1. Introduction
Infectious diseases caused by microbes are a significant global health concern, with an escalating economic burden. The continuous increase in antibiotic resistance rates, which continues to accelerate annually
[1], highlights the urgent need for innovative solutions. This concerning trend has resulted in higher mortality and morbidity rates among patients, leading the World Health Organization (WHO) to designate antimicrobial resistance as one of the top three global health challenges
[2]. The consequences of antimicrobial resistance extend across various aspects of healthcare, including cancer treatment, premature infant care, transplantation, and surgical procedures. All of these are susceptible to severe disruptions without effective strategies to combat drug-resistant bacteria
[3]. The challenges of rising treatment costs, prolonged treatment durations, and increased mortality rates due to antimicrobial resistance require the exploration of effective alternatives
[4]. Efforts to address these challenges involve the promotion of diverse novel approaches to regulate antimicrobial medications
[5]. These alternatives encompass a range of interventions, including vaccine development, phage therapy, immune stimulants, adjuvants, anti-treatment agents, probiotics, and their various derivatives
[6]. Pre-disease vaccination, which aims to prevent diseases by strengthening the human immunological response to bacterial infections, is also a viable strategy. However, widespread implementation faces obstacles related to cost and accessibility
[7]. Similarly, strategies like probiotics, prebiotics, symbiotics, and competitive exclusion have been established to deter pathogenic colonization but are constrained by variable efficacy and regulatory processes, such as those imposed by the Food and Drug Administration (FDA)
[8]. Furthermore, bacterial cells employ primary defense mechanisms, including enzyme function inhibition and efflux pumps, to reduce susceptibility to antibiotics
[9]. Consequently, the era of antibiotics teeters on the brink of extinction, demanding the innovation of new approaches to combat multidrug-resistant strains. Researchers are actively seeking alternative methods to address this pressing issue. In this contemporary context, nano-sized materials emerge as a promising alternative to traditional antimicrobial agents
[10]. Nanoparticles (NPs) exhibit distinctive physico-chemical properties, including their controllable small size (ranging from 1 to 100 nm), high reactivity, functionalized structure, and large surface area-to-mass ratio
[11]. Leveraging NPs as delivery vehicles for antimicrobial drugs has proven highly effective, mitigating numerous limitations associated with conventional antimicrobial therapies
[12]. Notably, microorganisms find it challenging to develop resistance to NPs because these versatile entities can concurrently target multiple cellular pathways
[13]. Consequently, NPs have the potential to replace traditional antibiotics and antifungal agents in the treatment of microorganism infections that have become resistant to standard therapies
[14]. The synthesis of NPs can be achieved through various methods, each with its own set of advantages and disadvantages. These methods encompass biological, physical, and chemical approaches
[15]. Physical methods often yield high quantities of NPs, making them attractive for large-scale production; however, they tend to be energy-intensive and cost-inefficient, which may limit their practicality for some applications
[16]. On the other hand, chemical synthesis methods are generally cost-effective and efficient in producing NPs, but they often involve the use of hazardous and volatile chemicals, which can pose environmental risks and safety concerns
[17]. In recent years, there has been a growing emphasis on environmentally friendly approaches to NPs synthesis. One such approach is green synthesis, which has garnered significant attention due to its cost-effectiveness, environmental sustainability, and relative simplicity
[18]. Green synthesis methods utilize plant extracts, bacteria, and fungi to produce NPs. Among these, plant extracts are particularly desirable as they eliminate the need for complex cell culture maintenance and downstream processing
[19]. Harnessing their exceptional chemical properties and physical characteristics, NPs offer a promising avenue for addressing the challenge posed by multidrug-resistant bacteria. In today’s challenging landscape of antibiotic resistance, the need for effective strategies to combat multidrug-resistant strains has reached a critical juncture. This pressing issue necessitates a comprehensive approach that spans research, development, and implementation.
2. Antibiotic Resistance
Antibiotics are used to combat microbial infections, utilizing a range of mechanisms such as inhibiting enzymes, altering membrane structure, and disrupting transcription and translation processes
[15]. However, some microbes have evolved to become resistant to antibiotics, posing a significant challenge to the efficacy of antimicrobial agents
[20]. Antibiotic resistance is a primary factor contributing to increased drug dosages, extended hospital stays, heightened toxicity, and elevated mortality rates
[21]. Multiple factors, such as the over-prescription, misuse, and excessive use of antibiotics, including their extensive use in agriculture, and the scarcity of new antibiotics, all contribute to the occurrence of antibiotic resistance
[22].
Antibiotic resistance is a formidable challenge in combating microbial infections caused by bacteria. However, it is important to note that antibiotic resistance is not confined solely to bacteria. Fungi, another category of microorganisms, can also develop mechanisms of resistance to antimicrobial agents
[23].
TWhile the primary focus of this section remains on bacterial resistance, the emergence of antifungal resistance is a significant concern in medical and agricultural settings. Fungi, like bacteria, can adapt and develop various strategies to resist the effects of antifungal drugs, posing additional complexities in the fight against microbial infections.
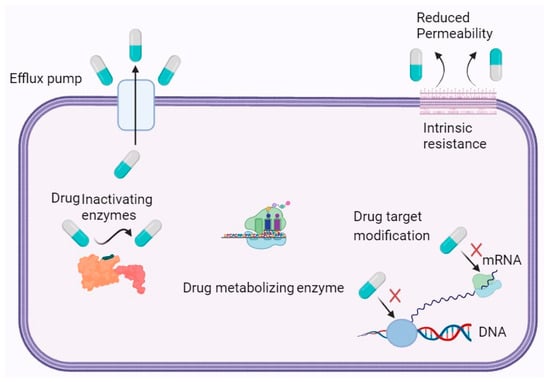
Bacteria employ multiple mechanisms for developing resistance to antibiotics, including intrinsic mechanisms that are typically genetically transmitted. Unlike eukaryotic cells, bacteria are prokaryotes and lack a nuclear membrane to protect their genetic material
[24][25]. As a result, random or spontaneous mutations can occur frequently in the bacterial genome due to the exposed nature of their genetic material. Excessive exposure of a bacterial strain to a particular antibiotic can lead to genetic mutations that give rise to a novel protein that helps the bacterium fight against the antibiotic
[25][26]. The bacterium employs various methods to develop intrinsic resistance to antibiotics, and one of these methods involves utilizing newly produced proteins as tools for survival
[26][27]. Specifically, the protein functions by obstructing the intake of the antibiotic into a bacterial cell, thereby conferring resistance
[27][28]. This involves the production of a mutated protein that triggers the efflux pump present in the bacterial cellular wall. As a result, after antibiotics enter a cell, they are recognized by the altered protein and subsequently pumped out from the cell through an efflux pump
[28][29]. Additionally, bacteria can utilize an enzymatic reaction to inactivate antibiotics as another intrinsic resistance strategy
[29][30]. Bacteria can acquire antibiotic resistance through various mechanisms, such as modifying the antibiotic target, evading the antibiotic target, and vertical gene transfer
[30][31]. The adaptive mechanism employed by bacteria during an infection involves the development of biofilms, which play a crucial role in their survival
[31][32]. Biofilms, complex communities of microorganisms, also interact with their environment. In the case of metalloids and heavy metals, these biofilms can act as both a protective barrier and a source of resistance. They have been found to sequester and immobilize metalloids and heavy metals, reducing their toxicity to the bacteria within the biofilm. These arrangements eventually lead to reduced buildup of antibiotics within the bacteria cells, leading to suboptimal therapeutic levels of the medication
[32][33]. Consequently, higher and more frequent doses of antibiotics may be necessary, which can have dangerous effects on humans and animals.
Figure 1 illustrates the mechanism of antibiotic resistance.
Figure 1. Schematic Illustration of Antibiotic Resistance Mechanisms in Bacteria.
3. Mechanistic Action of Metallic Nanoparticles against Bacteria
NPs exhibit unique properties that set them apart from their macroscopic counterparts, making them highly effective in various applications. These properties include crystallinity, exceptional stability, reduced size, the surface plasmon resonance effect, unique shapes, and a higher surface-to-volume ratio
[33][34]. These attributes bestow upon NPs exceptional antibacterial, antifungal, larvicidal, and antiprotozoal capabilities. Specifically, NPs’ distinct size, crystal structure, and reduced dimensions position them as superior alternatives to existing antibiotics, offering potential relief from the burden of antibiotic resistance
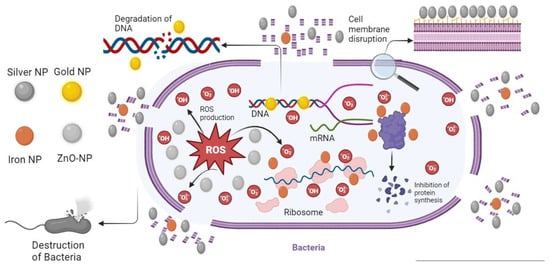
[34][35]. Additionally, NPs demonstrate manageable morphologies and excellent size uniformity. Anisotropy, a crucial property of NPs, implies that different facets of their crystal structure possess distinct reactivity. The optical properties of metallic salts transform when converted into nanoforms, enabling significant customization of these characteristics. These remarkable property alterations, combined with the ability to tailor them to specific requirements, establish NPs as a highly promising avenue for addressing antibiotic resistance. Moreover, these changes in properties, coupled with the potential for tailoring them to specific needs, position NPs as a highly promising avenue for addressing antibiotic resistance. Several factors contribute to the antibacterial activity of NPs against bacteria. These include their large surface area that comes into contact with bacteria, electrostatic interactions, or hydrophobic interactions
[35][36]. NPs that interfere with bacteria typically cause oxidative stress, enzyme inhibition, protein degradation, and changes in gene expression
[36][37]. Nevertheless, oxidative stress, metal release, and non-oxidative pathways are the most common antibacterial mechanisms
[37][38]. Among these mechanisms, Reactive Oxygen Species (ROS)-induced oxidative stress stands out as one of the main mechanisms supporting NPs in their antibacterial operation. In bacteria, ROS is produced primarily through aerobic respiration and is balanced by antioxidant cell machinery; however, an excess of ROS can lead to more significant insults, biomolecular oxidation, and cellular damage
[38][39]. Further, when investigating the generation of ROS by NPs, it is crucial to consider the environmental conditions under which this phenomenon occurs. ROS generation by NPs can vary significantly depending on whether it takes place under light or dark conditions
[39][40]. NPs can be viewed as reactive centers, particularly when exposed to electromagnetic activation, such as light conditions, which can greatly influence their intensity and kinetics. Metal ions are slowly released by metal oxides that are carried by the cell to the inner cell, where they interact with protein and nucleic acid functional groups
[40][41]. This interface alters the composition of the holding cell, blocks the enzyme, and intervenes with normal bacteria in the cellular processes
[41][42]. NPs that interfere with the bacteria cell wall create a focal stream of ions that continually emit ions and cause more toxicity to the cells
[42][43]. The surface morphological features of NPs have fundamental effects on their behavior, and where the surface of the NPs is rougher; dissolution happens more rapidly
[43][44]. The association of NPs with the cell wall is implicated in non-oxidative processes. In bacteria, the protective barriers to environmental defense are the cell membrane and cell wall. Different adsorbent ways for NPs are given by the components of the cell membrane and component
[44][45]. The Gram-negative bacterial cell wall comprises phospholipids, lipopolysaccharides, and lipoproteins, forming a barrier only for certain macro molecules to enter
[45][46]. The cell wall consists of a thin layer of peptidoglycans and abundant pores for the Gram-positive bacteria, which allow foreign molecules to penetrate contributing to the binding to proteins and other cellular components covalently that disrupt bacterial cell activity
[46][47]. Lipid polysaccharides on the Gram-negative bacterial cell wall create regions that are negatively charged and attract NPs
[47][48], and because teichoic acid is expressed only in Gram-positive bacterial strains, NPs are spread throughout the phosphate chain. The result is, therefore, more pronounced in Gram-positive bacteria more than that of Gram-negative bacteria
[36][37]. As an example, Zinc oxide NPs (ZnONPs) were synthesized, and their antimicrobial activity against various bacteria was assessed. The results revealed a reliance on the structure and composition of the bacterial cell wall for the antibacterial effects. These NPs exhibited more potent antibacterial activity against Gram-positive bacteria, while specific components appeared to resist the adhesion of ZnONP to bacterial membranes
[48][49].
Figure 2 show the mechanistic approach of NPs against bacteria.
Figure 2. Illustration of the Mechanistic Approach of NPs Against Bacteria.
3.1. Effect of Silver NPs against Bacteria
Among different types of NPs, silver NPs (AgNPs) stand out as one of the most potent antimicrobial agents
[49][50]. When considering the antimicrobial properties of AgNPs, it is vital to delve into the diverse mechanisms underlying their effectiveness. These mechanisms are influenced not only by NPs size but also by a spectrum of experimental conditions, including dissolution kinetics, pH levels in the medium, solution ionic strength, media composition, and the specific microorganisms or biofilms encountered. One fundamental aspect of AgNPs’ antimicrobial action is their interaction with the microbial cell membrane. AgNPs possess a remarkable ability to disrupt membrane permeability and interfere with cellular respiration
[50][51]. This disruption stems from AgNPs binding to and penetrating the cell membrane, ultimately affecting its integrity. Additionally, AgNPs can engage with thiol groups within microbial cells. This interaction has the potential to trigger the production of ROS, which can disrupt respiratory enzymes, ultimately leading to microbial cell death
[50][51]. For example, the bactericidal effect of AgNPs against
Escherichia coli has been observed, potentially attributed to the disruption of the proton motive force
[51][52]. The mode of action of AgNPs, although explored over the last few decades, remains a topic of ongoing research and discussion. Some studies suggest that AgNPs kill or inhibit the growth of resistant bacteria by producing Ag
+ ions. In this scenario, a redox reaction occurs within the bacterial cell when exposed to AgNPs, resulting in the production of silver ions due to their oxidation
[52][53]. These silver ions then alter the macromolecules which lead to the growth inhibition of bacteria. AgNPs tend to affect bacteria directly rather than producing silver ions. It is proposed that AgNPs degrade the cell membrane of bacteria as they are positively charged and have a higher affinity towards the negatively charged peptidoglycan layer of the bacterial cell membrane
[53][54]. It has also been concluded from some research studies that AgNPs react with the DNA of bacteria and control its replication
[50][51]. When AgNPs penetrate through the cell membrane of bacteria, they release silver ions, and these silver ions turn the DNA into its condensed form and alter its replication process
[54][55]. This alteration results in no replication, and cell death occurs as an endpoint of this reaction.
3.2. Effect of Zinc Oxide NPs against Bacteria
To combat the problem of antibiotic resistance, ZnONPs have shown promise in different applications, particularly in combating antibiotic resistance
[55][56]. Among the properties of ZnONP, chemical sensing, semiconducting, electric conductivity, and piezoelectric are included
[56][57]. ZnONP exhibits biocompatibility with human cells and demonstrates efficacy as an antimicrobial agent. For instance,
Bacillus subtilis and
Staphylococcus aureus, which are Gram-positive bacteria, werefound to be sensitive to ZnONP
[57][58].
Escherichia coli,
Pseudomonas aeruginosa, and
Campylobacter jejuni are some Gram-negative bacteria found to be influenced by ZnONP
[58][59]. The antibacterial potential of ZnONP depends on their particle size, morphology, and concentration
[59][60]. Various ZnONP structures have significantly different antibacterial activities. ZnONP of rods and wires shape can easily discern bacterial cell walls as compared to spherical shapes
[60][61]. Flower-shaped particles have been proven to be more efficient than both rod and spherical shaped when observed against
Staphylococcus aureus and
Escherichia coli [59][60]. Enhanced antibacterial activity of ZnONP with a larger surface area and increased concentration was reported
[61][62]. ZnONP of smaller sizes have been proven to be comparatively more effective in their antibacterial activities because of their enhanced potential for penetration
[62][63]. The ZnONP activity was also checked against
Staphylococcus aureus and
Escherichia coli and effective results were concluded due to their smaller particle size
[63][64]. These size-dependent activities of ZnONP were the same for both Gram-positive and Gram-negative strains
[64][65]. The concentration of ZnONP has noticeable effects on antibacterial activity
[65][66]. A higher concentration of ZnONP can have enhanced antibacterial activity to increase cell death
[66][67]. In a research study, 80 nm-sized ZnONP was employed to combat
Escherichia coli. The findings revealed that the suppression of bacterial growth was more pronounced at higher concentrations of ZnONP compared to lower concentrations
[67][68].
Studies about mechanisms through which ZnONP kills bacteria and acts as an antibacterial agents are very limited. Various modes of action of ZnONP are concluded from different research studies. Like AgNPs, ZnONP also inhibits the growth of bacteria by generating ROS such as hydrogen peroxides and hydroxyl radicals’ induction. ROS itself is the cause of different antibacterial mechanisms. For example, ZnO localized interaction causes cell wall damage
[68][69]. ZnONPs exhibit another mode of action by altering the cell membrane and permeability of bacteria. Additionally, they function as carriers of zinc ions, releasing these ions through an oxidation reaction once they enter the bacterial cell. These released zinc ions are highly toxic to bacteria, particularly by weakening the mitochondria
[69][70]. ROS alters several macromolecules in the cell. As a result, they express the oxidative stress gene causing growth inhibition and cell death
[70][71]. ZnONPs are used in the food packaging industries for their potential of incorporation into packaging materials as they release NPs against bacteria and prevent foodborne diseases
[71][72].
3.3. Effect of Gold NPs against Bacteria
Gold NPs (AuNPs) are a comparatively a more unique type of NPs. AuNPs have various clinical applications in the field of nanomedicine against different strains of bacteria
[72][73]. AuNPs synthesis can be achieved through various techniques, including physical methods such as laser ablation, chemical methods involving chloroauric acid, and biological approaches utilizing plants and their extracts. Among these methods, biological approaches, also known as green synthesis, are favored for their reduced environmental impact, cost-effectiveness, and ease of handling
[73][74]. AuNPs hold the potential for conjugation with other antibiotics, antibacterial peptides, and specific antigens
[74][75]. While AuNPs do exhibit antibacterial properties, they may not be as effective against bacteria as AgNPs
[75][76], However, it is worth noting that they can be cytotoxic and genotoxic to animal cells
[76][77]. AuNPs exhibit low toxicity and greater cytocompatibility when compared to AgNPs. Antibacterial activities of AuNPs against both Gram-negative and Gram-positive strains of bacteria have been investigated
[77][78]. For example, a study assessed the effects of these NPs on both
Mycobacterium tuberculosis and
Escherichia coli, revealing significant antibacterial activity against Gram-positive
Mycobacterium tuberculosis and Gram-negative
Escherichia coli [78][79]. Research into the antibacterial mechanisms of AuNPs is currently a topic of active investigation. A study reported that AuNPs can interact with the cell membranes of
Escherichia coli, leading to membrane degradation and growth inhibition. They also observed that the inhibitory effects varied with changes in surface modification agents
[79][80]. AuNPs with similar size and shape caused cell lysis when PAH (Poly-allylamine hydrochloride) was capped, but not in the case of citrate-capped particles
[80][81]. One of the modes of action of AuNPs involves interaction with cellular barriers, such as cell walls and cell membranes
[81][82]. Another study highlighted distinct interactions between AuNPs and the protective barriers of Gram-negative strains when compared to Gram-positive bacteria. In this investigation, it was observed that Au-DAPT-coated gold nanoparticles significantly increased the permeability of
E. coli membranes by 70%, leading to nucleic acid leakage. In contrast, the impact on
P. aeruginosa was measured at 42%
[82][83]. AuNPs can also affect bacteria by interacting with various biological substances like DNA and proteins, inhibiting enzyme activity and neutralizing plasmid movement within bacteria
[83][84]. For instance, it was proposed that AuNPs induce DNA fragmentation, ultimately resulting in bacterial cell death
[84][85]. Another study suggested that the photothermal effect of AuNPs is one of their convincing mechanisms, converting infrared light energy into local heat
[85][86]. This heat causes protein denaturation, cell fluid evaporation, and structural breakdown, resulting in bacterial growth inhibition or death. Furthermore, AuNPs can cause a redox imbalance, similar to AgNPs, by generating ROS that induce cell apoptosis and necrosis. They have the potential to disrupt the macromolecules of bacteria, leading to oxidative stress
[86][87]. Despite these findings, further research is needed to fully uncover the antibacterial mode of action of AuNPs.
3.4. Effect of Iron NPs against Bacteria
Like other metallic NPs, iron NPs (FeNPs) have demonstrated their antimicrobial efficacy against numerous pathogenic bacteria, suggesting their potential for use in combating microbial infections
[87][88]. FeNPs are significantly cost-effective compared to silver and gold NPs
[88][89], they are also slightly preferred over silver and other NPs as they are less toxic to humans unlike other NPs, especially silver, which can cause cytotoxicity in various human cell lines
[89][90]. FeNPs are considered less toxic and therefore, Ferumoxytol an intravenous Fe
3O
4 formulation, was also approved by the FDA as a treatment for iron-deficient patients. Apart from being less toxic, the byproduct of FeO-NPs, which is iron, can be stored by the body
[90][91]. Antibacterial activities of NPs vary from 80–100 nm of semi-crystalline biogenic Fe
3O
4 that have been developed from leaf extract of
T. procumbens and revealed to have bactericidal activity against Gram-negative bacterium
P. aeruginosa [91][92]. Similarly, it was reported that Fe
2O
3−NPs produced from
Skimmia laureola exhibit the highest antibacterial activity against
Ralstonia solanacearum by degrading the cell wall
[92][93]. In another study, it was suggested that rod-shaped FeNPs produced using
Eichhornia crassipes leaf extract exhibited the highest inhibition against
Pseudomonas fluorescens and
Staphylococcus aureus [93][94]. FeNPs synthesized from
Gardenia jasminoides and
Lawsonia inermis, were tested against
Staphylococcus aureus, and displayed a noticeable increase in the inhibition zone, going from 15 mm to 16 mm
[94][95]. The antimicrobial activity of FeNPs depends on its surface coating. Different NPs have a different surface coating, and this property of NPs seems to play a huge role in their antibacterial efficiency
[95][96]. FeNPs are used as antimicrobial agents and are usually capped with other compounds, such as alginate, for stability purposes because uncapped FeNPs are slightly unstable
[96][97][97,98]. FeNPs prove their efficacy through the presence of iron, resulting in the production of ROS, such as H
2O
2, that can damage the cell membrane once they enter the intracellular space, ultimately leading to bacterial cell death. The bactericidal activity of FeNPs is a result of the oxidative stress caused by FeNPs-generated ROS
[98][99]. Another possible mechanism of FeO-NPs is the damage of DNA through Fenton’s reaction
[99][100]. In Fenton’s reaction, the superoxide anion O- is dismutased to hydrogen peroxide H
2O
2, which releases Fe ions (in the Fe
2+ or Fe
3+ oxidation state) that can cause direct damage to bacterial DNA, leading to its growth inhibition
[100][101].