Acne vulgaris stands out as the most prevalent skin disorder among teenagers and young adults, causing physical discomfort and considerable economic and psychological burdens on individuals and society. Chemical peeling is a skin resurfacing technique designed to rebuild healthy skin using exfoliating substances, a simple and affordable process with various dermatological uses. Chemical peels, classified as superficial, medium, and deep, have been utilized for acne vulgaris and multiple other skin issues. In these chemical peels, a diverse range of chemical substances is employed, each with its unique mode of action. Among these, α-hydroxy and β-hydroxy acids have gathered attention for their efficacy in reducing acne lesions and enhancing overall skin appearance. Acids, such as salicylic acid, glycolic acid, or lactic acid, are commonly used in chemical peels due to their exfoliating and sebum-regulating properties. Despite the widespread use of these acids, there exists a lack of consensus regarding the most effective acid type and concentration for treating acne-prone skin.
1. Introduction
Acne vulgaris is the most common skin disease affecting adolescents and young adults. Although it is seen as a mild skin condition, it places a significant economic and psychological cost on individuals and society. Anxiety, depression, and poor self-esteem are common among patients suffering from acne vulgaris, negatively impacting their quality of life
[1,2][1][2].
Propionibacterium acnes colonization, inflammation, enhanced keratinocyte proliferation in the follicular infundibulum, and hypersensitive androgen-sensitive sebaceous glands are some of the components involved in the pathogenesis of acne. It is a disease of the pilosebaceous units, clinically characterized by seborrhea, comedones, papules, pustules, nodules, cysts, and, in some cases, scarring
[3,4][3][4].
Chemical peeling is a skin resurfacing technique frequently used for cosmetic and face rejuvenation. A new epidermal layer of the dermal tissues regenerates because the skin suffers controllable damage under the influence of a chemical agent. The amount of acid employed, the kind of vehicle, the amount of buffering, and the length of skin contact all affect how deep the peeling is. Chemical peels are divided into superficial (epidermis-papillary dermis), medium (papillary to upper reticular dermis), and deep peels (mid-reticular dermis), depending on how deeply they penetrate the skin
[14][5].
Medium-depth and superficial chemical peels are used frequently to treat acne vulgaris, as there are various substances and combinations of peels (alpha-hydroxy acids, beta-hydroxy acids, polyhydroxy acids, and bionic acids). Chemical peels can decrease sebum production and have antibacterial, anti-inflammatory, keratolytic, and comedolytic properties. Consequently, chemical peels have been utilized widely to treat acne vulgaris as an additional or maintenance therapy
[15,16][6][7].
Although chemical peels are commonly used to treat acne, there is a lack of high-quality studies to support their efficacy and safety. Some small studies have shown promising results. Thus, there is a need for larger, randomized, controlled trials to understand better the effectiveness of chemical peels in treating acne. Additionally, there is a need for more research to determine the optimal type of chemical peel and concentration of the active ingredient for treating acne. The available studies have used a variety of chemical peels, including salicylic acid, glycolic acid, and trichloroacetic acid, and the optimal concentration and duration of treatment may vary depending on the individual and the severity of the acne. Furthermore, there is a need for more research to determine the safety of chemical peels in treating acne, particularly in individuals with darker skin tones who may be at a higher risk of hyperpigmentation or scarring
[17,18][8][9].
32. Organic Acids Used in Chemical Peels for Acne Treatment
3.1. Classification of the Chemical Peel Agents Based on the Depth of Penetration
2.1. Classification of the Chemical Peel Agents Based on the Depth of Penetration
Chemical peeling agents can be classified based on the depth of penetration into the skin; there are three main types of chemical peels based on the depth of penetration: superficial, medium, and deep. The characteristics of these types of peeling are presented below.
32.1.1. Superficial Peels
Superficial peels penetrate only the outermost layer of the skin (epidermis) and are used to improve skin texture, reduce fine lines and wrinkles, and treat mild acne. These peels involve the application of a chemical solution to the skin’s surface, which helps to exfoliate dead skin cells, unclog pores, and stimulate the growth of new, healthier skin. These peels use weak acids like α-hydroxy acids (AHAs), such as glycolic acid and lactic acid, and β-hydroxy acids (BHAs), such as salicylic acid, to exfoliate the skin. Superficial peels have minimal downtime and are generally safe for all skin types
[19,20][10][11].
32.1.2. Medium-Depth Peels
Medium-depth peels penetrate the epidermis and upper layers of the dermis and treat more severe skin concerns such as sun damage, fine lines and wrinkles, and acne scars. Medium chemical peels are a more aggressive form of chemical peel compared to superficial peels. They are sometimes used to manage acne vulgaris, particularly for individuals with more severe or stubborn acne and acne scarring. These peels use trichloroacetic acid (TCA), Jessner’s solution, or a combination of TCA and glycolic acid to exfoliate the skin. Medium-depth peels have a more extended downtime and may require pain relief treatment during the procedure
[21,22][12][13].
32.1.3. Deep Peels
Deep peels penetrate the deeper layers of the dermis and are used to treat severe skin concerns such as deep wrinkles, scarring, and severe sun damage. These peels use phenol, TCA, or a combination of TCA and phenol to exfoliate the skin. Deep peels have the most extended downtime and are generally only recommended for fair-skinned individuals due to the risk of hypopigmentation or other adverse effects. Deep peels can also determine the appearance of second-degree burns; applying the solution is preferred based on phenols in this condition. The treatment must be used only once and with the utmost caution due to adverse reactions (the appearance of spots that will not disappear); this method is recommended only for people with severe dermatological problems and should only be performed by a dermatologist
[22,23][13][14].
32.1.4. Peeling Agents
Among the factors that influence the penetration capacity of the tissues by the chemical substances are the pH of the peeling agent, the number of layers applied, and the time given for the action before the substance is neutralized. The penetration depth is determined by the peeling agent’s nature, concentration, and pH.
43. Classification of the Chemical Peel Agents Based on Chemical Structure
4.1. Aliphatic Carboxylic Acids
3.1. Aliphatic Carboxylic Acids
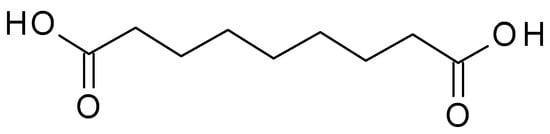
43.1.1. Azelaic Acid
Azelaic acid (C
9H
16O
4) (
Figure 1) is a naturally occurring dicarboxylic acid found in whole grain cereals, wheat, rye, and barley. It is a straight-chain, saturated fatty acid with a carbon chain length of nine carbon atoms. It is a white crystalline powder with a slight odour, sparingly soluble in water but more soluble in organic solvents. The pKa value of azelaic acid is around 4.55, indicating that it exists mainly in its non-ionized form at physiological pH. The pH of azelaic acid formulations typically ranges from 4 to 5, which is close to the skin’s natural pH level
[25][15].
Figure 1. Azelaic acid chemical structure and the IUPAC name: nonanedioic acid.
4.1.2. Thricloracetic Acid (TCA)
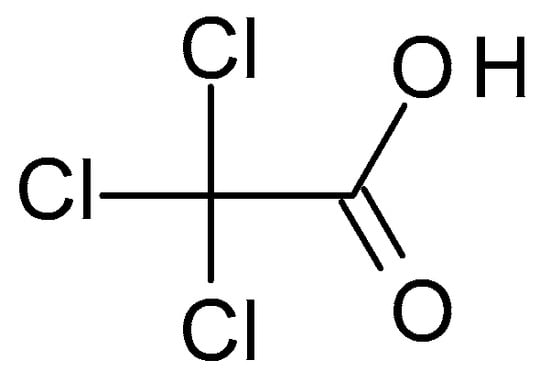
TCA (C Azelaic acid chemical structure and the IUPAC name: nonanedioic acid.
3.1.2. Thricloracetic Acid (TCA)
TCA (C
232Figure 2) is a structural analogue of acetic acid in which the hydrogen atoms in the methyl group have been replaced with chlorine atoms. It is a white crystalline solid highly soluble in water and alcohol. TCA has a pH of around 1.5 to 2, which makes it highly acidic [28].
) is a structural analogue of acetic acid in which the hydrogen atoms in the methyl group have been replaced with chlorine atoms. It is a white crystalline solid highly soluble in water and alcohol. TCA has a pH of around 1.5 to 2, which makes it highly acidic [16].
Figure 2. Thricloracetic acid chemical structure and the IUPAC name: 2,2,2-trichloroacetic acid.
4.2. Aliphatic Hydroxycarboxylic Acids
3.2. Aliphatic Hydroxycarboxylic Acids
4.2.1. Glycolic Acid
3.2.1. Glycolic Acid
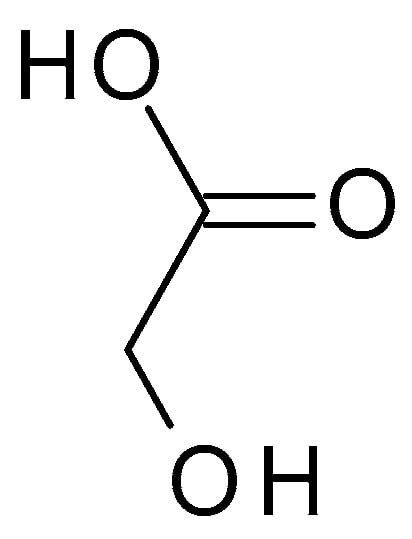
Glycolic acid (C
2H
4O
3) (
Figure 3) is the simplest AHA with the smallest molecular weight and size. It is a colourless, odourless, crystalline substance highly soluble in water. This AHA can be isolated from natural sources, such as sugarcane, sugar beets, pineapple, or cantaloupe. Glycolic acid has a pH of around 3.5 to 4, which makes it mildly acidic [32].) is the simplest AHA with the smallest molecular weight and size. It is a colourless, odourless, crystalline substance highly soluble in water. This AHA can be isolated from natural sources, such as sugarcane, sugar beets, pineapple, or cantaloupe. Glycolic acid has a pH of around 3.5 to 4, which makes it mildly acidic [17].
Figure 3. Glycolic acid chemical structure and the IUPAC name: 2-hydroxyacetic acid.
4.2.2. Lactic Acid
3.2.2. Lactic Acid
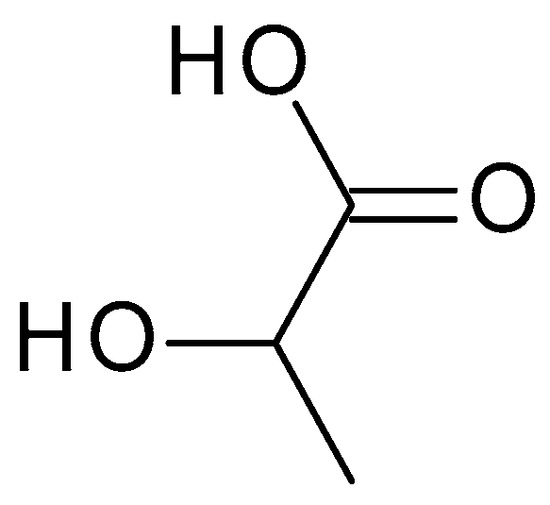
Lactic acid (C
3H
6O
3) (
Figure 4) is structurally similar to glycolic acid, having an additional methyl group. Lactic acid is produced via the bacterial fermentation of milk or carbohydrates. It is a colourless or yellowish liquid with a slightly acidic odour. The pH of lactic acid is around 3.5 to 4, which makes it mildly acidic [36].) is structurally similar to glycolic acid, having an additional methyl group. Lactic acid is produced via the bacterial fermentation of milk or carbohydrates. It is a colourless or yellowish liquid with a slightly acidic odour. The pH of lactic acid is around 3.5 to 4, which makes it mildly acidic [18].
Figure 4. Lactic acid chemical structure and the IUPAC name: 2-hydroxypropanoic acid.
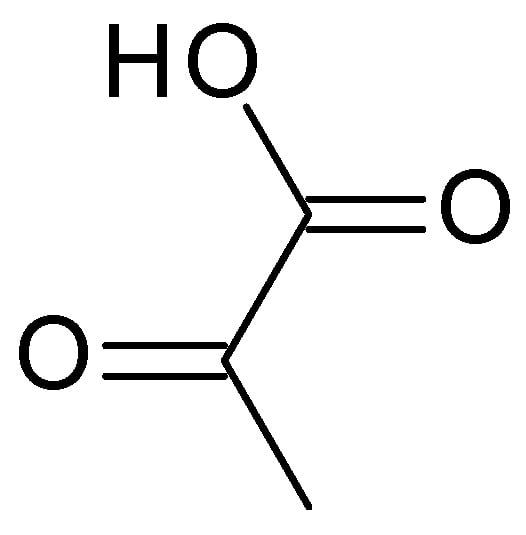
4.2.3. Pyruvic Acid
3.2.3. Pyruvic Acid
Pyruvic acid (C
3H
4O
3) (
Figure 5) is the simplest α-keto acid, with a functional ketone and carboxylic acid group. Pyruvic acid is a colourless to pale yellow liquid with a distinct odour, highly soluble in water and alcohol. Typically, it has a pH range of 2–3 when dissolved in water. It plays a crucial role in various biological processes, including energy metabolism and the synthesis of amino acids [39].) is the simplest α-keto acid, with a functional ketone and carboxylic acid group. Pyruvic acid is a colourless to pale yellow liquid with a distinct odour, highly soluble in water and alcohol. Typically, it has a pH range of 2–3 when dissolved in water. It plays a crucial role in various biological processes, including energy metabolism and the synthesis of amino acids [19].
Figure 5. Pyruvic acid chemical structure.
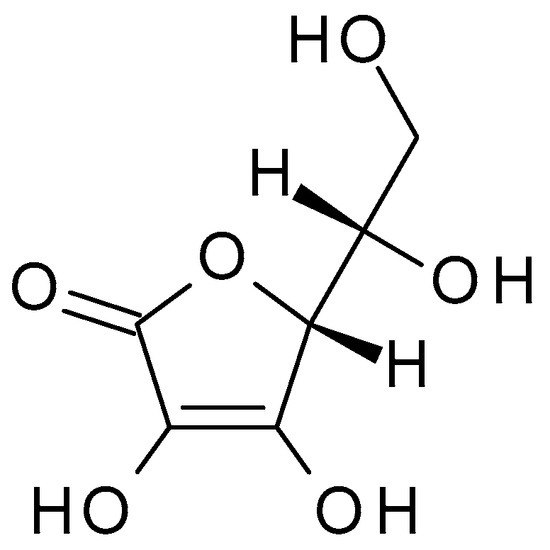
4.2.4. Ascorbic Acid
3.2.4. Ascorbic Acid
Ascorbic acid (C
6H
8O
6) (Vitamin C) (
Figure 6) is a water-soluble vitamin that plays a crucial role in various bodily functions. Ascorbic acid is susceptible to degradation, particularly under certain conditions such as exposure to heat, light, and oxygen. It is relatively unstable and can undergo oxidation, leading to the formation of dehydroascorbic acid. Ascorbic acid is an organic acid with a relatively low pKa (4.2); it can act as a weak acid, donating hydrogen ions in solution and lowering the pH. It is a good reducing agent and undergoes reversible oxidation-reduction reactions; it can donate electrons, making it an effective antioxidant that can scavenge and neutralize free radicals. Free radicals can contribute to inflammation and damage the skin’s barrier, worsening acne symptoms. It is an essential nutrient; the human body cannot synthesize it, and it must be obtained via diet or supplements. Ascorbic acid helps protect the skin against free radical damage caused by environmental factors such as UV radiation, pollution, and oxidative stress; this antioxidant activity can help prevent premature ageing and support overall skin health [41].) is a water-soluble vitamin that plays a crucial role in various bodily functions. Ascorbic acid is susceptible to degradation, particularly under certain conditions such as exposure to heat, light, and oxygen. It is relatively unstable and can undergo oxidation, leading to the formation of dehydroascorbic acid. Ascorbic acid is an organic acid with a relatively low pKa (4.2); it can act as a weak acid, donating hydrogen ions in solution and lowering the pH. It is a good reducing agent and undergoes reversible oxidation-reduction reactions; it can donate electrons, making it an effective antioxidant that can scavenge and neutralize free radicals. Free radicals can contribute to inflammation and damage the skin’s barrier, worsening acne symptoms. It is an essential nutrient; the human body cannot synthesize it, and it must be obtained via diet or supplements. Ascorbic acid helps protect the skin against free radical damage caused by environmental factors such as UV radiation, pollution, and oxidative stress; this antioxidant activity can help prevent premature ageing and support overall skin health [20].
Figure 6. Ascorbic acid chemical structure and the IUPAC name: ((2
R)-2-[(1
S)-1,2-dihydroxyethyl]-3,4-dihydroxy-2
H-furan-5-one).
4.2.5. Citric Acid
3.2.5. Citric Acid
Citric acid (C
6H
8O
7) (2-hydroxypropane-1,2,3-tricarboxylic acid) (
Figure 7) is a weak organic acid with three carboxylic acid functional groups naturally found in citrus fruits. Citric acid is a white crystalline powder with a tart, acidic taste, which contributes to the sourness of citrus fruits; it is highly soluble in water. Citric acid has a pH value of approximately 2.2 to 2.5 in a 1% aqueous solution. Citric acid has antioxidant properties, donating electrons to neutralize free radicals and prevent their harmful effects [43].) is a weak organic acid with three carboxylic acid functional groups naturally found in citrus fruits. Citric acid is a white crystalline powder with a tart, acidic taste, which contributes to the sourness of citrus fruits; it is highly soluble in water. Citric acid has a pH value of approximately 2.2 to 2.5 in a 1% aqueous solution. Citric acid has antioxidant properties, donating electrons to neutralize free radicals and prevent their harmful effects [21].
Figure 7. Citric acid chemical structure and the IUPAC name: 2-hydroxypropane-1,2,3-tricarboxylic acid.
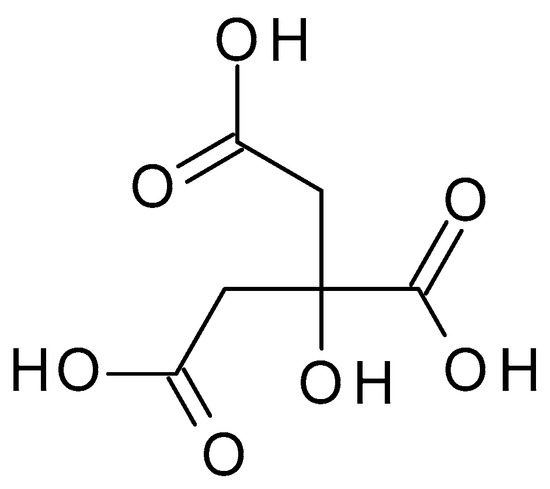
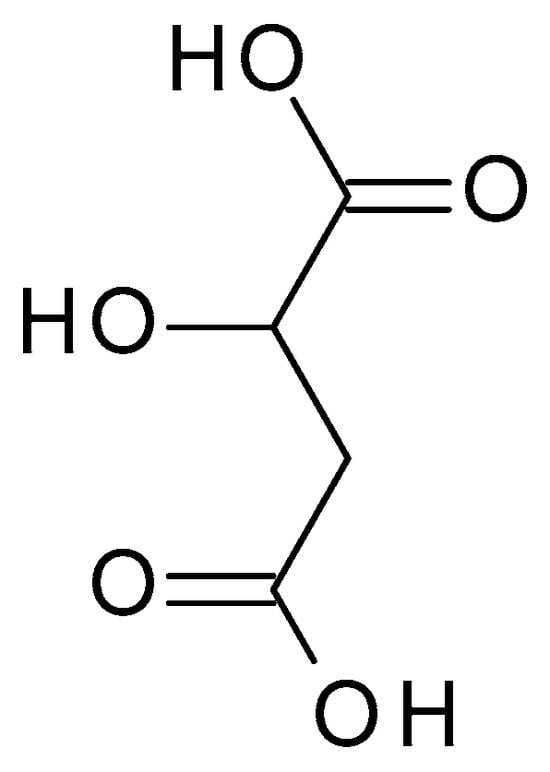
4.2.6. Malic Acid
3.2.6. Malic Acid
Malic acid (C
4H
6O
5) (
Figure 8) is a dicarboxylic acid occurring naturally in various fruits, such as apples, grapes, and citrus. Malic acid is a white crystalline powder with a pleasantly sour taste and is highly soluble in water. Malic acid has a pH value of approximately 2.83 in aqueous solution [45].) is a dicarboxylic acid occurring naturally in various fruits, such as apples, grapes, and citrus. Malic acid is a white crystalline powder with a pleasantly sour taste and is highly soluble in water. Malic acid has a pH value of approximately 2.83 in aqueous solution [22].
Figure 8. Malic acid chemical structure and the IUPAC name: 2-hydroxybutanedioic acid.
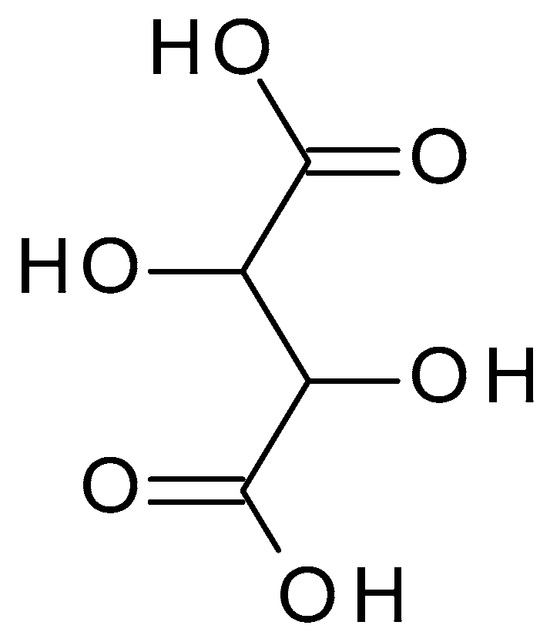
4.2.7. Tartaric Acid
3.2.7. Tartaric Acid
Tartaric acid (C
4H
6O
6) (2,3-dihydroxybutanedioic acid) (
Figure 9) is a dicarboxylic acid occurring naturally in various fruits, such as grapes or bananas. Tartaric acid is a white crystalline powder with a sour taste; it is highly soluble in water. It has a pH value of approximately 2.2 to 3.0 in aqueous solution [47].) is a dicarboxylic acid occurring naturally in various fruits, such as grapes or bananas. Tartaric acid is a white crystalline powder with a sour taste; it is highly soluble in water. It has a pH value of approximately 2.2 to 3.0 in aqueous solution [23].
Figure 9. Tartaric acid chemical structure and the IUPAC name: 2,3-dihydroxybutanedioic acid.
4.3. Aromatic Hydroxycarboxylic Acids
3.3. Aromatic Hydroxycarboxylic Acids
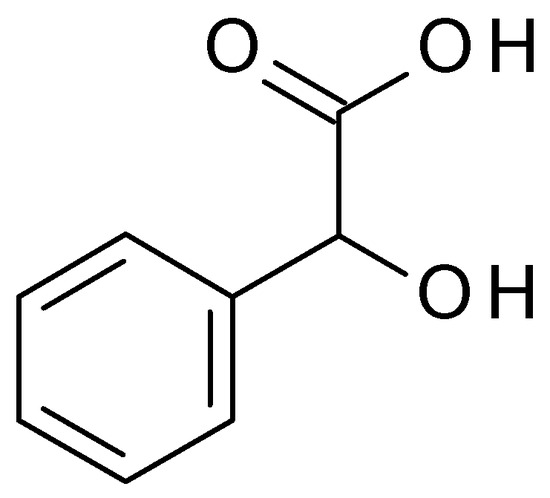
4.3.1. Mandelic Acid
3.3.1. Mandelic Acid
Mandelic acid (C
8H
8O
3) (2-hydroxy-2-phenylacetic acid) (
Figure 10) is an AHA derived from bitter almonds. Mandelic acid is a white crystalline powder, highly soluble in water. Mandelic acid has a pH value of approximately 2.8 to 3.2 in aqueous solution [48].) is an AHA derived from bitter almonds. Mandelic acid is a white crystalline powder, highly soluble in water. Mandelic acid has a pH value of approximately 2.8 to 3.2 in aqueous solution [24].
Figure 10. Mandelic acid chemical structure and the IUPAC name: 2-hydroxy-2-phenylacetic acid.
4.3.2. Salicylic Acid
3.3.2. Salicylic Acid
Salicylic acid (C