Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Alice Vilela and Version 2 by Catherine Yang.
The natural biopolymer chitin and its deacetylated derivative chitosan are abundant in nature. They are obtained from different sources, including the crustacean shells and the cell walls of fungi. Chitin and chitosan have various applications in the beverage industry, such as a flocculent to improve the clarification process, reduce metals and contaminates, and extend shelf-life. They are also used as material for immobilizing microorganisms and enzymes, allowing bioprocesses to develop that preserve the quality of alcoholic and non-alcoholic beverages.
- clarification agent
- contaminants reduction
- antioxidant activity
- antimicrobial activity
- immobilization
1. Introduction
Chitosan (deacetylated chitin) has been gaining increasing attention due to its biodegradability, renewability, nontoxicity, and non-allergenic properties in the case of fungal chitosan [1]. A vast quantity of living organisms synthesizes chitin and occurs as a building material in the cell walls of fungi and yeast or the exoskeleton of arthropods, crustaceans (shrimp and crabs), insects (beetles, ants, brachiopods, scorpions, and cockroaches) as well as in algae (green algae and brown algae) [2][3][2,3]. Chitosan is generally considered safe (GRAS) by the Food and Drug Administration (FDA). Chitosan has been described as having several applications in the beverage industry (alcoholic and non-alcoholic beverages), such as a chelating agent for metal ions, an antioxidant, antimicrobial, flocculant, and a clarifying agent [4][5][6][4,5,6]. Chitosan and chitin have been applied as flocculants [7]. They can be used for clarification of non-alcoholic and alcoholic beverages, such as fruit juices [8][9][10][11][8,9,10,11] and beer [4], since these increase the suspended particles sedimentation rate and remove particles that could, for example, impact in color and limpidity. Only fungal chitosan is authorized by the European authorities and by the International Organisation of Vine and Wine to be used in wines as a fining agent for clarification, reduction of heavy metals, prevention of iron and copper haze, reduction of the contaminant contents (particularly ochratoxin A), and for antimicrobial action (namely, Brettanomyces spp.) [12]. Several authors have studied the application of chitosan in wine, for example, to wine protein stabilization [13][14][15][16][13,14,15,16], for avoiding wine oxidation [17][18][19][20][17,18,19,20], as an antimicrobial agent [21][22][23][24][25][21,22,23,24,25], and for removing volatile phenolic compounds from red wine [26][27][28][26,27,28]. Recently, Castro-Marín et al. [29] published a review article concerning the different applications of chitosan in winemaking and summarizing the chemical mechanisms underlying its action.
The amino groups of chitosan are protonated in acidic environments, and chitosan is expected to show a performance characteristic of a polyelectrolyte [4]. Electrostatic interaction between chitosan (positively charged) and the acidic protein pepsin (enzyme) is dependent on pH values [30]. Moreover, the interaction between chitosan and pectin has been studied by Marudova et al. [31], describing the action of chitosan as an effective crosslinker at pH 5.6 and exhibiting gel behavior dependent on the pectin esterification degree. It has also been shown that the complexation of chitosan with alginate, pectin, or carrageenan produces coagulating agents with enhanced protein adsorption and a more significant limpidity increase than only chitosan application [32].
2. Chitin and Chitosan Structural Properties
Chitosan is a natural polysaccharide containing glucosamine and N-acetylglucosamine monomeric units. Chitin is produced by chemical treatments involving the extraction by acid treatment to dissolve the calcium carbonate, named demineralization, followed by the alkaline solution to dissolve proteins, called deproteinization [2][33][2,33]. The chitin extraction could also be performed by biological treatments using enzymes and microorganisms [34], namely proteolytic enzymes to digest the proteins, or a fermentation process using microorganisms, which permits the digestion of proteins and minerals [35][36][35,36]. Decolorization is also frequently performed to eliminate pigments to obtain colorless pure chitin [33]. This process should be adjusted according to the chitin source due to the diversity in the ultrastructure of the chitin material obtained from the different sources [33]. Chitosan can then be obtained by partial deacetylation of chitin through hydrolysis by a chitin deacetylase [37][38][39][37,38,39] or by a chemical procedure [40]. From a chemical point of view, both acids and alkalis can be used to deacetylate chitin; nevertheless, glycosidic bonds are highly vulnerable to an acid; thus, alkali deacetylation is more frequently used [41]. At an industrial scale, deacetylation is usually a nonenzymatic process whereby chitosan is obtained by removing R-NHCOCH3 residue by treating it with solid alkali (sodium hydroxide solution 40–50%) at high temperatures (100 °C) [40]. Deacetylation describes a reaction that removes an acetyl functional group. When deacetylation (expressed as a molar percentage) is higher than 50 mol%, the biopolymer becomes soluble in acidic aqueous solutions. It is called chitosan and behaves as a cationic polyelectrolyte due to the protonation of amine groups in the presence of H+ ions [42]. Therefore, chitosan—poly-β-(1,4)-d-glucosamine is a deacetylated cationic linear biopolymer of chitin—poly-β-(1,4)-N-acetyl-d-glucosamine (Figure 1), one of the most abundant natural polysaccharides found in the composition of fungi cell walls [43][44][45][43,44,45] and exoskeletons of insects [46][47][48][49][46,47,48,49], cephalopods, and crustaceans [49][50][51][52][53][54][49,50,51,52,53,54]. Chitosan is a linear polysaccharide composed of randomly distributed β-(1–4)-linked d-glucosamine and N-acetyl-d-glucosamine units. The ratio between these two monomeric units, expressed as the degree of acetylation or deacetylation, ranges from 40 to 99% (degree of deacetylation), and the molar weight ranges from 2 to more than 200 kDa [55]. The chitosan functional proprieties depend on structural characteristics, for instance, degree of deacetylation, molecular weight, and purity. Various chitosans are available commercially, which differ primarily in the degree of deacetylation and molecular weight.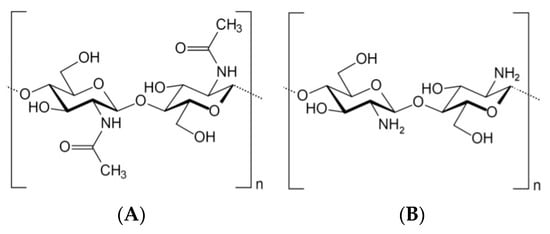
Figure 1.
Chitin (
A
) and chitosan (
B
) chemical structure.
3. Chitosan Applications in Beverages
3.1. Clarification Agent, Metals and Contaminants Reduction
In beverages like fruit juice, clarification is an essential step in the production process, mainly to remove pectins and other compounds in the fluid. Juice limpidity and homogeneity are the two significant characteristics of clarified juices and are achieved by eliminating all suspended solids [58]. Chitosan coagulates the anionic suspended particles such as pectin and protein, and consequently, their separation from beverages is fast, and their turbidity decreases [6][10][59][60][6,10,59,60]. This behavior is related to the chitosan physicochemical properties associated with the presence of amine functions [31]. According to Rizzo et al. [61], the concentration of chitosan used in the clarification process and pH, in addition to the initial juice turbidity to be treated, are essential variables for the coagulation process using chitosan and, consequently, to reach the desired limpidity. Domingues et al. [62] also showed that besides the chitosan concentration, the pH value is an essential factor for the juice turbidity to decrease, as all samples of passion fruit juice at pH 6 presented higher turbidity removal after the treatment with chitosan. Rao et al. [6] also showed that the optimal pH for green tea clarification using chitosan was at pH 5.5. It was observed to be concentration-dependent, leading to higher turbidity reduction in juices when chitosan content was increased [60][62][60,62]. It was also shown that chitosan with a higher deacetylation degree is more effective in protein flocculating; this could be related to the augmented charge density from the free amino groups [63].3.2. Extending the Shelf-Life
There is an increase in consumer demand for safe and healthier products; in this way, the consumption of freshly processed products has been observed, and the beverage industries need to search for compounds to extend the shelf life of these products by applying natural products. The antimicrobial and antioxidant activity of chitosan permits its application to open the beverage shelf life, as shelf life is constrained by microbial spoilage and oxidation [57][64][65][66][57,82,83,84]. The antioxidant activity of chitosan is related to the scavenging effect on free radicals [65][67][68][83,85,86]. Chitosan deacetylation degree influenced its antioxidant activity, and this activity rises with unsubstituted amino groups [57]. Therefore, the antioxidant activities of chitosan increased with an increase in deacetylation degree [69][87]. The molecular weight of chitosan has also been described to have a primary effect on its antioxidant activities; with low molecular weight (16 to 190, 127 kDa), chitosans have more marked scavenging effects on superoxide and hydroxyl radicals than those with high molecular weight (>300 kDa), probably related to the compact structure of the chitosan with a high molecular weight that limited the antiradical activity of the hydroxyl and amino groups [70][71][88,89]. For antimicrobial activity, the most important factors are the type of microorganism [66][72][73][74][84,90,91,92], the chitosan charge density, concentration, molecular weight [66][74][84,92], hydrophilic/hydrophobic characteristics, chelating capacity, and the degree of deacetylation [66][84]. It has been suggested that the polycationic nature of chitosan that forms from acidic solutions below pH 6.5 is a crucial factor. A higher positive charge density leads to electrostatic solid interaction. Therein, the positive charge is associated with the deacetylation degree of chitosan [75][93]. The chitosan antifungal and antimicrobial activity against different fungi, Gram-positive and Gram-negative bacteria, is related to the chitosan cationic properties in an acidic media at pH values below chitosan pKa. As protonated amino groups bind to the negatively charged carboxyl groups, such as bacterial cell wall surface peptidoglycans, altering their barrier properties, leading to permeabilization and destruction of external membranes [76][94]. Therefore, Chitosan is most active at the fungi or bacteria cell surface, leading to permeabilization [90,92,95,96,97], which results in intracellular material leakage and consequently cell death [77][78][79][96,98,99].4. Chitosan Immobilization
Chitosan, like alginate, forms a gel by ionotropic gelation (or coacervation) and is a polymer with numerous applications in immobilization technology of enzymes and microorganisms due to its nontoxic, biocompatible, biodegradable, and antimicrobial properties [80][111]. Chitosan-bearing protonated amino groups can interact with a wide variety of natural or synthetic anionic species, such as negatively charged proteins, DNA [81][82][112,113], and synthetic basic polymers, such as sodium tripolyphosphate (TPP) [83][84][114,115] to form ionic complexes. Ionotropic gelation has been used in the production of polymeric (micro and nan) particles for many applications, namely in biomedicine [85][116] and the pharmaceutical industry, including interferon [86][87][88][117,118,119] and antioxidant administration [89][120]. This technique is versatile and relatively simple. It is possible to produce particles in a wide range of sizes [90][121]. This ionic gelation method to prepare Chitosan/TPP nanoparticles presents the advantages of simple operation, low equipment requirements, low cost, good repeatability, environmentally friendly, and easy large-scale preparation [91][92][122,123], Figure 2.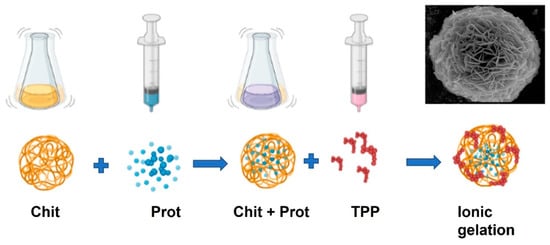
Figure 2. Schematic illustration of the preparation procedure for protein chitosan immobilization. An ionic gelation technique can prepare protein-loaded chitosan beads. Chitosan (Chit) is first mixed with protein (Prot) to provide a homogeneous solution (Chit + Prot). Then, it interacts with sodium tripolyphosphate (TPP) solution to encapsulate the protein during the ionic gelation process.
4.1. Chitosan Microorganism’s Immobilization
Microorganisms’ immobilization consists of the physical confinement of intact cells to a region of space with conservation of biological activity. The use of these methodologies for alcoholic fermentation offers many advantages over the use of the conventional free yeast cell method. The most studied methods for yeast immobilization include using organic supports, mainly alginate. Some benefits of the yeast-immobilization systems include high cell densities, product yield improvement, lowered risk of microbial contamination, better control, and reproducibility of the processes, as well as reuse of the immobilization system for batch fermentation and continuous fermentation technologies [93][94][130,131].
According to Martin and Etievant [95][132], for alcoholic beverage production, the cell carrier or immobilization matrix must fulfill specific requirements: (i) Large surface, with chemical groups favoring cells to adhere; (ii) Easy to handle and regenerate; (iii) High and retained cell viability and operational stability; (iv) Does not affect catalytic activity; (v) Uniform and controllable porosity to allow the free exchange of substrates, products, cofactors, and gases; (vi) Mechanical, chemical, thermal, and biological stability; (vii) Easy to handle, cost-effective, and amenable to scale-up immobilization technique; (viii) Does not affect product quality.
