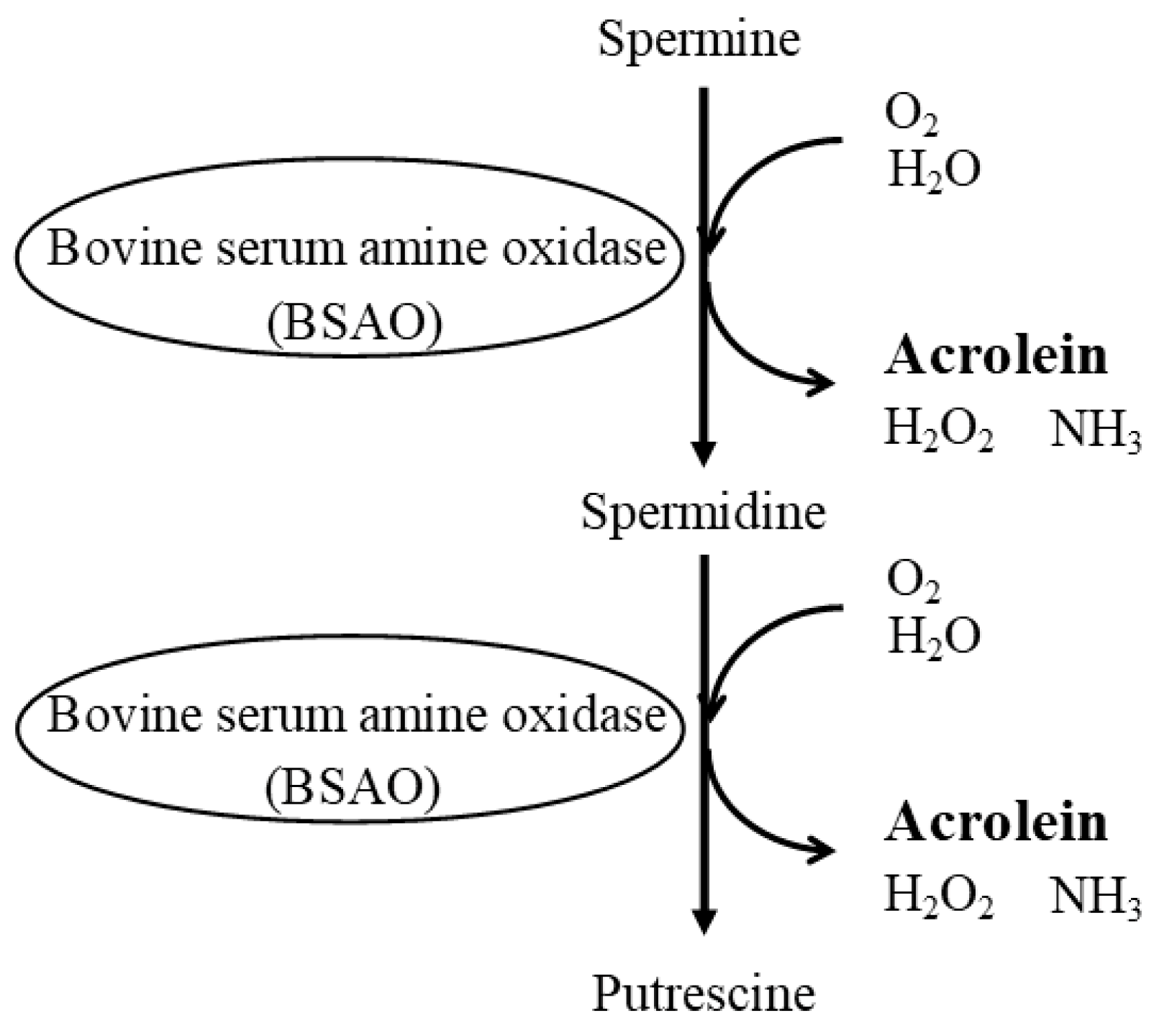
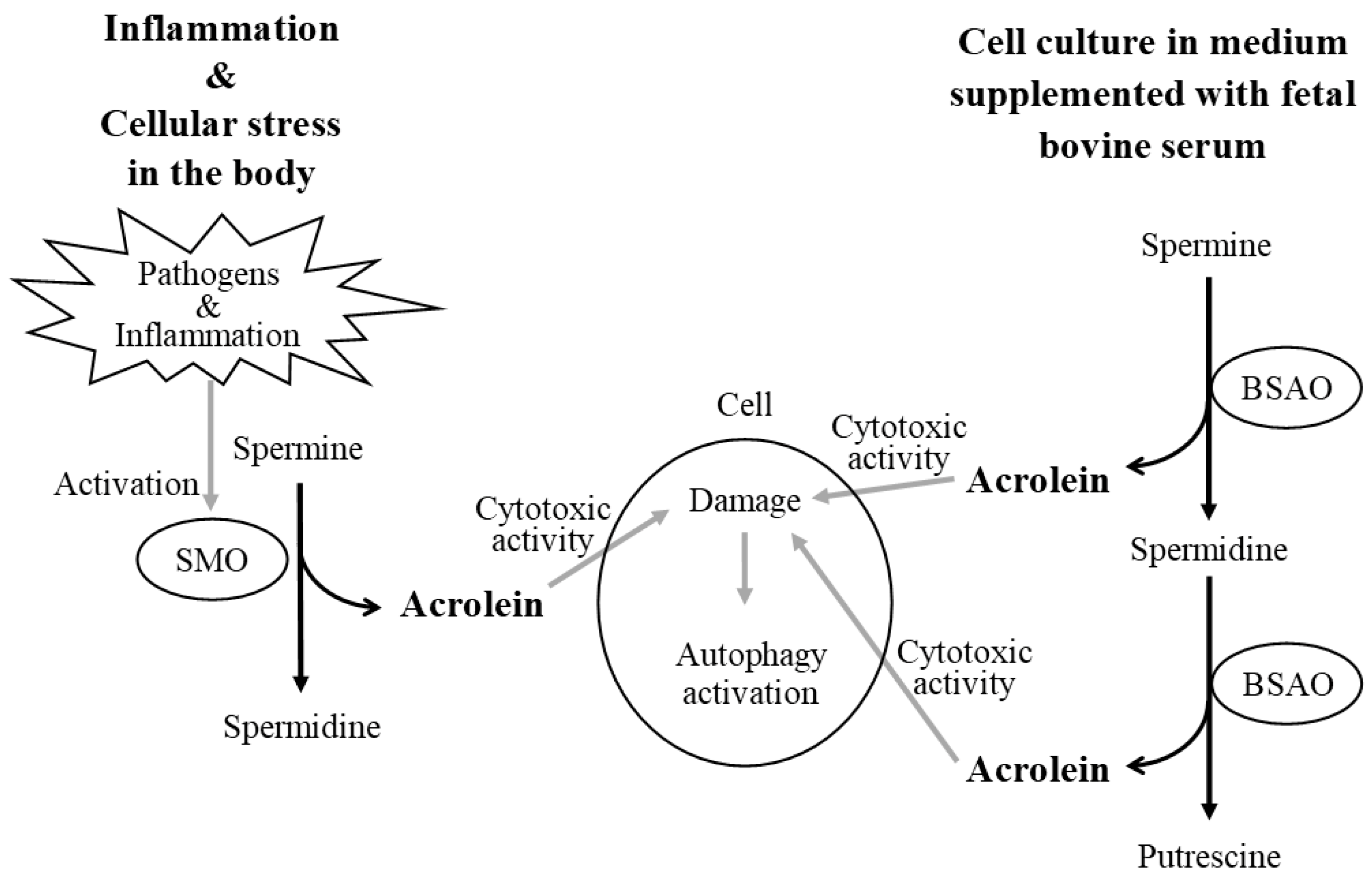
The relationship between polyamines and healthy longevity has received much attention in recent years. The most fundamental consideration in conducting polyamine studies is that bovine serum used for cell culture contains bovine serum amine oxidase. Bovine serum amine oxidase, which is not inactivated by heat treatment, breaks down spermine and spermidine to produce the highly toxic aldehyde acrolein, which causes cell damage and activates autophagy. Polyamine catabolism does not produce toxic aldehydes under normal conditions, but inflammation and some pathogens provoke an inducible enzyme, spermine oxidase, which only breaks down spermine to produce acrolein, resulting in cytotoxicity and the activation of autophagy. Therefore, spermine oxidase activation reduces spermine concentration and the ratio of spermine to spermidine, a feature recentlyre reported in patients with age-related diseases. Spermine, which is increased by a long-term, continuous high polyamine diet, suppresses aberrant gene methylation and the pro-inflammatory status that progress with age and are strongly associated with the development of several age-related diseases and senescence. Changes in spermine concentration and the spermine/spermidine ratio should be considered as indicators of human health status.
- polyamine
- spermine
- spermidine
- lifespan extension
- polyamine-rich food
- age-related disease
1. Polyamines
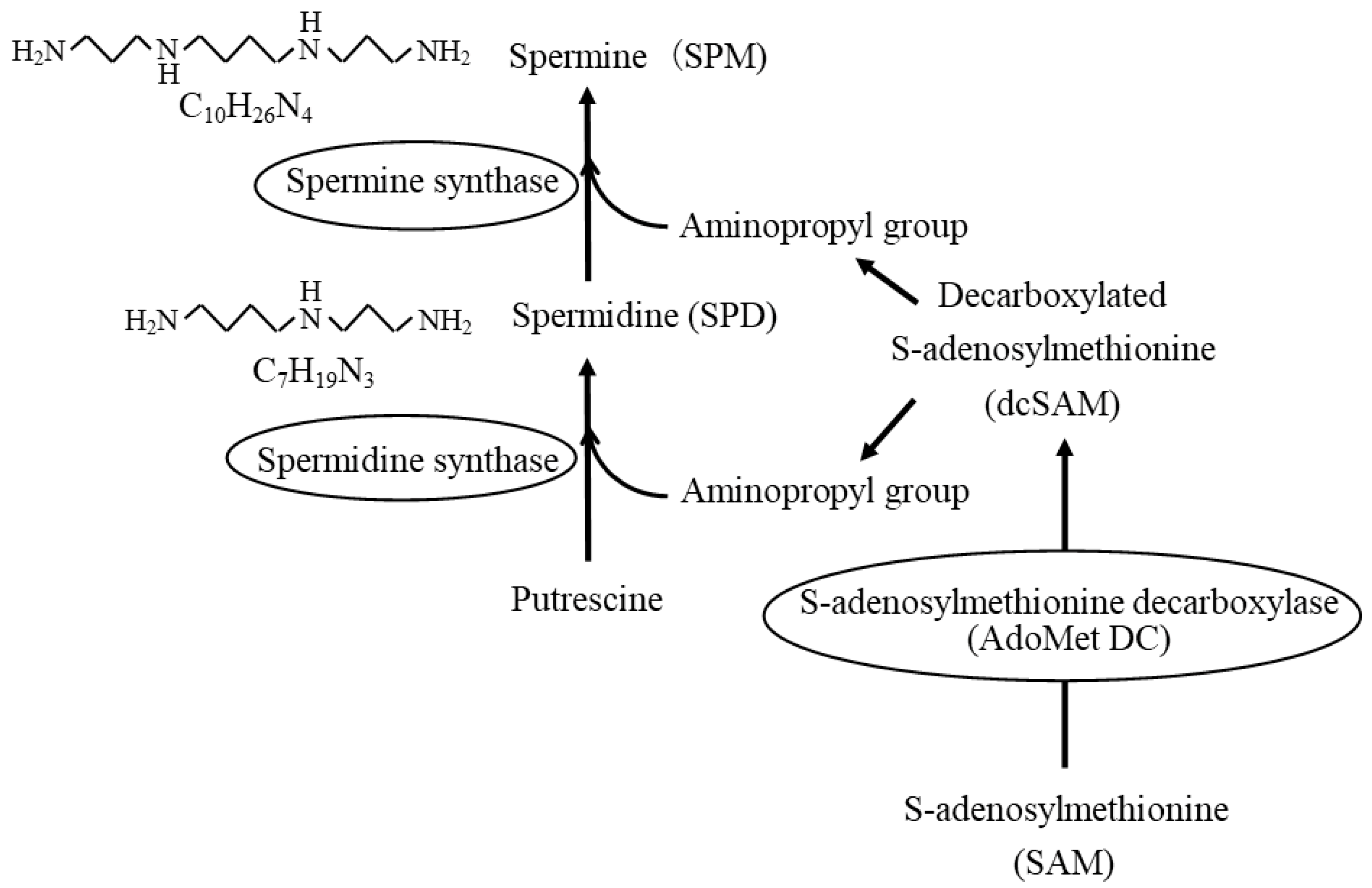
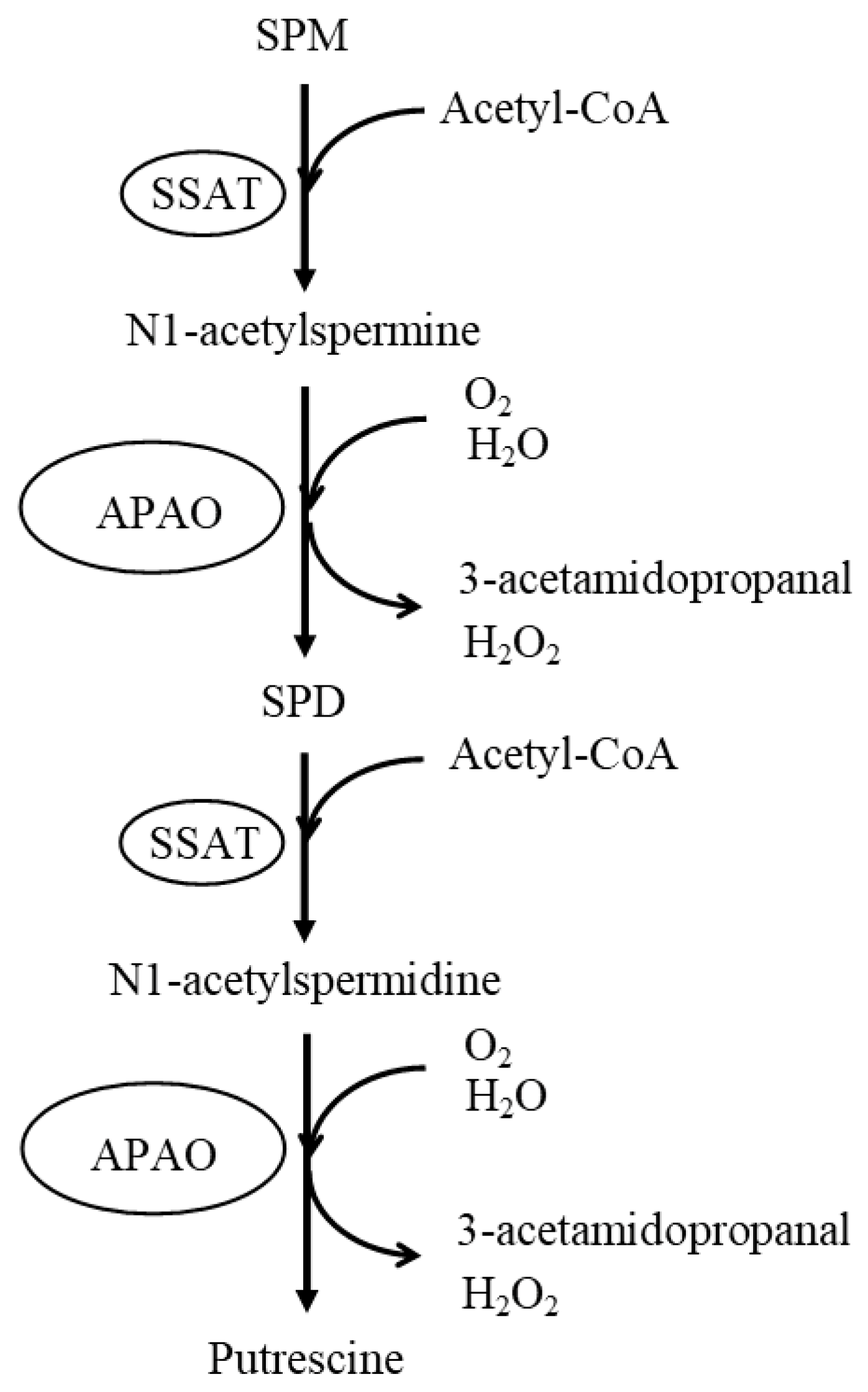
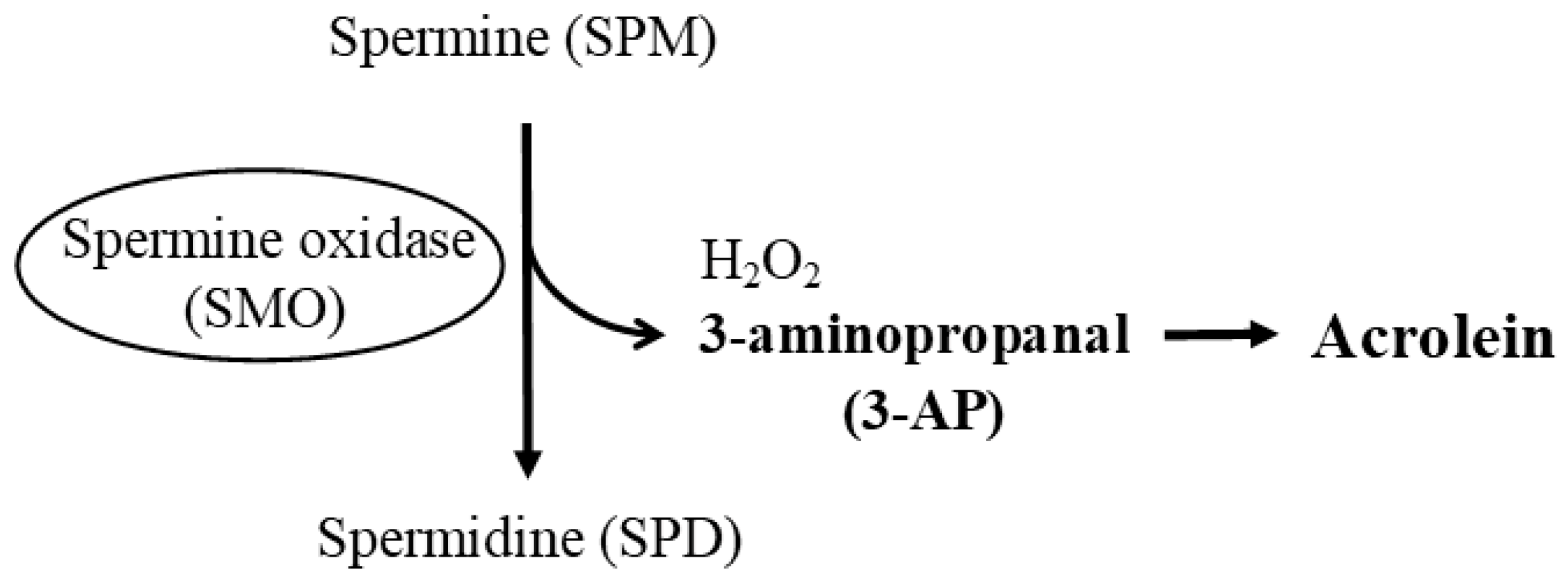
2. The Most Basic and Important Aspects of Conducting Polyamine Research
3. Age-Related Changes in Polyamine Concentrations
4. Age- and Disease-Related Changes in the Ratio of Spermine to Spermidine
In adults, changes in tissue and blood polyamine concentrations with age are not pronounced and do not decrease with age. However, SPM concentrations show a slight tendency to gradually decrease with age, so that the SPM/SPD ratio tends to decrease [23,26,27][16][18][19]. And this decline tends to be more pronounced in patients with age- and lifestyle-related diseases [23,26][16][18]. Similar changes in polyamine levels, i.e., a decrease in the SPM/SPD ratio and/or an increase in SPD levels, have been reported in other age-related diseases such as cerebral infarction, neurodegenerative diseases, and sarcopenia [26,27,29,30][18][19][20][21]. In patients with neurodegenerative diseases such as Alzheimer’s disease, SPD levels were elevated in the frontal and parietal lobes of the brain [31][22]. Plasma concentrations of PUT and SPD increased in stroke patients, while SPM concentrations remained unchanged, resulting in a significant decrease in the SPM/SPD ratio [32][23]. Chronic inflammation has been implicated in the background of these age-related chronic diseases [33][24]. This means that in patients with these diseases, inflammation-induced increases in SMO activity and 3-AP or acrolein levels should be noted. In fact, urinary acrolein levels were significantly higher in patients with diabetes mellitus than in those without [34][25]. An increase in plasma acrolein concentration and SMO activity has been observed in patients with chronic renal failure, such as diabetic nephropathy, chronic glomerulonephritis, and nephrosclerosis [35][26]. Chronic inflammation has been implicated in the onset and progression of several age- and lifestyle-related diseases, as well as protein-energy depletion leading to cardiovascular disease and sarcopenia [39,40,41][27][28][29]. In addition, the presence of chronic inflammation was a strong predictor of poor outcomes in dialysis patients [42][30]. In light of these scientific facts, the cytotoxic activity of acrolein, which results from the degradation of SPM via activated SMO in the presence of inflammation, may contribute to the development and progression of these diseases and the prognosis of patients [43,44][31][32].5. Polyamines as Nutritional Contributors to the Prevention of Age- and Lifestyle-Related Disease Development
Considering that the main source of polyamines is thought to be the gastrointestinal tract, i.e., polyamines in food and polyamines synthesized by intestinal bacteria are important, and that cells can take up extracellular polyamines, it is likely that polyamine levels in the body are affected by food intake and the state of intestinal bacteria. In fact, many studies have shown that reducing polyamine intake, as well as inhibiting the activity of gut bacteria with antibiotics, reduces blood polyamine levels [45,46][33][34]. When SPD was mixed with drinking water and administered to mice, an increase in blood SPD levels was reported [2][7]. SPM concentrations also appeared to have increased, as seen in the figure in the paper, but specific data are not shown. There are not many human intervention studies using high polyamine diets. The results of studies using high SPD supplements have been reported [49][35]. There was a 12-month study of supplementation in elderly patients between the ages of 60 and 90. This study showed a 10–20% increase in polyamine intake compared to normal dietary polyamine intake, yet the blood SPD levels did not change at all [49][35]. Thus, if there are clinical changes after being on a high SPD diet, it is not at all clear whether they are due to SPD or to other components that occurred at the same time [50][36]. Because polyamines are absorbed from the gastrointestinal tract without being broken down, and because many foods generally contain more SPD than SPM, it was thought that a diet high in polyamines would increase blood SPD. However, although limited research can be confirmed, there is very little evidence that continuous consumption of a high polyamine diet increases the blood levels of SPD. Instead, prolonged consumption of a diet high in polyamines (richer in SPD than in SPM) or even a short period of high SPD intake appears to increase SPM concentration, although there are individual differences. It is interesting to note that the age- and lifestyle-related diseases, which are associated with shorter life expectancy, decrease the SPM/SPD ratio, whereas diets rich in polyamines, which contribute to life extension, such as soy products, increase the SPM/SPD ratio.6. Biological Activity of Polyamines in Human Health and Disease
Polyamines are known to possess many biological activities that may counteract age-related conditions and senescence [6][1]. For example, they have anti-inflammatory and antioxidant properties [52,53][37][38] and protect cells and genes from damaging stimuli such as ionizing radiation, ultraviolet rays, toxic chemicals, and other stresses [6][1]. And some researchers, including us, have reported that increasing polyamine intake extends the lifespan of animals [1,5,54][10][39][40]. The anti-inflammatory effects of polyamines include the suppression of proinflammatory cytokine production by immune cells upon the stimulation and suppression of LFA-1 expression in the cell membrane [52,53][37][38]. Increased LFA-1 protein causes immune cells to respond to even minor stimuli, triggering the production of proinflammatory cytokines and provoking inflammation. SPM has strong physiological activity and therefore shows anti-inflammatory activity over a range of physiological concentration changes. SPD also shows similar biological activity to SPM, but requires a concentration change well beyond the physiological concentration change to confirm the effect [52,53][37][38]. Furthermore, the suppression of LFA-1 expression on immune cells by SPM is specific [52][37]. The amount of LFA-1 has been found to be related to the methylation status of the ITGAL, where the gene for LFA-1 is encoded. Increased levels of LFA-1 protein on immune cells with aging are associated with the progressive demethylation of ITGAL [55,56][41][42]. Gene methylation is a change that only occurs in cytosine, one of the four bases that make up a gene’s information, and is a mechanism that alters the reading of genetic information by adding or removing methyl groups from cytosine. In front of the genetic information, there is a site called the CpG land, which contains repeated sequences of cytosine and guanine. The methylation of cytosines within the CpG island results in decreased transcription and consequently decreased production of the protein encoded by the gene. Conversely, when cytosines within the CpG island are demethylated, transcription is more likely to occur, resulting in increased synthesis of the protein encoded by the gene. DNA methylation is regulated by DNA methyltransferases (DNMTs). DNMTs control the methylation state of cytosines by using methyl groups provided by SAM.References
- Soda, K. Overview of Polyamines as Nutrients for Human Healthy Long Life and Effect of Increased Polyamine Intake on DNA Methylation. Cells 2022, 11, 164.
- Starks, R.; Kirby, P.; Ciliberto, M.; Hefti, M. Snyder-Robinson syndrome. Autops. Case Rep. 2018, 8, e2018031.
- Wood, P.L.; Khan, M.A.; Moskal, J.R. The concept of “aldehyde load” in neurodegenerative mechanisms: Cytotoxicity of the polyamine degradation products hydrogen peroxide, acrolein, 3-aminopropanal, 3-acetamidopropanal and 4-aminobutanal in a retinal ganglion cell line. Brain Res. 2007, 1145, 150–156.
- Pegg, A.E. Toxicity of polyamines and their metabolic products. Chem. Res. Toxicol. 2013, 26, 1782–1800.
- Goodwin, A.C.; Destefano Shields, C.E.; Wu, S.; Huso, D.L.; Wu, X.; Murray-Stewart, T.R.; Hacker-Prietz, A.; Rabizadeh, S.; Woster, P.M.; Sears, C.L.; et al. Polyamine catabolism contributes to enterotoxigenic Bacteroides fragilis-induced colon tumorigenesis. Proc. Natl. Acad. Sci. USA 2011, 108, 15354–15359.
- Xu, H.; Chaturvedi, R.; Cheng, Y.; Bussiere, F.I.; Asim, M.; Yao, M.D.; Potosky, D.; Meltzer, S.J.; Rhee, J.G.; Kim, S.S.; et al. Spermine oxidation induced by Helicobacter pylori results in apoptosis and DNA damage: Implications for gastric carcinogenesis. Cancer Res. 2004, 64, 8521–8525.
- Eisenberg, T.; Abdellatif, M.; Schroeder, S.; Primessnig, U.; Stekovic, S.; Pendl, T.; Harger, A.; Schipke, J.; Zimmermann, A.; Schmidt, A.; et al. Cardioprotection and lifespan extension by the natural polyamine spermidine. Nat. Med. 2016, 22, 1428–1438.
- Chae, Y.B.; Kim, M.M. Activation of p53 by spermine mediates induction of autophagy in HT1080 cells. Int. J. Biol. Macromol. 2014, 63, 56–63.
- Zhang, H.; Alsaleh, G.; Feltham, J.; Sun, Y.; Napolitano, G.; Riffelmacher, T.; Charles, P.; Frau, L.; Hublitz, P.; Yu, Z.; et al. Polyamines Control eIF5A Hypusination, TFEB Translation, and Autophagy to Reverse B Cell Senescence. Mol. Cell 2019, 76, 110–125.e9.
- Xu, T.T.; Li, H.; Dai, Z.; Lau, G.K.; Li, B.Y.; Zhu, W.L.; Liu, X.Q.; Liu, H.F.; Cai, W.W.; Huang, S.Q.; et al. Spermidine and spermine delay brain aging by inducing autophagy in SAMP8 mice. Aging 2020, 12, 6401–6414.
- Tabor, C.W.; Tabor, H.; Rosenthal, S.M. Purification of amine oxidase from beef plasma. J. Biol. Chem. 1954, 208, 645–661.
- Holbert, C.E.; Dunworth, M.; Foley, J.R.; Dunston, T.T.; Stewart, T.M.; Casero, R.A., Jr. Autophagy induction by exogenous polyamines is an artifact of bovine serum amine oxidase activity in culture serum. J. Biol. Chem. 2020, 295, 9061–9068.
- Yamamoto, T.; Takabatake, Y.; Kimura, T.; Takahashi, A.; Namba, T.; Matsuda, J.; Minami, S.; Kaimori, J.Y.; Matsui, I.; Kitamura, H.; et al. Time-dependent dysregulation of autophagy: Implications in aging and mitochondrial homeostasis in the kidney proximal tubule. Autophagy 2016, 12, 801–813.
- Matsumoto, M.; Kitada, Y.; Naito, Y. Endothelial Function is improved by Inducing Microbial Polyamine Production in the Gut: A Randomized Placebo-Controlled Trial. Nutrients 2019, 11, 1118.
- Pekar, T.; Wendzel, A.; Flak, W.; Kremer, A.; Pauschenwein-Frantsich, S.; Gschaider, A.; Wantke, F.; Jarisch, R. Spermidine in dementia: Relation to age and memory performance. Wien. Klin. Wochenschr. 2020, 132, 42–46.
- Sanayama, H.; Ito, K.; Ookawara, S.; Uemura, T.; Sakiyama, Y.; Sugawara, H.; Tabei, K.; Igarashi, K.; Soda, K. Whole Blood Spermine/Spermidine Ratio as a New Indicator of Sarcopenia Status in Older Adults. Biomedicines 2023, 11, 1403.
- Soda, K.; Uemura, T.; Sanayama, H.; Igarashi, K.; Fukui, T. Polyamine-Rich Diet Elevates Blood Spermine Levels and Inhibits Pro-Inflammatory Status: An Interventional Study. Med. Sci. 2021, 9, 22.
- Saiki, S.; Sasazawa, Y.; Fujimaki, M.; Kamagata, K.; Kaga, N.; Taka, H.; Li, Y.; Souma, S.; Hatano, T.; Imamichi, Y.; et al. A metabolic profile of polyamines in parkinson disease: A promising biomarker. Ann. Neurol. 2019, 86, 251–263.
- Uemura, T.; Akasaka, Y.; Ikegaya, H. Correlation of polyamines, acrolein-conjugated lysine and polyamine metabolic enzyme levels with age in human liver. Heliyon 2020, 6, e05031.
- Gomes-Trolin, C.; Nygren, I.; Aquilonius, S.M.; Askmark, H. Increased red blood cell polyamines in ALS and Parkinson’s disease. Exp. Neurol. 2002, 177, 515–520.
- Els, T.; Bruckmann, J.; Rohn, G.; Daffertshofer, M.; Monting, J.S.; Ernestus, R.I.; Hennerici, M. Spermidine: A predictor for neurological outcome and infarct size in focal cerebral ischemia? Stroke 2001, 32, 43–46.
- Inoue, K.; Tsutsui, H.; Akatsu, H.; Hashizume, Y.; Matsukawa, N.; Yamamoto, T.; Toyo’oka, T. Metabolic profiling of Alzheimer’s disease brains. Sci. Rep. 2013, 3, 2364.
- Park, M.H.; Igarashi, K. Polyamines and their metabolites as diagnostic markers of human diseases. Biomol. Ther. 2013, 21, 1–9.
- Neves, J.; Sousa-Victor, P. Regulation of inflammation as an anti-aging intervention. FEBS J. 2020, 287, 43–52.
- Daimon, M.; Sugiyama, K.; Kameda, W.; Saitoh, T.; Oizumi, T.; Hirata, A.; Yamaguchi, H.; Ohnuma, H.; Igarashi, M.; Kato, T. Increased urinary levels of pentosidine, pyrraline and acrolein adduct in type 2 diabetes. Endocr. J. 2003, 50, 61–67.
- Sakata, K.; Kashiwagi, K.; Sharmin, S.; Ueda, S.; Igarashi, K. Acrolein produced from polyamines as one of the uraemic toxins. Biochem. Soc. Trans. 2003, 31, 371–374.
- Bielecka-Dabrowa, A.; Ebner, N.; Dos Santos, M.R.; Ishida, J.; Hasenfuss, G.; von Haehling, S. Cachexia, muscle wasting, and frailty in cardiovascular disease. Eur. J. Heart Fail. 2020, 22, 2314–2326.
- Ferrucci, L.; Fabbri, E. Inflammageing: Chronic inflammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 2018, 15, 505–522.
- Chhetri, J.K.; de Souto Barreto, P.; Fougere, B.; Rolland, Y.; Vellas, B.; Cesari, M. Chronic inflammation and sarcopenia: A regenerative cell therapy perspective. Exp. Gerontol. 2018, 103, 115–123.
- Sharif, S.; Van der Graaf, Y.; Cramer, M.J.; Kapelle, L.J.; de Borst, G.J.; Visseren, F.L.J.; Westerink, J.; SMART Study Group. Low-grade inflammation as a risk factor for cardiovascular events and all-cause mortality in patients with type 2 diabetes. Cardiovasc. Diabetol. 2021, 20, 220.
- Kashiwagi, K.; Igarashi, K. Molecular Characteristics of Toxicity of Acrolein Produced from Spermine. Biomolecules 2023, 13, 298.
- Igarashi, K.; Kashiwagi, K. Functional roles of polyamines and their metabolite acrolein in eukaryotic cells. Amino Acids 2021, 53, 1473–1492.
- Cipolla, B.; Guilli, F.; Moulinoux, J.P. Polyamine-reduced diet in metastatic hormone-refractory prostate cancer (HRPC) patients. Biochem. Soc. Trans. 2003, 31, 384–387.
- Nishimura, K.; Araki, N.; Ohnishi, Y.; Kozaki, S. Effects of dietary polyamine deficiency on Trypanosoma gambiense infection in rats. Exp. Parasitol. 2001, 97, 95–101.
- Schwarz, C.; Stekovic, S.; Wirth, M.; Benson, G.; Royer, P.; Sigrist, S.J.; Pieber, T.; Dammbrueck, C.; Magnes, C.; Eisenberg, T.; et al. Safety and tolerability of spermidine supplementation in mice and older adults with subjective cognitive decline. Aging 2018, 10, 19–33.
- Wirth, M.; Benson, G.; Schwarz, C.; Kobe, T.; Grittner, U.; Schmitz, D.; Sigrist, S.J.; Bohlken, J.; Stekovic, S.; Madeo, F.; et al. The effect of spermidine on memory performance in older adults at risk for dementia: A randomized controlled trial. Cortex 2018, 109, 181–188.
- Soda, K.; Kano, Y.; Nakamura, T.; Kasono, K.; Kawakami, M.; Konishi, F. Spermine, a natural polyamine, suppresses LFA-1 expression on human lymphocyte. J. Immunol. 2005, 175, 237–245.
- Zhang, M.; Caragine, T.; Wang, H.; Cohen, P.S.; Botchkina, G.; Soda, K.; Bianchi, M.; Ulrich, P.; Cerami, A.; Sherry, B.; et al. Spermine inhibits proinflammatory cytokine synthesis in human mononuclear cells: A counterregulatory mechanism that restrains the immune response. J. Exp. Med. 1997, 185, 1759–1768.
- Soda, K.; Dobashi, Y.; Kano, Y.; Tsujinaka, S.; Konishi, F. Polyamine-rich food decreases age-associated pathology and mortality in aged mice. Exp. Gerontol. 2009, 44, 727–732.
- Eisenberg, T.; Knauer, H.; Schauer, A.; Buttner, S.; Ruckenstuhl, C.; Carmona-Gutierrez, D.; Ring, J.; Schroeder, S.; Magnes, C.; Antonacci, L.; et al. Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 2009, 11, 1305–1314.
- Zhang, Z.; Deng, C.; Lu, Q.; Richardson, B. Age-dependent DNA methylation changes in the ITGAL (CD11a) promoter. Mech. Ageing Dev. 2002, 123, 1257–1268.
- Lu, Q.; Kaplan, M.; Ray, D.; Ray, D.; Zacharek, S.; Gutsch, D.; Richardson, B. Demethylation of ITGAL (CD11a) regulatory sequences in systemic lupus erythematosus. Arthritis Rheum. 2002, 46, 1282–1291.
