The research focused on the generation of clean energy has been boosted over the past decades, but the intermittent nature of these renewable resources requires an effective energy storage system (ESS) to store the generated energy. Among the various ESS technologies, rechargeable batteries have been considered the most feasible and reliable, and they already play a prominent role in our lives today. Among them, rechargeable lithium-ion batteries (LIBs) have dominated the application fields of portable devices, electric vehicles, and large-scale energy storage. But, limited lithium resources and the increasing prices of LIBs have aroused great concerns. Therefore, some cheaper rechargeable batteries have received a lot of attention, such as sodium-ion batteries (SIBs), potassium-ion batteries (KIBs), and so on. Despite the significant advancements in SIBs and KIBs, the widespread adoption of organic-based electrolytes, which are toxic, volatile, and flammable, continues to make batteries encounter safety and environmental hurdles. In contrast, aqueous rechargeable batteries offer the advantages of affordability, heightened safety, superior ionic conductivity, and simplified manufacturing. These attributes position aqueous batteries as the most auspicious contender for the forthcoming era of large-scale ESS.
while the reaction process in the chemical conversion reaction mechanism is as follows:
2. The Challenges of the Zinc Anode
Even though the research on high-performance ZIBs has achieved great progress, the widespread commercial implementation of metallic zinc anodes in secondary batteries has encountered substantial limitations. Generally, the limitations of ZIB development mainly come from the zinc anode side. The poor rechargeability and low Zn utilization are ascribed to dendrite growth
[13][14][15][16,17,18], hydrogen evolution
[16][17][18][19,20,21], and surface passivation
[19][20][21][22,23,24] occurring on the Zn anodes during the repetitive plating and stripping processes
(Figure 3). These factors significantly impede its progress in practical applications in next-generation batteries.
2.1. Dendrites and Protrusion
Currently, commercial zinc foils are used as the anode in the ZIBs. However, in practical scenarios, the Zn anodes consistently exhibit uneven and non-uniform surface morphology at the micro- or nanoscale, leading to heterogeneous current distribution and nucleation barriers. During the charge/discharge process, the factors that are responsible for the formation of dendrites are uneven Zn
2+ concentration distribution and non-uniform electric field distribution. It is always preferred for the nucleation to occur in locations that have high Zn
2+ concentration and a high electric field
[21][22][24,26]. The formed needle-like protrusion will further promote the growth of zinc dendrites because of the so-called “tip effect”. With repeated charge/discharge cycling, the dendrites will even puncture the separator and give rise to a short circuit during further cycling.
2.2. Hydrogen Evolution Reaction
In most of the studies, metallic Zn is employed as an anode material in ZIBs directly. However, as a relatively active transition metal, metallic Zn is easily affected by side reactions between Zn electrodes and aqueous electrolytes during the battery operation, which has severely hindered the practical applications of ZIBs. The hydrogen evolution reaction (HER) is the most frequent side reaction during the charge/discharge process in the battery operation. The electrochemical reaction which occurred on the surface of Zn anode is as follows:

The HER not only consumes the fresh electrolyte and Zn anode but also produces H
2 gas, which will lead to the battery swelling. Interestingly, according to ref.
[23][27], a high current density will alleviate the HER during the charge–discharge process. This is because the electrodeposited Zn crystal at high current density has a smaller specific surface area, which is critical for HER activity. As a result, the HER is significantly reduced at high current density.
2.3. Passivation Layer
The surface passivation process is always accompanied by the hydrogen evolution reaction and surface corrosion during the repeated plating/stripping cycling. With the hydrogen evolution, the local pH will be increased dramatically since the OH
− concentration increases rapidly
[24][25][26][29,30,31], and the generated OH
− will react with Zn
2+ and H
2O to form the passivation layer on the zinc surface. The non-conductive inactive and insoluble byproducts (such as ZnO and Zn
4SO
4(OH)
6‧xH
2O) precipitate onto the surface of the zinc anode. This cuts down the reversibility of the plating/stripping process, increases internal resistance, and limits zinc utilization. The accumulation of the non-conductive passivation layer will further reduce the reversibility and enhance the polarization, ultimately leading to the diminished recharging performance of the Zn anode.
3. The Strategies of Zinc Anode Protection
3.1. Surface Engineering
The first is constructing an artificial protecting layer that can work similarly to the SEI in ZIBs. If the commercial zinc foil is directly used as the anode without any modification, it leads to the occurrence of both dendrite growth and side reaction on the surface of the zinc surface. Therefore, an engineered surface layer is constructed to protect the anode from contacting the aqueous electrolyte, effectively curbing dendrite growth and the HER. Hence, building an artificial protection layer on the surface of the zinc anode, whether by an ex situ or in situ method, seems to be essential. Inorganic coating layers, such as CaCO3, [27][37] TiO2, [28][29][38,39] montmorillonite [30][31][40,41], carbon materials [32][42], and other materials [33][34][35][36][37][32,43,44,45,46] are extensively studied as the protecting layer. The porous nano-CaCO3 coating layer worked as a buffer layer, which can guide the deposition of Zn during the plating/stripping process [27][37]. The porous nature of the coating layer can confine the Zn plating reaction beneath the buffer layer, guide uniform electrolyte flux, and control the Zn plating rate, resulting in a uniform and dense Zn deposition layer that can effectively inhibit dendrite formation. In addition, porous nano-SiO2 and acetylene black were also tested and obtained similar results as the porous nano-CaCO3. The TiO2 insulating layers are also studied as the protecting layer on the Zn anode surface. By avoiding the direct contact between zinc anode and electrolyte, the undesired hydrogen evolution and corrosion process are significantly suppressed. This protective layer leads to an enhanced electrochemical performance of ZIBs. The protective layer prepared via ALD with (001) facet can effectively prevent the Zn dendrites from vertical growth and stabilize the interface between the electrode and electrolyte; hence, it exhibits a long-term cycle life in the test of plating/stripping performance [28][38]. The role of crystal orientation of the TiO2 protective layer was also studied in the paper, and DFT was used to calculate the interaction between Zn and different facets of TiO2. The calculation results show that the TiO2 coating layer with the (001) and (101) facets exposed possesses a relatively low Zn affinity [29][39]. In addition to the inorganic substances, such as zinc metal anode armor, polymer-based coating layers (such as polyacrylamide, polyamide, poly(vinyl butyral), polyvinylidene fluoride, and others) can serve as barriers to shield against H2O/O2 in the electrolyte and exhibit high protective performance in recent studies [38][39][40][41][42][43][47,48,49,50,51,52]. The polar functional groups contained in the polymers, such as C=O and -N-H2 bonds, can provide abundant adsorption or coordination sites and transfer Zn2+ to the reaction interface along the polymer chains, which can achieve the regulation of Zn2+ distribution from the molecular scale through the fast ion pathway. In addition, the polymer layers are water-insoluble and exhibit strong adhesion and excellent flexibility on the Zn surface. With the help of a protective layer, the side reactions with water can be significantly suppressed since the water from the Zn solvation shell was effectively removed during the penetration process, and dendrite growth was remarkably inhibited because of the electrostatic shield effect to avoid the accumulation of Zn2+/electrons on local regions.
The second method, manipulating the crystallographic orientation of zinc deposition, can effectively get rid of this problem. According to previously published articles [44][45][57,58], the (002) plane of metal Zn shows 0~30° alignment to the substrate, while the (100) and (110) planes have a higher angle of 70~90°. Hence, the (002) facet of Zn metal is less likely to facilitate the growth of dendrites when compared with the (100) and (110) planes. In addition, the (002) plane is the closest in hexagonal close-packed (hcp) metal, which has lower thermodynamic free energy than other planes. The decrease in activation energy of the (002) plane enhances the ability of Zn metal to exhibit strong resistance against surface corrosion. Therefore, controlling the growth of deposited zinc during the plating/stripping process is also regarded as one effective method to enhance Zn reversibility [46][47][48][49][50][51][52][35,59,60,61,62,63,64].
3.2. Electrolyte Modification
The use of water as an electrolyte solvent enabled the ZIBs to have the advantage of being affordable and nonflammable, with high ionic conductivity and enhanced kinetics. But similar to how every coin has two sides, the water molecules that serve as the solvent will be consumed during the charge/discharge process, and the decomposition of water is the root of the undesired side reaction. In addition, the byproduct and corrosion process will lead to the acceleration of Zn anode deterioration in the following galvanostatic charge/discharge (GCD) process. Due to the tendency of Zn
2+ ions to predominantly exist as hydrated [Zn(H
2O)
6]
2+ species in the electrolyte, the aqueous electrolyte exhibits an acidic nature. Additionally, the metal zinc is not thermodynamically stable when exposed to these electrolytes. Therefore, modifying the electrolyte to control the decomposition of water molecules and the behavior of Zn
2+ during the plating/stripping process is also an important strategy to prolong the lifespan of the Zn anode. The most common strategies to improve the batteries’ electrochemical performance and suppress the undesired side reaction derived from the electrolyte are mainly categorized into three approaches: highly concentrated electrolyte, hydrogel electrolyte, and functional additives.
3.2.1. Highly Concentrated Electrolyte
The “Water-in-salt” (WiS) electrolyte refers to the electrolyte in which the dissolved salt outnumbers the solvent by both weight and volume; the number of available water molecules that can participate in the ion solvation shell in the WiS electrolyte is far below that in conventional electrolyte
[53][67]. With the limited water content in the electrolyte, the undesired side reactions triggered by the decomposition of H
2O that happened on the surface of the zinc anode were highly reduced
[54][68]. In addition, the interaction of the metal ions in the WiS electrolyte replaces the hydrogen bonds with interionic attractions. The high de-solvation energy related to the metal ions with water molecules was significantly reduced, which can facilitate the plating/stripping process
[55][69]. The WiS electrolyte was first proposed in the field of aqueous lithium batteries by Suo et al. in 2015
[56][70]. With the limited water molecules and improved interionic attractions, the highly concentrated lithium bis(trifluoromethanesulfonyl)imide (LiTFSI)-based WiS electrolyte exhibits a wide electrochemical stable window up to 3.0 V and nearly 100% coulombic efficiency at both low and high charge–discharge rates. When this concept is transferred to ZIBs, super-concentrated electrolytes (such as 1 m Zn(TFSI)
2 with 20 m LiTSFI
[57][71], 1 m Zinc trifluoromethanesulfonate (Zn(CF
3SO
3)) with 21 m LiTFSI
[58][72], and 30 m ZnCl
2 [59][73]) were investigated, and all of these WiS electrolytes exhibit high suppression of dendrite growth and improved zinc anode performance. In addition, the high-concentration electrolyte can also alleviate the dissolution problem of cathode-active materials
[54][68]. But, the introduction of highly concentrated electrolytes will also increase the cost, which will hinder their practical applications.
These DES solutions are receiving more and more attention in ZIBs as electrolytes to improve the full battery performance, and DES electrolytes endow the ZIBs processes with excellent electrochemical performance even at extremely low temperatures. For example, Shi et al. prepared one kind of DES electrolyte by simply introducing and manipulating the molar ratio of water molecules into a ZnCl
2-acetamide electrolyte
[60][78]. The solvation structure of Zn
2+ in the electrolyte was changed, and the de-solvation energy barrier of Zn
2+ was reduced, which can facilitate uniform zinc nucleation during the plating process. With an optimal ratio of ZnCl
2/acetamide/H
2O = 1:3:1, the zinc anode shows a highly improved electrochemical performance, with the CE reaching 98% even after 1000 cycles, and the full battery can achieve high-capacity retention of 85.7% even cycled for 10,000 cycles. They also developed one kind of DES electrolyte composed of 3.5 M Mg(ClO
4)
2 and 1 M Zn(ClO
4)
2, which can be used at ultra-low temperatures
[61][79].
3.2.2. Hydrogel Electrolytes
Hydrogel electrolytes, which can serve both as electrolytes and separators, have attracted significant attention because of their numerous functional groups, good mechanical properties, and notably constrained active water content in comparison to WiS or DES electrolytes. For this type of electrolyte, zinc salt-containing electrolytes are embedded in the network of polymer chains. The possibility of electrolyte leakage, which always happens in liquid electrolytes, can be highly reduced. These features promote the hydrogel electrolyte to be the most promising candidate for flexible energy storage devices. Gel electrolytes based on polyvinyl alcohol (PVA)
[62][84], xanthan gum
[63][85], and carboxymethyl cellulose (CMC)
[64][86] have been investigated a lot, and all of them have shown an inhibition of dendrite formation and improvement in long-term cycling stability. But, the drawback of zinc salt selectivity and poor mechanical properties restrained their further application. However, the hydrogel electrolytes with cross-linking can effectively solve these problems. Gelatin-based hydrogel electrolyte exhibits much stronger mechanical properties compared with the abovementioned gel electrolytes with the help of hydrogen bonds between -OH, -CO, and -NH
2 functional groups
[65][87].
3.2.3. Functional Additives
The introduction of functional additives was recognized to be an economical and effective method to improve the Zn reversibility in ZIBs, and it has been proven that even a very small amount of additives can significantly change the morphology of deposited Zn
[66][90]. The additives can confine the 2D zinc-ion diffusion, regulate the local current distribution, and lower the electrolyte freezing point. According to recent reports, both inorganic ions and organic molecules introduced into electrolytes have shown high efficiency in inhibiting dendrite growth and alleviating the side reaction during the charge/discharge process. The inorganic additives, such as InSO
4,
[67][91] Boric acid,
[67][91] SnO
2,
[67][91] SnCl
2,
[68][92] Na
2SO
4,
[12][69][15,93], and LiCl
[70][94] were used to tune the Zn
2+ deposition process during the plating/stripping cycles. Specifically, the inorganic additives improve the Zn reversibility in the following ways: (1) Manipulating the crystallographic orientation. As discussed in
Section 3.1, the (002) plane has lower thermodynamic free energy and moderate angle alignment to the substrate compared with other planes. During the plating/stripping process, the Zn ions can be deposited uniformly, and the battery will show high electrochemical performance. (2) Electrostatic shielding. Due to the low reduction potential of Li
+ and Na
+, they were adsorbed or accumulated much easier than Zn
2+ ions in the local region with high current density. The first adsorbed metal ions will form an electrostatic shielding layer, and this layer will inhibit the tip effect that prevents the Zn
2+ ions from aggregating at these nuclei.
3.3. Three-Dimensional Structural Skeleton and Alloy Strategies
Building a 3D structural skeleton is another way to effectively restrict dendrite growth and increase the lifetime of the anode
[71][72][101,102]. Since Zn dendrites are mostly derived from the uneven local current density distribution, the 3D electrode or current collector can provide much more electroactive nucleation sites and homogeneous electrical field distribution when compared with the 2D planar electrode. However, certain conditions need to be met for the design of a 3D electrode or current collector. First, the as-prepared 3D substrate should have the advantages of a highly open architecture, high electronic conductivity, high electrochemical stability, and high corrosion resistance. Then, the 3D structure should also possess high hydrogen evolution overpotential. Based on these rules, metal substrates such as Cu
[73][97] and Ni
[74][103] have been employed in this strategy because they have excellent electronic conductivity and high HER overpotentials.

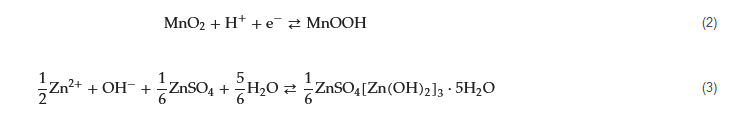
 The HER not only consumes the fresh electrolyte and Zn anode but also produces H2 gas, which will lead to the battery swelling. Interestingly, according to ref. [23][27], a high current density will alleviate the HER during the charge–discharge process. This is because the electrodeposited Zn crystal at high current density has a smaller specific surface area, which is critical for HER activity. As a result, the HER is significantly reduced at high current density.
The HER not only consumes the fresh electrolyte and Zn anode but also produces H2 gas, which will lead to the battery swelling. Interestingly, according to ref. [23][27], a high current density will alleviate the HER during the charge–discharge process. This is because the electrodeposited Zn crystal at high current density has a smaller specific surface area, which is critical for HER activity. As a result, the HER is significantly reduced at high current density.
