1. Historical, Botanical, and Taxonomic Characteristics of Mentha piperita
The genus
Mentha is a common taxonomic group in the Mediterranean flora
[1][2][3][4][5][6][16,19,24,31,41,42].
Mentha piperita (M. piperita) has been one of the most broadly used aromatic herbs for medicinal purposes since its discovery
[1][4][7][8][9][10][11][12][13][16,23,27,31,37,40,43,44,45]. The aromatic qualities of
M. piperita are connected with its hospitality in Greek mythology.
M. piperita was used as a room deodorizer for spreading over floors with the purpose of reducing the unpleasant smell of hard-packed soils
[7][23]. Pedanius Dioscorides (c. 40–90), a famous Greek physician and herbalist, first tried to determine the botanical classification of the plants in the genus
Mentha. Several centuries later, Carl Nilsson Linnaeus (1707–1778), in his work
Species Plantarum (1753), proposed a more formal classification of this genus
[9][10][37,40]. This genus was also described and named by Jussieu in 1789
[3][24].
The genus
Mentha was in a state of continuous change concerning its taxonomy, which is highly complex, and there is not always a consensus
[12][44]. This genus was redefined to comprise 18–20 worldwide species and 11 hybrids, which are divided into four sections, on the basis of the chromosome numbers, phylogenetic analysis, and major components of essential oil
[3][4][12][14][24,31,44,46]. Now the genus
Mentha can be classified into 42 species, 15 hybrids, and hundreds of subspecies, varieties, and cultivars, and it is often divided into five sections: Audibertia, Eriodontes, Mentha, Preslia, and Pulegium
[15][16][47,48]. The facets of the systematization of the genus
Mentha were not the subject of this re
svie
archw.
M. piperita has widespread overground and underground stolons. It has a square, erect, reddish, and branched stem that is relatively smooth or fringed, with a few spreading hairs, and is 2 or 3 feet in height. Its dark green leaves are arranged in opposite pairs, from oblong to lanceolate, approaching downy, and with sharp edges. The leaves are smooth or hairy on the underside, serrated, and borne on ciliated petioles. The flowers have prominent lip-like lower petals and are formed in false whorls, also known as verticillasters, with a purple color. Each flower has a two-lipped corolla with four lobes and its fruits have 1–4 seeds, covered with a stony layer. The whorls are few and lax, with the uppermost in a short, oblong, obtuse, reddish spike; and the lowermost remote, with the cymes shortly stalked. The bracts are subulate, the outer ones as long as the calyx. The pedicels are quite smooth. The calyxes are five-toothed, subulate, and erect. The corollas are four-cleft and tubular, with the broadest segment emarginated
[7][8][12][17][18][20,23,27,44,49].
2. Chemical Composition of Essential Oil
The whole dried leaves of
M. piperita contain not less than 12 mL/kg of essential oil. The cut dried leaves contain not less than 9 mL/kg of essential oil
[19][33].
Essential oils are secondary metabolites of aromatic plants with a complex composition consisting of a mixture of different chemical compounds, mainly oxygenated compounds and hydrocarbons, which are accumulated in the oil ducts, resin ducts, glands, or trichomes of plants
[6][8][14][20][27,42,46,50]. These compounds exert principal roles in the protection of plants against different assailants (insects, herbivores, and microorganisms). Moreover, these compounds have important pharmacological properties
[1][20][21][16,50,51].
The essential oil of
M. piperita is typically obtained through the steam distillation of aerial parts of the flowering plant
[1][6][8][14][21][22][16,27,28,42,46,51]. The raw essential oil of
M. piperita is usually subjected to further redistillation or rectification to remove unpleasant-smelling sulphur compounds, primarily dimethyl sulfide
[23][24][39,52]. This essential oil is a colorless to light yellow or greenish oily liquid with a characteristic fresh odor and taste
[21][25][51,53]. The principal components of the essential oil are
α-pinene,
β-pinene, sabinene, myrcene,
cis-ocimene,
β-caryophyllene, γ-terpinene, germacrene D, carvone (3.5%),
L–menthol (38–70%),
L–menthone (0.4–35%); isomenthone (1.5–10%), menthyl acetate (0–20%), eucalyptol (1,8-cineole) (0.4–12%), menthofuran (0.1–21%); limonene (0.6–4.5%), pulegone (traces–7%), and neoisomenthol
[1][2][8][19][21][24][26][9,16,19,27,33,51,52].
The essential oil yield is one of the most important quality parameters
[27][10]. As a rule, the essential oil yield ranges from 0.1 to 1% for air-dried parts
[28][8]. However, this range can be widened to 0.5–4%
[8][27][29][1,10,27]. In general, the yield and oil composition of
M. piperita depends on many factors, among which are the plant genotype; plant origin; soil and climatic conditions; harvest stage and techniques used; isolation methods; weed protection; pedoclimatic conditions; stress conditions; ontogenetic stage of the plant; photoperiod; organic farming conditions, including use of plant growth regulators and fertilizers; and analytical procedure used; etc.
[1][2][8][14][21][27][29][30][31][32][33][34][35][1,10,16,19,27,46,51,54,55,56,57,58,59]. For example, the yield of essential oil of
M. piperita of Egyptian origin was (0.40 ± 0.01) mL or (1.21 ± 0.08) mL of essential oil per 100 g of fresh weight and air-dried herb, respectively
[28][8]. The yield of essential oil of
M. piperita cultivated in Santa Catarina (Brazil) was 2.5 mL per 100 g of air-dried herb (2.5%
v/m)
[29][1].
In general, the quality of essential oil of
M. piperita mostly relies on the proportionate balance of its various components, especially menthol, menthone, menthofuran, and pulegone, and the essential oil’s quality is improved with higher menthol and menthone content and lower menthofuran and pulegone content
[31][34][55,58]. The essential oil of
M. piperita is characterized by the following quality indexes: consistency in the range of 0.900 to 0.916 g/cm
3, an optical versatility in the range of −10 to −30°, a refractive index in the range of 1.457–1.467, and an acidity number of not more than 1.4 mg KOH/g
[8][27]. Menthol has colorless, needle-shaped or prismatic crystals. It also has a strong odor and aromatic taste, followed by a sensation of cold when the air is drawn into the mouth. Menthol is supposed to be the component imparting a specific taste and odor to essential oil and the whole herb of
M. piperita [8][27].
According to the E/S/C/O/P Monographs
Menthae piperitae folium, the principal component is usually menthol in the form of (−)-menthol (30.0–55.0%), with smaller amounts of stereoisomers such as (+)-neomenthol (2.5–3.5%) and (+)-iso-menthol (approx. 3%)
[19][33].
RWe
searchers summarized some publications from the point of view of the chemical composition and origin of the essential oil of
M. piperita [2][8][35][36][37][38][39][40][41][42][43][44][45][46][47][19,27,59,60,61,62,63,64,65,66,67,68,69,70,71]. Sústriková and Šalamon, using gas chromatography–mass spectrometry (GC-MS) and GC-FID, determined the chemical composition of the essential oil of six samples of
M. piperita grown in different parts of Eastern Slovakia (1999–2001). The following contents of the constituents were revealed: menthol (38.3–69.1%), menthone (0.4–20.9%), menthyl acetate (3.5–4.5%), iso-menthone (0.8–8.8%), linalool (0.6–5.1%), and limonene (2.50–6.70%). The essential oil of several cultivars of
M. piperita had a very similar composition
[8][27]. The menthol content varied from 32.92% to 39.65%, 34.29% to 42.83%, and 22.56% to 32.77% during the crop growth in three cultivars, viz., Kukrail’, ‘CIM-Madhurus’, and ‘CIM-Indus’, of
M. piperita from Uttarakhand hills (India), which were analyzed using GC-FID
[35][59]. Kehili et al. (2020) determined the content of menthol (53.29%) and found a high content of oxygenated monoterpene compounds (92.75%) in the essential oil of
M. piperita grown in the Cherchell region (Tipaza, Algeria)
[36][60]. Twenty-two components, accounting for 95.6% of the total essential oil, were identified in the
M. piperita essential oil of Turkish origin using GC-MS. (+)-Menthol (38.1%), menthol (35.6%), neomenthol (6.7%), and eucalyptol (1,8-cineole) (3.6%) were the main components
[37][61]. Al-Mijali et al. (2022) determined the main compounds of the essential oil of Moroccan
M. piperita using GC-MS. Among them were menthone (29.24%), levomenthol (38.73%), menthol (2.71%), and eucalyptol (6.75%)
[38][62]. The chemical composition of the essential oil from
M. piperita, analyzed using GC/FID and GC-MS, showed that the main constituents were menthol (40.7%) and menthone (23.4%)
[39][63]. Using GC-MS, Gharib and Silva determined such components as
p-menthone (36.6%) and neo-menthol (40.5%)
[28][8]. It is worth mentioning that there are some problems with the identifications and nomenclature of the isomers of menthol in some publications related to
M. piperita as, for instance, (+)-menthol stated in paper
[37][61] is not specific for
M. piperita.
The essential oil composition differs depending on the ontogenetic stage of the plant and growing conditions. For instance, young plant oils generally contain a higher concentration of menthone (40–55%) than menthol (20–30%). Later, menthone is converted to menthol, and during the blooming period the content of the latter considerably increases. After the blooming period, in October or November, the menthone content considerably decreases. The toxic components, pulegone and menthofuran, are the most abundant in essential oil during the flowering stage, with menthofuran present mostly in flowers (even up to a content of 15%)
[24][40][41][52,64,65]. In other words,
M. piperita harvested at the flowering phase has a higher content of menthol compared to during the bud phase, when larger amounts of menthone are produced. In addition, stress conditions connected to light, temperature, and moisture status tend to promote the accumulation of pulegone and menthofuran. Moreover, menthofuran synthase expression (particularly overexpression), which leads to the production of high levels of menthofuran, results in a notable decrease in pulegone reductase message, accompanied by a decrease in reductase activity, with the consequence of increasing in the pulegone concentration in the essential oil
[34][58].
Using GC-MS, Nilo et al. detected 2.3% of menthyl acetate, 3.9% of eucalyptol (1,8-cineole), 10.5% of pulegone, 12.0% of menthofuran, 12.0% of isomenthol, 14.1% of menthol, and 31.4% of menthone. These authors considered menthofuran as an original marker to identify an authentic essential oil of
M. piperita [29][1]. In addition to this statement, menthofuran was found at low levels (traces) in the essential oil of
M. arvensis L.
[29][1], and was even absent in
M. spicata [1][26][37][9,16,61],
M. × gentilis L.
[26][9],
M. crispa L.
[26][9], and
M. gracilis R.Br.
[1][16]. Moreover, according to the requirements of the monograph of the European Pharmacopeia, using GC-FID analysis it should be present, but its content can range widely (1.0-8.0%)
[25][53]. However, according to the analysis performed in some publications, it was revealed that it could be non-identified in the essential oil of
M. piperita [2][28][8,19]. Finally, essential oil
M. spicata can be considered adulterant for peppermint
[22][28].
Menthol is a cyclic monoterpene alcohol with three asymmetric carbons
[34][45][48][58,69,72]. The main stereoisomer of menthol found in nature and in
M. piperita is levomenthol ((−)-menthol), with a configuration of (1R,3R,4S). Other isomers include isomenthol (0.3–0.4%), neomenthol (0.1–6.5%), and neoisomenthol (0.2–1.5%)
[49][50][51][38,73,74]. Menthol isomers, especially levomenthol, have a widely appreciated strong, cooling, and refreshing aroma and flavor. Its cooling effect is attributed to its ability to activate the cold-sensitive transient receptor potential cation channel (TRPM8)
[50][52][73,75]. (−)-Menthone, the second most abundant monoterpenoid in the essential oil of
M. piperita, is a critical intermediate in menthol biosynthesis and possesses a minty aroma and various acknowledged biological activities. (−)-Menthone is directly synthesized from (+)-pulegone through the reduction of the C2–C8 alkene double bond, catalyzed using (+)-pulegone reductase
[53][76]. (+)-Pulegone is the precursor of (−)-menthone, (−)-menthol, and (+)-menthofuran
[34][53][58,76]. Pulegone and its metabolite menthofuran can adversely affect the quality of the essential oil
[34][48][52][54][58,72,75,77]. These components are known to display hepatotoxicity and, therefore, their content is limited by some regulatory authorities. According to the EC1334/2008 regulations, a joint maximum daily intake of 0.1 mg/kg body weight was established for pulegone and menthofuran. The content of menthofuran in foods and beverages should not be more than 20 mg/kg, with the exceptions of mint/peppermint-flavored alcoholic beverages (100 mg/kg), mint/peppermint-flavored confectionery (200 mg/kg), and mint/peppermint-flavored chewing gum (1000 mg/kg). The content of pulegone in foods and beverages should not be more than 20 mg/kg, with the exceptions of mint/peppermint-flavored alcoholic beverages (100 mg/kg), mint/peppermint-flavored confectionery 100 mg/kg, intensely strong mint/peppermint-flavored confectionery (200 mg/kg), and mint/peppermint flavored chewing gum 30 mg/kg
[54][77]. Therefore, it is preferred that the essential oil of
M. piperita contain a high concentration of menthol, moderate levels of menthone, and low amounts of pulegone and menthofuran
[34][41][58,65].
The essential oil of
M. piperita is among the most important essential oils used in the pharmaceutical and cosmetics industries
[1][22][32][55][56][57][58][16,28,56,78,79,80,81]. Therefore, there is an International Standard Organization (ISO) standard ISO 856:2006 “Oil of peppermint (
Mentha ×
piperita L.)”
[59][82]. This essential oil is also included in many pharmacopeias
[9][23][37,39] and WHO monographs on selected medicinal plants
[60][83]. The United States Pharmacopeia–National Formulary (USP-NF)
[61][84] peppermint oil monograph does not set any specifications for the chromatographic profile of the essential oil of
M. piperita. The normative ranges for the main components of this essential oil are presented in
Table 1. The European Pharmacopeia regulates fewer components and generally permits wider ranges than the ISO
[25][53].
Table 1.
Chromatographic profiles of essential oil of
M. piperita
specified by international standards.
It should be mentioned that the chemical composition of the essential oil of
M. piperita and its yield depend on plant hormones, fertilizers, and abiotic and biotic factors. Among the abiotic factors are climate, soil, sunlight, salinity, etc.
[11][13][14][32][43,45,46,56]. For instance, salinity stress reduced the plant stem and root length, the internode length, the shoot and root fresh and dry weight, and the yield of essential oil, while there was an increase in the proline content, possibly because of a decrease in proline oxidase activity in saline conditions. Four levels of salinity were formed by four solutions containing 0, 50, 100, and 200 mmol/L of sodium chloride for irrigation plantings in a greenhouse
[11][43]. Among the consequences of soil moisture stress, at which the plants grew at 75% and 50% FC, there was a decrease in the fresh and dry weight of leaves, leaf width and length, and shoot length compared to the control plants; however, there was an increase in the TPC and TFC
[13][45].
Plant hormones had an impact on the percentage of particular compounds in the composition of volatile organic substances. Some compounds or their combination were used for the studies (6-benzyloadeninopurine + indolyl-3-acetic acid (MS-1), 2-isopentinloadenine + indolyl-3-butyric acid (MS-2), indolyl-3-butyric acid (MS-3), 6-benzyloadeninopurine (MS-4), along with a control medium without the addition of plant hormones (MS-K). The menthofuranolactone share was significantly increased in sample MS-2 while the limonene and eucalyptol mixture share was decreased. It should be noted that the authors used the special chemotype of
M. piperita, which was characterized by high amounts of limonene, eucalyptol (1,8-cineole), and menthofuranolactone. In their studies, there was no menthol or menthone. These authors explained the lack of most characteristic components of essential oil of
M. piperita (menthone, menthol, and their derivatives) as a result of the very early stage of the plant development in the in vitro culture
[32][56]. In general, this partly conforms to the statement that the menthol content increases with the leaf and oil gland’s maturity
[34][58]. Menthol and menthone contents were different in response to the different fertilization treatments. The highest percentage of menthol was obtained in response to the treatment of the combination of organic fertilizers (poultry manure + cattle manure) and effective microorganisms (41.5%). Contrastingly, the lowest percentage of menthol was obtained in the plants fertilized with cattle and poultry manures as organic fertilizers (39.1% and 39.6%, respectively). However, this content was higher compared to the control (38.2%)
[33][57].
The studies with plant growth regulators and fertilizers indicated that farming conditions should be standardized for growing M. piperita for the pharmaceutical and food industries.
3. Chemical Composition of Extracts of Mentha piperita
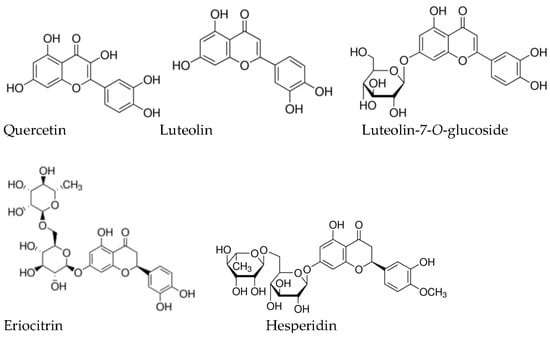
The hydroalcoholic and other extracts of
M. piperita contain flavonoids from different groups (epicatechin; rutin; quercetin; naringenin; naringenin-7-
O-glucoside; kaemferol; hesperidin (3′,5,7-trihydroxy-4′-methoxyflavanone 7-rutinoside); luteolin (3′,4′,5,7-tetrahydroxyflavone); luteolin-7-
O-glucoside; eriocitrin ((
S)-3′,4′,5,7-tetrahydroxyflavanone-7-[6-
O-(α-L-rhamnopyranosyl)-β-
d-glucopyranosid], apigenin, etc.)
[6][58][62][63][64][65][35,42,81,85,86,87]; and phenolic acids, including cafeic acid
[63][64][65][85,86,87], rosmarinic acid
[64][65][66][67][17,86,87,88], trierpenoids (ursolic acid), etc.
[63][85]. Their amounts in extracts depend first of all on the solvent chosen for extraction
[64][86]. The chemical structure of main flavonoids of
M. piperira is provided on
Figure 1.
Figure 1.
The chemical structure of main flavonoids of
M. piperira
.
The TFC content was in the range of 29.2 to 53.2 mg of catechin equivalents in one gram of dry mass depending on conditions of the vacuum drying process of
M. piperita. Therefore, processes of drying are important for the pharmaceutical industry, as they have an impact on the content of biologically active substances
[68][21]. The reflux extraction of
M. piperita with methanol in 45 min at a temperature of 70 °C resulted in an extraction yield of 0.9%. The obtained extract, evaluated using high-performance liquid chromatography (HPLC), indicated that rosmarinic acid was present at a high concentration (1.9 mg/mL in the extract)
[66][17].
Eriocitrin (2.7–182.6 mg/g), luteolin-7-
O-glucoside (3.2–90.8 mg/g), and rosmarinic acid (10.6–176.8 mg/g) were the most abundant components identified in the leaves of
M. piperita, whilst naringenin-7-
O-glucoside was the least abundant component found only in the ethylacetate extract (0.8 mg/g). Eriocitrin was found in the largest and smallest amounts in the acetonitrile and aqueous extracts, respectively (182.6 and 2.7 mg/g). Luteolin-7-
O-glucoside was found in the largest and smallest amounts in the methanolic and aqueous extracts, respectively (90.8 and 3.2 mg/g). Among the identified compounds were caffeic acid (1.4–1.8 mg/g), hesperidin (3.8–32.6 mg/g), eriodictyol, and luteolin. Moreover, these studies showed that methanol and water were the solvents which extracted the most and the least active substances, respectively. In addition, the ethylacetate, acetonitrile, and aqueous extracts were the most active in such tests as the iron (III) reduction and DPPH test, ABTS test, and hydroxyl free radical scavenging properties. The aqueous and dichloromethane extracts had the most active antioxidant activity in the β-carotene-linoleic acid bleaching inhibition assay. The light petroleum, methanolic, and aqueous extracts showed moderate iron (II) chelating activity
[64][86]. According to the requirements of the Pharmacopeias, it is necessary to avoid acetonitrile, dichlormethane, and methanol in the manufacture of medicinal products
[25][69][53,89]. Therefore, these studies have more of a theoretical meaning concerning the dependence of the amounts of extractive substances on the nature of the solvents used.
In another study, it was reported that the aqueous extracts (teas) of
M. piperita contain phenolic compounds such as hesperidin (18.64 ± 0.16 mg/100 mL of the tea), rosmarinic acid (4.91 ± 0.18 mg/100 mL of the tea), eriocitrin (1.55 ± 0.08 mg/100 mL of the tea), chlorogenic acid (traces), and luteolin 7-
O-β-glucuronide 3.68 ± 0.01 mg/100 mL
[29][1]. This
res
earch tudy was in line with Dorman et al.
[64][86]. Similar results were obtained for
M. piperita of Greek origin. As a result of the maceration of
M. piperita using a combined solvent (90% of water and 10 % of ethanol), the following phenolics were extracted: luteolin 27.64 ± 0.32 ppm, quercetin 6.25 ± 0.46 ppm, apigenin 0.10 ± 0.03 ppm, rutin 8.32 ± 0.59 ppm, eriodictyol 32.23 ± 0.75 ppm, rosmarinic acid 6.03 ± 0.52 ppm,
p-coumaric acid 35.21 ± 1.02 ppm, ferulic acid 31.92 ± 0.83 ppm, caffeic acid 116.89 ± 0.28 ppm, hydroxybenzoic acid 30.86 ± 0.92 ppm, and benzoic acid 41.92 ± 0.61 ppm
[65][87].
Athanasiadis et al. confirmed the presence of phenolics such as caffeic acid, eriocitrin, luteolin-7-
O-rutinoside, luteolin-7-
O-glucoronide, hesperidin, rosmarinic acid, and luteolin derivate. This fact has been observed in many scientific publications
[65][70][71][87,90,91]. Sinapic acid, gallic acid, syringic acid, kaemferol, quercetin, epigallocatechin, and quercetin-rhamno-di-hexoside were found in
M. piperita of Pakistani origin
[71][91]. Athanasiadis et al. also revealed that adding
β-cyclodextrin increased the yield of some polyphenols of
M. piperita in aqueous extracts compared to aqueous extracts without
β-cyclodextrin. Moreover, these authors compared the
β-cyclodextrin aqueous extracts to the other aqueous and ethanolic extracts. It was confirmed that there were three principal components in all the extracts, namely, eriocitrin, luteolin-7-
O-rutinoside, and rosmarinic acid. In general, ethanol extracted the phenolics the best. However, the
β-cyclodextrin aqueous extract was richer in some phenolics compared to the aqueous extract
[70][90]. Chlorogenic acid, rosmarinic acid, epicatechin, quercetin, gallic acid, epigallocatechin, syringic acid, kaempferol, and caffeic acid were identified in the methanolic extract and aqueous extracts of
M. piperita [72][92].
Therefore, caffeic acid, rosmarinic acid, eriocitrin, luteolin-7-
O-rutinoside, luteolin-7-
O-glucoronide, and hesperidin can be used as the markers for the quantitative and qualitative analysis of extracts of
M. piperita using the spectrophotometric method for measuring TPC and TFC, high-performance thin-layer chromatography, and HPLC as it was previously published for the extracts of
Satureja montana L.
[73][4]. Moreover, some authors attribute anti-inflammatory and antiviral activity of extracts of
M. piperita to rosmarinic acid, luteolin-7-
O-rutinoside, and hesperidin
[74][12].
Among the main pharmacological properties of
M. piperita extracts and essential oil are antioxidant
[1][26][74][9,12,16], anti-inflammatory
[2][75][19,93], immunomodulatory
[63][85], hepatorenalprotective
[76][94], and antimicrobial activities
[17][74][77][78][7,12,20,95].
4. Antioxidant Activity
M. piperita extracts and essential oil possess significant antioxidant potential and related biological activities, among which is anti-inflammatory activity
[1][2][26][36][55][64][66][74][76][79][9,12,16,17,19,60,78,86,94,96].
All of the analyzed publications provide essential oil and extracts of M. piperita as DPPH•-, ABTS•+, and OH•-scavengers. During in vivo tests there are pro- and antioxidant effects of essential oil depending on the concentration used. This topic needs further study.
5. Anti-Inflammatory Activity
Studies of plant-based extracts and essential oils as anti-inflammatory preparations arouse a lot of interest, considering the adverse reactions of non-steroidal anti-inflammatory medicinal products [1][2][36][55][57][74][80][81][82][83][84][85][86][12,16,19,60,78,80,100,101,102,103,104,105,106].
Inflammation is a natural complex biological response to pathogens, damaged cells (injury), or irritants, which involves immune cells, blood vessels, excessive phagocyte activation, molecular mediators, and the production of hydroxyl, superoxide anions, and non-free-radical species such as H2O2 [55][74][86][12,78,106]. In addition, inflammation is usually connected to edema and pain at the place of an injury or wound [60,101]. Moreover, unrestricted inflammation and chronic inflammation can lead to various diseases, such as rheumatoid arthritis, asthma, cancer, and CVDs, or even to the loss of functions or tissue structure [2][27][36][74][86][10,12,19,60,106]. M. piperita extracts and essential oil exhibit anti-inflammatory activity, which can help to reduce inflammation and prevent the development of chronic diseases [2][36][74][82][12,19,60,102].
Researchers suggest that essential oil and extracts of M. piperita are promising herbal preparations due to their anti-inflammatory and wound-healing properties for use in skin care products, care products for oral cavities, and even in preparations for systemic administration in combination with synthetic anti-inflammatory or antischistomatosis drug substances in tablets and capsules.
6. Antimicrobial Activity
M. piperita extracts and essential oil were also reported to exhibit antimicrobial and antiviral activity [46][74][83][84][87][88][12,13,25,70,103,104]. This activity can help to prevent or treat infections at least on the level of complementary therapy. Antimicrobial properties make M. Piperita extracts and essential oil promising candidates for developing new antimicrobial drug products.
7. Anticancer Activity
Searching for alternative and complementary medical products from plants is a topical issue for the treatment of cancer. Therefore, there are many studies directed at the study of different plant resources in the search for anticancer active substances or active substances for cancer prevention [2][20][55][58][89][90][91][92][93][94][95][19,50,78,81,108,109,110,111,112,113,114]. The results of numerous studies showed that the essential oils and different extracts of several species of Lamiaceae can inhibit the growth of cancer cells, indicating the potential effects for the complementary treatment of cancer [2][55][91][19,78,110]. Among the advantages of essential oils in the fight against cancer is their lipophilic character, which facilitates their crossing through cell membranes and reaching the inner side of the cell [20][21][90][91][50,51,109,110].
Summing up, preparations of M. piperita possess anticancer properties of different extents and could be regarded as the basis for further laboratory research concerning the potential use of M. piperita preparations or active substances for anticancer treatments.
8. Cardiovascular Diseases
M. piperita preparations showed hypotensive, vasorelaxant, and antiplatelet activities [94][96][97][98][99][100][101][26,29,36,113,115,116,117]. The inflammatory activity of M. piperita may be responsible for diminishing risks of CVDs because patients with these diseases have high inflammation [97][98][29,36].
Summing up, preparations of M. piperita have cardiprotective effects due to their antioxidant activities [99][115], increased high-density lipoprotein cholesterol levels [94][99][113,115], and a reduction in arterial smooth muscle tonicity [100][116], etc. Therefore, preparations of M. piperita can be components of medicinal products with cardioprotective activity for the decrease in the blood pressure or prevention of acute myocardial infarction.
9. Other Activities (Gastrointestinal Effects, Protective Activity, Larvicidal, and Repellent Activities) and Other Applications
M. piperita preparations were reported to have neuroprotective, hepatorenalprotective, and gastrointestinal effects such as anti-spasmodic and anti-ulcer activity, and antispasmodic activity of smooth muscles [58][76][102][103][104][105][106][107][108][109][81,94,118,119,120,121,122,123,124,125].