The ADAMTS (A Disintegrin and Metalloproteinase with Thrombospondin Motifs) family genes code for key metalloproteinases in the remodeling process of different ECM. Several genes of this family encode for proteins with important functions in reproductive processes; in particular, ADAMTS1, 4, 5 and 9 are genes that are differentially expressed in cell types and the physiological stages of reproductive tissues. ADAMTS enzymes degrade proteoglycans in the ECM of the follicles so that the oocytes can be released and regulate follicle development during folliculogenesis, favoring the action of essential growth factors, such as FGF-2, FGF-7 and GDF-9. The transcriptional regulation of ADAMTS1 and 9 in preovulatory follicles occurs because of the gonadotropin surge in preovulatory follicles, via the progesterone/progesterone receptor complex. In addition, in the case of ADAMTS1, pathways involving protein kinase A (PKA), extracellular signal regulated protein kinase (ERK1/2) and the epidermal growth factor receptor (EGFR) might contribute to ECM regulation. Different Omic studies indicate the importance of genes of the ADAMTS family from a reproductive aspect. ADAMTS genes could serve as biomarkers for genetic improvement and contribute to enhance fertility and animal reproduction.
- animal reproduction
- ADAMTS
- extracellular matrix
- genetic improvement
1. Introduction
2. Structural Features of ADAMTS Genes
ADAMTS proteinases are multidomain enzymes with highly conserved structures [17]. ADAMTS1 was the first gene of this family to be described in mice [18], and later, other genes were identified in other species. In mammalian genomes, 19 ADAMTS genes have been identified and named ADAMTS1 to ADAMTS20. It was later discovered that ADAMTS5 and ADAMTS11 are the same gene, and ADAMTS11 is no longer used [19][20]. The expansion in the number of ADAMTS genes in mammals seems to have occurred due to gene duplication, thus generating sub-functionalization or neo-functionalization regarding the physiological processes in which they participate [13]. Rose et al. [21], in their excellent review, explain that Gon-1 is the only ADAMTS orthologous gene in Caenorhabditis elegans, and it has similarity to ADAMTS9 and ADAMTS20 in humans. The six ADAMTS proteases in the ascidian Ciona intestinalis represent the central evolutionary clades in chordates from which gene expansion into vertebrates occurs [22], along with the evolution of the ECM. Phylogenetic analysis clearly suggests the gene duplication of the ADAMTS genes [21]. The signature domains of the ADAMTS proteins are: a signal peptide, necessary for protein trafficking and secretion, and an inhibitory prodomain that must be cleaved from the ADAMTS zymogens to render them catalytically active. Such cleavage occurs in Golgi, the cell surface or extracellularly. The size of this prodomain comprises about 200 residues in all ADAMTS proteases, with the exception of ADAMTS13, which has a short prodomain that does not need to be removed for the protease to be active [23]. Interestingly, the removal of the ADAMTS9 prodomain reduces the protease catalytic activity upon versican, its substrate; a disintegrin-like domain; a thrombospondin type 1 repeat (TSR-1) motif; and a cysteine-rich domain followed by a spacer region. The TSR-1 domains and the spacer domain appear to be involved in ECM anchoring. The description of the organization of these proteins is based on the structure of ADAMTS4, the other members of the family vary mainly at the C-terminus, with either, more or fewer repeats of the TSR-1 motifs [24]. Figure 1 illustrates the structure and localization of these proteins.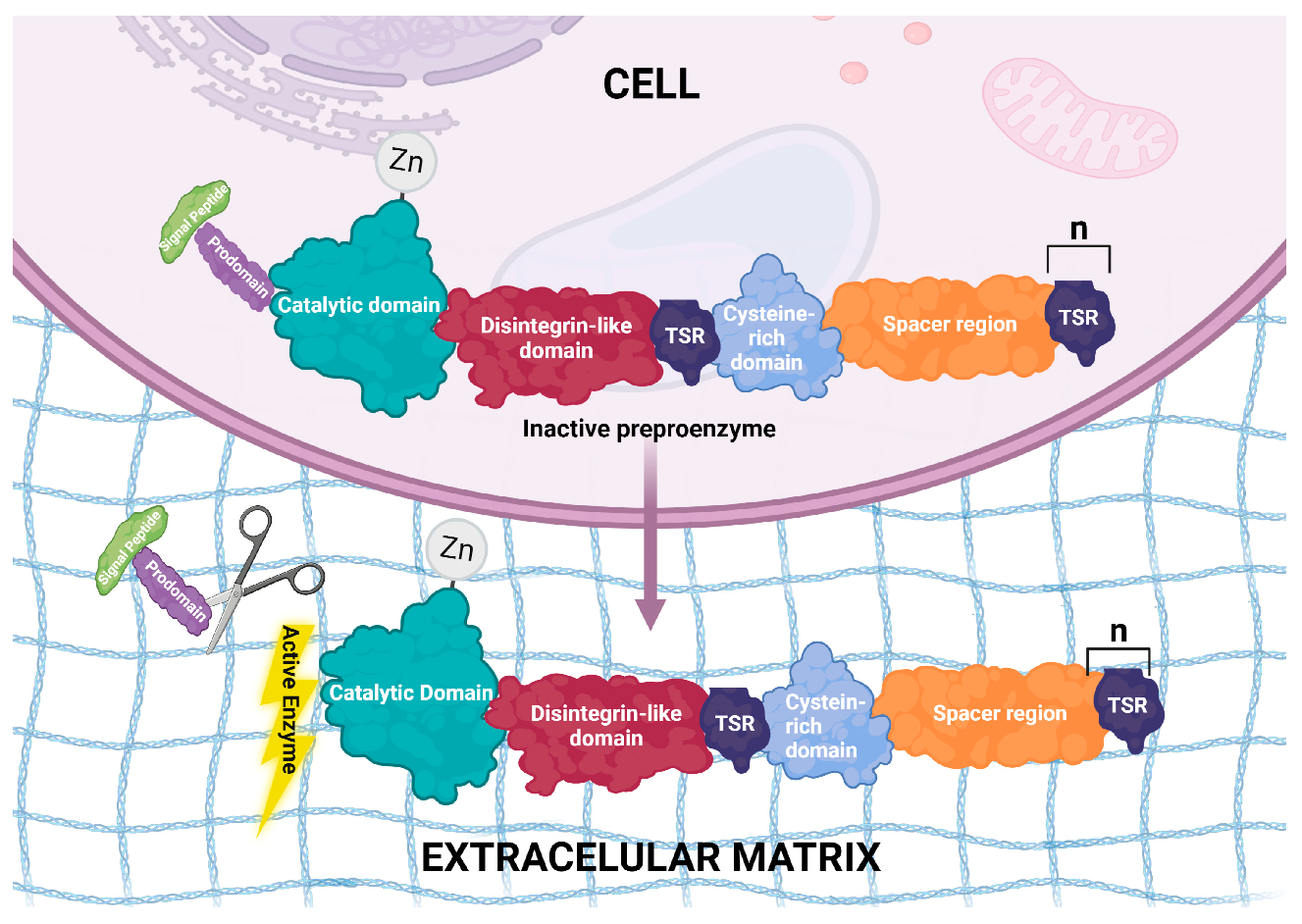
3. ADAMTS and Fertility in Females
3.1. Folliculogenesis
The process of development and maturation of follicles, termed folliculogenesis, is necessary for ovulation to occur [25]. The ADAMTS family genes seem to be related to this process, inferred by the relative abundance of mRNA transcripts in the follicles and corpus lutea of several mammalian species. ADAMTS1 have been reported to be expressed in granulosa cells in cows [26], horses [27] and pigs [28]. According to Brown et al. [29], ADAMTS1 functions are necessary for the structural changes of the ECM to occur during follicular development. The proteoglycans present in the ECM can inhibit the action of certain growth factors, such as FGF-2, FGF-7 and GDF-9 [30], which are essential for various exquisite reproductive processes to occur. For example, FGF-2 stimulates angiogenesis and granulosa cell proliferation and function in cattle [31] and buffalo [32]. FGF-2 also stimulates the initiation and development of follicular growth in sheep and goats [33][34]. Thus, the functions of ADAMTS1 and 4 are thought to enhance these processes by controlling the amount and location of various proteoglycans [35]. Shozu et al. [36] inactivated the ADAMTS1 gene and reported that the absence of ADAMTS1 led to follicular atresia. Versican is an abundant ECM proteoglycan that is hormonally regulated by the ovary. Versican abundance varies throughout the several stages of follicular growth but particularly during ovulation in rodents [37]. The presence of versican in bovine and porcine follicular basement membrane [38][39], suggests that ADAMTS1 may also regulate the development of follicles during folliculogenesis.3.2. Ovulation
The preovulatory surge of gonadotropins induces a series of biochemical processes within the dominant follicle that culminate in ovulation and, subsequently, in the formation of the corpus luteum. Ovulation is associated with the degradation of the follicular basement membrane and the fragmentation of the ECM at the apex of the follicle wall, resulting in the release of the oocyte [40]. Metalloproteinases enzymes are responsible for the degradation of the follicular ECM during ovulation [9]. ADAMTS1 degrades versican, aggrecan and brevican, proteoglycans present in the ECM of the follicle. Such degradation of the follicular wall allows oocyte release [41][42]. Indeed, ADAMTS1 was reported to have a fundamental function in ovulation, as reported by Mittaz et al. [43], based on results from a study with female mice lacking the ADAMTS1. In this study, exon 2 was deleted to disrupt the ADAMTS1 gene and a selectable marker gene was inserted in intron 1. The modified ADAMTS1 allele was functionally null. The authors reported that these females were subfertile due to impaired ovulation, resulting in the mature oocytes not being released from the follicles, as would typically occur during ovulation. Likewise, Brown et al. [44] reported that the ovulation rate was 77% less in female mice lacking the ADAMTS1 enzyme, compared to the wild-type animals. The finding was explained by the lack of versican degradation during the matrix expansion of the cumulus–oocyte complex. Hu et al. [45] reported that SNP-type polymorphisms in ADAMTS1 are related to litter size in goats; therefore, they could be used as molecular markers for the selection of litter size. ADAMTS1, 4 and 9 are considered to be of great importance for animal production, where reproductive prolificacy is a determinant for sustainability. Figure 2 depicts the possible functions of ADAMTS proteins in folliculogenesis and ovulation in livestock.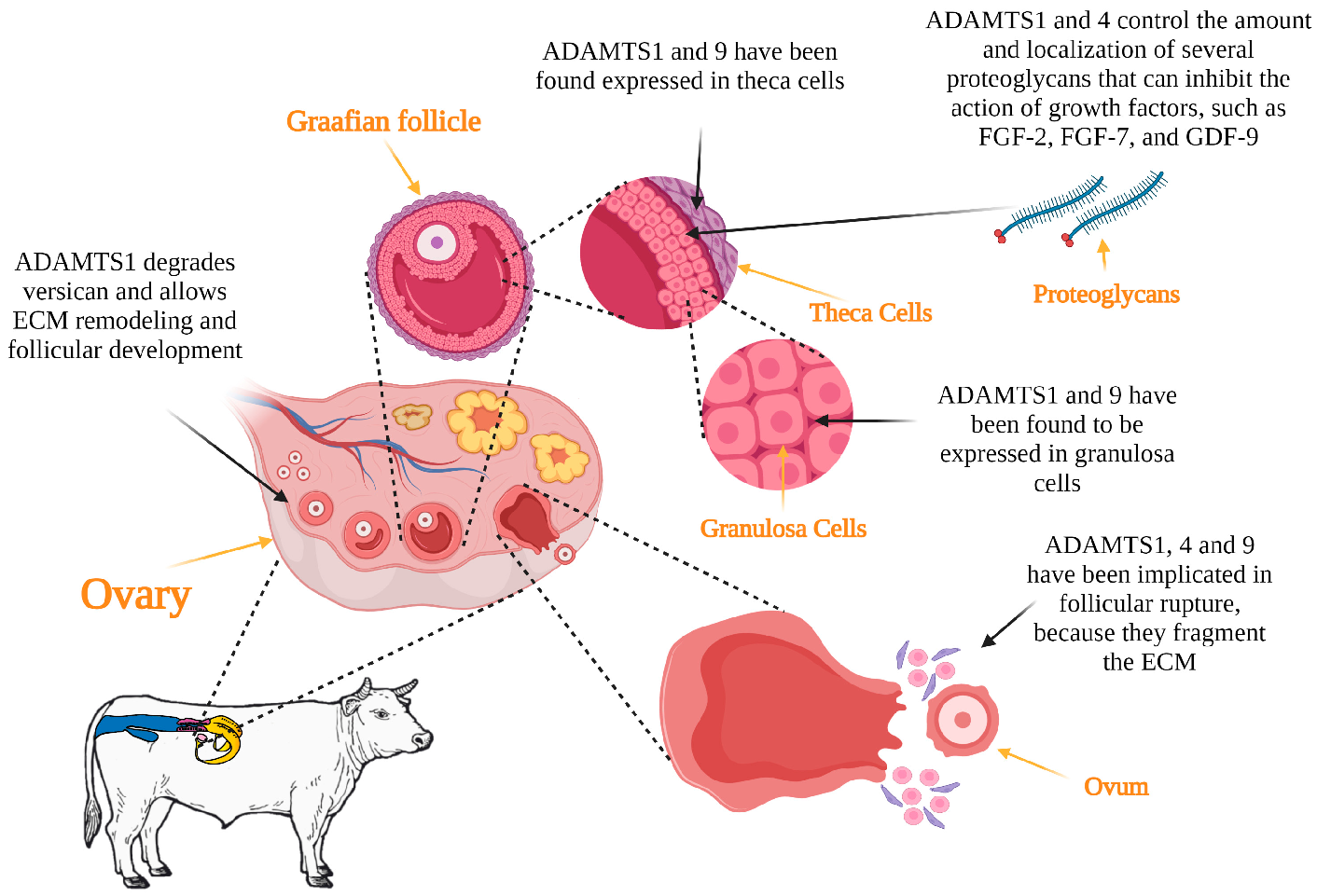
3.3. Implantation, Placentation and Parturition
Similar to the constant remodeling of the ECM required for the cyclical transformations of the ovary, the uterus also undergoes the cyclic development and remodeling of the endometrial tissue matrix. This remodeling is necessary for implantation and placentation [46]. Implantation is a critical process for the establishment of pregnancy and begins with essential signaling from the blastocyst to the endometrium, which must be prepared to respond [47][48]. The attachment of the blastocyst to the uterus and subsequent trophoblast cell invasion occurs through ECM remodeling [49]. Uterine tissue remodeling is also required for placental cotyledon formation and angiogenesis near trophoblast tissue in sheep, as well as a decrease in endometrial thickness during implantation [50][51]. Another member of this family of metalloproteinases, ADAMTS9, has been reported to contribute to remodeling in the uterus at the time of parturition. The extracellular matrix undergoes remodeling during late gestation to allow smooth muscle cells to connect to each other and effect uterine contractions at the time of parturition [52]. ADAMTS9 is present in all reproductive states and contributes to uterine tissue remodeling. The accumulation of versican from the extracellular matrix in the uterus leads to abnormal contractions. Mead et al. [53] reported that there are abnormally large concentrations of versican in mice that do not produce ADAMTS9, leading to abnormal parturition processes. This abnormality was due to a reduction in focal adhesions between cells that interact with one another to generate uterine contractions. Thus, ADAMTS9 contributes to the remodeling of the uterine extracellular matrix through the degradation of versican, and its null or poor functionality disrupts parturition processes. The possible functions of the ADAMTS proteases in implantation, placentation and parturition in livestock is illustrated in Figure 3.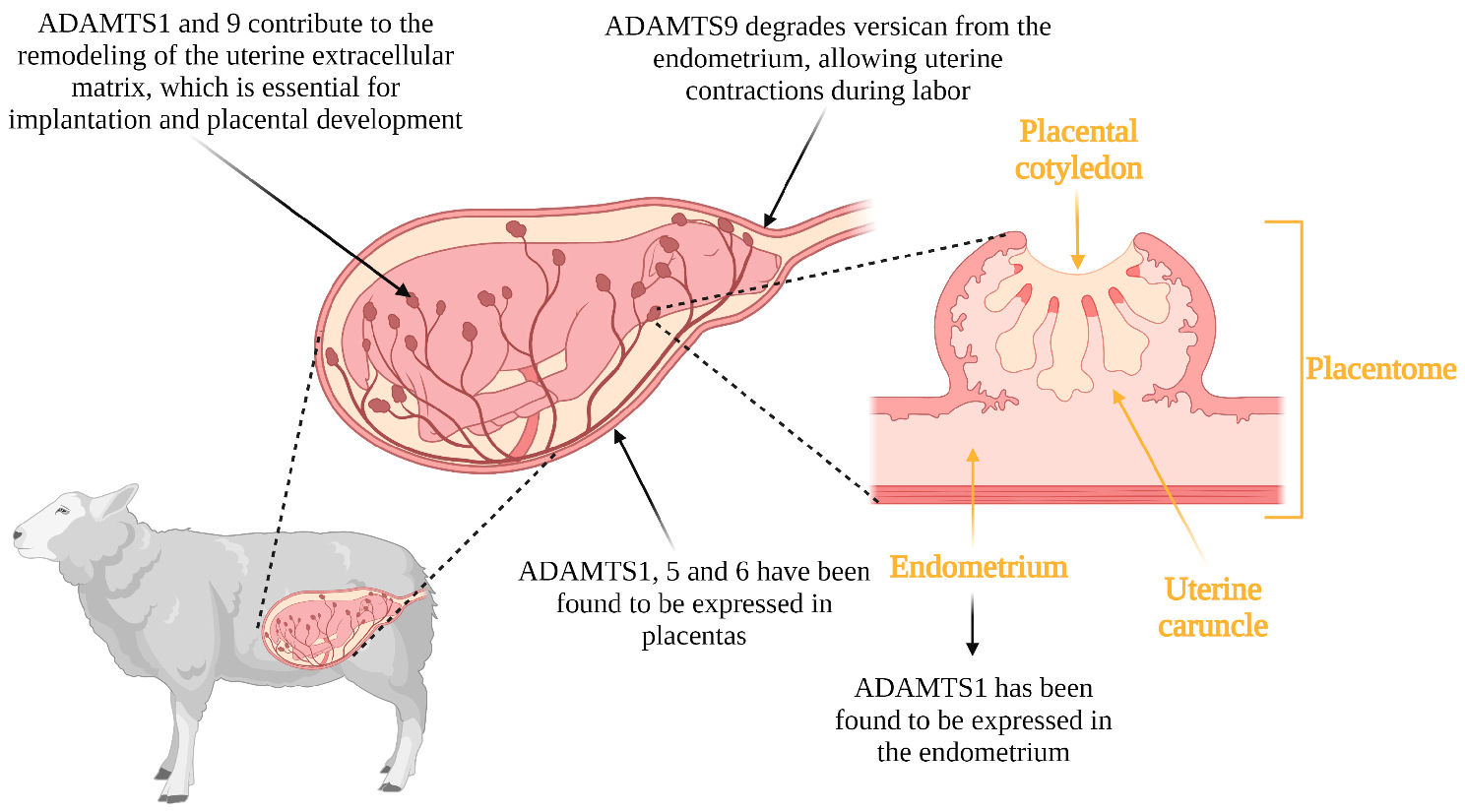
4. ADAMTS and Fertility in Males
4.1. Testicular Development
The expression of ADAMTS family genes, in addition to being related to fertility in females, has also been linked to reproductive capacity in males, specifically to testicular development [54]. The testicles develop in the abdomen during the embryonic and fetal stages. Subsequently, the testes pass through the inguinal canal into the scrotum [7], with this process requiring the tissue remodeling of the ECM [6]; however, regarding this process, the literature is limited to only a few studies of rodents. Jacobi et al. [55] reported there was expression of ADAMTS16 in the testes of mouse embryos. Likewise, alterations in ADAMTS16 led to cryptorchidism and infertility in male rats [54][56].4.2. Spermatogenesis
Spermatogenesis is the process through which germ cells multiply and differentiate to produce sperm in the seminiferous tubules [57]. The presence of ADAMTS10 expression in the testis, epididymis and ejaculated spermatozoa of Asian buffalo (Bubalus bubalis) [58] suggests a possible function in the sperm maturation process [59]. ADAMTS2 has also been linked to sperm maturation in mice, as shown by Li et al. [60]. In transgenic mice homozygous for the inactive alleles of ADAMTS2, there was less sperm maturation and activity compared to those in the control group; however, more research is needed to determine the functions of these proteins in sperm maturation. In bulls with besnoitiosis compared to healthy bulls, ADAMTS1 mRNA abundance is lower in scrotal skin, the pampiniform plexus and testicular parenchyma [61]. This disease causes the fibrosis and thickening of the skin of the scrotum, which leads to failure in thermoregulation and to the inhibition of spermatogenesis, ultimately causing infertility [62]. Wu et al. [63] performed a transcriptomic analysis of yak and cattleyak testes to investigate the genetic causes of hybrid animal sterility, and several ADAMTS genes were differentially expressed. ADAMTS1, 10, 12, 3, 5 and 14 were upregulated, whereas ADAMTS16, 20, 6 and 18 were downregulated; thus, these proteins could be involved in cattleyak sterility. However, how these proteins are associated with hybrid animal sterility is still unclear.4.3. Fertilization
ADAMTS is apparently involved in sperm and egg fertilization processes. In a study conducted by Dun et al. [8] in mice, ADAMTS10 was expressed during the late stages of spermatogenesis, and the protein was incorporated into the acrosome of developing spermatids. ADAMTS10 presence in the acrosome is thought to function by inducing sperm adhesion to the zona pellucida. The zona pellucida is an ECM that surrounds the oocytes and must be crossed by spermatozoa to penetrate the oocyte and carry out fertilization. ADAMTS10 has important functions in this process by acting in the degradation of the zona pellucida [64]; however, further research in farm animals is needed to understand the role of ADAMTS10 in fertilization.References
- Hill, W.G. Is continued genetic improvement of livestock sustainable? Genetics 2016, 202, 877–881.
- Taylor, J.F.; Schnabel, R.D.; Sutovsky, P. Genomics of bull fertility. Animal 2018, 12, 172–183.
- Notter, D. Genetic aspects of reproduction in sheep. Reprod. Domest. Anim. 2008, 43, 122–128.
- Albert, F.W.; Kruglyak, L. The role of regulatory variation in complex traits and disease. Nat. Rev. Genet. 2015, 16, 197–212.
- Demircan, K.; Comertoglu, I.; Akyol, S.; Yigitoglu, B.N.; Sarikaya, E. A new biological marker candidate in female reproductive system diseases: Matrix metalloproteinase with thrombospondin motifs (ADAMTS). J. Turk. Ger. Gynecol. Assoc. 2014, 15, 250–255.
- Russell, D.L.; Brown, H.M.; Dunning, K.R. ADAMTS proteases in fertility. Matrix Biol. 2015, 44–46, 54–63.
- Copp, H.L.; Shortliffe, L.D. Undescended testes and testicular tumors. In Ashcraft’s Pediatric Surgery; Elsevier: Amsterdam, The Netherlands, 2010; pp. 676–686.
- Dun, M.D.; Anderson, A.L.; Bromfield, E.G.; Asquith, K.L.; Emmett, B.; McLaughlin, E.A.; Aitken, R.J.; Nixon, B. Investigation of the expression and functional significance of the novel mouse sperm protein, a disintegrin and metalloprotease with thrombospondin type 1 motifs number 10 (ADAMTS10). Int. J. Androl. 2012, 35, 572–589.
- Fata, J.; Ho, A.-V.; Leco, K.; Moorehead, R.; Khokha, R. Cellular turnover and extracellular matrix remodeling in female reproductive tissues: Functions of metalloproteinases and their inhibitors. CMLS 2000, 57, 77–95.
- Blobel, C.P.; Apte, S. ADAMs and ADAMTSs. In Encyclopedia of Respiratory Medicine; Elsevier: Amsterdam, The Netherlands, 2020; pp. 568–573.
- Sternlicht, M.D.; Werb, Z. How matrix metalloproteinases regulate cell behavior. Annu. Rev. Cell Dev. Biol. 2001, 17, 463–516.
- Apte, S.S.; Parks, W.C. Metalloproteinases: A parade of functions in matrix biology and an outlook for the future. Matrix Biol. 2015, 44–46, 1–6.
- Dubail, J.; Apte, S.S. Insights on ADAMTS proteases and ADAMTS-like proteins from mammalian genetics. Matrix Biol. 2015, 44, 24–37.
- Menke, D.B.; Koubova, J.; Page, D.C. Sexual differentiation of germ cells in XX mouse gonads occurs in an anterior-to-posterior wave. J. Dev. Biol. 2003, 262, 303–312.
- Doyle, K.M.; Russell, D.L.; Sriraman, V.; Richards, J.S. Coordinate transcription of the ADAMTS-1 gene by luteinizing hormone and progesterone receptor. Mol. Endocrinol. 2004, 18, 2463–2478.
- Richards, J.S.; Hernandez-Gonzalez, I.; Gonzalez-Robayna, I.; Teuling, E.; Lo, Y.; Boerboom, D.; Falender, A.E.; Doyle, K.H.; LeBaron, R.G.; Thompson, V. Regulated expression of ADAMTS family members in follicles and cumulus oocyte complexes: Evidence for specific and redundant patterns during ovulation. Biol. Reprod. 2005, 72, 1241–1255.
- Hurskainen, T.L.; Hirohata, S.; Seldin, M.F.; Apte, S.S. ADAM-TS5, ADAM-TS6, and ADAM-TS7, Novel Members of a New Family of Zinc Metalloproteases. J. Biol. Chem. 1999, 274, 25555–25563.
- Kuno, K.; Kanada, N.; Nakashima, E.; Fujiki, F.; Ichimura, F.; Matsushima, K. Molecular cloning of a gene encoding a new type of metalloproteinase-disintegrin family protein with thrombospondin motifs as an inflammation associated gene. J. Biol. Chem. 1997, 272, 556–562.
- Porter, S.; Clark, I.M.; Kevorkian, L.; Edwards, D.R. The ADAMTS metalloproteinases. Biochem. J. 2005, 386, 15–27.
- Apte, S.S. A disintegrin-like and metalloprotease (reprolysin-type) with thrombospondin type 1 motif (ADAMTS) superfamily: Functions and mechanisms. J. Biol. Chem. 2009, 284, 31493–31497.
- Rose, K.W.; Taye, N.; Karoulias, S.Z.; Hubmacher, D. Regulation of ADAMTS proteases. Front. Mol. Biosci. 2021, 8, 701959.
- Huxley-Jones, J.; Apte, S.S.; Robertson, D.L.; Boot-Handford, R.P. The characterisation of six ADAMTS proteases in the basal chordate Ciona intestinalis provides new insights into the vertebrate ADAMTS family. Int. J. Biochem. Cell Biol. 2005, 37, 1838–1845.
- Longpré, J.-M.; Leduc, R. Identification of prodomain determinants involved in ADAMTS-1 biosynthesis. J. Biol. Chem. 2004, 279, 33237–33245.
- Kelwick, R.; Desanlis, I.; Wheeler, G.N.; Edwards, D.R. The ADAMTS (A Disintegrin and Metalloproteinase with Thrombospondin motifs) family. Genome Biol. 2015, 16, 113.
- Gershon, E.; Dekel, N. Newly identified regulators of ovarian folliculogenesis and ovulation. Int. J. Mol. Sci. 2020, 21, 4565.
- Madan, P.; Bridges, P.J.; Komar, C.M.; Beristain, A.G.; Rajamahendran, R.; Fortune, J.E.; MacCalman, C.D. Expression of messenger RNA for ADAMTS subtypes changes in the periovulatory follicle after the gonadotropin surge and during luteal development and regression in cattle. Biol. Reprod. 2003, 69, 1506–1514.
- Boerboom, D.; Russell, D.L.; Richards, J.S.; Sirois, J. Regulation of transcripts encoding ADAMTS-1 (a disintegrin and metalloproteinase with thrombospondin-like motifs-1) and progesterone receptor by human chorionic gonadotropin in equine preovulatory follicles. J. Mol. Endocrinol. 2003, 31, 473–485.
- Shimada, M.; Nishibori, M.; Yamashita, Y.; Ito, J.; Mori, T.; Richards, J.S. Down-regulated expression of A disintegrin and metalloproteinase with thrombospondin-like repeats-1 by progesterone receptor antagonist is associated with impaired expansion of porcine cumulus-oocyte complexes. Endocrinology 2004, 145, 4603–4614.
- Brown, H.M.; Dunning, K.R.; Robker, R.L.; Pritchard, M.; Russell, D.L. Requirement for ADAMTS-1 in extracellular matrix remodeling during ovarian folliculogenesis and lymphangiogenesis. Dev. Biol. 2006, 300, 699–709.
- Park, P.W.; Reizes, O.; Bernfield, M. Cell surface heparan sulfate proteoglycans: Selective regulators of ligand-receptor encounters. J. Biol. Chem. 2000, 275, 29923–29926.
- Berisha, B.; Sinowatz, F.; Schams, D. Expression and localization of fibroblast growth factor (FGF) family members during the final growth of bovine ovarian follicles. Mol. Reprod. Dev. 2004, 67, 162–171.
- Mishra, S.R.; Thakur, N.; Somal, A.; Parmar, M.S.; Reshma, R.; Rajesh, G.; Yadav, V.P.; Bharti, M.K.; Bharati, J.; Paul, A.; et al. Expression and localization of fibroblast growth factor (FGF) family in buffalo ovarian follicle during different stages of development and modulatory role of FGF2 on steroidogenesis and survival of cultured buffalo granulosa cells. Res. Vet. Sci. 2016, 108, 98–111.
- Matos, M.H.; Lima-Verde, I.B.; Bruno, J.B.; Lopes, C.A.; Martins, F.S.; Santos, K.D.; Rocha, R.M.; Silva, J.R.; Bao, S.N.; Figueiredo, J.R. Follicle stimulating hormone and fibroblast growth factor-2 interact and promote goat primordial follicle development in vitro. Reprod. Fertil. Dev. 2007, 19, 677–684.
- Santos, J.M.; Menezes, V.G.; Barberino, R.S.; Macedo, T.J.; Lins, T.L.; Gouveia, B.B.; Barros, V.R.; Santos, L.P.; Goncalves, R.J.; Matos, M.H. Immunohistochemical localization of fibroblast growth factor-2 in the sheep ovary and its effects on pre-antral follicle apoptosis and development in vitro. Reprod. Domest. Anim. 2014, 49, 522–528.
- Richards, J.S.; Russell, D.L.; Ochsner, S.; Espey, L.L. Ovulation: New dimensions and new regulators of the inflammatory-like response. Annu. Rev. Physiol. 2002, 64, 69–92.
- Shozu, M.; Minami, N.; Yokoyama, H.; Inoue, M.; Kurihara, H.; Matsushima, K.; Kuno, K. ADAMTS-1 is involved in normal follicular development, ovulatory process and organization of the medullary vascular network in the ovary. J. Mol. Endocrinol. 2005, 35, 343–355.
- Russell, D.L.; Ochsner, S.A.; Hsieh, M.; Mulders, S.; Richards, J.S. Hormone-regulated expression and localization of versican in the rodent ovary. Endocrinology 2003, 144, 1020–1031.
- McArthur, M.E.; Irving-Rodgers, H.F.; Byers, S.; Rodgers, R.J. Identification and immunolocalization of decorin, versican, perlecan, nidogen, and chondroitin sulfate proteoglycans in bovine small-antral ovarian follicles. Biol. Reprod. 2000, 63, 913–924.
- Rodgers, R.J.; Irving-Rodgers, H.F. Formation of the ovarian follicular antrum and follicular fluid. Biol. Reprod. 2010, 82, 1021–1029.
- Richards, J.S.; Russell, D.L.; Robker, R.L.; Dajee, M.; Alliston, T.N. Molecular mechanisms of ovulation and luteinization. Mol. Cell. Endocrinol. 1998, 145, 47–54.
- Russell, D.L.; Doyle, K.M.; Ochsner, S.A.; Sandy, J.D.; Richards, J.S. Processing and localization of ADAMTS-1 and proteolytic cleavage of versican during cumulus matrix expansion and ovulation. J. Biol. Chem. 2003, 278, 42330–42339.
- Curry, T.E.; Smith, M.F. Impact of extracellular matrix remodeling on ovulation and the folliculo-luteal transition. Semin. Reprod. Med. 2006, 24, 228–241.
- Mittaz, L.; Russell, D.; Wilson, T.; Brasted, M.; Tkalcevic, J.; Salamonsen, L.A.; Hertzog, P.J.; Pritchard, M.A. Adamts-1 is essential for the development and function of the urogenital system. Biol. Reprod. 2004, 70, 1096–1105.
- Brown, H.M.; Dunning, K.R.; Robker, R.L.; Boerboom, D.; Pritchard, M.; Lane, M.; Russell, D.L. ADAMTS1 cleavage of versican mediates essential structural remodeling of the ovarian follicle and cumulus-oocyte matrix during ovulation in mice. Biol. Reprod. 2010, 83, 549–557.
- Hu, W.; Tang, J.; Zhang, Z.; Tang, Q.; Yan, Y.; Wang, P.; Wang, X.; Liu, Q.; Guo, X.; Jin, M.; et al. Polymorphisms in the ASMT and ADAMTS1 gene may increase litter size in goats. Vet. Med. Sci. 2020, 6, 775–787.
- Guillomot, M. Changes in extracellular matrix components and cytokeratins in the endometrium during goat implantation. Placenta 1999, 20, 339–345.
- Nardo, L.G.; Li, T.C.; Edwards, R.G. Introduction: Human embryo implantation failure and recurrent miscarriage: Basic science and clinical practice. Reprod. Biomed. Online 2006, 13, 11–12.
- Guzeloglu-Kayisli, O.; Basar, M.; Arici, A. Basic aspects of implantation. Reprod. Biomed. Online 2007, 15, 728–739.
- Das, S.K.; Yano, S.; Wang, J.; Edwards, D.R.; Nagase, H.; Dey, S.K. Expression of matrix metalloproteinases and tissue inhibitors of metalloproteinases in the mouse uterus during the peri-implantation period. Dev. Genet. 1997, 21, 44–54.
- Smith, S.E.; Cullen, W.C.; Godkin, J.D. Ultrastructural morphometric analysis of the uterine epithelium during early pregnancy in the sheep. J. Reprod. Fertil. 1990, 89, 517–525.
- Reynolds, L.P.; Redmer, D.A. Growth and microvascular development of the uterus during early pregnancy in ewes. Biol. Reprod. 1992, 47, 698–708.
- Riemer, R.K.; Heymann, M.A. Regulation of uterine smooth muscle function during gestation. Pediatr. Res. 1998, 44, 615–627.
- Mead, T.J.; Du, Y.; Nelson, C.M.; Gueye, N.A.; Drazba, J.; Dancevic, C.M.; Vankemmelbeke, M.; Buttle, D.J.; Apte, S.S. ADAMTS9-Regulated Pericellular Matrix Dynamics Governs Focal Adhesion-Dependent Smooth Muscle Differentiation. Cell Rep. 2018, 23, 485–498.
- Abdul-Majeed, S.; Mell, B.; Nauli, S.M.; Joe, B. Cryptorchidism and infertility in rats with targeted disruption of the Adamts16 locus. PLoS ONE 2014, 9, e100967.
- Jacobi, C.L.; Rudigier, L.J.; Scholz, H.; Kirschner, K.M. Transcriptional regulation by the Wilms tumor protein, Wt1, suggests a role of the metalloproteinase Adamts16 in murine genitourinary development. J. Biol. Chem. 2013, 288, 18811–18824.
- Sarila, G.; Bao, T.; Abeydeera, S.A.; Li, R.; Mell, B.; Joe, B.; Catubig, A.; Hutson, J. Interplay between collagenase and undescended testes in Adamts16 knockout rats. J. Pediatr. Surg. 2020, 55, 1952–1958.
- Staub, C.; Johnson, L. Review: Spermatogenesis in the bull. Animal 2018, 12, 27–35.
- Gurupriya, V.S.; Roy, S.C.; Javvaji, P.K.; Dhali, A.; Badami, S.; Rahim, F.; Divyashree, B.C.; Panda, A.P. Expression of ADAMTS10 in male reproductive tract of buffaloes (Bubalus bubalis). J. Exp. Biol. Agric. Sci. 2018, 6, 800–807.
- Cornwall, G.A. New insights into epididymal biology and function. Hum. Reprod. Update 2009, 15, 213–227.
- Li, S.W.; Arita, M.; Fertala, A.; Bao, Y.; Kopen, G.C.; Langsjo, T.K.; Hyttinen, M.M.; Helminen, H.J.; Prockop, D.J. Transgenic mice with inactive alleles for procollagen N-proteinase (ADAMTS-2) develop fragile skin and male sterility. Biochem. J. 2001, 355, 271–278.
- González-Barrio, D.; Diezma-Díaz, C.; Gutiérrez-Expósito, D.; Tabanera, E.; Jiménez-Meléndez, A.; Pizarro, M.; González-Huecas, M.; Ferre, I.; Ortega-Mora, L.M.; Álvarez-García, G. Identification of molecular biomarkers associated with disease progression in the testis of bulls infected with Besnoitia besnoiti. Vet. Res. 2021, 52, 106.
- Olias, P.; Schade, B.; Mehlhorn, H. Molecular pathology, taxonomy and epidemiology of Besnoitia species (Protozoa: Sarcocystidae). Infect. Genet. Evol. 2011, 11, 1564–1576.
- Wu, S.; Mipam, T.; Xu, C.; Zhao, W.; Shah, M.A.; Yi, C.; Luo, H.; Cai, X.; Zhong, J. Testis transcriptome profiling identified genes involved in spermatogenic arrest of cattleyak. PLoS ONE 2020, 15, e0229503.
- Le Goff, C.; Cormier-Daire, V. The ADAMTS(L) family and human genetic disorders. Hum. Mol. Genet. 2011, 20, R163–R167.
