The intestinal barrier is a precisely regulated semi-permeable physiological structure that absorbs nutrients and protects the internal environment from infiltration of pathological molecules and microorganisms. Bile acids are small molecules synthesized from cholesterol in the liver, secreted into the duodenum, and transformed to secondary or tertiary bile acids by the gut microbiota. Bile acids interact with bile acid receptors (BARs) or gut microbiota, which plays a key role in maintaining the homeostasis of the intestinal barrier.
- bile acid
- bile acid receptor
- gut microbiota
- intestinal barrier
1. Introduction
2. Bile Acids and Intestinal Barrier
The integrity of the intestinal barrier requires constant renewal of the epithelial boundary, maintenance of tight junctions (physical barrier), mucus secretion and normal gut microbiota (biochemical barrier), and a finely regulated intestinal lamina propria immune system (immune barrier). Because of the complexity and heterogeneity of the intestinal barrier, specific mechanisms underlying the dysfunction of the intestinal barrier are still far from clear. That bile acids may play a pivotal role in many aspects of the maintenance of intestinal barrier integrity is a novel concept (Figure 1).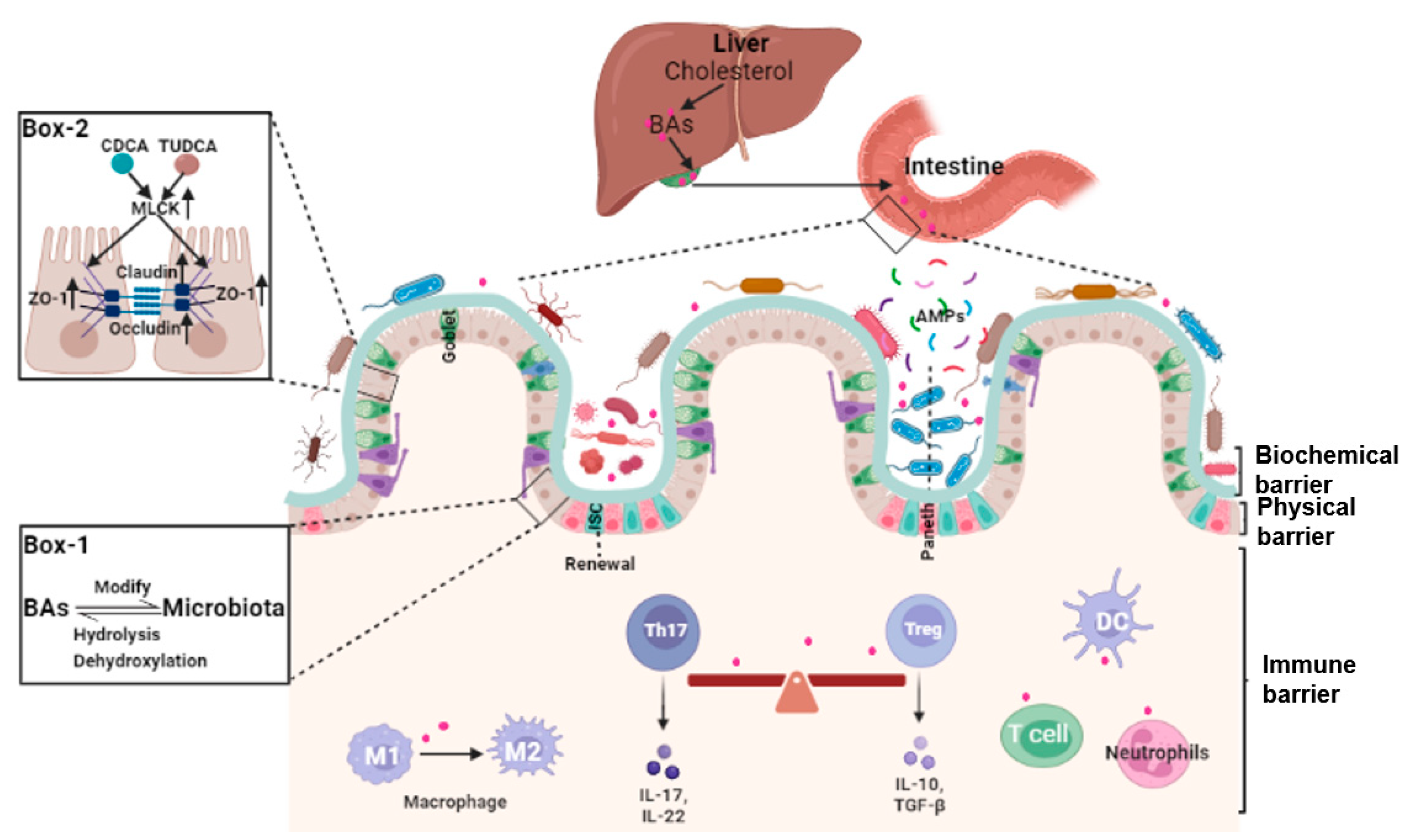
2.1. Bile Acids and Intestinal Epithelial Cells Tight Junctions
2.2. Bile Acids and Gut Microbiota
2.3. Bile Acids, Intestinal Stem Cells (ISCs), and Epithelial Injury Repair
Balance between intestinal epithelial proliferation and cell death from damage, stress, and other pathological conditions maintain normal intestinal barrier function. Intestinal epithelial cells are self-renewed every 3–5 days, in part, from Lgr5+ intestinal stem cell (ISCs) as at counteract intestinal barrier damage and different stress stimuli [30][17]. Lgr5+ ISCs replenish damaged epithelial cells and generate progenitors of goblet and Paneth cells. These cells secrete mucus and antimicrobial peptides to support the integrity of the intestinal mucus layer [31][18]. Data demonstrated that bile acids metabolism plays a potential role in the self-renewal function of Lgr5+ ISCs. Based on pathway synthesis and chemical structures, bile acids are grouped into 12α-hydroxylated (OH) bile acids and non-12α-OH bile acids [3]. What the two groups exert varies, and, at times, they have opposing effects on ISCs. For instance, the 12α-OH bile acid CA can inhibit the activity of peroxisome proliferator-activated receptor alpha (PPARα), impeding fatty acid oxidation (FAO), and the self-renewal of Lgr5+ ISCs [32][19]. An HFD-driven increase in DCA decreased ISC proliferation and differentiate into goblet cells through pathologic endoplasmic reticulum stress [33][20].2.4. Bile Acids and Intestinal Local Immune Homeostasis
The intestinal lamina propria is colonized by a variety of innate and adaptive immune cells and gut-associated lymphoid tissue and is termed the intestinal immune barrier [37,38,39][21][22][23]. Under normal conditions, this microecosystem is tightly and finely regulated. Environmental factors and the gut microbiota and their metabolites (microorganism-associated molecular patterns and pathogen-associated molecular patterns) are recognized by specific receptors (Toll-like receptors, TLRs) on immune cells, leading to intestinal immune homeostasis and self-tolerance [39][23]. Bile acids modulate immune responses in the intestine through BARs, including TGR5, FXR, VDR, CAR, and retinoic-acid-receptor-related orphan nuclear receptor-γt (RORγt). Unique lymphocyte populations function cooperatively to maintain the intestinal immune system, especially the balance between pro-inflammatory T helper 17 (Th17) cells and anti-inflammatory Treg cells [40][24]. Two derivatives of LCA, 3-oxoLCA and isoalloLCA, inhibited RORγt to suppress Th17 cell differentiation and increased the differentiation of Treg cells through the production of mitochondrial reactive oxygen species [41][25]. The secondary bile acid, isoLCA, via VDR, modulated colonic FOXP3 + Treg cells expressing RORγt and ameliorated their susceptibility to inflammatory colitis [42][26].3. Bile Acid and Intestinal-Barrier-Dysfunction-Related Diseases
3.1. Inflammatory Bowel Diseases (IBDs)
Some bile acids altered the expression of tight junction proteins and the renewal of intestinal stem cells, leading to the intestinal barrier injury, increasing the incidence of IBD [32,50][19][27]. In contrast, some other bile acids may maintain intestinal immune barrier homeostasis by activating BARs such as FXR and TGR5. The DCA-mediated activation of FXR inhibited Paneth cell function and type I interferon signaling in mice with Crohn’s disease [51][28]. Nuclear xenobiotic receptor CAR signaling altered the transcriptome of Teff cells that infiltrated the small intestine lamina propria (siLP) and suppressed Crohn’s disease-like small bowel inflammation [80][29].3.2. Gut Origin of Sepsis
The role of bile acids in intestinal barrier retention is well confirmed. However, there is also a close relationship between the metabolism of bile acids and gut-originating sepsis. Consistent with this view, the inflammatory mediators released during sepsis inhibited hepatobiliary transporter gene expression, resulting in hyperbilirubinemia and cholestasis [90][30]. Serum bile acid concentrations were significantly higher in animals and humans with sepsis. Thus, bile acids may be a potential marker for early sepsis [52,53][31][32]. Of interest is the analysis of plasma from septic individuals found to be glycochenodeoxycholate- and phenylalanine-associated with survival of sepsis [91][33].3.3. Non-Alcoholic Fatty Liver Disease (NAFLD)
Bile acids may also affect the progression of NAFLD through adjusting the intestinal barrier [105,106,107][34][35][36]. Long-term HFD intake can lead to gut dysbiosis and the aberrant metabolism of bile acids. Bidirectional crosstalk between gut microbiota and bile acids could impair the intestinal epithelial function, increasing the translocation of gut-derived endotoxins such as LPS to the blood and lymphatics to activate hepatic TLR-4/NF-κB signaling and promote NAFLD or NASH [108,109][37][38]. HFD-mediated changes in bile acids decreased FXR and TGR5 signaling and degraded the intestinal barrier [110,111][39][40].3.4. Colorectal Cancer (CRC)
The dysregulation of bile acids-BAR pathways also plays a pivotal role in the progression of CRC. Decreased FXR-FGF15 signaling and overexpressed TGR5 were observed in azoxymethane (AOM)/dextran sodium sulfate (DSS)-induced CAC mice [68][41]. In contrast, the activation of FXR protected the intestinal barrier, decreased inflammation, and restricted tumor growth [126][42]. HFD and dysregulated Wnt signaling (APC mutation) altered bile acids profiles, increased the malignant transformation of Lgr5+ cancer stem cells, and promoted adenocarcinoma progression which was counteracted by the activation of FXR [127][43].3.5. Aging
Even though longevity is the people’s long-standing pursuit, to clarify the exact mechanism and pick out the ‘Mr. Key’ in this process is still the most important to success. Fortunately, many lines of evidence have clearly demonstrated that modifying the bile acids profile can delay aging effectively. A Mediterranean diet or calorie restriction altered the gut microbiota and bile acids, which was posited to improve health during aging [135,136,137,138][44][45][46][47]. In rodents, methionine restriction increased macroautophagy/autophagy and altered bile acid conjugation and levels to lengthen lifespan [139,140][48][49]. Modifying the bile acid profile with medications or fecal transplantation also relieved age-associated metabolic dysregulation in mice [141,142][50][51].
4. Conclusions
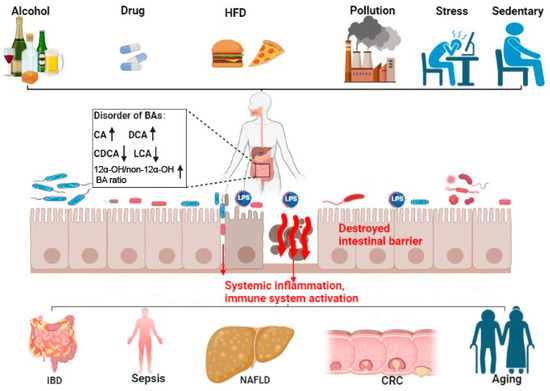
A well-functioning intestinal barrier maintains normal function of the digestive tract and positively impacts the entire individual. This is not surprising as the intestinal barrier is the largest interface between the individual and the environment. It is sensitive to changes in many external and internal factors such as diet, pollution, alcohol, drugs, stress, and life cycle [143,144,145][52][53][54]. Bile acids are the only small molecules synthesized de novo and metabolized by the digestive system. Bile acids traverse the enterohepatic circulation 6–8 times per day and have a complex dynamic interaction with the gut microbiota [3]. It is reasonable to ascribe a central role to bile acids in intestinal homeostasis. Given the physiological significance of bile acids signaling, greater insight into the complex relationship between bile acids and the intestinal barrier could uncover safe therapies for intestinal, hepatobiliary, and age-related diseases. Encouraging is the considerable progress achieved in modifying bile acid signaling through the use of bile acids and their derivatives (e.g., UDCA and OCA), targeting BARs, and through the regulation of the gut microbiota (e.g., by fecal microbiota transplant). However, considering interindividual variability in disease status and tolerance to treatment, personalized medicines are still in need. Bile acid metabolism is a multi-step physiological process, except for 12a-hydroxylated, many other modification processes, such as 7a-dehydroxylation, hydrolysis, and epimerization, may also play significant roles in the physiochemical and signaling properties of BAs. For example, some derivatives of LCA, formed by isomerization, can affect the function of the intestinal immune barrier through suppressing Th17 cell differentiation and increasing the differentiation of Treg [40,41][24][25]. Even the composition of BAs in mice differs from that of humans because the majority of CDCA in mice is typically converted to MCA [135][44]. But, according to the most recent research, both have been reported to improve the function of gut barrier [18,146,147][55][56][57]. In summary, fine-tuning the metabolism of bile acids is important in the homeostasis of the intestinal barrier. Disorders of bile acids and BAR signaling are involved in intestinal barrier dysfunction. Targeting bile acids and bile acid pathways may provide treatments to the related diseases arising from the deterioration of the intestine barrier. Still, further studies are warranted to elucidate the underlying mechanisms of action. Large, controlled, longitudinal clinical studies will assist in this.References
- Breugelmans, T.; Oosterlinck, B.; Arras, W.; Ceuleers, H.; De Man, J.; Hold, G.L.; De Winter, B.Y.; Smet, A. The role of mucins in gastrointestinal barrier function during health and disease. Lancet Gastroenterol. Hepatol. 2022, 7, 455–471.
- Maynard, C.L.; Elson, C.O.; Hatton, R.D.; Weaver, C.T. Reciprocal interactions of the intestinal microbiota and immune system. Nature 2012, 489, 231–241.
- Cai, J.W.; Rimal, B.; Jiang, C.T.; Chiang, J.Y.L.; Patterson, A.D. Bile acid metabolism and signaling, the microbiota, and metabolic disease. Pharmacol. Ther. 2022, 237, 108238.
- Bhargava, P.; Smith, M.D.; Mische, L.; Harrington, E.; Fitzgerald, K.C.; Martin, K.; Kim, S.; Reyes, A.A.; Gonzalez-Cardona, J.; Volsko, C.; et al. Bile acid metabolism is altered in multiple sclerosis and supplementation ameliorates neuroinflammation. J. Clin. Investig. 2020, 130, 3467–3482.
- Sun, R.C.; Xu, C.J.; Feng, B.S.; Gao, X.; Liu, Z.J. Critical roles of bile acids in regulating intestinal mucosal immune responses. Ther. Adv. Gastroenterol. 2021, 14, 17562848211018098.
- Li, C.G.; Wang, Y.; Liu, D.B.; Wong, C.C.; Coker, O.O.; Zhang, X.; Liu, C.A.; Zhou, Y.F.; Liu, Y.L.; Kang, W.; et al. Squalene epoxidase drives cancer cell proliferation and promotes gut dysbiosis to accelerate colorectal carcinogenesis. Gut 2022, 71, 2253–2265.
- Sato, Y.; Atarashi, K.; Plichta, D.R.; Arai, Y.; Sasajima, S.; Kearney, S.M.; Suda, W.; Takeshita, K.; Sasaki, T.; Okamoto, S.; et al. Novel bile acid biosynthetic pathways are enriched in the microbiome of centenarians. Nature 2021, 599, 458–464.
- Ding, L.; Yang, L.; Wang, Z.T.; Huang, W.D. Bile acid nuclear receptor FXR and digestive system diseases. Acta Pharm. Sin. B 2015, 5, 135–144.
- Wang, Y.D.; Chen, W.D.; Huang, W.D. FXR, a target for different diseases. Histol. Histopathol. 2008, 23, 621–627.
- Allam-Ndoul, B.; Castonguay-Paradis, S.; Veilleux, A. Gut Microbiota and Intestinal Trans-Epithelial Permeability. Int. J. Mol. Sci. 2020, 21, 6402.
- Liu, L.; Dong, W.X.; Wang, S.N.; Zhang, Y.J.; Liu, T.Y.; Xie, R.X.; Wang, B.M.; Cao, H.L. Deoxycholic acid disrupts the intestinal mucosal barrier and promotes intestinal tumorigenesis. Food Funct. 2018, 9, 5588–5597.
- He, Z.Y.; Ma, Y.L.; Yang, S.R.; Zhang, S.Y.; Liu, S.; Xiao, J.X.; Wang, Y.J.; Wang, W.; Yang, H.J.; Li, S.L.; et al. Gut microbiota-derived ursodeoxycholic acid from neonatal dairy calves improves intestinal homeostasis and colitis to attenuate extended-spectrum beta-lactamase-producing enteroaggregative Escherichia coli infection. Microbiome 2022, 10, 79.
- Gadaleta, R.M.; Garcia-Irigoyen, O.; Cariello, M.; Scialpi, N.; Peres, C.; Vetrano, S.; Fiorino, G.; Danese, S.; Ro, B.; Luo, J.; et al. Fibroblast Growth Factor 19 modulates intestinal microbiota and inflammation in presence of Farnesoid X Receptor. Ebiomedicine 2020, 54, 102719.
- Nakanishi, T.; Fukui, H.; Wang, X.; Nishiumi, S.; Yokota, H.; Makizaki, Y.; Tanaka, Y.; Ohno, H.; Tomita, T.; Oshima, T.; et al. Effect of a High-Fat Diet on the Small-Intestinal Environment and Mucosal Integrity in the Gut-Liver Axis. Cells 2021, 10, 3168.
- Arifuzzaman, M.; Won, T.H.; Li, T.T.; Yano, H.; Digumarthi, S.; Heras, A.F.; Zhang, W.; Parkhurst, C.N.; Kashyap, S.; Jin, W.B.; et al. Inulin fibre promotes microbiota-derived bile acids and type 2 inflammation. Nature 2022, 611, 578–584.
- Anhe, F.F.; Nachbar, R.T.; Varin, T.V.; Trottier, J.; Dudonne, S.; Le Barz, M.; Feutry, P.; Pilon, G.; Barbier, O.; Desjardins, Y.; et al. Treatment with camu camu (Myrciaria dubia) prevents obesity by altering the gut microbiota and increasing energy expenditure in diet-induced obese mice. Gut 2019, 68, 453–464.
- Williams, J.M.; Duckworth, C.A.; Burkitt, M.D.; Watson, A.J.M.; Campbell, B.J.; Pritchard, D.M. Epithelial Cell Shedding and Barrier Function: A Matter of Life and Death at the Small Intestinal Villus Tip. Vet. Pathol. 2015, 52, 445–455.
- Liu, C.Y.; Cham, C.M.; Chang, E.B. Epithelial wound healing in inflammatory bowel diseases: The next therapeutic frontier. Transl. Res. 2021, 236, 35–51.
- Chen, L.; Jiao, T.Y.; Liu, W.W.; Luo, Y.H.; Wang, J.; Guo, X.Z.; Tong, X.; Lin, Z.M.; Sun, C.Y.; Wang, K.L.; et al. Hepatic cytochrome P450 8B1 and cholic acid potentiate intestinal epithelial injury in colitis by suppressing intestinal stem cell renewal. Cell Stem Cell 2022, 29, 1366–1381.
- Huang, D.; Xiong, M.L.; Xu, X.J.; Wu, X.W.; Xu, J.X.; Cai, X.B.; Lu, L.G.; Zhou, H. Bile acids elevated by high-fat feeding induce endoplasmic reticulum stress in intestinal stem cells and contribute to mucosal barrier damage. Biochem. Biophys. Res. Commun. 2020, 529, 289–295.
- Mowat, A.M.; Agace, W.W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 2014, 14, 667–685.
- Morbe, U.M.; Jorgensen, P.B.; Fenton, T.M.; von Burg, N.; Riis, L.B.; Spencer, J.; Agace, W.W. Human gut-associated lymphoid tissues (GALT); diversity, structure, and function. Mucosal Immunol. 2021, 14, 793–802.
- Di Tommaso, N.; Gasbarrini, A.; Ponziani, F.R. Intestinal Barrier in Human Health and Disease. Int. J. Environ. Res. Public Health 2021, 18, 12836.
- Tanoue, T.; Atarashi, K.; Honda, K. Development and maintenance of intestinal regulatory T cells. Nat. Rev. Immunol. 2016, 16, 295–309.
- Paik, D.; Yao, L.N.; Zhang, Y.C.; Bae, S.; D’Agostino, G.D.; Zhang, M.H.; Kim, E.; Franzosa, E.A.; Avila-Pacheco, J.; Bisanz, J.E.; et al. Human gut bacteria produce T(H)17-modulating bile acid metabolites. Nature 2022, 603, 907–912.
- Song, X.Y.; Sun, X.M.; Oh, S.F.; Wu, M.; Zhang, Y.B.; Zheng, W.; Geva-Zatorsky, N.; Jupp, R.; Mathis, D.; Benoist, C.; et al. Microbial bile acid metabolites modulate gut ROR gamma(+) regulatory T cell homeostasis. Nature 2020, 577, 410–415.
- Zhang, P.; Zheng, L.; Duan, Y.; Gao, Y.; Gao, H.; Mao, D.; Luo, Y. Gut microbiota exaggerates triclosan-induced liver injury via gut-liver axis. J. Hazard. Mater. 2022, 421, 126707.
- Liu, T.C.; Kern, J.T.; Jain, U.; Sonnek, N.M.; Xiong, S.; Simpson, K.F.; VanDussen, K.L.; Winkler, E.S.; Haritunians, T.; Malique, A.; et al. Western diet induces Paneth cell defects through microbiome alterations and farnesoid X receptor and type I interferon activation. Cell Host Microbe 2021, 29, 988–1001.e6.
- Chen, M.L.; Huang, X.S.; Wang, H.T.; Hegner, C.; Liu, Y.J.; Shang, J.S.; Eliason, A.; Diao, H.T.; Park, H.; Frey, B.; et al. CAR directs T cell adaptation to bile acids in the small intestine. Nature 2021, 593, 147–151.
- Horvatits, T.; Drolz, A.; Rutter, K.; Roedl, K.; Langouche, L.; Van den Berghe, G.; Fauler, G.; Meyer, B.; Hulsmann, M.; Heinz, G.; et al. Circulating bile acids predict outcome in critically ill patients. Ann. Intensive Care 2017, 7, 48.
- Baptista, L.; Pollard, D.; Di Bella, A. Evaluation of Resting Serum Bile Acid Concentrations in Dogs with Sepsis. Vet. Sci. 2022, 9, 627.
- Zohrer, E.; Meinel, K.; Fauler, G.; Moser, V.A.; Greimel, T.; Zobl, J.; Schlagenhauf, A.; Jahnel, J. Neonatal sepsis leads to early rise of rare serum bile acid tauro-omega-muricholic acid (TOMCA). Pediatr. Res. 2018, 84, 66–70.
- Kosyakovsky, L.B.; Somerset, E.; Rogers, A.J.; Sklar, M.; Mayers, J.R.; Toma, A.; Szekely, Y.; Soussi, S.; Wang, B.; Fan, C.S.; et al. Machine learning approaches to the human metabolome in sepsis identify metabolic links with survival. Intensive Care Med. Exp. 2022, 10, 24.
- Sun, J.Y.; Fan, J.M.; Li, T.T.; Yan, X.X.; Jiang, Y.H. Nuciferine Protects Against High-Fat Diet-Induced Hepatic Steatosis via Modulation of Gut Microbiota and Bile Acid Metabolism in Rats. J. Agric. Food Chem. 2022, 70, 12014–12028.
- Fiaschini, N.; Mancuso, M.; Tanori, M.; Colantoni, E.; Vitali, R.; Diretto, G.; Rebenaque, L.L.; Stronati, L.; Negroni, A. Liver Steatosis and Steatohepatitis Alter Bile Acid Receptors in Brain and Induce Neuroinflammation: A Contribution of Circulating Bile Acids and Blood-Brain Barrier. Int. J. Mol. Sci. 2022, 23, 14254.
- Rubio, C.; Puerto, M.; Garcia-Rodriquez, J.J.; Lu, V.B.; Garcia-Martinez, I.; Alen, R.; Sanmartin-Salinas, P.; Toledo-Lobo, M.V.; Saiz, J.; Ruperez, J.; et al. Impact of global PTP1B deficiency on the gut barrier permeability during NASH in mice. Mol. Metab. 2020, 35, 100954.
- Rohr, M.W.; Narasimhulu, C.A.; Rudeski-Rohr, T.A.; Parthasarathy, S. Negative Effects of a High-Fat Diet on Intestinal Permeability: A Review. Adv. Nutr. 2020, 11, 77–91.
- Ahmad, M.I.; Ijaz, M.U.; Hussain, M.; ul Haq, I.; Zhao, D.; Li, C.B. High-Fat Proteins Drive Dynamic Changes in Gut Microbiota, Hepatic Metabolome, and Endotoxemia-TLR-4-NF kappa B-Mediated Inflammation in Mice. J. Agric. Food Chem. 2020, 68, 11710–11725.
- Finn, P.D.; Rodriguez, D.; Kohler, J.; Jiang, Z.; Wan, S.; Blanco, E.; King, A.J.; Chen, T.; Bell, N.; Dragoli, D.; et al. Intestinal TGR5 agonism improves hepatic steatosis and insulin sensitivity in Western diet-fed mice. Am. J. Physiol.-Gastrointest. Liver Physiol. 2019, 316, G412–G424.
- Zhai, Y.Y.; Zhou, W.L.; Yan, X.; Qiao, Y.; Guan, L.L.; Zhang, Z.C.; Liu, H.; Jiang, J.Z.; Liu, J.; Peng, L. Astragaloside IV ameliorates diet-induced hepatic steatosis in obese mice by inhibiting intestinal FXR via intestinal flora remodeling. Phytomedicine 2022, 107, 154444.
- Liu, L.; Yang, M.; Dong, W.X.; Liu, T.Y.; Song, X.L.; Gu, Y.; Wang, S.N.; Liu, Y.; Abla, Z.; Qiao, X.M.; et al. Gut Dysbiosis and Abnormal Bile Acid Metabolism in Colitis-Associated Cancer. Gastroenterol. Res. Pract. 2021, 2021, 6645970.
- Zhou, M.J.; Wang, D.F.; Li, X.; Cao, Y.; Yi, C.X.; Ocansey, D.K.W.; Zhou, Y.L.; Mao, F. Farnesoid-X receptor as a therapeutic target for inflammatory bowel disease and colorectal cancer. Front. Pharmacol. 2022, 13, 1016836.
- Fu, T.; Coulter, S.; Yoshihara, E.; Oh, T.G.; Fang, S.; Cayabyab, F.; Zhu, Q.Y.; Zhang, T.; Leblanc, M.; Liu, S.H.; et al. FXR Regulates Intestinal Cancer Stem Cell Proliferation. Cell 2019, 176, 1098–1112.
- Chiang, J.Y. Bile acid metabolism and signaling. Compr. Physiol. 2013, 3, 1191–1212.
- Ghosh, T.S.; Rampelli, S.; Jeffery, I.B.; Santoro, A.; Neto, M.; Capri, M.; Giampieri, E.; Jennings, A.; Candela, M.; Turroni, S.; et al. Mediterranean diet intervention alters the gut microbiome in older people reducing frailty and improving health status: The NU-AGE 1-year dietary intervention across five European countries. Gut 2020, 69, 1218–1228.
- Green, C.L.; Soltow, Q.A.; Mitchell, S.E.; Derous, D.; Wang, Y.C.; Chen, L.N.; Han, J.D.J.; Promislow, D.E.L.; Lusseau, D.; Douglas, A.; et al. The Effects of Graded Levels of Calorie Restriction: XIII. Global Metabolomics Screen Reveals Graded Changes in Circulating Amino Acids, Vitamins, and Bile Acids in the Plasma of C57BL/6 Mice. J. Gerontol. A-Biol. 2019, 74, 16–26.
- Faits, T.; Walker, M.E.; Rodriguez-Morato, J.; Meng, H.C.; Gervis, J.E.; Galluccio, J.M.; Lichtenstein, A.H.; Johnson, W.E.; Matthan, N.R. Exploring changes in the human gut microbiota and microbial-derived metabolites in response to diets enriched in simple, refined, or unrefined carbohydrate-containing foods: A post hoc analysis of a randomized clinical trial. Am. J. Clin. Nutr. 2020, 112, 1631–1641.
- Barcena, C.; Quiros, P.M.; Durand, S.; Mayoral, P.; Rodriguez, F.; Caravia, X.M.; Marino, G.; Garabaya, C.; Fernandez-Garcia, M.T.; Kroemer, G.; et al. Methionine Restriction Extends Lifespan in Progeroid Mice and Alters Lipid and Bile Acid Metabolism. Cell Rep. 2018, 24, 2392–2403.
- Barcena, C.; Lopez-Otin, C.; Kroemer, G. Methionine restriction for improving progeria: Another autophagy-inducing anti-aging strategy? Autophagy 2019, 15, 558–559.
- Barcena, C.; Valdes-Mas, R.; Mayoral, P.; Garabaya, C.; Durand, S.; Rodriguez, F.; Fernandez-Garcia, M.T.; Salazar, N.; Nogacka, A.M.; Garatachea, N.; et al. Healthspan and lifespan extension by fecal microbiota transplantation into progeroid mice. Nat. Med. 2019, 25, 1234–1242.
- Wei, D.Q.; Li, Y.Z.; Che, M.; Li, C.W.; Wu, Q.; Sun, C. Melatonin relieves hepatic lipid dysmetabolism caused by aging via modifying the secondary bile acid pattern of gut microbes. Cell. Mol. Life Sci. 2022, 79, 527.
- Gustafsson, J.K.; Davis, J.E.; Rappai, T.; McDonald, K.G.; Kulkarni, D.H.; Knoop, K.A.; Hogan, S.P.; Fitzpatrick, J.A.J.; Lencer, W.I.; Newberry, R.D. Intestinal goblet cells sample and deliver lumenal antigens by regulated endocytic uptake and transcytosis. eLife 2021, 10, e67292.
- Camilleri, M. Leaky gut: Mechanisms, measurement and clinical implications in humans. Gut 2019, 68, 1516–1526.
- Gohir, W.; Kennedy, K.M.; Wallace, J.G.; Saoi, M.; Bellissimo, C.J.; Britz-McKibbin, P.; Petrik, J.J.; Surette, M.G.; Sloboda, D.M. High-fat diet intake modulates maternal intestinal adaptations to pregnancy and results in placental hypoxia, as well as altered fetal gut barrier proteins and immune markers. J. Physiol. 2019, 597, 3029–3051.
- Song, M.; Ye, J.Y.; Zhang, F.L.; Su, H.; Yang, X.H.; He, H.W.; Liu, F.F.; Zhu, X.T.; Wang, L.N.; Gao, P.; et al. Chenodeoxycholic Acid (CDCA) Protects against the Lipopolysaccharide-Induced Impairment of the Intestinal Epithelial Barrier Function via the FXR-MLCK Pathway. J. Agric. Food Chem. 2019, 67, 8868–8874.
- Hasan, M.N.; Chen, J.; Wang, H.; Du, Y.; Clayton, Y.D.; Gu, L.; Li, T. Glycine-β-Muricholic Acid Improves Liver Fibrosis and Gut Barrier Function by Reducing Bile Acid Pool Size and Hydrophobicity in Male Cyp2c70 Knockout Mice. Cells 2023, 12, 1371.
- Jian, Y.P.; Yang, G.; Zhang, L.H.; Liang, J.Y.; Zhou, H.L.; Wang, Y.S.; Xu, Z.X. Lactobacillus plantarum alleviates irradiation-induced intestinal injury by activation of FXR-FGF15 signaling in intestinal epithelia. J. Cell. Physiol. 2022, 237, 1845–1856.
