presented the following trend: Pt0 > Pd0 > Au0 > Ag0, in line with a higher Schottky barrier upon TiO2 contact [50][32]. Gäßler et al. produced SiO2, TiO2, and SiO2-TiO2 mixture with DN-FSP deposition of Co3O4 (see Figure 8c–f) [65][46]: titania, comprising anatase and rutile phases, the SiO2-TiO2 mixed support, with separate anatase and silica phases. H2O adsorption varies significantly based on the support: SiO2 < SiO2-TiO2 < TiO2. CH4 formation rate increased with higher TiO2 fractions, while CO formation rate peaked in the mixed support. Psathas et al. used DN-FSP to engineer heterojunctions of perovskite SrTiO3 with deposited CuO nanoparticles (0.5 to 2 wt%) [58][47]. Higher CuO deposition led to larger SrTiO3 particle sizes due to increased enthalpy from the second flame [40][22]. Scanning TEM depicted small CuO particles (<2 nm), mainly found on the surface of SrTiO3. The dopant concentration significantly controlled the selective production of H2 or CH4 from H2O/CH3OH. CuO incorporation drastically shifted production to CH4, achieving a rate of 1.5 mmol g−1 h−1 for the La:SrTiO3/CuO catalyst (0.5 wt%) [58][47]. Other particles: Tada et al., using DN-FSP, produced a ZrO2/CuO heterostructure [69][48]. Changing the geometrical parameters of DN-FSP altered the proportion of interfacial sites vs. copper surface sites. As active sites are primarily at the metal–oxide interface, ZrO2/CuO with smaller CuO clusters exhibited higher activity in methanol synthesis via CO2 hydrogenation. Gockeln et al., by a combination of DN-FSP and a lamination technique [73][49], synthesized in situ carbon-coated nano-Li4Ti5O12 Li-ion battery electrodes. Li et al. synthesized LiMn2O4 spinel as a cathode material for Li-ion batteries via screening 16 different precursor–solvent combinations [70][50]. To overcome the drawback of capacity fading, the deposition of AlPO4 (1–5%) via DN-FSP was homogeneously mixed with LiMn2O4. The optimal 1% AlPO4 with LiMn2O4 demonstrated an energy density of 116.1 mA h g−1 at 1 C (one-hour discharge). Henning et al. used DN-FSP to engineer luminescent biosensors CeO2:Eu3+/Y2O3:Tb3+ [66][51]. CeO2:Eu3+ nanoparticles (6 nm, 22 wt%) and Y2O3:Tb3+ nanoparticles (32.5 nm, 78 wt%) were shown to function as robust optical-based ratiometric H2O2 biosensors (see Figure 8g,h). Based on the collective effect, H2O2 caused significant luminescence quenching in CeO2:Eu3+ nanocrystals, but Y2O3:Tb3+ nanoparticles were unaffected [48][30]. Asymmetric Double Flame: Lovell et al. utilized asymmetric-DN-FSP geometry to control the SiO2 interaction with Ce0.7Zr0.3O2 nanoparticles [71][52]. Tuning the intersection distance during DN-FSP (18.5 to 28.5 cm) prevented silica coating. Short intersection distances led to high surface-area silica encapsulating ceria-zirconia, while longer distances suppressed this encapsulation. The material at longer intersection distances, used as Ni support for dry methane reforming, showed enhanced oxygen storage capacity and basicity, yielding a highly selective catalyst. Psathas et al. used asymmetrical-DN-FSP-deposited NiO or Pt0 nanomaterials on the surface of Ta2O5 or the perovskite NaTaO3 [72][53]. Single-step synthesis of the smallest produced NaTaO3 (<15 nm), with finely dispersed NiO or Pt0 (<3 nm). NaTaO3/NiO produced from FSP had half the photocatalytic hydrogen production than those from DN-FSP. Also, DN-FSP had a ten times higher yield than the conventional deposition of wet-impregnated NiO. Similar results were found for the photocatalytic efficiency of NaTaO3/Pt0, which was 30% more photocatalytically active than the conventional liquid-Pt photo-deposition method [54][36].
Figure 7. (a) Schematic example of SN-FSP where two precursors are mixed before being fed to the flame. (b) Geometry parameters of DN-FSP. (c) Example of a symmetrical DN-FSP, used for engineering of La-doped SrTiO3, with surface deposition of CuO. (d) Example of asymmetrical DN-FSP.TiO2: Grossmann et al., through the utilization of DN-FSP, produced TiO2 with deposited Pt particles [67][44]. Geometric configurations in DN-FSP strongly influenced Pt particle size and distribution on TiO2. Larger intersection distances and smaller angles result in nonuniform large and broadly distributed Pt clusters on TiO2. Conversely, smaller distances and larger angles enhance Pt dispersion and a uniform mixing, akin to single flame; however, DN-FSP allows for individual tuning of compound particle sizes. Solakidou et al. produced {TiO2-Noble metal} nanohybrids, with deposition of Pt0, Pd0, Au0, or Ag0 [68][45]. As shown, DN-FSP is superior vs. single-nozzle-FSP for finely dispersing noble metals on TiO2 support, achieving a narrower size distribution [50][32]. DN-FSP promoted intraband states in TiO2/noble metal, reducing the band gap. Efficient H2 generation presented the following trend: Pt0 > Pd0 > Au0 > Ag0, in line with a higher Schottky barrier upon TiO2 contact [50][32]. Gäßler et al. produced SiO2, TiO2, and SiO2-TiO2 mixture with DN-FSP deposition of Co3O4 (see Figure 8c–f) [65][46]: titania, comprising anatase and rutile phases, the SiO2-TiO2 mixed support, with separate anatase and silica phases. H2O adsorption varies significantly based on the support: SiO2 < SiO2-TiO2 < TiO2. CH4 formation rate increased with higher TiO2 fractions, while CO formation rate peaked in the mixed support. Psathas et al. used DN-FSP to engineer heterojunctions of perovskite SrTiO3 with deposited CuO nanoparticles (0.5 to 2 wt%) [58][47]. Higher CuO deposition led to larger SrTiO3 particle sizes due to increased enthalpy from the second flame [40][22]. Scanning TEM depicted small CuO particles (<2 nm), mainly found on the surface of SrTiO3. The dopant concentration significantly controlled the selective production of H2 or CH4 from H2O/CH3OH. CuO incorporation drastically shifted production to CH4, achieving a rate of 1.5 mmol g−1 h−1 for the La:SrTiO3/CuO catalyst (0.5 wt%) [58][47]. Other particles: Tada et al., using DN-FSP, produced a ZrO2/CuO heterostructure [69][48]. Changing the geometrical parameters of DN-FSP altered the proportion of interfacial sites vs. copper surface sites. As active sites are primarily at the metal–oxide interface, ZrO2/CuO with smaller CuO clusters exhibited higher activity in methanol synthesis via CO2 hydrogenation. Gockeln et al., by a combination of DN-FSP and a lamination technique [73][49], synthesized in situ carbon-coated nano-Li4Ti5O12 Li-ion battery electrodes. Li et al. synthesized LiMn2O4 spinel as a cathode material for Li-ion batteries via screening 16 different precursor–solvent combinations [70][50]. To overcome the drawback of capacity fading, the deposition of AlPO4 (1–5%) via DN-FSP was homogeneously mixed with LiMn2O4. The optimal 1% AlPO4 with LiMn2O4 demonstrated an energy density of 116.1 mA h g−1 at 1 C (one-hour discharge). Henning et al. used DN-FSP to engineer luminescent biosensors CeO2:Eu3+/Y2O3:Tb3+ [66][51]. CeO2:Eu3+ nanoparticles (6 nm, 22 wt%) and Y2O3:Tb3+ nanoparticles (32.5 nm, 78 wt%) were shown to function as robust optical-based ratiometric H2O2 biosensors (see Figure 8g,h). Based on the collective effect, H2O2 caused significant luminescence quenching in CeO2:Eu3+ nanocrystals, but Y2O3:Tb3+ nanoparticles were unaffected [48][30]. Asymmetric Double Flame: Lovell et al. utilized asymmetric-DN-FSP geometry to control the SiO2 interaction with Ce0.7Zr0.3O2 nanoparticles [71][52]. Tuning the intersection distance during DN-FSP (18.5 to 28.5 cm) prevented silica coating. Short intersection distances led to high surface-area silica encapsulating ceria-zirconia, while longer distances suppressed this encapsulation. The material at longer intersection distances, used as Ni support for dry methane reforming, showed enhanced oxygen storage capacity and basicity, yielding a highly selective catalyst. Psathas et al. used asymmetrical-DN-FSP-deposited NiO or Pt0 nanomaterials on the surface of Ta2O5 or the perovskite NaTaO3 [72][53]. Single-step synthesis of the smallest produced NaTaO3 (<15 nm), with finely dispersed NiO or Pt0 (<3 nm). NaTaO3/NiO produced from FSP had half the photocatalytic hydrogen production than those from DN-FSP. Also, DN-FSP had a ten times higher yield than the conventional deposition of wet-impregnated NiO. Similar results were found for the photocatalytic efficiency of NaTaO3/Pt0, which was 30% more photocatalytically active than the conventional liquid-Pt photo-deposition method [54][36].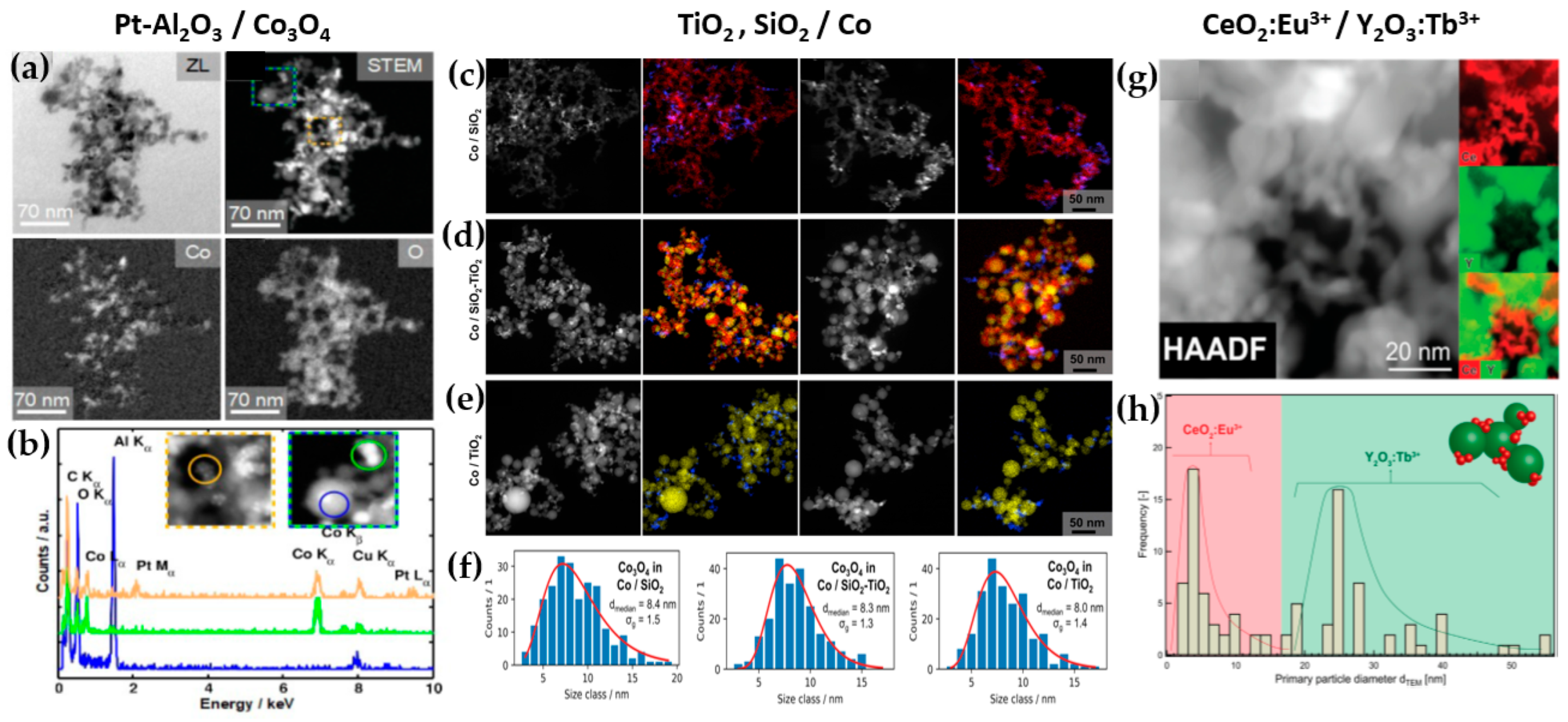 Figure 8. (a) TEM images revealing the local distribution of cobalt and oxygen for Pt-Al2O3/Co3O4, (b) EDX measurements for chemical composition. DΝ-FSP-prepared (c) SiO2/Co, (d) SiO2-TiO2/Co, (e) and TiO2/Co; left images show STEM-HAADF and right images show EDX mappings of the elements Co (blue), Si (red) and Ti (yellow). (f) Particle size distributions of Co3O4 for the materials SiO2, SiO2-TiO2, and TiO2. (g) STEM-HAADF of the nano-mixed CeO2:Eu3+/Y2O3:Tb3+ and its elemental mapping for Ce in red and Y in green, (h) dTEM distribution of CeO2:Eu3+ and Y2O3:Tb3+.
Figure 8. (a) TEM images revealing the local distribution of cobalt and oxygen for Pt-Al2O3/Co3O4, (b) EDX measurements for chemical composition. DΝ-FSP-prepared (c) SiO2/Co, (d) SiO2-TiO2/Co, (e) and TiO2/Co; left images show STEM-HAADF and right images show EDX mappings of the elements Co (blue), Si (red) and Ti (yellow). (f) Particle size distributions of Co3O4 for the materials SiO2, SiO2-TiO2, and TiO2. (g) STEM-HAADF of the nano-mixed CeO2:Eu3+/Y2O3:Tb3+ and its elemental mapping for Ce in red and Y in green, (h) dTEM distribution of CeO2:Eu3+ and Y2O3:Tb3+.
References
- Meierhofer, F.; Fritsching, U. Synthesis of Metal Oxide Nanoparticles in Flame Sprays: Review on Process Technology, Modeling, and Diagnostics. Energy Fuels 2021, 35, 5495–5537.
- Teoh, W.Y.; Amal, R.; Mädler, L. Flame Spray Pyrolysis: An Enabling Technology for Nanoparticles Design and Fabrication. Nanoscale 2010, 2, 1324–1347.
- Ulrich, G.D. Special Report. Chem. Eng. News Arch. 1984, 62, 22–29.
- Liu, S.; Mohammadi, M.M.; Swihart, M.T. Fundamentals and Recent Applications of Catalyst Synthesis Using Flame Aerosol Technology. Chem. Eng. J. 2021, 405, 126958.
- Malekzadeh, M.; Swihart, M.T. Vapor-Phase Production of Nanomaterials. Chem. Soc. Rev. 2021, 50, 7132–7249.
- Rsanchez. Technology Readiness Assessment Guide—DOE Directives, Guidance, and Delegations. Available online: https://www.directives.doe.gov/directives-documents/400-series/0413.3-EGuide-04a (accessed on 25 October 2023).
- Workie, A.B.; Ningsih, H.S.; Shih, S.-J. An Comprehensive Review on the Spray Pyrolysis Technique: Historical Context, Operational Factors, Classifications, and Product Applications. J. Anal. Appl. Pyrolysis 2023, 170, 105915.
- Teoh, W. A Perspective on the Flame Spray Synthesis of Photocatalyst Nanoparticles. Materials 2013, 6, 3194–3212.
- Strobel, R.; Pratsinis, S.E. Flame Aerosol Synthesis of Smart Nanostructured Materials. J. Mater. Chem. 2007, 17, 4743.
- CRC. Handbook of Chemistry and Physics, 95th ed.; Haynes, W.M., Ed.; CRC Press: Boca Raton, FL, USA, 2014; ISBN 978-1-4822-0867-2.
- Sokolowski, M.; Sokolowska, A.; Michalski, A.; Gokieli, B. The “in-Flame-Reaction” Method for Al2O3 Aerosol Formation. J. Aerosol Sci. 1977, 8, 219–230.
- Karthikeyan, J.; Berndt, C.C.; Tikkanen, J.; Wang, J.Y.; King, A.H.; Herman, H. Nanomaterial Powders and Deposits Prepared by Flame Spray Processing of Liquid Precursors. Nanostructured Mater. 1997, 8, 61–74.
- Bickmore, C.R.; Waldner, K.F.; Treadwell, D.R.; Laine, R.M. Ultrafine Spinel Powders by Flame Spray Pyrolysis of a Magnesium Aluminum Double Alkoxide. J. Am. Ceram. Soc. 1996, 79, 1419–1423.
- Tikkanen, J.; Gross, K.A.; Berndt, C.C.; Pitkänen, V.; Keskinen, J.; Raghu, S.; Rajala, M.; Karthikeyan, J. Characteristics of the Liquid Flame Spray Process. Surf. Coat. Technol. 1997, 90, 210–216.
- Mädler, L.; Kammler, H.K.; Mueller, R.; Pratsinis, S.E. Controlled Synthesis of Nanostructured Particles by Flame Spray Pyrolysis. J. Aerosol Sci. 2002, 33, 369–389.
- Mädler, L. Liquid-Fed Aerosol Reactors for One-Step Synthesis of Nano-Structured Particles. KONA Powder Part. J. 2004, 22, 107–120.
- Camenzind, A.; Caseri, W.R.; Pratsinis, S.E. Flame-Made Nanoparticles for Nanocomposites. Nano Today 2010, 5, 48–65.
- Sotiriou, G.A. Biomedical Applications of Multifunctional Plasmonic Nanoparticles. WIREs Nanomed. Nanobiotechnol. 2013, 5, 19–30.
- Koirala, R.; Pratsinis, S.E.; Baiker, A. Synthesis of Catalytic Materials in Flames: Opportunities and Challenges. Chem. Soc. Rev. 2016, 45, 3053–3068.
- Li, S.; Ren, Y.; Biswas, P.; Tse, S.D. Flame Aerosol Synthesis of Nanostructured Materials and Functional Devices: Processing, Modeling, and Diagnostics. Prog. Energy Combust. Sci. 2016, 55, 1–59.
- Schneider, F.; Suleiman, S.; Menser, J.; Borukhovich, E.; Wlokas, I.; Kempf, A.; Wiggers, H.; Schulz, C. SpraySyn—A Standardized Burner Configuration for Nanoparticle Synthesis in Spray Flames. Rev. Sci. Instrum. 2019, 90, 085108.
- Pokhrel, S.; Mädler, L. Flame-Made Particles for Sensors, Catalysis, and Energy Storage Applications. Energy Fuels 2020, 34, 13209–13224.
- Venkatesan, S.; Mitzel, J.; Wegner, K.; Costa, R.; Gazdzicki, P.; Friedrich, K.A. Nanomaterials and Films for Polymer Electrolyte Membrane Fuel Cells and Solid Oxide Cells by Flame Spray Pyrolysis. Renew. Sustain. Energy Rev. 2022, 158, 112080.
- Tran-Phu, T.; Daiyan, R.; Ta, X.M.C.; Amal, R.; Tricoli, A. From Stochastic Self-Assembly of Nanoparticles to Nanostructured (Photo)Electrocatalysts for Renewable Power-to-X Applications via Scalable Flame Synthesis. Adv. Funct. Mater. 2022, 32, 2110020.
- John, A.T.; Tricoli, A. Flame Assisted Synthesis of Nanostructures for Device Applications. Adv. Phys. X 2022, 7, 1997153.
- Grass, R.N.; Stark, W.J.; Athanassiou, E.-K. Reducing Flame Spray Pyrolysis Method for the Production of Metal, Non-Oxidic, Ceramic and Reduced Metal Oxide Powders and Nano-Powders, European Patent Office. European Patent EP1760043A1, 7 March 2007.
- Grass, R.N.; Albrecht, T.F.; Krumeich, F.; Stark, W.J. Large-Scale Preparation of Ceria/Bismuth Metal-Matrix Nano-Composites with a Hardness Comparable to Steel. J. Mater. Chem. 2007, 17, 1485.
- Grass, R.N.; Stark, W.J. Flame Spray Synthesis under a Non-Oxidizing Atmosphere: Preparation of Metallic Bismuth Nanoparticles and Nanocrystalline Bulk Bismuth Metal. J. Nanopart. Res. 2006, 8, 729–736.
- Athanassiou, E.K.; Grass, R.N.; Stark, W.J. Large-Scale Production of Carbon-Coated Copper Nanoparticles for Sensor Applications. Nanotechnology 2006, 17, 1668–1673.
- Grass, R.N.; Athanassiou, E.K.; Stark, W.J. Covalently Functionalized Cobalt Nanoparticles as a Platform for Magnetic Separations in Organic Synthesis. Angew. Chem. Int. Ed. 2007, 46, 4909–4912.
- Athanassiou, E.K.; Grass, R.N.; Osterwalder, N.; Stark, W.J. Preparation of Homogeneous, Bulk Nanocrystalline Ni/Mo Alloys with Tripled Vickers Hardness Using Flame-Made Metal Nanoparticles. Chem. Mater. 2007, 19, 4847–4854.
- Athanassiou, E.K.; Grass, R.N.; Stark, W.J. One-Step Large Scale Gas Phase Synthesis of Mn2+ Doped ZnS Nanoparticles in Reducing Flames. Nanotechnology 2010, 21, 215603.
- Strobel, R.; Pratsinis, S.E. Direct Synthesis of Maghemite, Magnetite and Wustite Nanoparticles by Flame Spray Pyrolysis. Adv. Powder Technol. 2009, 20, 190–194.
- Kumfer, B.M.; Shinoda, K.; Jeyadevan, B.; Kennedy, I.M. Gas-Phase Flame Synthesis and Properties of Magnetic Iron Oxide Nanoparticles with Reduced Oxidation State. J. Aerosol Sci. 2010, 41, 257–265.
- Kumfer, B.M.; Skeen, S.A.; Axelbaum, R.L. Soot Inception Limits in Laminar Diffusion Flames with Application to Oxy–Fuel Combustion. Combust. Flame 2008, 154, 546–556.
- Deligiannakis, Y.; Mantzanis, A.; Zindrou, A.; Smykala, S.; Solakidou, M. Control of Monomeric Vo’s versus Vo Clusters in ZrO2−x for Solar-Light H2 Production from H2O at High-Yield (millimoles gr−1 h−1). Sci. Rep. 2022, 12, 15132.
- Zindrou, A.; Belles, L.; Solakidou, M.; Boukos, N.; Deligiannakis, Y. Non-Graphitized Carbon/Cu2O/Cu0 Nanohybrids with Improved Stability and Enhanced Photocatalytic H2 Production. Sci. Rep. 2023, 13, 13999.
- Strobel, R.; Mädler, L.; Piacentini, M.; Maciejewski, M.; Baiker, A.; Pratsinis, S.E. Two-Nozzle Flame Synthesis of Pt/Ba/Al2O3 for NOx Storage. Chem. Mater. 2006, 18, 2532–2537.
- Minnermann, M.; Grossmann, H.K.; Pokhrel, S.; Thiel, K.; Hagelin-Weaver, H.; Bäumer, M.; Mädler, L. Double Flame Spray Pyrolysis as a Novel Technique to Synthesize Alumina-Supported Cobalt Fischer–Tropsch Catalysts. Catal. Today 2013, 214, 90–99.
- Høj, M.; Pham, D.K.; Brorson, M.; Mädler, L.; Jensen, A.D.; Grunwaldt, J.-D. Two-Nozzle Flame Spray Pyrolysis (FSP) Synthesis of CoMo/Al2O3 Hydrotreating Catalysts. Catal. Lett. 2013, 143, 386–394.
- Schubert, M.; Pokhrel, S.; Thomé, A.; Zielasek, V.; Gesing, T.M.; Roessner, F.; Mädler, L.; Bäumer, M. Highly Active Co–Al2O3 -Based Catalysts for CO2 Methanation with Very Low Platinum Promotion Prepared by Double Flame Spray Pyrolysis. Catal. Sci. Technol. 2016, 6, 7449–7460.
- Horlyck, J.; Pokhrel, S.; Lovell, E.; Bedford, N.M.; Mädler, L.; Amal, R.; Scott, J. Unifying Double Flame Spray Pyrolysis with Lanthanum Doping to Restrict Cobalt–Aluminate Formation in Co/Al2O3 Catalysts for the Dry Reforming of Methane. Catal. Sci. Technol. 2019, 9, 4970–4980.
- Stahl, J.; Ilsemann, J.; Pokhrel, S.; Schowalter, M.; Tessarek, C.; Rosenauer, A.; Eickhoff, M.; Bäumer, M.; Mädler, L. Comparing Co-catalytic Effects of ZrOx, SmOx, and Pt on COx Methanation over Co-based Catalysts Prepared by Double Flame Spray Pyrolysis. ChemCatChem 2021, 13, 2815–2831.
- Grossmann, H.K.; Grieb, T.; Meierhofer, F.; Hodapp, M.J.; Noriler, D.; Gröhn, A.; Meier, H.F.; Fritsching, U.; Wegner, K.; Mädler, L. Nanoscale Mixing during Double-Flame Spray Synthesis of Heterostructured Nanoparticles. J. Nanopart. Res. 2015, 17, 174.
- Solakidou, M.; Georgiou, Y.; Deligiannakis, Y. Double-Nozzle Flame Spray Pyrolysis as a Potent Technology to Engineer Noble Metal-TiO2 Nanophotocatalysts for Efficient H2 Production. Energies 2021, 14, 817.
- Gäßler, M.; Stahl, J.; Schowalter, M.; Pokhrel, S.; Rosenauer, A.; Mädler, L.; Güttel, R. The Impact of Support Material of Cobalt-Based Catalysts Prepared by Double Flame Spray Pyrolysis on CO2 Methanation Dynamics. ChemCatChem 2022, 14, e202200286.
- Psathas, P.; Zindrou, A.; Papachristodoulou, C.; Boukos, N.; Deligiannakis, Y. In Tandem Control of La-Doping and CuO-Heterojunction on SrTiO3 Perovskite by Double-Nozzle Flame Spray Pyrolysis: Selective H2 vs. CH4 Photocatalytic Production from H2O/CH3OH. Nanomaterials 2023, 13, 482.
- Tada, S.; Larmier, K.; Büchel, R.; Copéret, C. Methanol Synthesis via CO2 Hydrogenation over CuO–ZrO2 Prepared by Two-Nozzle Flame Spray Pyrolysis. Catal. Sci. Technol. 2018, 8, 2056–2060.
- Gockeln, M.; Pokhrel, S.; Meierhofer, F.; Glenneberg, J.; Schowalter, M.; Rosenauer, A.; Fritsching, U.; Busse, M.; Mädler, L.; Kun, R. Fabrication and Performance of Li4Ti5O12/C Li-Ion Battery Electrodes Using Combined Double Flame Spray Pyrolysis and Pressure-Based Lamination Technique. J. Power Sources 2018, 374, 97–106.
- Li, H.; Erinmwingbovo, C.; Birkenstock, J.; Schowalter, M.; Rosenauer, A.; La Mantia, F.; Mädler, L.; Pokhrel, S. Double Flame-Fabricated High-Performance AlPO4/LiMn2O4 Cathode Material for Li-Ion Batteries. ACS Appl. Energy Mater. 2021, 4, 4428–4443.
- Henning, D.F.; Merkl, P.; Yun, C.; Iovino, F.; Xie, L.; Mouzourakis, E.; Moularas, C.; Deligiannakis, Y.; Henriques-Normark, B.; Leifer, K.; et al. Luminescent CeO2:Eu3+ Nanocrystals for Robust in Situ H2O2 Real-Time Detection in Bacterial Cell Cultures. Biosens. Bioelectron. 2019, 132, 286–293.
- Lovell, E.C.; Großman, H.; Horlyck, J.; Scott, J.; Mädler, L.; Amal, R. Asymmetrical Double Flame Spray Pyrolysis-Designed SiO2/Ce0.7Zr0.3O2 for the Dry Reforming of Methane. ACS Appl. Mater. Interfaces 2019, 11, 25766–25777.
- Psathas, P.; Moularas, C.; Smykała, S.; Deligiannakis, Y. Highly Crystalline Nanosized NaTaO3/NiO Heterojunctions Engineered by Double-Nozzle Flame Spray Pyrolysis for Solar-to-H2 Conversion: Toward Industrial-Scale Synthesis. ACS Appl. Nano Mater. 2023, 6, 2658–2671.
