1. Synthesis of Fatty Acids
Fatty acids can either be derived from the diet or biosynthesized within the cytosol and endoplasmic reticulum (ER) through an indigenous pathway known as de novo fatty acid synthesis. This synthesis is a complex process influenced by several determinants, including species, transcription genes, dietary composition, age, gender, stored lipids, and both endogenous (metabolic and interactive molecules) and exogenous (environmental) factors. A multitude of genes regulate the synthesis of fatty acids, which can vary among different species. In eukaryotic organisms, nearly 5% of the overall genes are responsible for a significant proportion of lipid structures
[1][255]. Remarkably, the liver X factor (LXR) contributes to the regulation and synthesis of saturated, mono-, and polyunsaturated fatty acids by targeting their transcriptional genes
[2][256]. It also indirectly influences encoding factors involved in lipogenesis, such as sterol regulatory element-binding protein 1c (SREBP1c)
[3][257], peroxisome proliferator-activated receptor gamma (PPAR-γ)
[4][258], and carbohydrate response element-binding protein (ChREBP)
[5][259].
Numerous organisms can produce a wide variety of fatty acids, but only a limited number of molecular structures are synthesized in significant quantities at the natural physiological rate
[6][7][260,261]. Generally, the synthesis activity of fatty acids is relatively low in normal adult cells, with the exception of certain tissues, including the brain, liver, adipose, and lungs
[8][9][262,263]. The liver, known as a lipogenic organ, is predominantly responsible for the de novo pathway, although the white adipose tissue (which consists of lipogenic cells) and mammary glands in animals and humans also possess the capability to produce fatty acids through de novo lipogenesis
[10][11][12][13][264,265,266,267]. Under conditions of energy equilibrium, the liver takes up a remarkable proportion (30–50%) of free fatty acids continually absorbed from the diet. These assimilated lipids are either directly incorporated into phospholipids and triglycerides (TAGs) or subjected to modifications (including elongation and/or desaturation) to produce new/modified fatty acids.
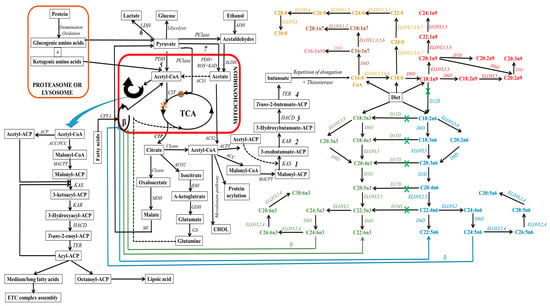
Lipogenic cells can synthesize fatty acids endogenously from non-fat molecules, such as glucose or amino acids (see
Figure 15). In this process, pyruvate, a metabolite of glucose, enters the mitochondria, where it undergoes oxidative decarboxylation through the pyruvate dehydrogenase complex to form acetyl-CoA. Within the mitochondria, acetyl-CoA can also be derived from the degradation of ethanol, proteins (deamination and oxidation), and fatty acids undergoing β-oxidation. Subsequently, these produced acetyl-CoA enter the tricarboxylic acid cycle (TCA, citrate cycle, or Szent–Györgyi–Krebs cycle). This event is very crucial since mitochondrial acetyl-CoA molecules are not permeable to mitochondrial membranes. Thus, they are initially located within the mitochondria, whereby they endure a condensation reaction with oxaloacetate to form citrate, a process catalyzed by citrate synthase during TCA
[14][15][268,269]. The citrate molecule is then expelled from the TCA cycle to the inner mitochondrial membrane, and subsequently to the cytosol. This citrate transporting event requires a dicarboxylate antiporter solute carrier family 25 (SLC25A1)
[16][270].
Figure 15. Schematic depiction of eukaryotic fatty acid biosynthesis and modification pathways, with emphasis on enzyme involvement (in italics). In this schematic, mitochondria are denoted by the red box, while proteasome/ribosome components are indicated by the orange box. However, light orange text delineates the elongation pathway for diverse saturated fatty acids. Within the diagram, both green and blue hues, accompanied by corresponding colored arrows, elucidating the discrete pathways for n3 and n6-fatty acid synthesis, respectively. On the other hand, text with color spectrum transitioning from pink to red designates the pathways for the synthesis of various monounsaturated fatty acids. The green “X” indicates the unattainability of this pathway in mammals, particularly higher eukaryotes, owing to the absence of a specific enzyme. ELOVL8 is a fish-specific elongase. Abbreviations: 1, condensation; 2, reduction; 3, dehydrogenation; 4, reduction; 5, aerobic conditions; 6, hypoxia or anaerobic conditions; 7, aerobic conditions; ACC, acetyl-CoA carboxylase; ACP, acyl carrier protein; ACPT, acyl carrier protein transacylase; ACS1, acetyl-CoA synthetases-1; ACS2, acetyl-CoA synthetases-2; ADH, alcohol dehydrogenase; ALDH, aldehyde dehydrogenase; ACO2, aconitase; β, beta oxidation; CHOL, cholesterol; CIT, citrate synthase; Clyase, citrate lyase; CPT-I, carnitine-palmitoyl transferase-I; CTP, citrate transporter protein, EAR, enoyl-ACP reductase; ETC, electron transport chain; GDH, glutamate dehydrogenase; GS, glutamine synthetase; HACD, β-hydroxyacyl-ACP dehydrase; IDH, isocitrate dehydrogenase; KAD, keto acid dehydrogenase; KAR, β-ketoacyl-ACP reductase; KAS, β-ketoacyl-ACP synthetase; MACPT, malonyl-CoA:ACP transacylase; MDH, malate dehydrogenase, MI, malic enzyme, PCase, pyruvate carboxylase; PCC, propionyl-CoA carboxylase; PDH, pyruvate dehydrogenase; ROS, reactive oxygen species; TCA, tricarboxylic acid cycle; TER, trans-enoyl-ACP reductase; O, oxaloacetate; *, citric acid).
In the cytosol, citrate can undergo distinct metabolic pathways to generate various metabolites (see
Figure 15). For instance, adenosine triphosphate (ATP)-citrate lyase enzymatically cleaves citrate into acetyl-CoA, which leads to the carboxylation of acetyl-CoA and the formation of malonyl-CoA. Within cellular cytoplasm, acetyl-CoA (an active form of acetate) and malonyl-CoA serve as the primary substrates that initiate the process of carbon chain elongation. It is worth noting that propionyl and short-branched acyl units for priming can also be utilized in specific cases. This occurs, for instance, when adipose tissue contains monomethyl-branched fatty acids
[17][271]. Additionally, the elongation process during the synthesis of branched fatty acids in specific glands requires the incorporation of methylmalonyl units
[18][272].
It is imperative to emphasize the critical role played by the acyl carrier protein (ACP), which binds to acetyl-CoA and malonyl-CoA, a process catalyzed by malonyl-CoA:ACP transacylase. This integration event facilitates cytosolic elongation in higher eukaryotes by sequentially transferring these substrates from one enzyme/enzyme domain to another throughout sequential biosynthetic cycles. This active participation of ACP is vital for fatty acid biosynthesis and the functions of fatty acid synthase (FAS), which is a multi-enzyme system regulated by the encoded FASN gene. This cytosolic de novo pathway comprises a series of reactions catalyzed by acetyl-CoA carboxylase (ACC) and FAS, which serve as rate-limiting enzymes
[19][20][21][273,274,275]. These reactions include ATP-dependent carboxylation of acetyl-CoA to form malonyl-CoA, Claisen condensation to extend malonyl-ACP and form 3-oxobutanoate, ketoreduction to yield 3-hydroxybutanoate, dehydration to yield butenoate, enoyl reduction to yield butanoate, and repeating elongation reactions (see
Figure 15). This process results in the elongation of carbon chains up to the length of C16 or C18 in the cytosol
[22][23][24][276,277,278].
ACC, the rate-limiting enzyme in the de novo pathway, facilitates the irreversible decarboxylation of acetyl-CoA through the addition of CO
2 to produce malonyl-CoA. The resulting malonyl-CoA attaches to ACP and also serves as a two-carbon donor within a cyclic sequence of reactions facilitated by FAS, leading to the generation of a variety of fatty acid species. The end products (acyl-ACP) of cytosolic de novo synthesis are primarily palmitic acids (C16:0), with lower extents of myristic (C14:0) or stearic (C18:0) acids originating from acetyl-CoA. The determination of chain length during cytosolic de novo biosynthesis involves three enzymes: acyltransferases, ketosynthases, and thioesterases
[25][279]. It is essential to highlight that there are two ACC isoforms: ACC1 (also known as ACCα), which is highly expressed in adipose and hepatic tissues, and ACC2 (also known as ACCβ), which is highly expressed in the heart and skeletal muscles
[26][280]. The mechanism for the conversion of acetyl-CoA to malonyl-CoA by ACC is suggested to differ depending on the ACC type due to their different expression locations
[27][281]. ACC1 is a cytosolic enzyme, whereas ACC2 is located in the outer mitochondrial membrane. On the other hand, FAS enzymes are exclusively cytosolic and can catalyze the formation of C16:0 from acetyl-ACP (substrate) and malonyl-ACP (2-carbon donor)
[20][21][274,275]. FAS comprises numerous large-multifunctional protein domains (type I FAS) in eukaryotic and specific bacteria, while a monofunctional polypeptide domain is present in the case of major bacteria (type II FAS)
[25][28][29][30][31][279,282,283,284,285].
In animals, the FAS pathway undergoes termination through a process involving a thioesterase, resulting in the liberation of the free fatty acid as the final product. The termination of the repeating elongation process exhibits the greatest degree of variation in fatty acid biosynthesis. Nevertheless, the conversion of the cytosolic elongated product to the CoA-ester is vital for further biosynthetic pathways that generate new fatty acid structures.
1.1. Elongating Fatty Acids through Non-Cytosolic Mechanisms
Generally, the incorporation of the product into lipid structures or its involvement in subsequent elongation and/or desaturation processes is contingent upon the specific requirements of the organism at a given time. Further elongation pathways are not exclusive to fatty acids derived from de novo fatty acid synthesis in the cytosol; they also act on fatty acids derived from the diet, further extending and/or desaturating them to produce longer saturated, monounsaturated, or polyunsaturated fatty acids that are vital for all biomembranes. Following the production of palmitic acid in the cytosol, further modifications of this fatty acid may occur within cell organelles. In mammals, these modifications involve elongation (chains of 18 carbons or longer) and/or desaturation (formation of monenoic/monounsaturated fatty acids).
The ER and mitochondria regulate the modification (elongation) of fatty acids
[24][278] in order to provide sufficient specific signals and functions. Already existing saturated fatty acids are elongated by the sequential addition of two carbon atoms, resulting in the formation of new fatty acids
[32][286]. The principal fatty acid elongation pathway at the cytosolic side of the ER involves a series of four independent reactions: (1) condensation, (2) reduction, (3) dehydration, and (4) a final reduction step
[33][287]. Major enzymes involved in the elongation process include 3-ketoacyl-CoA synthases (elongase enzymes or ELOVLs for reaction 1), 3-ketoacyl-CoA reductase (for reaction 2), hydroxyacyl-CoA dehydratase (for reaction 3), and
trans-2,3-enoyl-CoA reductase (for reaction 4). The ELOVL family, sometimes referred to as type III FAS, serves as the rate-limiting enzyme family in the elongation pathway. This family consists of seven subtypes in mice, rats, and humans, and their regulation is governed by ELOVL-encoded genes.
ELOVLs catalyze the condensation of acyl-CoA and malonyl-CoA, which is responsible for elongating fatty acids and determining their carbon chain length, thereby influencing the cell’s fatty acid composition and signaling. ELOVLs exhibit variation in substrate specificity, tissue distribution, and regulation
[34][288]. Based on their final products (see
Figure 15), ELOVLs are classified into groups: ELVOLs that elongate saturated and monounsaturated fatty acids (ELOVL1, 3, 6, and 7), ELVOLs that produce very long-chain polyunsaturated fatty acids (ELOVL2 and 4), ELVOL5, which acts on a wide range of substrates with carbon chains ranging from 16 and 22, and ELOVL8, which acts on a wide range of substrates with carbon chains ranging from 16 to 20. ELOVL8 is a distinct subtype that has been recently discovered but is believed to be specific to fish
[35][289]. It is widely acknowledged that these genes are primarily regulated at the transcriptional level; however, additional regulatory mechanisms may exist, including allosteric inhibition. In mammals, ELOVL4 is the sole enzyme capable of catalyzing the formation of fatty acids with more than 26 carbons
[36][290]. These polyunsaturated fatty acids with more than 28 carbon atoms are primarily found in the retina, brain
[37][38][39][40][291,292,293,294], testis
[41][42][295,296], spermatozoa
[43][297], epidermis
[44][298], meibomian gland
[45][46][299,300], and Vernix caseosa
[47][301].
An additional pathway for elongating fatty acids occurs in non-cytosolic fatty acid synthesis, especially in the mitochondria. Both animals and yeasts possess mitochondria that contain FAS II enzymes (mtFAS II), which appear to interact with ACP-linked molecules
[48][49][302,303]. It should be noted that the ACC enzyme has not been identified in the mitochondria of most mammalian species, including humans. However, a recent isoform of ACC1 has been identified in the mitochondria of mice
[50][304]. Thus, isoforms of ACC1, and potentially mitochondrial propionyl-CoA carboxylase
[51][305], are believed to regulate the decarboxylation of acetyl-CoA to yield malonyl-CoA within mammalian mitochondria. Nevertheless, these reactions occur at a limited rate, suggesting that imported malonate may play a role in mitochondrial fatty acid synthesis.
Mitochondrial fatty acid elongation relies on nicotinamide adenine dinucleotide phosphate (NADPH)-dependent enoyl-ACP reductase, with acetyl-ACP and acyl-ACP serving as substrates
[52][306]. This pathway appears to be energetically unfavorable and represents a minor pathway in eukaryotes
[24][278], primarily contributing to the generation of fatty acids used in the lipogenesis of mitochondrial membranes and cellular respiration. Unlike animals, where thioesterase-mediated termination is involved, mitochondrial termination entails channeling the mitochondrial acyl-ACP into the lipid biosynthetic pathway
[53][307]. The primary generated product of mtFAS II activity is an octanoyl chain, which serves as a substrate for lipoic acid synthesis—a vitamin that acts as a scavenger for free radicals
[54][55][56][308,309,310] and enhances energy metabolism as a cofactor
[57][311]. Although this pathway can also generate medium and long fatty acids
[58][312], their exact biological roles remain uncertain. However, studies by Nowinski et al.
[10][264] and Angerer et al.
[59][313] have suggested that these mitochondrial long-chain fatty acids are involved in the electron transport chain (ETC) complex assembly.
Furthermore, a similar pathway for fatty acid elongation is proximal fatty acid elongation, which is characterized by reversible β-oxidation. In this pathway, acetyl-CoA acts as the carbon donor, and peroxisomal
trans-2-enoyl-CoA reductase substitutes acyl-CoA dehydrogenase to facilitate a thermodynamically favorable reaction
[60][314]. It is important to emphasize that CoA is implicated in the fatty acid catabolism of the reversible pathway, while ACP plays a role in mitochondrial fatty acid elongation. However, the precise functions of fatty acid elongation within peroxisomes remain insufficiently characterized from a scientific standpoint. In practice, the extent of elongation is typically assessed using the elongase estimated index, which is determined by the ratio of C16:0 to C18:0.
1.2. Desaturation of Fatty Acids
Within the context of de novo fatty acid synthesis, a process characterized by the removal of two hydrogen atoms to create a double bond often intersects with the fatty acid elongation pathway. This synergy ensures the production of long-chain and very long-chain unsaturated fatty acids, both mono- and polyunsaturated. Notably, the enzymes responsible for fatty acid desaturation, known as fatty acid desaturases, are ubiquitous across all domains of life with the exception of archaea, where they are notably absent
[61][315]. However, it is of significant importance to underscore that the synthesis of polyunsaturated fatty acids can also occur independently of the classical series of desaturase and elongase enzymes. Several studies, including those conducted by Smith and Tsai
[62][316], Kaulmann and Hertweck
[63][317], Napier
[64][318], and Metz et al.
[65][319], have extensively documented an alternative pathway for the biosynthesis of long-chain polyunsaturated fatty acids in both prokaryotes and lower eukaryotes. This alternative route relies on semi-fatty acid synthesis systems, specifically known as polyketide synthases (PKS). PKS employs the same four fundamental reactions as FAS. However, the PKS cycle is frequently condensed, resulting in the formation of highly modified carbon chains featuring numerous keto and hydroxy groups, along with trans-configured double bonds that exhibit various functional roles
[66][67][320,321].
Among the plethora of desaturase families found in different species, researchers have categorized them into three distinct types, as described by Cerone and Smith
[68][322]. The first family, acyl-acyl carrier protein desaturase, is exclusive to plastids of higher plants. The second family, acyl-lipid desaturases, is found in the ER membranes of higher plants and cyanobacteria. The third family is the family of acyl-CoA desaturases, which can be present in both eukaryotes and prokaryotes, and these enzymes use a cytochrome b5-like system during oxygen reactions
[69][323]. Within the context of this review, with a primary emphasis on mammals, the discussion will be specifically on the acyl-CoA desaturase families. In mammals, a critical in vivo biosynthetic route for the production of long-chain polyunsaturated fatty acids is known as the ‘Sprecher pathway’
[70][324]. This pathway relies on two fatty acid desaturase enzymes, two ELOVLs, and a peroxisomal β-oxidation process.
The desaturase pathway encompasses diverse enzyme families, such as stearoyl-CoA desaturase (SCD)
[71][325] and fatty acid desaturase enzymes (FADS)
[72][326]. Each of these enzymes acts on distinct substrates. For example, FADS primarily targets polyunsaturated substrates, while SCD predominantly acts on saturated substrates. SCD, also referred to as delta-9 desaturase (∆9-desaturase, or D9D), is an ER enzyme that catalyzes the formation of monounsaturated fatty acids, including palmitoleic (C16:1 n7) and oleic (C18:1 n9) acids, from saturated fatty acids, such as palmitic and stearic acids, respectively. These enzymes exhibit varying specificities and can work on a range of fatty acids with different carbon chain lengths, from 16 to 26 carbon chains. This variation hinges on the specific isoform present, with some species harboring multiple homologues of D9D. For instance, two homologues (D9D-1 and D9D-2) have been identified in marine copepods
[73][327].
Basically, within the D9D reaction, a double bond is introduced at the 9th position from the carboxyl group of the fatty acid. In addition, a multitude of desaturase enzymes present in plants, bacteria, and insects perform the initial double bond introduction on saturated fatty acids at various positions, including ∆3, ∆5, ∆7, and ∆11
[74][75][76][77][78][328,329,330,331,332]. The activity of D9D is modulated by dietary fatty acid intake and is subject to up-regulation by the expressions of SCD gene isoforms
[79][333]. Since Bloch’s 1960 pioneering discovery of SCD
[61][315], a plethora of gene isoforms have come to light. In mice, the SCD exhibits four distinct isoforms (SCD-1–4)
[80][334], whereas in humans, only two isoforms (SCD-1 and SCD-5) have been identified
[81][335]. These gene isoforms can vary in substrate preference, ∆ position, and double bond conformation
[82][336]. SCD-1 is the most studied isoform among them
[71][325]. The importance of the SCD pathway is underscored by its involvement in cellular stress, lipid metabolism, inflammation, and autoimmunity. Among the isoforms, SCD-1 is the most extensively studied and is associated with various physiological processes
[83][84][85][86][87][88][337,338,339,340,341,342].
Fatty acids featuring a ∆9 double bond are eligible for elongation, but it is important to note that these fatty acids may also be derived from the diet. However, D9D activity is indirectly determined by assessing the ratio between C18:0 and oleic acid. Notably, the de novo elongation of oleic acid results in the formation of longer chains of monounsaturated fatty acids. Furthermore, oleic acid, in particular, may undergo a desaturation process often followed by elongation, where the double bond is introduced before the ∆9 position. Nevertheless, unlike in plants and a limited number of animal species, mammals lack the enzymatic capability to catalyze the introduction of the second double bond in oleic acid, particularly not after the Δ9 position.
Fatty acid desaturase genes (FADs) comprise a gene family responsible for encoding a variety of desaturase enzymes. These enzymes play a significant role in catalyzing the synthesis of polyunsaturated fatty acids by introducing multiple double bonds at positions within fatty acids. Among the genes involved in fatty acid desaturation, mammals have been identified with four distinct FADs
[81][335]: (1) FAD-1, (2) FAD-2, (3) FAD-3, and (4) FAD-6. Each of these FAD types tends to have alternative transcriptions
[89][90][91][343,344,345], which can express distinct desaturase activities at different ∆-positions. Generally, FAD enzymes can be categorized into FAD-1, responsible for generating omega-3 (n3) fatty acids; FAD-2, involved in generating omega-x (nx) fatty acids; and FAD-3, which contributes to the formation of omega-6 (n6) fatty acids. While little is known about the substrates of FAD-6 and their major roles have not yet been defined, it appears to be homologous to human FAD-2
[92][93][94][346,347,348] and likely plays a role in the synthesis of polyunsaturated fatty acids.
The transcription factor FAD-6 has been described to possess Δ4-, Δ5-, and Δ8-desaturation activities, with a notable impact on docosahexaenoic acid (C22:6 n3, or DHA) synthesis from n3-docosapentaenoic acid (C22:5 n3, or DPA-n3) in golden pompano fish
[95][349]. On the other hand, Park et al.
[91][345] detailed the existence of nine alternative transcriptions for FAD-3, potentially arising from splicing events. Initially identified through cloning efforts by Marquardt et al.
[96][350], FAD-3 has been traditionally classified as a non-polyunsaturated desaturase, primarily due to its limited substrates, namely, vaccenic acid (C18:1 trans-11) and sphingoid bases. In this regard, it should be noted that FAD-3 may facilitate the unanticipated ∆13-desaturation of trans-vaccenate
[97][351]. These limited substrates give rise to the production of 11E,13Z-octadecadienoic acid
[98][352] and 4E,14Z-sphingodienine
[99][353], respectively.
In contrast to FAD-3, FAD-1 and FAD-2 are the principal contributors to the biosynthesis of polyunsaturated fatty acids in mammals. Nevertheless, mammals lack two crucial desaturases, namely delta-12 desaturase (∆12-desaturase, or D12D) and delta-15 desaturase (∆15-desaturase, or D15D)
[100][101][102][354,355,356]. These enzymes are often referred to as “methyl-end desaturases” due to their ability to introduce a new double bond between an existing unsaturated bond and the methyl terminus (–CH
3) of the fatty acid. Thus, mammals are de novo incapable of introducing a new double bond after the ∆9 position of oleic acid. Hence, mammals must obtain polyunsaturated fatty acids from their diet, leading to the concept of essential fatty acids. These essential fatty acids, such as linoleic acid (C18:2 n6, or LA) and α-linolenic acid (C18:3 n3, or ALA), cannot be endogenously synthesized by mammals and must be sourced from dietary intake
[103][104][105][357,358,359]. Nonetheless, the enzyme activities of FAD-1 (∆5-desaturase, or D5D) and FAD-2 (∆6-desaturase, or D6D) have been identified in mammals. These enzymes are responsible for introducing double bonds at the ∆5 and ∆6 positions, respectively
[106][107][360,361]. Notably, both D6D and D6D are highly expressed in the liver, with D6D exhibiting particularly high expression levels
[108][362].
In mammals, dietary LA, ALA, and other unsaturated fatty acids, whether from the diet or synthesized endogenously, serve as substrates for the generation of polyunsaturated fatty acids characterized by an increased number of double bonds and longer carbon chains. The enzyme D6D plays a crucial role in the initial steps of synthesizing arachidonic acid (C20:4 n6, or AA), eicosapentaenoic acid (C20:5 n3, or EPA), and DHA by catalyzing the conversion of LA and ALA into γ-linolenic acid (C18:3 n6) and stearidonic acid (C18:4 n3), respectively
[108][362]. However, the biosynthesis of AA and EPA, in particular, involves an additional desaturase enzyme, delta-5 desaturase (D5D), which utilizes the substrates dihomo-γ-linolenic acid (C20:3 n6, or DGLA) and eicosatetraenoic acid (C20:4 n3) to yield AA
[106][360] and EPA
[109][363], respectively. According to Vagner and Santigosa
[110][364], the substrate affinities of D6D appear to exhibit contrasting and debatable characteristics. Consequently, further investigations are imperative to substantiate a definitive conclusion. It is essential to emphasize that the distinctive substrate affinities of D6D play a critical role in determining the ratio of n6- to n3-polyunsaturated fatty acids (referred to as n6/n3 or n6:n3). The role of FAD-6 in determining the ratio of n6/n3-polyunsaturated fatty acids and its influence on the metabolic flux of these fatty acids have been highlighted
[111][112][365,366].
Remarkably, D6D and D5D are also involved in the synthesis of n9-polyunsaturated fatty acids, specifically, Mead acid (C20:3 n9), which is produced in the absence of LA and ALA
[113][367], when only monounsaturated fatty acids are available as substrates. Elevated levels of Mead acid are matched with the proportional depletion of n6- and n3-polyunsaturated fatty acids and serve as a biomarker for LA and ALA deficiency in diets. For instance, the ratio between trienoic and tetraenoic acids (such as Mead acid:AA) functions as a biomarker for the lack of dietary essential fatty acids
[114][115][368,369]. Park et al.
[116][370] have identified two pathways for Mead acid production, involving D6D and D5D. These pathways vary in substrates of D6D, which can either utilize oleic acid or gondoic acid (C20:1 n9) to yield C18:2 n9 and C20:2 n9, respectively. D5D catalyzes the direct conversion of C20:2 n9 into Mead acid by introducing a double bond at the ∆5 position. Furthermore, authors have also reported the novel activity of ∆7-desaturase (D7D, regulated by FAD-1), leading to the formation of C20:2 n9 from C20:1 n9.
Notably, AA, EPA, and DHA are biologically essential fatty acids with enormous contributions to membrane phospholipids. In the Sprecher pathway, the primary fatty acid synthetic pathway in mammals, the biosynthesis of DHA from EPA is not direct but rather involves a series of reactions: (1) elongation to a 24-carbon chain, (2) desaturation via D6D, and (3) peroxisomal β-oxidation for chain shortening. However, the synthesis of n6-docosapentaenoic acid (C22:5 n6, or DPA-n6) and DHA in eukaryotes also occurs through ∆4-desaturase (D4D) pathways, depending on the species. In lower eukaryotes, marine vertebrates, and humans, D4D (encoded by the FAD-2 gene), which is expressed to a lesser extent, plays an essential role in introducing a double bond at position ∆4, directly producing DPA-n6 and DHA from docosatetraenoic acid (C22:4 n6, or adrenic acid) and DPA-n3 substrates, respectively
[117][118][119][120][371,372,373,374]. It is noteworthy that this reaction bears resemblance to that facilitated by FAD-6 expression, suggesting the possibility of FAD-6 up-regulating D4D activities.
A further marked expression of FAD-2 identified within mammalian cells is ∆8-desaturase (D8D). This enzyme establishes an autonomous pathway distinct from D6D, introducing an additional double bond to substrates like ALA, LA, and their elongated polyunsaturated fatty acids, resulting in the formation of very long-chain polyunsaturated fatty acids
[121][122][375,376]. For instance, the emergence of D8D was observed when DGLA and eicosatetraenoic acid were derived from eicosadienoic acid (C20:2 n6) and eicosatrienoic acid (C20:3 n3), respectively
[123][377]. In a study conducted on mouse liver, D8D expressed low activity, which is primarily associated with n3-unsaturated fatty acids, such as eicosatetraenoic acid (C20:4 n3), EPA, DPA-n3, DHA, and nisinic acid (C24:6 n3)
[121][375].
In general, more than a hundred FAD-2-related desaturase enzymes have been identified in various animal species, although they are notably absent in mammals. For instance, the ∆17 (17-desaturase, or D17D) and ∆19 (19-desaturase, or D19D) desaturases have been identified in lower animal families (excluding mammals) and algae
[124][125][378,379]. These enzymes play an essential role in the production of EPA and docosapentaenoic acid-n3 (C22:5 n3, or DPA-n3) from AA and adrenic acid, respectively. The activities of enzymes involved in lipogenesis are subject to intricate regulation by a matrix of genes and exogenous factors. Notably, polyunsaturated fatty acids have been shown to inhibit the transcription of hepatocellular genes responsible for encoding lipogenic enzymes
[126][127][380,381]. It is a common practice to assess desaturase enzyme activity indirectly by determining their coefficients based on the ratio of the fatty acids generated to the substrates utilized.
Desaturase enzymes exhibit a broader scope of activities beyond their involvement with fatty acid substrates incorporated into phospholipids, as they are also active in sphingolipids (SLs). Ordinarily, SLs are characterized by their predominantly very long saturated or monounsaturated nature. The determination of fatty acid chain length within SLs is intricately governed by the activities of the ceramide synthase (CerS) type
[128][382]. However, it is worth noting that polyunsaturated fatty acids show a slight accumulation in SLs within testes and spermatozoa
[129][130][383,384] as compared to phospholipids within the same tissues. The classification of desaturases responsible for introducing double bonds into ceramide (Cer) structures has been presented by Nachtschatt et al.
[131][385]. This classification delineates three distinct categories of desaturases: (1) α-hydroxylases
[132][386], (2) D4D and C4-hydroxylases
[133][387], and (3) D8D
[134][388]. These desaturases play a pivotal role in diversifying the composition of SLs, particularly in terms of the introduction of double bonds, which contribute to the functional and structural heterogeneity of these important lipid molecules.
2. Incorporation of Fatty Acids into Lipids of Biomembranes
Fatty acid synthesis and their subsequent incorporation into biomembranes represent critical processes in the life of all organisms. The one exception to this rule is the archaea domain, which employs isoprenoids as membrane lipid side chains rather than fatty acids
[135][389]. Understanding how fatty acids are incorporated into membrane lipids is of paramount importance. Fatty acids may become part of phospholipids either through acylation of glycerol-3-phosphate during the biosynthesis of phospholipids or through the action of lysophosphatidic acyltransferases and phospholipases that remodel the structure of pre-existing phospholipids
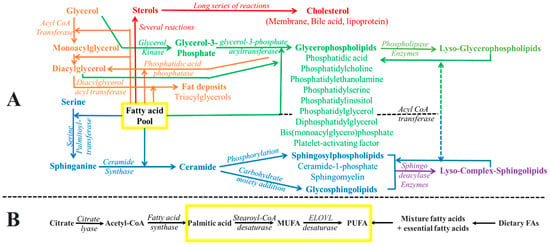
[136][137][390,391] (which can be seen in
Figure 26).
Figure 26. Schematic representation of (A) fatty acids incorporated into various lipids and (B) the origins of fatty acid pool formation (the de novo pathway and dietary sources). The depiction employs color coding to symbolize distinct metabolic pathways. The yellow box designates the fatty acid pool, signifying its integration into diverse membrane lipids. The brown shade denotes processes related to non-membrane and non-polar lipid formation. The red color represents the metabolic routes responsible for sterol production. Additionally, the green color signifies the integration of fatty acids into various phospholipids, while the blue hue corresponds to the incorporation of fatty acids into diverse sphingolipids.
The vital nature of these processes is underscored by the coexistence of diverse fatty acid structures within complex biomembrane lipids
[138][392]. This structural diversity arises from various factors, including the preferences of
sn-positions for particular fatty acids, the substrate preferences of biosynthetic enzymes, and the dynamic lipid remodeling program. Notably, studies by Shindou et al.
[139][393] and Coleman et al.
[140][394] have elucidated the preferences of specific enzymes for distinct fatty acids. For instance, acyl-CoA synthetase long-chain family members 3 and 4 demonstrate preferences for AA and EPA, while 1-acylglycerol-3-phosphate O-acyltransferase-α prefers myristic acid, palmitic acid, and LA. Meanwhile, 1-acylglycerol-3-phosphate O-acyltransferase-β exhibits a preference for AA.
In the context of lipid remodeling, the replacement of fatty acids within existing phospholipids plays a central role. For instance, the incorporation of C20 fatty acids generally takes place post-de novo synthesis, necessitating the employment of the remodeling pathway
[141][395]. Hence, this process involves the conversion of one distinct phospholipid into another
[142][85], highlighting the importance of the Lands cycle in this process. The Lands cycle
[143][144][396,397] is a central component of the remodeling process, enabling the attainment of specific structural configurations required for the generation of particular cellular signals. In this pathway, phospholipases initiate cleavage (deacylation) of fatty acids from phospholipids, resulting in the formation of free fatty acids and lysophosphatidate. On the other hand, acyltransferases function in a contrasting manner, acylating the requisite fatty acids into lysophosphatidate to generate phospholipids. This process is CoA-dependent, which is primarily due to the extensive utilization of CoA intermediates as substrates. Other remodeling pathways include the CoA-independent transacylation pathway and the direct transacylation pathway, which are specifically suited for highly unsaturated fatty acids such as AA, EPA, and DHA. In these pathways, transacylases catalyze the migration of fatty acids between molecular species of phospholipids. Remarkably, these remodeling pathways exhibit substantial variability across different tissues
[141][395], as comprehensively reviewed
[136][390].
Additionally, it is noteworthy that fatty acids are predominantly incorporated into SLs by the rate-limiting enzyme CerS. However, a remodeling mechanism can also come into play, modulating specific signaling and functional roles. For example, the work of Markham et al.
[145][398] suggests that the accumulation of very long-chain fatty acids contributes to the formation of microdomains through increased hydrophobicity, membrane leaflet interdigitation, and the transition from a fluid to a gel phase. These structural transformations have significant implications for cellular function and signaling within membranes.
The movement of membrane phospholipids between bilayers involves the activities of various transmembrane lipid transporter proteins, namely, flippase, floppase, and scramblase. These proteins play distinct roles in the translocation of lipids and exhibit differential mechanisms of action. For instance, flippase facilitates the translocation of lipids from the exo-leaflet to the inner-leaflet, contrasting with the activity of floppase, which operates in the opposite direction, moving lipids from the inner-leaflet to the exo-leaflet. Notably, both flippase and floppase depend on ATP as an energy source for their functions. Conversely, scramblase functions as an ATP-independent transporter and orchestrates the bidirectional movement of lipids across membranes in a non-specific manner, allowing for the concurrent translocation of lipids from the inner to the outer leaflet and from the outer to the inner leaflet. Comprehensive insights into these membrane fatty acid transporters have been provided by the extensive reviews conducted by Samovski et al.
[146][399] and Glatz et al.
[147][16]. These reviews offer a detailed discussion of the mechanisms and significance of these proteins in lipid translocation processes within biological membranes.
3. Fatty Acid Composition in Biomembranes
The fatty acid composition of biomembranes plays a critical role in maintaining cellular homeostasis and ensuring proper functionality. It is worth noting that this composition is not static but can be modified/remodeled in response to homeoviscous adaptation. This process was initially described in algae
[148][400] and has been later extended to non-homeothermic animals. In contrast, mammals are generally considered to have stable biophysical properties and lipid order within cellular membranes, but they can respond to changes in their dietary fatty acid compositions. Furthermore, certain mammalian cells potentially exhibit a lower degree of homeoviscous adaptation in response to the membrane curvature elastic stress
[149][401]. The fact that there is not a single standardized composition for fatty acids in biomembranes should not be viewed as a flaw but rather as an indication of the intricate and dynamic nature of biological systems. The complexity and adaptability of fatty acid composition in biomembranes underscore the potential evolutionary advantage conferred by the ability to alter lipid structures.
The available literature, which will be discussed in subsequent sections, provides extensive data regarding the proportions of specific fatty acids in various lipid complexes within biomembranes. This section merely focuses on the major fatty acids identified in phospholipids and SLs, as these fractions are the central aspects of this review. Regardless, the length and degree of saturation of the fatty acyl side chain in cholesterol can vary between different cells. This variation is primarily influenced by dietary factors and cell metabolism, and it has a direct impact on the stability and fluidity of the cellular membrane.
3.1. Fatty Acid Profile of Phospholipids
Commonly, the acyl chains of phospholipids found at the
sn-1 and
sn-2 positions of the glycerol backbone are typically composed of a saturated fatty acid (such as C16:0 or C18:0) and an unsaturated fatty acid (with carbon chains of 18, 20, 22, or 24), respectively. Furthermore, these acyl chains exhibit variations in terms of their length, the number of double bonds, and the position of hydrogen atoms adjacent to these double bonds (whether in
cis or
trans configuration). It is important to note that
trans-double bonds are relatively rare within mammalian membranes, while
cis-double bonds are frequently abundant. In fact, the occurrence of
trans-double bonds in mammals is far less frequent than in bacteria
[150][151][402,403]. In some instances, identical acyl chains can be found at both
sn-1 and
sn-2 positions of glycerol
[152][153][404,405]. It is worth highlighting that even when the number of carbons is the same, a slight mismatch may occur due to the
sn-positions. This mismatch is a consequence of the
sn-2 chain bending perpendicular to the membrane’s plane
[154][155][406,407].
In the case of palmitic acid (PA), the composition of the two fatty acids can vary across different cell types and constituents. Typically, dietary fatty acids and metabolic disorders play a substantial role in determining the composition of these acyl chains. PA is primarily composed of saturated and monounsaturated fatty acids, with carbon chains ranging from 14 to 24. Concerning phosphatidylglycerol (PG), this class exhibits variations in the length and number of unsaturation of its fatty acids, depending on the cell type and the dietary fatty acids involved. In the PG of rat lungs, palmitic acid is the most abundant fatty acid, with unsaturated fatty acids constituting a smaller portion
[156][408]. PG is unique among phospholipids due to its higher degree of unsaturation, with unsaturated fatty acids more likely to occupy the
sn-1 position. Research by Xie et al.
[157][409] suggests that the degree of unsaturation in PG may have varying effects on mouse keratinocyte proliferation.
Commonly, the fatty acid composition of phosphatidylcholines (PCs) is typically determined post-synthesis, with various events, such as deacylation and reacylation, taking place during its remodeling
[143][144][396,397]. These events, part of the Lands cycle, can also impact the composition of other phospholipids due to homeostatic mechanisms or metabolic implications
[158][410]. Generally, PC exhibits variations in its fatty acid composition across species and cell types
[159][160][161][162][163][164][411,412,413,414,415,416]. Saturated fatty acids, like palmitic or stearic acids, are typically abundant at the
sn-1 position, while C18 unsaturated chains or longer polyunsaturated fatty acids like AA and DHA are more prevalent at the
sn-2 position.
The fatty acid composition of phosphatidylethanolamine (PE) is highly dependent on the particular cell, tissue, and physiological conditions. In contexts like chicken egg, rat liver, and brain, palmitic and stearic acids tend to occupy the
sn-1 position, while AA, oleic, and DHA are more common at the
sn-2 position
[159][160][165][411,412,417]. Notably, PE in the erythrocyte membrane exhibits a greater tendency for the accumulation/recruitment of polyunsaturated fatty acids compared to PC
[166][418]. Consequently, PE in this context contains more polyunsaturated fatty acids, primarily AA and DHA, at the
sn-2 position, despite its diacyl structure bearing similarities to that of PC.
The composition of fatty acids in phosphatidylserine (PS) differs depending on the tissue type. Nonetheless, lipid remodeling and selective insertion of fatty acids are common processes that occur along the biosynthetic pathway. Therefore, the fatty acid composition of newly synthesized PS differs from that of its precursors, PE and PC. When PS was isolated from rat liver and cow brain and analyzed regiospecifically
[160][162][412,414], it was observed that stearic acid was more abundant at the
sn-1 position, while the proportion of palmitic acid was lower here. These data underscore the distinct fatty acid incorporation pattern exhibited by PS in comparison to PC and PE. Furthermore, the acylation of fatty acids at the
sn-2 position has been shown to be tissue-specific, with high proportions of AA and DHA in the livers of rats and the brains of bovines, respectively. Similar findings regarding DHA in brain regions of mice and rats have been reported by Kim et al.
[167][419] and Hamilton et al.
[168][420]. However, stearic acid was the most abundant fatty acid at the
sn-1 position in these cases. Remarkably, the incorporation of these unsaturated fatty acids into PS appears to be more extensive than in the case of PC, as revealed by these studies. The fatty acid composition of PS at its
sn-positions plays a crucial role in determining its functional properties to varying degrees
[169][170][93,421].
In mammalian cells, the composition of phosphatidylinositol (PI) is characterized by the presence of stearic acid and AA in the
sn-1 and
sn-2 positions, respectively, as consistently demonstrated in various studies
[159][160][171][172][411,412,422,423]. These two fatty acids collectively account for a substantial portion of PI acyls, typically ranging from 50% to 80% in the brain and liver. Additionally, oleic acid is frequently identified as the third most abundant fatty acid in the rat brain, while palmitic acid and DHA exhibit lower levels of acylation in this phospholipid. However, it is worth noting that Ulmann et al.
[173][424] reported a distinct fatty acid composition in the rat brain, where oleic acid was the most prevalent, followed by stearic and palmitic acids. This variability in PI composition across studies may be attributed to a range of factors, including dietary influences and genetic variations. In general, PI exhibits a strong preference for AA in its acyl composition. Remodeling processes can lead to the deacylation of pre-existing PI, resulting in the formation of lyso-PI via the action of phospholipases. The incorporation of AA into lyso-PI is facilitated by lysophosphatidylinositol acyltransferase 1
[174][425].
The structure of fatty acids within cardiolipin (CL), which is also referred to as diphosphatidylglycerol (DPG), greatly influences its shape and properties. Saturated chains tend to produce lamellar forms of CL, whereas unsaturated chains contribute to non-lamellar structures
[175][38]. Thus, the distinctive fatty acid composition of CL is believed to play a critical role in its diverse biological functions across different cellular and subcellular membranes. CL remodeling is an essential process for CL formation, during which LA and DHA are primarily incorporated, influencing susceptibility to oxidation
[176][177][178][179][426,427,428,429]. LA has been reported as the majority of CL fatty acids, often accounting for 80–90% of the composition
[180][430]. Although C18 polyunsaturated fatty acids are the predominant constituents of CL, dietary fatty acid intake can influence its composition. Increased dietary supplementation levels of C20 polyunsaturated fatty acids and DHA have been shown to elevate their proportions in mammalian CL, as indicated by Berger et al.
[181][431] and Wolff and Entressangles
[182][432].
The structure of bis(monacylglycero)phosphate (BMP), which is also known as lysobisphosphatidic acid, exhibits variations in chain length and the degree of fatty acid unsaturation. Oleic acid is frequently identified as the most abundant fatty acid in BMP
[183][184][185][126,132,433]. However, in certain cell types, polyunsaturated fatty acids such as LA and/or EPA and DHA have been reported to be highly accumulative
[186][187][188][189][434,435,436,437]. In alveolar macrophages, for instance, Holbrook et al.
[190][438] noted that oleic acid predominates along with either AA or DHA.
In intact tissues (e.g., neural tissue), palmitic acid typically represents the most abundant fatty acid within the alkyl group of platelet-activating factor (PAF). Stearic and oleic acids may also be present but to a relatively lesser extent. The
sn-2 position of PAF is often esterified with the acetyl group or other short-chain fatty acids
[191][439]. However, in rat nervous tissue,
sn-2 is predominantly occupied by unsaturated fatty acids, primarily AA and adrenic acid (C22:4n6)
[192][440], indicating the profound impact of
sn-2 composition on PAF activities. These longer n6-type fatty acids serve as potent precursors for eicosanoids.
3.2. Fatty Acid Profile of Sphingolipids
In general, SLs exhibit a prevalent composition of very long-chain saturated and/or monounsaturated fatty acids, typically ranging from 18 to 34 carbon chains. Notably, some sphingolipid structures have been reported to contain odd-numbered fatty acid chains
[193][194][201,441], suggesting a possible proportional elevation of these chains in the tissues of ruminants and coprophagous species. Despite the fact that the enzyme SPT utilizes palmitoyl-Coenzyme A to produce sphinganine (SA), it has the capability to utilize various other fatty-CoA substrates as well
[195][442]. Sphingoid bases within SLs predominantly feature saturated aliphatic chains, with some instances of mono- and di-unsaturated chains. Sphingosine (SO) possesses a fixed
trans-double bond between carbons 4 and 5. The chain length of sphingoid bases’ fatty acids typically falls within the range of 14 to 32 carbons
[196][443].
Within the context of Cer, the variability in chain length is attributed to the diversity of CerS isoforms. In most scenarios, CerS5 and CerS6 predominantly provide chains with 14–18 carbons, CerS1, CerS4, and CerS2 contribute chains with 18–24 carbons, and CerS3 supplies chains with up to 34 carbons
[128][382]. Cer features an acyl chain linked to an amide that is frequently saturated and considerably longer compared to those found in SLs containing sugar moieties
[197][240]. The length of the Cer fatty acid is determined by ER elongase and desaturase complexes, rather than the Golgi apparatus, which produces complex SLs. Cer and sphingomyelin (SM) are primarily acylated by palmitoleic and oleic acids, in addition to long and very long-chain fatty acids. In most organisms, including mammals, Cer can contain 2-hydroxylated fatty acids
[198][199][444,445], which is a characteristic favoring interaction with sphingomyelin synthase-2 (SMS2) over sphingomyelin synthase-1 (SMS1)
[200][446].
SM structures are characterized by long fatty acids with relatively high chain length inequality, leading to their interdigitation sensitivity. The cause of interdigitation is not fully understood but may involve the regulation and/or induction of proteins in response to specific fatty acids within SM under certain membrane constituents. In comparison to PC, SM typically contains more saturated and longer acyl chains, typically ranging from 16 to 24 carbons
[163][201][415,447], and may include odd-numbered fatty acids
[202][448]. The palmitic, stearic, behenic (C22:0), lignoceric (C24:0), and nervonic (C24:1 n9) acids are among the most frequently occurring fatty acids in SM
[163][201][202][203][204][415,447,448,449,450]. Mammalian germ cells, in particular, are abundant in very long-chain fatty acids with up to 34 carbons
[43][205][297,451], some of which may exist in a 2-hydroxylated form in certain mammals
[206][452]. Double bonds are relatively rare in SM fatty acids, and, when present, they are often located at a distance from the membrane–water interface
[201][447].
Glycosphingolipids (GSLs) exhibit a greater variation in the chain length of their fatty acids compared to phospholipids, with some GSLs containing fatty acids with more than 16 carbons
[207][453]. Although it is challenging to detect long and/or unsaturated fatty acids in GSL, a few studies have reported the presence of high proportions of long and very long fatty acids. For instance, stearic acid has been found to be more dominant than palmitic acid in the GSLs of the mouse brain
[208][454]. Furthermore, the majority of galactosylceramide (GalCer) in the brain is composed of very long fatty acids
[209][455]. In gangliosides, lignoceric acid (C24:0) is the most abundant acyl chain, constituting up to 70% of total fatty acids in myelin
[210][456], while stearic acid is the predominant component, making up 80% of total fatty acids in the human brain
[211][457].