In EC, the expression of ErbB receptors is significantly different, compared with the premenopausal and postmenopausal endometrium, mainly because of the increased transcriptional activity of ErbB encoding genes in EC cells. Moreover, there are some differences in ErbB-2 receptor profile among EC subgroups that could be explained by the alterations in pathophysiology and clinical behavior of various EC histologic subtypes. The fact that ErbB-2 receptor expression is more common in aggressive EC histologic subtypes (papillary
serous and clear cell) could indicate a future role of ErbB-targeted therapies in well-defined EC subgroups with overexpression of ErbB receptors.
In EC, the expression of ErbB receptors is significantly different, compared with the premenopausal and postmenopausal endometrium, mainly because of the increased transcriptional activity of ErbB encoding genes in EC cells. Moreover, there are some differences in ErbB-2 receptor profile among EC subgroups that could be explained by the alterations in pathophysiology and clinical behavior of various EC histologic subtypes. The fact that ErbB-2 receptor expression is more common in aggressive EC histologic subtypes (papillary serous and clear cell) could indicate a future role of ErbB-targeted therapies in well-defined EC subgroups with overexpression of ErbB receptors.
- ErbB receptors
- EGF system
- physiology
- signaling pathways
1. Introduction
2. Physiology of ErbB Receptors
The EGF system is present in various human organs and plays a significant role in cell proliferation, differentiation, migration and apoptosis during embryogenesis and postnatal development [39][43][44].2.1. ErbB Receptors
ErbB receptors are members of the subclass I superfamily of receptor tyrosine kinases (RTKs) [37][39][45]. In humans, the EGF system consists of the following ErbB receptors: epidermal growth factor receptor (EGFR), ErbB-2, ErbB-3 and ErbB-4 [37][39][44][45][46]. These receptors are trans-membrane glycoproteins that catalyze the transferring of γ phosphate of ATP to hydroxyl groups of tyrosines in target proteins [47]. However, ErbB-3 has no intrinsic tyrosine kinase activity, and it depends on another ErbB receptor (usually ErbB-2) for intracellular signaling [45][48]. Regarding their structure, ErbB receptors have an extracellular ligand-binding domain, a transmembrane domain, a short juxtamembrane section, an intracellular bilobed tyrosine kinase domain and a tyrosine-containing C-terminal tail (Figure 1) [44][45][46][49].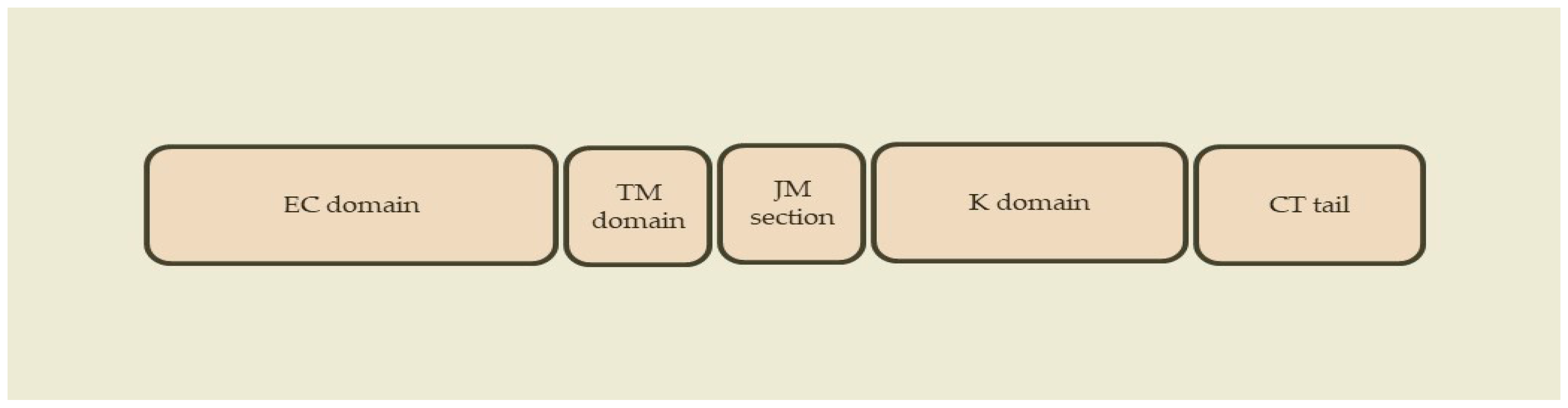
2.2. ErbB Ligands
In humans, the EGF system has the following ErbB peptide mediators (ligands): EGF, transforming growth factor-a (TGF-a), amphiregulin (AR), heparin-binding growth factor (HB-EGF), betacellulin (BTC), epigen, epiregulin (EPR), neuregulin-1 (NRG-1), neuregulin-2 (NRG-2), neuregulin-3 (NRG-3), neuregulin-4 (NRG-4), neuroglycan C and tomoregulin [39][44][45][46]. Ligand binding to the extracellular domain of the ErbB receptor results in conformational changes and induces homodimerization and heterodimerization of receptors [37][44][45][46]. However, the ErbB-2 receptor fails to bind any ligands [37][44][45][46]. Based on their affinity for one or more receptors, ErbB ligands could be further classified into the following subgroups: 1. Ligands with binding specificity for EGFR only: EGF, TGF-a and AR [44][45][46]. 2. Ligands with dual binding specificity for EGFR and ErbB4: HB-EGF, BTC and EPR [44][45][46]. 3. Ligands with binding specificity for ErbB-3 only: neuroglycan C [44][45][46]. 4. Ligands with binding specificity for ErbB-4 only: NRG-3, NRG-4 and tomoregulin [44][45][46]. 5. Ligands with dual binding specificity for ErbB-3 and ErbB-4: NRG-1, NRG-2 [44][45][46].2.3. Receptor Homodimerization and Heterodimerization
There are two distinct conformations of the extracellular ligand-binding domain, based on the activation status of EGFR, ErbB-3 and ErbB-4 receptors: 1. Closed conformation. When ErbB receptors are inactive, there are intramolecular interactions between the cysteine-rich subdomains CR1 and CR2, causing closed conformation of the extracellular ligand-binding domain [44][45][46][50][51]. 2. Open conformation. When ErbB receptors become active, the leucine-rich subdomains L1 and L2 create a ligand-binding pocket, allowing interactions with a single ligand, while the extracellular ligand-binding domain takes an open conformation and the β-hairpin loop dimerization arm of subdomain CR1 is exposed [44][45][46][50][51]. It seems that there is equilibrium between both conformations of the extracellular ligand-binding domain, related directly to ligand presence and subsequent ligand binding [50][51][52]. More specifically, ligand binding to the leucine-rich subdomains L1 and L2 stabilises the extracellular ligand-binding domain to an open conformation, exposes the β-hairpin loop dimerization arm of subdomain CR1 and allows receptor homodimerization and heterodimerization [44][45][46][51][52][53]. Subsequently, ErbB receptor dimerization induces conformational changes of the intracellular bilobed tyrosine kinase domain [44][45][46][54][55]. In contrast, the extracellular ligand-binding domain of the ErbB-2 receptor has an extended conformation that is not suitable for ligand binding, as there is close proximity of the leucine-rich subdomains L1 and L2, abolishing the ligand-binding site [44][45][46][56][57][58]. However, the extended conformation of the ErbB-2 receptor is necessary for interaction with other ErbB receptors and subsequent ligand-independent heterodimerization and signaling [44][45][46][56][57][58]. Moreover, abnormal overexpression of the ErbB-2 receptor permits ligand-independent receptor homodimerization [44][46][57]. Overall, homodimerization and heterodimerization of ErbB receptors represents an essential part in the pathophysiology of the EGF system signaling network [44][45][46][54][55]. Furthermore, the ErbB-2 and ErbB-3 heterodimer is the most transforming and mitogenic receptor complex [59].2.4. Intracellular Tyrosine Kinase Activation
Following homodimerization and heterodimerization of ErbB receptors, conformational changes of the intracellular tyrosine kinase domain take place, which in turn cause tyrosine kinase activation and phosphorylation of the tyrosine-containing C-terminal tail [44][45][46][54][55]. As already mentioned, the intracellular tyrosine kinase domain has a bilobed structure, with ATP binding between the N and C lobes [44][45][46][55]. More specifically, the C-lobe of an intracellular tyrosine kinase domain (activator) allosterically interacts with the N-lobe of another intracellular tyrosine kinase domain (receiver) within the same dimerization pair [44][45][46][55]. This interaction induces conformational changes in the N-lobe of the receiver tyrosine kinase and finally causes its activation [44][45][46][55]. Subsequently, the activated receiver tyrosine kinase catalyzes phosphorylation of tyrosine residues in the tyrosine-containing C-terminal tail of the activator tyrosine kinase [44][45][46][55]. These phosphorylated tyrosine residues serve as docking sites for adaptor proteins, enzymes and various signaling molecules containing Src homology 2 (SH2) and phosphotyrosine binding (PTB) domains [38][44][45][46][55][60][61].3. ErbB Receptors in Endometrial Cancer
During the menstrual cycle, there is a wide variation in the profile of ErbB receptors, indicating a central role of the EGF system in the regulation of endometrial cyclical growth and shedding [62][63]. In EC, the expression of ErbB receptors is significantly different, compared with the premenopausal and postmenopausal endometrium [62][64][65]. This is mainly because of the increased transcriptional activity of ErbB encoding genes in EC cells [65].3.1. Profile of ErbB Receptors in Endometrial Cancer
Overall, EGFR overexpression is reported in 43–67% of unselected EC cases [65][66][67][68][69][70][71][72][73][74][75][76]. EGFR overexpression is present in approximately 46% of type I EC (endometrioid) cases [70][72][77]. EGFR overexpression is observed in 34–50% of type II EC (papillary serous, clear cell, undifferentiated) cases [70][72][77][78][79][80]. ErbB-2 overexpression and ΕrbB-2 gene amplification represents a very rare event in unselected EC cases [65][70][74][75][76]. However, ΕrbB-2 overexpression and ΕrbB-2 gene amplification are present in only 8–15% and 3% of type I EC cases, respectively [65][70][77][81][82][83][84]. In contrast, ΕrbB-2 overexpression and ErbB-2 gene amplification are more common in type II EC cases [70][72][77][78][79][80]. Moreover, the exact frequency of ErbB-2 overexpression and ΕrbB-2 gene amplification in type II EC remains controversial, as there are many racial differences [70][72][78][85][86]. More specifically, ErbB-2 overexpression and ΕrbB-2 gene amplification are more common in African—American patients with type II EC, when compared with Caucasian individuals [85][86]. Likewise, ErbB-2 overexpression and ΕrbB-2 gene amplification have significant variations among different histologic subtypes of type II EC [70][72][78][81][86][87][88]. ErbB-2 overexpression and ΕrbB-2 gene amplification are reported in 18–80% and 17–47% of papillary serous EC cases, respectively [70][72][81][84][86][87][88], and 33% and 16–50% of clear cell EC cases, respectively [70][72][81][88]. ErbB-3 overexpression is reported in 30% of unselected EC cases [62][74]. More specifically, ErbB-3 overexpression is more common in well differentiated tumors when compared with moderately and poorly differentiated ones [62]. Similarly, ErbB-4 overexpression is reported in 15% of unselected EC cases [62][74]. Overall, there are some differences in ErbB-2 receptor profile in selected EC patients (EC histologic subtypes and racial—ethnic subgroups) [64][71][78][85]. ErbB-2 receptor expression is more common in papillary serous and clear cell EC cases [64][71][78]. This is mainly based on differences in the pathophysiology and clinical behavior of various EC histologic subtypes [64][71][78].3.2. Clinical Role in Endometrial Cancer
The relationship of the ErbB receptors profile with disease stage, tumor grade and response to treatment remains controversial in EC cases [70][74]. In particular, the clinical role of EGFR overexpression has not been studied thoroughly in EC patients [70][74]. Some studies demonstrate an association between EGFR overexpression and poor clinical outcome, while others report otherwise [66][67][68][69]. It seems that EGFR overexpression may have a dual role in EC cases [70]. EGFR overexpression in type I EC is associated with less aggressive disease and more favorable outcomes [70][72][74][78]. In contrast, EGFR overexpression in type II EC is associated with more aggressive disease and adverse clinical outcomes [70][72][74][78]. However, the clinical significance of ΕrbB-2 overexpression and ΕrbB-2 gene amplification has been studied extensively in EC patients [72][78][81][86][87][89][90][91]. ΕrbB-2 overexpression and ΕrbB-2 gene amplification are indicators of a more aggressive disease with reduced response to treatment and less favorable outcomes, especially in patients with type II EC [72][74][78][81][86][87][89][90][91][92]. Furthermore, the clinical role of ErbB-3 and ErbB-4 overexpression has not been studied extensively in patients with EC [62][64][71][72][73][74][78]. It becomes apparent that ErbB-2 receptor expression is more common in aggressive EC histologic subtypes (papillary serous and clear cell) [64][71][78]. This possibly indicates a future role of ErbB-targeted therapies in well-defined EC subgroups with overexpression of ErbB receptors [71][78][93].References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424.
- WHO Globocan. Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2020; International Agency for Research on Cancer: Lyon, France, 2020.
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386.
- Gitsch, G.; Hanzal, E.; Jensen, D.; Hacker, N.F. Endometrial cancer in premenopausal women 45 years and younger. Obstet. Gynecol. 1995, 85, 504–508.
- Duska, L.R.; Garrett, A.; Rueda, B.R.; Haas, J.; Chang, Y.; Fuller, A.F. Endometrial Cancer in Women 40 Years Old or Younger. Gynecol. Oncol. 2001, 83, 388–393.
- Erkanli, S.; Ayhan, A. Fertility-sparing therapy in young women with endometrial cancer: 2010 update. Int. J. Gynecol. Cancer 2010, 20, 1170–1187.
- Androutsopoulos, G. Current treatment options in patients with endometrial cancer. J. Community Med. Health Educ. 2012, 2, e113.
- Androutsopoulos, G.; Decavalas, G. Management of endometrial cancer. Int. J. Transl. Community Dis. 2013, 1, 101.
- Koufopoulos, N.; Carrer, D.; Koureas, N.; Sofopoulos, M.; Paraoulakis, I.; Androutsopoulos, G.; Arnogiannaki, N.; Zygouris, D.; Derdelis, G.; Terzakis, E. Pathological data on 19 cases of endometrioid carcinoma of the endometrium in women of reproductive age. Int. J. Gynecol. Cancer 2013, 23 (Suppl. 1), 322.
- Androutsopoulos, G.; Decavalas, G. Endometrial cancer: Current treatment strategies. World J. Oncol. Res. 2014, 1, 1–4.
- Androutsopoulos, G.; Michail, G.; Adonakis, G.; Decavalas, G. Current treatment approach of endometrial cancer. Int. J. Clin. Ther. Diagn. 2015, S1, 8–11.
- Androutsopoulos, G.; Adonakis, G.; Decavalas, G. Present and future in endometrial cancer treatment. Obstet. Gynecol. Int. J. 2015, 2, 00031.
- ACOG. ACOG practice bulletin No. 149: Endometrial cancer. Obstet. Gynecol. 2015, 125, 1006–1026.
- Colombo, N.; Creutzberg, C.; Amant, F.; Bosse, T.; Gonzalez-Martin, A.; Ledermann, J.; Marth, C.; Nout, R.; Querleu, D.; Mirza, M.R.; et al. ESMO-ESGO-ESTRO Consensus Conference on Endometrial Cancer: Diagnosis, treatment and follow-up. Ann. Oncol. 2016, 27, 16–41.
- Androutsopoulos, G.; Michail, G.; Decavalas, G. New insights in endometrial cancer treatment. Clin. Oncol. Endometrial Cancer 2016, 1, 1040.
- Androutsopoulos, G.; Decavalas, G. Standard and novel therapies in endometrial cancer. J. Gynecol. Women’s Health 2016, 1, 555564.
- Androutsopoulos, G.; Kotsopoulos, I.; Decavalas, G. Fertility preservation in young patients with endometrial cancer. World J. Oncol. Res. 2016, 3, 36–39.
- Sundar, S.; Balega, J.; Crosbie, E.; Drake, A.; Edmondson, R.; Fotopoulou, C.; Gallos, I.; Ganesan, R.; Gupta, J.; Johnson, N.; et al. BGCS uterine cancer guidelines: Recommendations for practice. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 213, 71–97.
- Androutsopoulos, G.; Kotsopoulos, I.; Korompelis, P.; Michail, G.; Adonakis, G.; Decavalas, G. Systematic lymphadenectomy or sentinel lymph node dissection in endometrial cancer: A clinical dilemma. Hell. J. Obst. Gynecol. 2017, 16, 14–19.
- Androutsopoulos, G.; Kotsopoulos, I.; Adonakis, G.; Decavalas, G. Conservative management of young patients with early stage endometrial cancer. J. Gynecol. Women’s Health 2017, 2, 555586.
- Androutsopoulos, G.; Adonakis, G.; Decavalas, G. ErbB targeted therapy in endometrial cancer. In Endometrial Cancer: Current Epidemiology, Detection and Management; Farghaly, S., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2014.
- Androutsopoulos, G.; Kotsopoulos, I.; Korompelis, P.; Michail, G.; Adonakis, G.; Decavalas, G. Conservative therapeutic approach in young patients with endometrial cancer: Is it really possible? Hell. J. Obst. Gynecol. 2017, 16, 7–23.
- Michail, G.D. Endometrial Cancer-Diagnosis. Int. J. Clin. Ther. Diagn. 2015, 1, 17–27.
- Weimer, J.; Hüttmann, M.; Nusilati, A.; Andreas, S.; Röseler, J.; Tribian, N.; Rogmans, C.; Stope, M.B.; Dahl, E.; Mustea, A.; et al. Fluorescence in situ hybridization test for detection of endometrial carcinoma cells by non-invasive vaginal swab. J. Cell. Mol. Med. 2023, 27, 379–391.
- Pouliakis, A.; Damaskou, V.; Margari, N.; Karakitsou, E.; Pergialiotis, V.; Valasoulis, G.; Michail, G.; Chrelias, C.; Chrelias, G.; Sioulas, V.; et al. Artificial Intelligence and Image Analysis for the Identification of Endometrial Malignancies: A Comparative Study. In Research Anthology on Medical Informatics in Breast and Cervical Cancer; I.R. Management Association, Ed.; IGI Global: Hershey, PA, USA, 2023; pp. 1–30.
- Pouliakis, A.; Damaskou, V.; Margari, N.; Karakitsou, E.; Pergialiotis, V.; Valasoulis, G.; Michail, G.; Chrelias, C.; Chrelias, G.; Sioulas, V.; et al. Artificial Intelligence and Image Analysis for the Identification of Endometrial Malignancies: A Comparative Study. In Quality Assurance in the Era of Individualized Medicine; Moumtzoglou, A.S., Ed.; IGI Global: Hershey, PA, USA, 2020; pp. 110–146.
- Pouliakis, A.; Margari, N.; Karakitsou, E.; Valasoulis, G.; Koufopoulos, N.; Koureas, N.; Alamanou, E.; Pergialiotis, V.; Damaskou, V.; Panayiotides, I.G. Artificial Intelligence via Competitive Learning and Image Analysis for Endometrial Malignancies: Discriminating Endometrial Cells and Lesions. Int. J. Reliab. Qual. E Healthc. 2019, 8, 38–54.
- Piedimonte, S.; Rosa, G.; Gerstl, B.; Sopocado, M.; Coronel, A.; Lleno, S.; Vicus, D. Evaluating the use of machine learning in endometrial cancer: A systematic review. Int. J. Gynecol. Cancer 2023, 33, 1383–1393.
- Purandare, N.C.; Trevisan, J.; Patel, I.I.; Gajjar, K.; Mitchell, A.L.; Theophilou, G.; Valasoulis, G.; Martin, M.; von Bünau, G.; Kyrgiou, M.; et al. Exploiting biospectroscopy as a novel screening tool for cervical cancer: Towards a framework to validate its accuracy in a routine clinical setting. Bioanalysis 2013, 5, 2697–2711.
- Theophilou, G.; Morais, C.L.M.; Halliwell, D.E.; Lima, K.M.G.; Drury, J.; Martin-Hirsch, P.L.; Stringfellow, H.F.; Hapangama, D.K.; Martin, F.L. Synchrotron- and focal plane array-based Fourier-transform infrared spectroscopy differentiates the basalis and functionalis epithelial endometrial regions and identifies putative stem cell regions of human endometrial glands. Anal. Bioanal. Chem. 2018, 410, 4541–4554.
- Jacobs, I.; Gentry-Maharaj, A.; Burnell, M.; Manchanda, R.; Singh, N.; Sharma, A.; Ryan, A.; Seif, M.W.; Amso, N.N.; Turner, G.; et al. Sensitivity of transvaginal ultrasound screening for endometrial cancer in postmenopausal women: A case-control study within the UKCTOCS cohort. Lancet Oncol. 2010, 12, 38–48.
- Bokhman, J.V. Two pathogenetic types of endometrial carcinoma. Gynecol. Oncol. 1983, 15, 10–17.
- Doll, A.; Abal, M.; Rigau, M.; Monge, M.; Gonzalez, M.; Demajo, S.; Colás, E.; Llauradó, M.; Alazzouzi, H.; Planagumá, J.; et al. Novel molecular profiles of endometrial cancer-new light through old windows. J. Steroid. Biochem. Mol. Biol. 2008, 108, 221–229.
- Kandoth, C.; Schultz, N.; Cherniack, A.; Akbani, R.; Liu, Y.; Shen, H.; Robertson, A.; Pashtan, I.; Shen, R.; Benz, C.; et al. Integrated genomic characterization of endometrial carcinoma. Nature 2013, 497, 67–73.
- Talhouk, A.; McConechy, M.; Leung, S.; Yang, W.; Lum, A.; Senz, J.; Boyd, N.; Pike, J.; Anglesio, M.; Kwon, J.; et al. Confirmation of ProMisE: A simple, genomics-based clinical classifier for endometrial cancer. Cancer 2017, 123, 802–813.
- Le Gallo, M.; Bell, D. The emerging genomic landscape of endometrial cancer. Clin. Chem. 2014, 60, 98–110.
- Holbro, T.; Civenni, G.; Hynes, N.E. The ErbB receptors and their role in cancer progression. Exp. Cell Res. 2003, 284, 99–110.
- Marmor, M.D.; Skaria, K.B.; Yarden, Y. Signal transduction and oncogenesis by ErbB/HER receptors. Int. J. Radiat. Oncol. 2004, 58, 903–913.
- Überall, I.; Kolář, Z.; Trojanec, R.; Berkovcová, J.; Hajdúch, M. The status and role of ErbB receptors in human cancer. Exp. Mol. Pathol. 2008, 84, 79–89.
- Lemmon, M.A.; Schlessinger, J. Cell Signaling by Receptor Tyrosine Kinases. Cell 2010, 141, 1117–1134.
- McDonell, L.M.; Kernohan, K.D.; Boycott, K.M.; Sawyer, S.L. Receptor tyrosine kinase mutations in developmental syndromes and cancer: Two sides of the same coin. Hum. Mol. Genet. 2015, 24, R60–R66.
- Du, Z.; Lovly, C.M. Mechanisms of receptor tyrosine kinase activation in cancer. Mol. Cancer 2018, 17, 58.
- Casalini, P.; Iorio, M.V.; Galmozzi, E.; Ménard, S. Role of HER receptors family in development and differentiation. J. Cell. Physiol. 2004, 200, 343–350.
- Wieduwilt, M.J.; Moasser, M.M. The epidermal growth factor receptor family: Biology driving targeted therapeutics. Cell. Mol. Life Sci. 2008, 65, 1566–1584.
- Linggi, B.; Carpenter, G. ErbB receptors: New insights on mechanisms and biology. Trends Cell Biol. 2006, 16, 649–656.
- Roskoski, R., Jr. The ErbB/HER family of protein-tyrosine kinases and cancer. Pharmacol. Res. 2014, 79, 34–74.
- Hunter, T. The Croonian Lecture 1997. The phosphorylation of proteins on tyrosine: Its role in cell growth and disease. Philos. Trans. R. Soc. B Biol. Sci. 1998, 353, 583–605.
- Mass, R.D. The HER receptor family: A rich target for therapeutic development. Int. J. Radiat. Oncol. 2004, 58, 932–940.
- Ogiso, H.; Ishitani, R.; Nureki, O.; Fukai, S.; Yamanaka, M.; Kim, J.H.; Saito, K.; Sakamoto, A.; Inoue, M.; Shirouzu, M.; et al. Crystal structure of the complex of human epidermal growth factor and receptor extracellular domains. Cell 2002, 110, 775–787.
- Ferguson, K.M.; Berger, M.B.; Mendrola, J.M.; Cho, H.-S.; Leahy, D.J.; Lemmon, M.A. EGF Activates Its Receptor by Removing Interactions that Autoinhibit Ectodomain Dimerization. Mol. Cell 2003, 11, 507–517.
- Dawson, J.P.; Berger, M.; Lin, C.-C.; Schlessinger, J.; Lemmon, M.A.; Ferguson, K.M. Epidermal Growth Factor Receptor Dimerization and Activation Require Ligand-Induced Conformational Changes in the Dimer Interface. Mol. Cell. Biol. 2005, 25, 7734–7742.
- Özcan, F.; Klein, P.; Lemmon, M.A.; Lax, I.; Schlessinger, J. On the nature of low- and high-affinity EGF receptors on living cells. Proc. Natl. Acad. Sci. USA 2006, 103, 5735–5740.
- Olayioye, M.; Neve, R.; Lane, H.; Hynes, N. The ErbB signaling network: Receptor heterodimerization in development and cancer. EMBO J. 2000, 19, 3159–3167.
- Qian, X.; LeVea, C.; Freeman, J.; Dougall, W.; Greene, M. Heterodimerization of epidermal growth factor receptor and wild-type or kinase-deficient Neu: A mechanism of interreceptor kinase activation and transphosphorylation. Proc. Natl. Acad. Sci. USA 1994, 91, 1500–1504.
- Zhang, X.; Gureasko, J.; Shen, K.; Cole, P.A.; Kuriyan, J. An allosteric mechanism for activation of the kinase domain of epidermal growth factor receptor. Cell 2006, 125, 1137–1149.
- Graus-Porta, D.; Beerli, R.R.; Daly, J.M.; Hynes, N.E. ErbB-2, the preferred heterodimerization partner of all ErbB receptors, is a mediator of lateral signaling. EMBO J. 1997, 16, 1647–1655.
- Garrett, T.P.; McKern, N.M.; Lou, M.; Elleman, T.C.; Adams, T.E.; Lovrecz, G.O.; Kofler, M.; Jorissen, R.N.; Nice, E.C.; Burgess, A.W.; et al. The Crystal Structure of a Truncated ErbB2 Ectodomain Reveals an Active Conformation, Poised to Interact with Other ErbB Receptors. Mol. Cell 2003, 11, 495–505.
- Citri, A.; Skaria, K.B.; Yarden, Y. The deaf and the dumb: The biology of ErbB-2 and ErbB-3. Exp. Cell Res. 2003, 284, 54–65.
- Yarden, Y.; Sliwkowski, M.X. Untangling the ErbB signalling network. Nat. Rev. Mol. Cell Biol. 2001, 2, 127–137.
- Zhou, S.; Shoelson, S.E.; Chaudhuri, M.; Gish, G.; Pawson, T.; Haser, W.G.; King, F.; Roberts, T.; Ratnofsky, S.; Lechleider, R.J.; et al. SH2 domains recognize specific phosphopeptide sequences. Cell 1993, 72, 767–778.
- Zhang, H.; Berezov, A.; Wang, Q.; Zhang, G.; Drebin, J.; Murali, R.; Greene, M.I. ErbB receptors: From oncogenes to targeted cancer therapies. J. Clin. Investig. 2007, 117, 2051–2058.
- Srinivasan, R.; Benton, E.; McCormick, F.; Thomas, H.; Gullick, W. Expression of the c-erbB-3/HER-3 and c-erbB-4/HER-4 growth factor receptors and their ligands, neuregulin-1 alpha, neuregulin-1 beta, and betacellulin, in normal endometrium and endometrial cancer. Clin. Cancer Res. 1999, 5, 2877–2883.
- Ejskjaer, K.; Sørensen, B.; Poulsen, S.; Mogensen, O.; Forman, A.; Nexø, E. Expression of the epidermal growth factor system in human endometrium during the menstrual cycle. Mol. Hum. Reprod. 2005, 11, 543–551.
- Ejskjaer, K.; Sorensen, B.S.; Poulsen, S.S.; Forman, A.; Nexo, E.; Mogensen, O. Expression of the epidermal growth factor system in endometrioid endometrial cancer. Gynecol. Oncol. 2007, 104, 158–167.
- Brys, M.; Semczuk, A.; Rechberger, T.; Krajewska, W.M. Expression of erbB-1 and erbB-2 genes in normal and pathological human endometrium. Oncol. Rep. 2007, 18, 261–265.
- Reinartz, J.; George, E.; Lindgren, B.; Niehans, G. Expression of p53, transforming growth factor alpha, epidermal growth factor receptor, and c-erbB-2 in endometrial carcinoma and correlation with survival and known predictors of survival. Hum. Pathol. 1994, 25, 1075–1083.
- Khalifa, M.A.; Mannel, R.S.; Haraway, S.D.; Walker, J.; Min, K.-W. Expression of EGFR, HER-2/neu, P53, and PCNA in Endometrioid, Serous Papillary, and Clear Cell Endometrial Adenocarcinomas. Gynecol. Oncol. 1994, 53, 84–92.
- Scambia, G.; Panici, P.B.; Ferrandina, G.; Battaglia, F.; Distefano, M.; D'Andrea, G.; De Vincenzo, R.; Maneschi, F.; Ranelletti, F.O.; Mancuso, S. Significance of epidermal growth factor receptor expression in primary human endometrial cancer. Int. J. Cancer 2007, 56, 26–30.
- Niikura, H.; Sasano, H.; Kaga, K.; Sato, S.; Yajima, A. Expression of epidermal growth factor family proteins and epidermal growth factor receptor in human endometrium. Hum. Pathol. 1996, 27, 282–289.
- Konecny, G.E.; Santos, L.; Winterhoff, B.; Hatmal, M.; Keeney, G.L.; Mariani, A.; Jones, M.; Neuper, C.; Thomas, B.; Muderspach, L.; et al. HER2 gene amplification and EGFR expression in a large cohort of surgically staged patients with nonendometrioid (type II) endometrial cancer. Br. J. Cancer 2008, 100, 89–95.
- Adonakis, G.; Androutsopoulos, G.; Koumoundourou, D.; Liava, A.; Ravazoula, P.; Kourounis, G. Expression of the epidermal growth factor system in endometrial cancer. Eur. J. Gynaecol. Oncol. 2008, 29, 450–454.
- Adonakis, G.; Androutsopoulos, G. The role of ErbB receptors in endometrial cancer. In Cancer of the Uterine Endometrium—Advances and Controversies; Saldivar, J.S., Ed.; IntechOpen: London, UK, 2012; pp. 23–38.
- Androutsopoulos, G.; Adonakis, G.; Gkermpesi, M.; Gkogkos, P.; Ravazoula, P.; Kourounis, G. Expression of the epidermal growth factor system in endometrial cancer after adjuvant tamoxifen treatment for breast cancer. Eur. J. Gynaecol. Oncol. 2006, 27, 490–494.
- Reyes, H.D.; Thiel, K.W.; Carlson, M.J.; Meng, X.; Yang, S.; Stephan, J.-M.; Leslie, K.K. Comprehensive profiling of EGFR/HER receptors for personalized treatment of gynecologic cancers. Mol. Diagn. Ther. 2014, 18, 137–151.
- Androutsopoulos, G.; Michail, G.; Adonakis, G.; Decavalas, G. Molecular mechanisms, expression and clinical role of ErbB receptors in endometrial cancer. Int. J. Clin. Ther. Diagn. 2015, S1, 28–32.
- Michail, G.; Styliara, I.; Panas, P.; Markatos, F.; Koumoundourou, D.; Ravazoula, P.; Adonakis, G.; Androutsopoulos, G. EP472 ErbB receptors profile in non-selected patients with endometrial cancer. Int. J. Gynecol. Cancer 2019, 29, A298–A299.
- Styliara, I.; Zarogianni, E.; Panas, P.; Michail, G.; Koumoundourou, D.; Ravazoula, P.; Adonakis, G.; Androutsopoulos, G. 299 EGF System receptors profiling in various histologic subgroups of endometrial cancer. Eur. J. Obstet. Gynecol. Reprod. Biol. 2022, 270, e85–e86.
- Androutsopoulos, G.; Adonakis, G.; Liava, A.; Ravazoula, P.; Decavalas, G. Expression and potential role of ErbB receptors in type II endometrial cancer. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 168, 204–208.
- Michail, G.; Panas, P.; Markatos, F.; Styliara, I.; Koumoundourou, D.; Ravazoula, P.; Adonakis, G.; Androutsopoulos, G. ErbB receptors profiling in selected patients with type II endometrial cancer. Int. J. Gynecol. Cancer 2019, 29 (Suppl. S4), A299.
- Zarogianni, E.; Panas, P.; Styliara, I.; Michail, G.; Koumoundourou, D.; Ravazoula, P.; Adonakis, G.; Androutsopoulos, G. EGF system receptors status in aggressive subtypes of endometrial cancer. Eur. J. Obstet. Gynecol. Reprod. Biol. 2022, 270, e86.
- Morrison, C.; Zanagnolo, V.; Ramirez, N.; Cohn, D.; Kelbick, N.; Copeland, L.; Maxwell, G.; Fowler, J. HER-2 is an independent prognostic factor in endometrial cancer: Association with outcome in a large cohort of surgically staged patients. J. Clin. Oncol. 2006, 24, 2376–2385.
- Engelsen, I.; Stefansson, I.; Beroukhim, R.; Sellers, W.; Meyerson, M.; Akslen, L.; Salvesen, H. HER-2/neu expression is associated with high tumor cell proliferation and aggressive phenotype in a population based patient series of endometrial carcinomas. Int. J. Oncol. 2008, 32, 307–316.
- Coronado, P.; Vidart, J.; Lopez-asenjo, J.; Fasero, M.; Furio-bacete, V.; Magrina, J.; Escudero, M. P53 overexpression predicts endometrial carcinoma recurrence better than HER-2/neu overexpression. Eur. J. Obstet. Gynecol. Reprod. Biol. 2001, 98, 103–108.
- Halperin, R.; Zehavi, S.; Habler, L.; Hadas, E.; Bukovsky, I.; Schneider, D. Comparative immunohistochemical study of endometrioid and serous papillary carcinoma of endometrium. Eur. J. Gynaecol. Oncol. 2001, 22, 122–126.
- Santin, A.; Bellone, S.; Siegel, E.; Palmieri, M.; Thomas, M.; Cannon, M.; Kay, H.; Roman, J.; Burnett, A.; Pecorelli, S. Racial differences in the overexpression of epidermal growth factor type II receptor (HER2/neu): A major prognostic indicator in uterine serous papillary cancer. Am. J. Obstet. Gynecol. 2005, 192, 813–818.
- Santin, A.; Bellone, S.; Van Stedum, S.; Bushen, W.; Palmieri, M.; Siegel, E.; De Las Casas, L.; Roman, J.; Burnett, A.; Pecorelli, S. Amplification of c-erbB2 oncogene: A major prognostic indicator in uterine serous papillary carcinoma. Cancer 2005, 104, 1391–1397.
- Slomovitz, B.; Broaddus, R.; Burke, T.; Sneige, N.; Soliman, P.; Wu, W.; Sun, C.; Munsell, M.; Gershenson, D.; Lu, K. Her-2/neu overexpression and amplification in uterine papillary serous carcinoma. J. Clin. Oncol. 2004, 22, 3126–3132.
- Grushko, T.; Filiaci, V.; Mundt, A.; Ridderstrale, K.; Olopade, O.; Fleming, G. An exploratory analysis of HER-2 amplification and overexpression in advanced endometrial carcinoma: A Gynecologic Oncology Group study. Gynecol. Oncol. 2008, 108, 3–9.
- Lukes, A.; Kohler, M.; Pieper, C.; Kerns, B.; Bentley, R.; Rodriguez, G.; Soper, J.; Clarke-Pearson, D.; Bast, R., Jr.; Berchuck, A. Multivariable analysis of DNA ploidy, p53, and HER-2/neu as prognostic factors in endometrial cancer. Cancer 1994, 73, 2380–2385.
- Odicino, F.; Bignotti, E.; Rossi, E.; Pasinetti, B.; Tassi, R.; Donzelli, C.; Falchetti, M.; Fontana, P.; Grigolato, P.; Pecorelli, S. HER-2/neu overexpression and amplification in uterine serous papillary carcinoma: Comparative analysis of immunohistochemistry, real-time reverse transcription-polymerase chain reaction, and fluorescence in situ hybridization. Int. J. Gynecol. Cancer 2008, 18, 14–21.
- Togami, S.; Sasajima, Y.; Oi, T.; Ishikawa, M.; Onda, T.; Ikeda, S.; Kato, T.; Tsuda, H.; Kasamatsu, T. Clinicopathological and prognostic impact of human epidermal growth factor receptor type 2 (HER2) and hormone receptor expression in uterine papillary serous carcinoma. Cancer Sci. 2012, 103, 926–932.
- Díaz-Montes, T.; Ji, H.; Smith Sehdev, A.; Zahurak, M.; Kurman, R.; Armstrong, D.; Bristow, R. Clinical significance of Her-2/neu overexpression in uterine serous carcinoma. Gynecol. Oncol. 2006, 100, 139–144.
- Androutsopoulos, G.; Styliara, I.; Zarogianni, E.; Michail, G.; Adonakis, G. Is it time to reconsider the clinical role of ErbB targeted therapy in endometrial cancer? In Endometrial Cancer; Farghaly, S., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2022; pp. 299–327.
