Nerve growth factor (NGF) plays a crucial immunomodulatory role and increased levels are found in numerous tissues during autoimmune states. NGF directly modulates innate and adaptive immune responses of B and T cells and causes the release of neuropeptides and neurotransmitters controlling the immune system activation in inflamed tissues. Evidence suggests that NGF is involved in the pathogenesis of numerous immune diseases including autoimmune thyroiditis, chronic arthritis, multiple sclerosis, systemic lupus erythematosus, mastocytosis, and chronic granulomatous disease. Furthermore, as NGF levels have been linked to disease severity, it could be considered an optimal early biomarker to identify therapeutic approach efficacy. In conclusion, by gaining insights into how these molecules function and which cells they interact with, future studies can devise targeted therapies to address various neurological, immunological, and other disorders more effectively. This knowledge may pave the way for innovative treatments based on NGF manipulation aimed at improving the quality of life for individuals affected by diseases involving neurotrophins.
- arthritis
- autoimmunity
- mastocytosis
- multiple sclerosis
- neurotrophins
- autoimmune thyroiditis
- NGF
- systemic lupus erythematosus
1. NGF and Autoimmune Diseases
| Disease | Clinical Manifestations | Role of NGF | Ref. |
|---|---|---|---|
| Autoimmune thyroiditis | Hyperthyroidism (e.g., Graves’ Disease) and hypothyroidism (e.g., Hashimoto Thyroiditis) with a variety of associated symptoms such as humoral psychotic symptoms, intolerance to cold/hot temperature, weight changes, difficulty in concentration, and eye disorders. |
|
[122,123,124,125,126,127][2][3][4][5][6][7] |
| Chronic arthritis | Chronic inflammation and damage to joints and surrounding tissues, chronic pain and reduced quality of life, asthenia, psychological and social symptoms |
|
[13,47,128,129,130,131,132,][11][12][133,13134,135,][14136,137][8][9][10][15][16][17][18][19] |
| Multiple sclerosis | Periods of relative well-being alternate with episodes of symptom deterioration with gradual worsening over time. Tingling, numbness, pain, burning, itching, reduced sense of touch, loss of strength or dexterity in a limb, vision disorders. |
|
[86,89,90,91,108,][23][24][109,25110,111,][26112,113][20][21][2292,93,94,95,96,97,][2798,][2899,29][30][31]100,[101,102,32][103,104,]105,106,[107,33][34][35][36][37][38][39][40][41][42][43][44][45] |
| Chronic granulomatous disease | Recurrent infections, multiorgan granulomatous lesions, abscesses, lymphadenitis, hypergammaglobulinemia, anemia. |
|
[46,138,139,140,141,142][46][47][48][49][50][51] |
| Systemic lupus erythematosus | Fever, fatigue, butterfly rash on the face, erythematous lesions in areas exposed to the sun, hair loss, purple-red lesions of the hard and nasal palate, cutaneous vasculitis, and multiorgan involvement. |
|
[143,144,145,146,147,148,149,150,151][52][53][54][55][56][57][58][59][60] |
| Mastocytosis | Itching, dyspnea, urticaria, dizziness, sense of fainting, multi-organ dysfunctions. |
|
[2,152,153,154][61][62][63][64] |
2. Autoimmune Thyroiditis
3. Chronic Arthritis
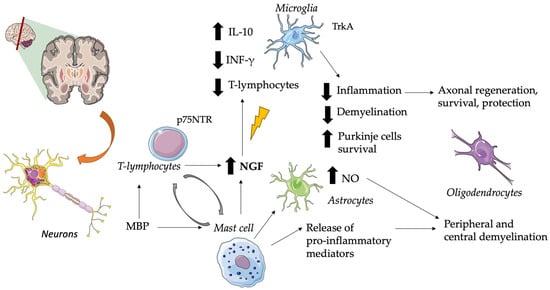
4. Multiple Sclerosis
5. Systemic Lupus Erythematosus
6. Mastocytosis
7. Chronic Granulomatous Disease
References
- Aloe, L.; Bracci-Laudiero, L.; Bonini, S.; Manni, L.; Aloe, L. The expanding role of nerve growth factor: From neurotrophic activity to immunologic diseases. Allergy 1997, 52, 883–994.
- Dicou, E.; Hurez, D.; Nerrière, V. Natural autoantibodies against the nerve growth factor in autoimmune diseases. J. Neuroimmunol. 1993, 47, 159–167.
- Fas, S.C.; Fritzsching, B.; Suri-Payer, E.; Krammer, P.H. Death receptor signaling and its function in the immune system. In Current Directions in Autoimmunity; KARGER: Basel, Switzerland, 2006; Volume 9, pp. 1–17.
- Dicou, E.; Nerrière, V. Evidence that natural autoantibodies against the nerve growth factor (NGF) may be potential carriers of NGF. J. Neuroimmunol. 1997, 75, 200–203.
- Dicou, E.; Masson, C.; Jabbour, W.; Nerriere, V. Increased frequency of NGF in sera of rheumatoid arthritis and systemic lupus erythematosus patients. Neuroreport 1993, 5, 321–324.
- Yoon, J.S.; Choi, S.H.; Lee, J.H.; Lee, S.J.; Lee, S.Y. Ocular surface inflammation, and nerve growth factor level in tears in active thyroid-associated ophthalmopathy. Graefes Arch. Clin. Exp. Ophthalmol. 2010, 248, 271–276.
- Molnár, I.; Bokk, Á. Decreased nerve growth factor levels in hyperthyroid Graves’ ophthalmopathy highlighting the role of neuroprotective factor in autoimmune thyroid diseases. Cytokine 2006, 35, 109–114.
- Minnone, G.; De Benedetti, F.; Bracci-Laudiero, L. NGF and Its Receptors in the Regulation of Inflammatory Response. Int. J. Mol. Sci. 2017, 18, 1028.
- Farina, L.; Minnone, G.; Alivernini, S.; Caiello, I.; MacDonald, L.; Soligo, M.; Manni, L.; Tolusso, B.; Coppola, S.; Zara, E.; et al. Pro Nerve Growth Factor and Its Receptor p75NTR Activate Inflammatory Responses in Synovial Fibroblasts: A Novel Targetable Mechanism in Arthritis. Front. Immunol. 2022, 13, 818630.
- Wise, B.L.; Seidel, M.F.; Lane, N.E. The evolution of nerve growth factor inhibition in clinical medicine. Nat. Rev. Rheumatol. 2021, 17, 34–46.
- Oo, W.M.; Hunter, D.J. Nerve Growth Factor (NGF) Inhibitors and Related Agents for Chronic Musculoskeletal Pain: A Comprehensive Review. BioDrugs 2021, 35, 611–641.
- Seidel, M.F.; Herguijuela, M.; Forkert, R.; Otten, U. Nerve Growth Factor in Rheumatic Diseases. Semin. Arthritis Rheum. 2010, 40, 109–126.
- Vincent, T.L. Mechanoflammation in osteoarthritis pathogenesis. Semin. Arthritis Rheum. 2019, 49, S36–S38.
- Yu, H.; Huang, T.; Lu, W.W.; Tong, L.; Chen, D. Osteoarthritis Pain. Int. J. Mol. Sci. 2022, 23, 4642.
- Ohashi, Y.; Uchida, K.; Fukushima, K.; Satoh, M.; Koyama, T.; Tsuchiya, M.; Saito, H.; Takahira, N.; Inoue, G.; Takaso, M. NGF Expression and Elevation in Hip Osteoarthritis Patients with Pain and Central Sensitization. Biomed. Res. Int. 2021, 2021, 9212585.
- Schmelz, M.; Mantyh, P.; Malfait, A.-M.; Farrar, J.; Yaksh, T.; Tive, L.; Viktrup, L. Nerve growth factor antibody for the treatment of osteoarthritis pain and chronic low-back pain: Mechanism of action in the context of efficacy and safety. Pain 2019, 160, 2210–2220.
- Bannwarth, B.; Kostine, M. Nerve Growth Factor Antagonists: Is the Future of Monoclonal Antibodies Becoming Clearer? Drugs 2017, 77, 1377–1387.
- Bannwarth, B.; Kostine, M. Targeting Nerve Growth Factor (NGF) for Pain Management: What Does the Future Hold for NGF Antagonists? Drugs 2014, 74, 619–626.
- Enomoto, M.; Mantyh, P.W.; Murrell, J.; Innes, J.F.; Lascelles, B.D.X. Anti-nerve growth factor monoclonal antibodies for the control of pain in dogs and cats. Vet. Rec. 2019, 184, 23.
- Genot, E.; Cantrell, D.A. Ras regulation and function in lymphocytes. Curr. Opin. Immunol. 2000, 12, 289–294.
- Hall, D.J.; Cui, J.; Bates, M.E.; Stout, B.A.; Koenderman, L.; Coffer, P.J.; Bertics, P.J. Transduction of a dominant-negative H-Ras into human eosinophils attenuates extracellular signal-regulated kinase activation and interleukin-5-mediated cell viability. Blood 2001, 98, 2014–2021.
- Futosi, K.; Fodor, S.; Mócsai, A. Neutrophil cell surface receptors and their intracellular signal transduction pathways. Int. Immunopharmacol. 2013, 17, 638–650.
- Nguyen, N.; Lee, S.B.; Lee, Y.S.; Lee, K.-H.; Ahn, J.-Y. Neuroprotection by NGF and BDNF against neurotoxin-exerted apoptotic death in neural stem cells are mediated through Trk receptors, activating PI3-kinase and MAPK pathways. Neurochem. Res. 2009, 34, 942–951.
- Soltoff, S.P.; Rabin, S.L.; Cantley, L.C.; Kaplan, D.R. Nerve growth factor promotes the activation of phosphatidylinositol 3-kinase and its association with the trk tyrosine kinase. J. Biol. Chem. 1992, 267, 17472–17477.
- Ross, S.H.; Cantrell, D.A. Signaling and Function of Interleukin-2 in T Lymphocytes. Annu. Rev. Immunol. 2018, 36, 411–433.
- Preite, S.; Gomez-Rodriguez, J.; Cannons, J.L.; Schwartzberg, P.L. T and B-cell signaling in activated PI3K delta syndrome: From immunodeficiency to autoimmunity. Immunol. Rev. 2019, 291, 154–173.
- Barker, S.A.; Lujan, D.; Wilson, B.S. Multiple roles for PI 3-kinase in the regulation of PLCgamma activity and Ca2+ mobilization in antigen-stimulated mast cells. J. Leukoc. Biol. 1999, 65, 321–329.
- Blatt, K.; Herrmann, H.; Mirkina, I.; Hadzijusufovic, E.; Peter, B.; Strommer, S.; Hoermann, G.; Mayerhofer, M.; Hoetzenecker, K.; Klepetko, W.; et al. The PI3-kinase/mTOR-targeting drug NVP-BEZ235 inhibits growth and IgE-dependent activation of human mast cells and basophils. PLoS ONE 2012, 7, e29925.
- Ma, B.; Athari, S.S.; Mehrabi Nasab, E.; Zhao, L. PI3K/AKT/mTOR and TLR4/MyD88/NF-κB Signaling Inhibitors Attenuate Pathological Mechanisms of Allergic Asthma. Inflammation 2021, 44, 1895–1907.
- Pan, T.; Sun, S.; Chen, Y.; Tian, R.; Chen, E.; Tan, R.; Wang, X.; Liu, Z.; Liu, J.; Qu, H. Immune effects of PI3K/Akt/HIF-1α-regulated glycolysis in polymorphonuclear neutrophils during sepsis. Crit. Care 2022, 26, 29.
- Gudasheva, T.A.; Logvinov, I.O.; Nikolaev, S.V.; Antipova, T.A.; Povarnina, P.Y.; Seredenin, S.B. Dipeptide Mimetics of Different NGF and BDNF Loops Activate PLC-γ1. Dokl. Biochem. Biophys. 2020, 494, 244–247.
- Braiman, A.; Barda-Saad, M.; Sommers, C.L.; Samelson, L.E. Recruitment and activation of PLCgamma1 in T cells: A new insight into old domains. EMBO J. 2006, 25, 774–784.
- Wen, R.; Chen, Y.; Schuman, J.; Fu, G.; Yang, S.; Zhang, W.; Newman, D.K.; Wang, D. An important role of phospholipase Cgamma1 in pre-B-cell development and allelic exclusion. EMBO J. 2004, 23, 4007–4017.
- Gilfillan, A.M.; Rivera, J. The tyrosine kinase network regulating mast cell activation. Immunol. Rev. 2009, 228, 149–169.
- Bae, Y.-S.; Lee, H.Y.; Jung, Y.S.; Lee, M.; Suh, P.-G. Phospholipase Cγ in Toll-like receptor-mediated inflammation and innate immunity. Adv. Biol. Regul. 2017, 63, 92–97.
- Bayas, A.; Kruse, N.; Moriabadi, N.F.; Weber, F.; Hummel, V.; Wohleben, G.; Gold, R.; Toyka, K.V.; Rieckmann, P. Modulation of cytokine mRNA expression by brain-derived neurotrophic factor and nerve growth factor in human immune cells. Neurosci. Lett. 2003, 335, 155–158.
- Vega, J.A.; García-Suárez, O.; Germanà, A. Vertebrate thymus and the neurotrophin system. Int. Rev. Cytol. 2004, 237, 155–204.
- Dae, J.K.; Yang, W.I.; Se, H.K.; Park, I.K.; Kyung, Y.C. Expression of neurotrophin receptors in surgically resected thymic epithelial tumors. Eur. J. Cardio-Thorac. Surg. 2005, 28, 611–616.
- Hillis, J.; O’Dwyer, M.; Gorman, A.M. Neurotrophins and B-cell malignancies. Cell. Mol. Life Sci. 2016, 73, 41–56.
- Vega, J.A.; García-Suárez, O.; Hannestad, J.; Pérez-Pérez, M.; Germanà, A. Neurotrophins and the immune system. J. Anat. 2003, 203, 1–19.
- Dubanet, L.; Bentayeb, H.; Petit, B.; Olivrie, A.; Saada, S.; De La Cruz-Morcillo, M.A.; Lalloué, F.; Gourin, M.P.; Bordessoule, D.; Faumont, N.; et al. Anti-apoptotic role and clinical relevance of neurotrophins in diffuse large B-cell lymphomas. Br. J. Cancer 2015, 113, 934–944.
- Bellanger, C.; Dubanet, L.; Lise, M.C.; Fauchais, A.L.; Bordessoule, D.; Jauberteau, M.O.; Troutaud, D. Endogenous neurotrophins and Trk signaling in diffuse large B cell lymphoma cell lines are involved in sensitivity to rituximab-induced apoptosis. PLoS ONE 2011, 6, e27213.
- Theofilopoulos, A.N.; Kono, D.H.; Baccala, R. The multiple pathways to autoimmunity. Nat. Immunol. 2017, 18, 716–724.
- Crooks, A.; Meeker, R. The new wave of p75 neurotrophin receptor targeted therapies. Neural Regen. Res. 2022, 17, 95.
- Bonini, S.; Lambiase, A.; Bonini, S.; Levi-Schaffer, F.; Aloe, L. Nerve Growth Factor: An Important Molecule in Allergic Inflammation and Tissue Remodelling. Int. Arch. Allergy Immunol. 1999, 118, 159–162.
- Ferraguti, G.; Terracina, S.; Micangeli, G.; Lucarelli, M.; Tarani, L.; Ceccanti, M.; Spaziani, M.; D’Orazi, V.; Petrella, C.; Fiore, M. NGF and BDNF in pediatrics syndromes. Neurosci. Biobehav. Rev. 2023, 145, 105015.
- Nayernia, Z.; Colaianna, M.; Robledinos-Antón, N.; Gutzwiller, E.; Sloan-Béna, F.; Stathaki, E.; Hibaoui, Y.; Cuadrado, A.; Hescheler, J.; José Stasia, M.; et al. Decreased neural precursor cell pool in NADPH oxidase 2-deficiency: From mouse brain to neural differentiation of patient derived iPSC. Redox Biol. 2017, 13, 82–93.
- Cole, T.; McKendrick, F.; Cant, A.; Pearce, M.; Cale, C.; Goldblatt, D.; Gennery, A.; Titman, P. Cognitive ability in children with chronic granulomatous disease: A comparison of those managed conservatively with those who have undergone hematopoietic stem cell transplant. Neuropediatrics 2013, 44, 230–232.
- Pao, M.; Wiggs, E.A.; Anastacio, M.M.; Hyun, J.; DeCarlo, E.S.; Miller, J.T.; Anderson, V.L.; Malech, H.L.; Gallin, J.I.; Holland, S.M. Cognitive Function in Patients with Chronic Granulomatous Disease: A Preliminary Report. Psychosomatics 2004, 45, 230–234.
- Becker, S.; Wasser, S.; Hauses, M.; Hossle, J.P.; Ott, M.G.; Dinauer, M.C.; Ganser, A.; Hoelzer, D.; Seger, R.; Grez, M. Correction of respiratory burst activity in X-linked chronic granulomatous cells to therapeutically relevant levels after gene transfer into bone marrow CD34+ cells. Hum. Gene Ther. 1998, 9, 1561–1570.
- Grez, M.; Becker, S.; Saulnier, S.; Knöβ, H.; Ott, M.G.; Maurer, A.; Dinauer, M.C.; Hoelzer, D.; Seger, R.; Hossle, J.P. Gene therapy of chronic granulomatous disease. Bone Marrow Transplant. 2000, 25, S99–S104.
- Otten, U.; Gadient, R.A. Neurotrophins and cytokines—Intermediaries between the immune and nervous systems. Int. J. Dev. Neurosci. 1995, 13, 147–151.
- Bracci-Laudiero, L.; Aloe, L.; Levi-Montalcini, R.; Galeazzi, M.; Schilter, D.; Scully, J.L.; Otten, U.; Bracci Laudiero, L.; Aloe, L.; Levi Montalcini, R.; et al. Increased levels of NGF in sera of systemic lupus erythematosus patients. Neuroreport 1993, 4, 563–565.
- Fauchais, A.-L.; Lise, M.-C.; Marget, P.; Lapeybie, F.-X.; Bezanahary, H.; Martel, C.; Dumonteil, S.; Sparsa, A.; Lalloué, F.; Ly, K.; et al. Serum and Lymphocytic Neurotrophins Profiles in Systemic Lupus Erythematosus: A Case-Control Study. PLoS ONE 2013, 8, e79414.
- Kalinowska-Łyszczarz, A.; Pawlak, M.A.; Wyciszkiewicz, A.; Pawlak-Buś, K.; Leszczyński, P.; Puszczewicz, M.; Paprzycki, W.; Kozubski, W.; Michalak, S. Immune Cell Neurotrophin Production Is Associated with Subcortical Brain Atrophy in Neuropsychiatric Systemic Lupus Erythematosus Patients. Neuroimmunomodulation 2017, 24, 320–330.
- Zusen, X.; Yanlei, C. Determination of serum interleukin-13 and nerve growth factor in patients with systemic lupus erythematosus and clinical significance. J. Huazhong Univ. Sci. Technol. Med. Sci. 2005, 25, 360–361.
- Bracci-Laudiero, L.; Lundeberg, T.; Stenfors, C.; Theodorsson, E.; Tirassa, P.; Aloe, L. Modification of lymphoid and brain nerve growth factor levels in systemic lupus erythematosus mice. Neurosci. Lett. 1996, 204, 13–16.
- Aloe, L.; Tuveri, M.A. Nerve growth factor and autoimmune rheumatic diseases. Clin. Exp. Rheumatol. 1997, 15, 433–438.
- Aalto, K.; Korhonen, L.; Lahdenne, P.; Pelkonen, P.; Lindholm, D. Nerve growth factor in serum of children with systemic lupus erythematosus is correlated with disease activity. Cytokine 2002, 20, 136–139.
- Welle, S.; Wolf, A.M.; Dernbach, C.; Klarmann-Schulz, U.; Seidel, M.F. Disease activity-dependent expression of nerve growth factor TRKA and P75 receptors on elevated dendritic cells and peripheral leucocytes in patients with systemic lupus erythematosus. Lupus 2020, 29, 970–975.
- Skaper, S.D. Nerve growth factor: A neuroimmune crosstalk mediator for all seasons. Immunology 2017, 151, 1–15.
- Leon, A.; Buriani, A.; Dal Toso, R.; Fabris, M.; Romanello, S.; Aloe, L.; Levi-Montalcini, R. Mast cells synthesize, store, and release nerve growth factor. Proc. Natl. Acad. Sci. USA 1994, 91, 3739–3743.
- Peng, W.M.; Maintz, L.; Allam, J.P.; Raap, U.; Gutgemann, I.; Kirfel, J.; Wardelmann, E.; Perner, S.; Zhao, W.; Fimmers, R.; et al. Increased circulating levels of neurotrophins and elevated expression of their high-Affinity receptors on skin and gut mast cells in mastocytosis. Blood 2013, 122, 1779–1788.
- Dutta, P.; Koch, A.; Breyer, B.; Schneider, H.; Dittrich-Breiholz, O.; Kracht, M.; Tamura, T. Identification of novel target genes of nerve growth factor (NGF) in human mastocytoma cell line (HMC-1 (V560G c-Kit)) by transcriptome analysis. BMC Genom. 2011, 12, 196.
- Ralli, M.; Angeletti, D.; Fiore, M.; D’Aguanno, V.; Lambiase, A.; Artico, M.; de Vincentiis, M.; Greco, A. Hashimoto’s thyroiditis: An update on pathogenic mechanisms, diagnostic protocols, therapeutic strategies, and potential malignant transformation. Autoimmun. Rev. 2020, 19, 102649.
- Street, S. Thyroid Disease and Autoimmune Thyroiditis; Heart of Minnesota Great Dane Club: St. Paul, MN, USA, 2004; p. 90403.
- Ragusa, F.; Fallahi, P.; Elia, G.; Gonnella, D.; Paparo, S.R.; Giusti, C.; Churilov, L.P.; Ferrari, S.M.; Antonelli, A. Hashimotos’ thyroiditis: Epidemiology, pathogenesis, clinic and therapy. Best Pract. Res. Clin. Endocrinol. Metab. 2019, 33, 101367.
- Eschler, D.C.; Hasham, A.; Tomer, Y. Cutting edge: The etiology of autoimmune thyroid diseases. Clin. Rev. Allergy Immunol. 2011, 41, 190–197.
- Stefan, M.; Jacobson, E.M.; Huber, A.K.; Greenberg, D.A.; Li, C.W.; Skrabanek, L.; Conception, E.; Fadlalla, M.; Ho, K.; Tomer, Y. Novel variant of thyroglobulin promoter triggers thyroid autoimmunity through an epigenetic interferon alpha-modulated mechanism. J. Biol. Chem. 2011, 286, 31168–31179.
- Ceccanti, M.; De Nicolò, S.; Mancinelli, R.; Chaldakov, G.; Carito, V.; Ceccanti, M.; Laviola, G.; Tirassa, P.; Fiore, M. NGF and BDNF long-term variations in the thyroid, testis and adrenal glands of a mouse model of fetal alcohol spectrum disorders. Ann. Ist. Super. Sanita 2013, 49, 383–390.
- Yang, C.R.; Ding, H.J.; Yu, M.; Zhou, F.H.; Han, C.Y.; Liang, R.; Zhang, X.Y.; Zhang, X.L.; Meng, F.J.; Wang, S.; et al. proBDNF/p75NTR promotes rheumatoid arthritis and inflammatory response by activating proinflammatory cytokines. FASEB J. 2022, 36, e22180.
- Schnitzer, T.J.; Marks, J.A. A systematic review of the efficacy and general safety of antibodies to NGF in the treatment of OA of the hip or knee. Osteoarthr. Cartil. 2015, 23, S8–S17.
- Sánchez-Robles, E.M.; Girón, R.; Paniagua, N.; Rodríguez-Rivera, C.; Pascual, D.; Goicoechea, C. Monoclonal Antibodies for Chronic Pain Treatment: Present and Future. Int. J. Mol. Sci. 2021, 22, 10325.
- Aloe, L.; Tuveri, M.A.; Levi-Montalcini, R. Studies on carrageenan-induced arthritis in adult rats: Presence of nerve growth factor and role of sympathetic innervation. Rheumatol. Int. 1992, 12, 213–216.
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2008, 372, 1502–1517.
- Amato, M.P.; Derfuss, T.; Hemmer, B.; Liblau, R.; Montalban, X.; Soelberg Sørensen, P.; Miller, D.H.; Alfredsson, L.; Aloisi, F.; Amato, M.P.; et al. Environmental modifiable risk factors for multiple sclerosis: Report from the 2016 ECTRIMS focused workshop. Mult. Scler. J. 2018, 24, 590–603.
- Ascherio, A.; Munger, K.L.; Lünemann, J.D. The initiation and prevention of multiple sclerosis. Nat. Rev. Neurol. 2012, 8, 602–612.
- Oh, J.; Vidal-Jordana, A.; Montalban, X. Multiple sclerosis: Clinical aspects. Curr. Opin. Neurol. 2018, 31, 752–759.
- Olsson, Y. Mast cells in plaques of multiple sclerosis. Acta Neurol. Scand. 2009, 50, 611–618.
- Villoslada, P.; Hauser, S.L.; Bartke, I.; Unger, J.; Heald, N.; Rosenberg, D.; Cheung, S.W.; Mobley, W.C.; Fisher, S.; Genain, C.P. Human Nerve Growth Factor Protects Common Marmosets against Autoimmune Encephalomyelitis by Switching the Balance of T Helper Cell Type 1 and 2 Cytokines within the Central Nervous System. J. Exp. Med. 2000, 191, 1799–1806.
- Kaplan, A.P.; Reddigari, S.; Baeza, M.; Kuna, P. Histamine Releasing Factors and Cytokine-Dependent Activation of Basophils and Mast Cells. Adv. Immunol. 1991, 50, 237–260.
- Askenase, P.W.; Van Loverent, H. Delayed-type hypersensitivity: Activation of mast cells by antigen-specific T-cell factors initiates the cascade of cellular interactions. Immunol. Today 1983, 4, 259–264.
- Moalem, G.; Gdalyahu, A.; Shani, Y.; Otten, U.; Lazarovici, P.; Cohen, I.R.; Schwartz, M. Production of Neurotrophins by Activated T Cells: Implications for Neuroprotective Autoimmunity. J. Autoimmun. 2000, 15, 331–345.
- Acosta, C.; Cortes, C.; Altaweel, K.; MacPhee, H.; Hoogervorst, B.; Bhullar, H.; MacNeil, B.; Torabi, M.; Burczynski, F.; Namaka, M. Immune System Induction of Nerve Growth Factor in an Animal Model of Multiple Sclerosis: Implications in Re-Myelination and Myelin RepairATION AND MYELIN REPAIR. CNS Neurol. Disord.-Drug Targets 2015, 14, 1069–1078.
- Skaper, S.D.; Pollock, M.; Facci, L. Mast cells differentially express and release active high molecular weight neurotrophins. Mol. Brain Res. 2001, 97, 177–185.
- Laudiero, L.B.; Aloe, L.; Levi-Montalcini, R.; Buttinelli, C.; Schilter, D.; Gillessen, S.; Otten, U. Multiple sclerosis patients express increased levels of beta-nerve growth factor in cerebrospinal fluid. Neurosci. Lett. 1992, 147, 9–12.
- Suzaki, I.; Hara, T.; Maegaki, Y.; Narai, S.; Takeshita, K. Nerve Growth Factor Levels in Cerebrospinal Fluid From Patients With Neurologic Disorders. J. Child Neurol. 1997, 12, 205–207.
- Monteleone, F.; Nicoletti, C.G.; Stampanoni Bassi, M.; Iezzi, E.; Buttari, F.; Furlan, R.; Finardi, A.; Marfia, G.A.; Centonze, D.; Mori, F. Nerve growth factor is elevated in the CSF of patients with multiple sclerosis and central neuropathic pain. J. Neuroimmunol. 2018, 314, 89–93.
- Micera, A.; De Simone, R.; Aloe, L. Elevated levels of nerve growth factor in the thalamus and spinal cord of rats affected by experimental allergic encephalomyelitis. Arch. Ital. Biol. 1995, 133, 131–142.
- Micera, A.; Vigneti, E.; Aloe, L. Changes of NGF presence in nonneuronal cells in response to experimental allergic encephalomyelitis in Lewis rats. Exp. Neurol. 1998, 154, 41–46.
- Valdo, P.; Stegagno, C.; Mazzucco, S.; Zuliani, E.; Zanusso, G.; Moretto, G.; Raine, C.S.; Bonetti, B. Enhanced Expression of NGF Receptors in Multiple Sclerosis Lesions. J. Neuropathol. Exp. Neurol. 2002, 61, 91–98.
- Dowling, P.; Ming, X.; Raval, S.; Husar, W.; Casaccia-Bonnefil, P.; Chao, M.; Cook, S.; Blumberg, B. Up-regulated p75NTR neurotrophin receptor on glial cells in MS plaques. Neurology 1999, 53, 1676–1682.
- Nataf, S.; Naveilhan, P.; Sindji, L.; Darcy, F.; Brachet, P.; Montero-Menei, C.N. Low affinity NGF receptor expression in the central nervous system during experimental allergic encephalomyelitis. J. Neurosci. Res. 1998, 52, 83–92.
- Aguado, F.; Ballabriga, J.; Pozas, E.; Ferrer, I. TrkA immunoreactivity in reactive astrocytes in human neurodegenerative diseases and colchicine-treated rats. Acta Neuropathol. 1998, 96, 495–501.
- Damarjian, T.G.; Craner, M.J.; Black, J.A.; Waxman, S.G. Upregulation and colocalization of p75 and Nav1.8 in Purkinje neurons in experimental autoimmune encephalomyelitis. Neurosci. Lett. 2004, 369, 186–190.
- Micera, A.; Lambiase, A.; Rama, P.; Aloe, L. Altered nerve growth factor level in the optic nerve of patients affected by multiple sclerosis. Mult. Scler. J. 1999, 5, 389–394.
- Jiang, Y.; Yang, Y.; Zhang, B.; Peng, F.; Bao, J.; Hu, X. Cerebrospinal fluid levels of iodothyronines and nerve growth factor in patients with multiple sclerosis and neuromyelitis optica. Neuro Endocrinol. Lett. 2009, 30, 85–90.
- Calza, L.; Fernandez, M.; Giuliani, A.; Aloe, L.; Giardino, L. Thyroid hormone activates oligodendrocyte precursors and increases a myelin-forming protein and NGF content in the spinal cord during experimental allergic encephalomyelitis. Proc. Natl. Acad. Sci. USA 2002, 99, 3258–3263.
- Caggiula, M.; Batocchi, A.P.; Frisullo, G.; Angelucci, F.; Patanella, A.K.; Sancricca, C.; Nociti, V.; Tonali, P.A.; Mirabella, M. Neurotrophic Factors and Clinical Recovery in Relapsing-Remitting Multiple Sclerosis. Scand. J. Immunol. 2005, 62, 176–182.
- Kalinowska-Łyszczarz, A.; Pawlak, M.A.; Michalak, S.; Losy, J. Cognitive deficit is related to immune-cell beta-NGF in multiple sclerosis patients. J. Neurol. Sci. 2012, 321, 43–48.
- Akkad, D.A.; Kruse, N.; Arning, L.; Gold, R.; Epplen, J.T. Genomic NGFBvariation and multiple sclerosis in a case control study. BMC Med. Genet. 2008, 9, 107.
- Acosta, C.M.R.; Cortes, C.; MacPhee, H.; Namaka, M.P. Exploring the Role of Nerve Growth Factor in Multiple Sclerosis: Implications in Myelin Repair. CNS Neurol. Disord.-Drug Targets 2014, 12, 1242–1256.
- Hsieh, T.Y.; Chang, Y.; Wang, S.J. Piperine Provides Neuroprotection against Kainic Acid-Induced Neurotoxicity via Maintaining NGF Signalling Pathway. Molecules 2022, 27, 2638.
- Colafrancesco, V.; Viloslada, P. Targeting NGF-pathway for developing neuroprotective therapies for multiple sclerosis and other neurological diseases. Arch. Ital. Biol. 2011, 149, 183–192.
- Razavi, S.; Nazem, G.; Mardani, M.; Esfandiari, E.; Esfahani, S.; Salehi, H. Neurotrophic factors and their effects in the treatment of multiple sclerosis. Adv. Biomed. Res. 2015, 4, 53.
- Massaro, A.R.; Tonali, P. Cerebrospinal fluid markers in multiple sclerosis: An overview. Mult. Scler. J. 1998, 4, 1–4.
- Villoslada, P.; Genain, C.P. Role of nerve growth factor and other trophic factors in brain inflammation. Prog. Brain Res. 2004, 146, 403–414.
- Biernacki, K.; Antel, J.P.; Blain, M.; Narayanan, S.; Arnold, D.L.; Prat, A. Interferon Beta Promotes Nerve Growth Factor Secretion Early in the Course of Multiple Sclerosis. Arch. Neurol. 2005, 62, 563.
- Caggiula, M.; Batocchi, A.P.; Frisullo, G.; Angelucci, F.; Patanella, A.K.; Sancricca, C.; Nociti, V.; Tonali, P.A.; Mirabella, M. Neurotrophic factors in relapsing remitting and secondary progressive multiple sclerosis patients during interferon beta therapy. Clin. Immunol. 2006, 118, 77–82.
- Boutros, T.; Croze, E.; Yong, V.W. Interferon-beta is a potent promoter of nerve growth factor production by astrocytes. J. Neurochem. 1997, 69, 939–946.
- Rahimlou, M.; Hosseini, S.A.; Majdinasab, N.; Haghighizadeh, M.H.; Husain, D. Effects of long-term administration of Multi-Strain Probiotic on circulating levels of BDNF, NGF, IL-6 and mental health in patients with multiple sclerosis: A randomized, double-blind, placebo-controlled trial. Nutr. Neurosci. 2022, 25, 411–422.
- Mokhtarzade, M.; Motl, R.; Negaresh, R.; Zimmer, P.; Khodadoost, M.; Baker, J.S.; Patel, D.; Majdinasab, N.; Ranjbar, R. Exercise-induced changes in neurotrophic factors and markers of blood-brain barrier permeability are moderated by weight status in multiple sclerosis. Neuropeptides 2018, 70, 93–100.
- Diechmann, M.D.; Campbell, E.; Coulter, E.; Paul, L.; Dalgas, U.; Hvid, L.G. Effects of Exercise Training on Neurotrophic Factors and Subsequent Neuroprotection in Persons with Multiple Sclerosis—A Systematic Review and Meta-Analysis. Brain Sci. 2021, 11, 1499.
- Naghibzadeh, M.; Ranjbar, R.; Tabandeh, M.; Habibi, A. Effects of Two Training Programs on Transcriptional Levels of Neurotrophins and Glial Cells Population in Hippocampus of Experimental Multiple Sclerosis. Int. J. Sports Med. 2018, 39, 604–612.
- Gold, S.M.; Schulz, K.-H.; Hartmann, S.; Mladek, M.; Lang, U.E.; Hellweg, R.; Reer, R.; Braumann, K.-M.; Heesen, C. Basal serum levels and reactivity of nerve growth factor and brain-derived neurotrophic factor to standardized acute exercise in multiple sclerosis and controls. J. Neuroimmunol. 2003, 138, 99–105.
- Bansi, J.; Bloch, W.; Gamper, U.; Kesselring, J. Training in MS: Influence of two different endurance training protocols (aquatic versus overland) on cytokine and neurotrophin concentrations during three week randomized controlled trial. Mult. Scler. J. 2013, 19, 613–621.
- Bansi, J.; Bloch, W.; Gamper, U.; Riedel, S.; Kesselring, J. Endurance training in MS: Short-term immune responses and their relation to cardiorespiratory fitness, health-related quality of life, and fatigue. J. Neurol. 2013, 260, 2993–3001.
- Houshmand, F.; Barati, M.; Golab, F.; Ramezani-sefidar, S.; Tanbakooie, S.; Tabatabaei, M.; Amiri, M.; Sanadgol, N. Metformin-induced AMPK activation stimulates remyelination through induction of neurotrophic factors, downregulation of NogoA and recruitment of Olig2+ precursor cells in the cuprizone murine model of multiple sclerosis. DARU J. Pharm. Sci. 2019, 27, 583–592.
- Jiang, Y.; Zou, Y.; Chen, S.; Zhu, C.; Wu, A.; Liu, Y.; Ma, L.; Zhu, D.; Ma, X.; Liu, M.; et al. The anti-inflammatory effect of donepezil on experimental autoimmune encephalomyelitis in C57 BL/6 mice. Neuropharmacology 2013, 73, 415–424.
- Zheng, X.P.; Zhang, H.L.; Li, H.F.; Zhang, M.Z.; Qiu, W.; Hu, X.Q. Neuroprotective potential beyond immunoregulation of helminth infection as a therapeutic target in multiple sclerosis. Med. Hypotheses 2012, 78, 95–97.
- Feng, M.; Shu, Y.; Yang, Y.; Zheng, X.; Li, R.; Wang, Y.; Dai, Y.; Qiu, W.; Lu, Z.; Hu, X. Ulinastatin attenuates experimental autoimmune encephalomyelitis by enhancing anti-inflammatory responses. Neurochem. Int. 2014, 64, 64–72.
- Chen, X.; Pi, R.; Zou, Y.; Liu, M.; Ma, X.; Jiang, Y.; Mao, X.; Hu, X. Attenuation of experimental autoimmune encephalomyelitis in C57 BL/6 mice by osthole, a natural coumarin. Eur. J. Pharmacol. 2010, 629, 40–46.
- Penkowa, M.; Hidalgo, J. Retracted: Treatment with metallothionein prevents demyelination and axonal damage and increases oligodendrocyte precursors and tissue repair during experimental autoimmune encephalomyelitis. J. Neurosci. Res. 2003, 72, 574–586.
- Yin, L.L.; Lin, L.L.; Zhang, L.; Li, L. Epimedium flavonoids ameliorate experimental autoimmune encephalomyelitis in rats by modulating neuroinflammatory and neurotrophic responses. Neuropharmacology 2012, 63, 851–862.
- Tabakman, R.; Lecht, S.; Sephanova, S.; Arien-Zakay, H.; Lazarovici, P. Interactions between the cells of the immune and nervous system: Neurotrophins as neuroprotection mediators in CNS injury. Prog. Brain Res. 2004, 146, 385–401.
- Parvaneh Tafreshi, A. Nerve growth factor prevents demyelination, cell death and progression of the disease in experimental allergic encephalomyelitis. Iran. J. Allergy Asthma Immunol. 2006, 5, 177–181.
- Aloe, L.; Micera, A. A role of nerve growth factor in oligodendrocyte growth and differentiation of EAE affected rats. Arch. Ital. Biol. 1998, 136, 247–256.
- Oderfeld-Nowaki, B.; Zarembai, M.; Lipkowski, A.W.; Kwiatkowska-Patzer, B.; Triaca, V.; Aloe, L. High-affinity NGF receptor in the rat spinal cord during acute and chronic phases of experimental autoimmune encephalomyelitis: A possible functional significance. Arch. Ital. Biol. 2003, 141, 103–116.
- Bonetto, G.; Charalampopoulos, I.; Gravanis, A.; Karagogeos, D. The novel synthetic microneurotrophin BNN27 protects mature oligodendrocytes against cuprizone-induced death, through the NGF receptor TrkA. Glia 2017, 65, 1376–1394.
- Zhao, M.; Tang, J.; Gao, F.; Wu, X.; Liang, Y.; Yin, H.; Lu, Q. Hypomethylation of IL10 and IL13 Promoters in CD4 + T Cells of Patients with Systemic Lupus Erythematosus. J. Biomed. Biotechnol. 2010, 2010, 931018.
- Baghdadi, M.; Umeyama, Y.; Hama, N.; Kobayashi, T.; Han, N.; Wada, H.; Seino, K. Interleukin-34, a comprehensive review. J. Leukoc. Biol. 2018, 104, 931–951.
- El-Banna, H.S.; El Khouly, R.M.; Gado, S.E. Elevated serum interleukin-34 level in juvenile systemic lupus erythematosus and disease activity. Clin. Rheumatol. 2020, 39, 1627–1632.
- Hussain, S.H. Pediatric mastocytosis. Curr. Opin. Pediatr. 2020, 32, 531–538.
- Carter, M.C.; Metcalfe, D.D.; Komarow, H.D. Mastocytosis. Immunol. Allergy Clin. N. Am. 2014, 34, 181–196.
- Yang, M.; Pan, Z.; Huang, K.; Büsche, G.; Feuerhake, F.; Chaturvedi, A.; Nie, D.; Heuser, M.; Thol, F.; von Neuhoff, N.; et al. Activation of TRKA receptor elicits mastocytosis in mice and is involved in the development of resistance to KIT-targeted therapy. Oncotarget 2017, 8, 73871–73883.
