Due to their potent immunoregulatory and angio-modulatory properties, mesenchymal stem cells (MSCs) and their exosomes (MSC-Exos) have emerged as potential game-changers in regenerative ophthalmology, particularly for the personalized treatment of inflammatory diseases. NLRP3 inflammasome, composed of Nod-like receptor family pyrin domain-containing protein 3 (NLRP3), apoptosis-associated speck-like protein containing a caspase activation and recruitment domain (CARD) (ASC), and pro-caspase-1 is dysregulated in almost all inflammatory eye disease. Accordingly, a large number of experimental findings suggested that this multi-protein complex could be considered an important intracellular target for MSC-based therapy of inflammatory eye disorders.
- NLRP3 inflammasome
- mesenchymal stem cell
- exosomes
- therapy
- inflammatory eye diseases
1. Introduction
2. Activation of NLRP3 Inflammasome in Eye-Infiltrated Immune Cells as an Initial Step in the Development of Inflammatory Eye Disease
The activation of the NLRP3 inflammasome in eye-infiltrated immune cells leads to the enhanced production of pro-inflammatory cytokines (interleukin-1β (IL-1β) and IL-18), which elicit inflammatory processes in ocular structures [10][6]. The activation of the NLRP3 inflammasome in immune cells is a tightly regulated, multi-step process (Figure 1). It requires two steps: priming and activation. Since some immune cells do not naturally express pro-IL-1 and have inadequate amounts of NLRP3 for inflammasome activation, one of the main functions of the priming step is to stimulate the transcriptional production of NLRP3, pro-IL-1β, and pro-IL-18. This initial step involves the activation of transcriptional factors (the nuclear factor kappa B (NF-κB), c-Jun, ATF-2, and CREB) that, upon activation, translocate to the nucleus to induce the enhanced transcription of pro-inflammatory genes, resulting in the increased synthesis of NLRP3 and pro-IL-1β. The priming step of NLRP3 inflammasome activation is elicited by the activation of pattern recognition receptors (PRRs), including Toll-like receptors (TLR). These membrane-bound receptors are expressed on immune cells where recognized pathogen-associated molecular patterns (PAMPs) of microbial antigens or damage-associated molecular patterns (DAMPs) are released from injured cells [10,11][6][7]. Signals generated from activated PRRs, particularly TLR-2, TLR-4, TLR-5, and TLR-6, initiate phosphorylation and the consequent activation of extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK), and p38 mitogen-activated protein kinase (MAPK), which, in turn, phosphorylate c-Jun, ATF-2, and CREB transcription factors that bind to the promoter regions of NLRP3 and pro-IL-1β, leading to their transcriptional upregulation [10,11][6][7]. Additionally, upon binding to TLRs, TLR ligands elicit the MyD88-driven activation of IL-1R-associated kinase 1 (IRAK-1), which, in a TNFR-associated factor 6 (TRAF6)-dependent manner, induces the activation of transforming growth factor-beta-activated kinase 1 (TAK-1), a key protein kinase responsible for the optimal activation of NF-κB. Activated TAK-1 phosphorylates and activates the IκB kinase (IKK) complex, which consists of IKKα, IKKβ, and IKKγ (also known as NEMO). The activated IKK complex phosphorylates the Inhibitor of κB (IκB), which, within cytosol, binds to NF-κB and prevents its nuclear translocation. The phosphorylation of IκB leads to its degradation via the ubiquitin–proteasome pathway. This degradation releases NF-κB, allowing it to translocate into the nucleus. Once in the nucleus, NF-κB binds to specific DNA sequences called κB sites, promoting the transcription of NF-κB-dependent genes, such as NLRP3, pro-IL-1β, and pro-IL-18, which are necessary for inflammasome activation.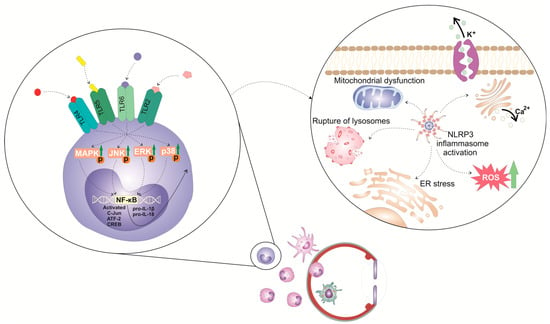
3. Molecular Mechanisms Responsible for NLRP3/IL-1β/IL-18-Dependent Generation of Detrimental Immune Response in Inflamed Eyes
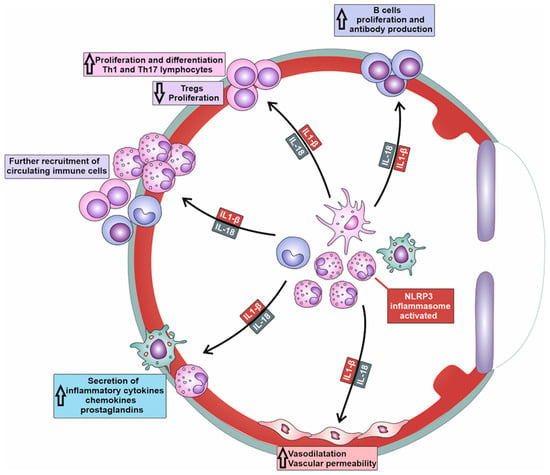
NLRP3/IL-1β/IL-18-dependent recruitment of immune cells is a complex process involving the activation of endothelial cells (ECs), the release of chemotactic factors, the modulation of adhesion molecules, and changes in vascular dynamics (Table 1) [9][10]. NLRP3-generated IL-1β and IL-18 induce increased expression of adhesion molecules (intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), E and P-selectins) on the membrane of ECs [9][10]. Additionally, the activation of NLRP3 inflammasome in immune cells enhances the affinity and avidity of integrin–ligand interactions and facilitates firm adhesion of immune cells to ECs. Precisely, the NLRP3/IL-1β/IL-18 axis upregulates the expression of integrins (lymphocyte function-associated antigen-1 (LFA-1) and very late antigen-4 (VLA-4)) on immune cells, which interact with their ligands on ECs, further promoting the adhesion and subsequent transmigration of immune cells into the inflamed tissues of affected eyes [9][10].| Cytokine/Chemokine | Cell Source | Mechanisms of Action | Biological Effects | Ref. No. |
|---|---|---|---|---|
| IL-1β, IL-18, TNF-α, CCL2, CCL3, CCL5, CXCL8, CXCL10 | monocytes/macrophages DCs neutrophils |
increased expression of adhesion molecules on the membrane of ECs within inflamed eyes | increased recruitment of circulating leukocytes in inflamed eyes | [9][10] |
| IL-1β, IL-6, IL-8, IL-18, PGE2 | monocytes/macrophages DCs neutrophils |
Increased vascular permeability | increased extravasation and accumulation of inflammatory immune cells in inflamed eyes | [13][9] |
| IL-1β and IL-18 | monocytes/macrophages | increased production of MMP-2, MMP-3 and MMP-9 | tissue damage and disruption of blood-aqueous and blood-retinal barriers | [14][11] |
| IL-1β, IL-18, IL-12, IL-23 | DCs | activation of naïve T cells and their differentiation in effector, inflammatory Th1 and Th17 cells | generation of Th1 and Th17 cell-driven eye inflammation | [10,11,15][6][7][12] |
| IL-1β and IL-18 | DCs | suppressed expression and activity of the transcription factor FoxP3 in T cells | attenuated expansion of Tregs | [16][13] |
| IL-1β, IL-6, IL-18 | monocytes/macrophages DCs neutrophils |
increased AID expression | increased production of high-affinity antibodies | [17][14] |
References
- Miller, J.R.; Hanumunthadu, D. Inflammatory eye disease: An overview of clinical presentation and management. Clin. Med. 2022, 22, 100–103.
- Tempest-Roe, S.; Joshi, L.; Dick, A.D.; Taylor, S.R. Local therapies for inflammatory eye disease in translation: Past, present and future. BMC Ophthalmol. 2013, 13, 39.
- Denniston, A.K.; Dick, A.D. Systemic therapies for inflammatory eye disease: Past, Present and Future. BMC Ophthalmol. 2013, 13, 1–8.
- Harrell, C.R.; Jovicic, N.; Djonov, V.; Arsenijevic, N.; Volarevic, V. Mesenchymal Stem Cell-Derived Exosomes and Other Extracellular Vesicles as New Remedies in the Therapy of Inflammatory Diseases. Cells 2019, 8, 1605.
- Harrell, C.R.; Volarevic, V.; Djonov, V.; Volarevic, A. Therapeutic Potential of Exosomes Derived from Adipose Tissue-Sourced Mesenchymal Stem Cells in the Treatment of Neural and Retinal Diseases. Int. J. Mol. Sci. 2022, 23, 4487.
- Zhuang, D.; Misra, S.L.; Mugisho, O.O.; Rupenthal, I.D.; Craig, J.P. NLRP3 Inflammasome as a Potential Therapeutic Target in Dry Eye Disease. Int. J. Mol. Sci. 2023, 24, 10866.
- Soliman, A.M.; Sim, R.H.; Das, S.; Mahakkanukrauh, P. Therapeutic Targeting of Inflammatory Pathways with Emphasis on NLRP3 Inflammasomes by Natural Products: A Novel Approach for the Treatment of Inflammatory Eye Diseases. Curr. Med. Chem. 2022, 29, 2891–2912.
- Wang, T.; Xu, H.; Dong, R.; Wu, S.; Guo, Y.; Wang, D. Effectiveness of targeting the NLRP3 inflammasome by using natural polyphenols: A systematic review of implications on health effects. Food Res. Int. 2023, 165, 112567.
- Zhang, Y.; Jiao, Y.; Li, X.; Gao, S.; Zhou, N.; Duan, J.; Zhang, M. Pyroptosis: A New Insight Into Eye Disease Therapy. Front. Pharmacol. 2021, 12, 797110.
- Yerramothu, P.; Vijay, A.K.; Willcox, M.D.P. Inflammasomes, the eye and anti-inflammasome therapy. Eye 2018, 32, 491–505.
- Bai, B.; Yang, Y.; Wang, Q.; Li, M.; Tian, C.; Liu, Y.; Aung, L.H.H.; Li, P.-F.; Yu, T.; Chu, X.-M. NLRP3 inflammasome in endothelial dysfunction. Cell Death Dis. 2020, 11, 1–18.
- Blevins, H.M.; Xu, Y.; Biby, S.; Zhang, S. The NLRP3 Inflammasome Pathway: A Review of Mechanisms and Inhibitors for the Treatment of Inflammatory Diseases. Front. Aging Neurosci. 2022, 14, 879021.
- Chernikov, O.V.; Moon, J.-S.; Chen, A.; Hua, K.-F. Editorial: NLRP3 Inflammasome: Regulatory Mechanisms, Role in Health and Disease and Therapeutic Potential. Front. Immunol. 2021, 12, 765199.
- Hsu, M.L.; Zouali, M. Inflammasome is a central player in B cell development and homing. Life Sci. Alliance 2022, 6, e202201700.
- Coyle, S.; Khan, M.N.; Chemaly, M.; Callaghan, B.; Doyle, C.; Willoughby, C.E.; Atkinson, S.D.; Gregory-Ksander, M.; McGilligan, V. Targeting the NLRP3 Inflammasome in Glaucoma. Biomolecules 2021, 11, 1239.
- Wang, Z.; Zhang, S.; Xiao, Y.; Zhang, W.; Wu, S.; Qin, T.; Yue, Y.; Qian, W.; Li, L. NLRP3 Inflammasome and Inflammatory Diseases. Oxidative Med. Cell. Longev. 2020, 2020, 4063562.
