Early-stage disease diagnosis is of particular importance for effective patient identification as well as their treatment. Lack of patient compliance for the existing diagnostic methods, however, limits prompt diagnosis, rendering the development of non-invasive diagnostic tools mandatory. One of the most promising non-invasive diagnostic methods that has also attracted great research interest is breath analysis; the method detects gas-analytes such as exhaled volatile organic compounds (VOCs) and inorganic gases that are considered to be important biomarkers for various disease-types. The diagnostic ability of gas-pattern detection using analytical techniques and especially sensors has been widely discussed in the literature; however, the incorporation of novel nanomaterials in sensor-development has also proved to enhance sensor performance, for both selective and cross-reactive applications.
- breath analysis
- volatile organic compounds
- sensors
- nanomaterials
- differential diagnosis
1. Introduction
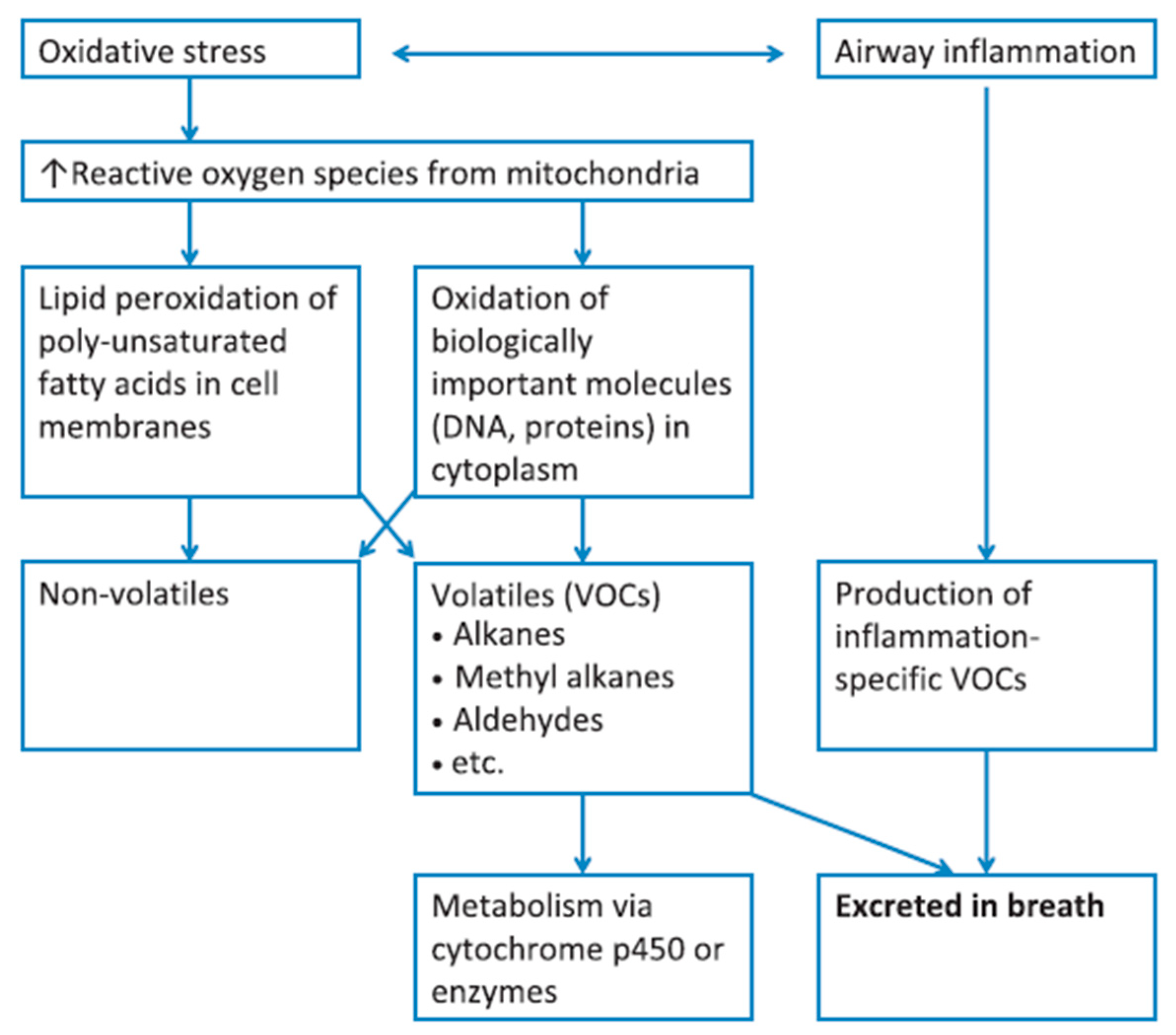 Figure 1. Diagram summarizing the correlation of VOCs present in the exhaled breath, with oxidative stress and inflammatory conditions; metabolic breakdown of larger molecules leads to the formation of exhaled VOCs. Reprinted with permission from ref. [17]. Copyright © 2012 John Wiley & Sons Ltd.
Figure 1. Diagram summarizing the correlation of VOCs present in the exhaled breath, with oxidative stress and inflammatory conditions; metabolic breakdown of larger molecules leads to the formation of exhaled VOCs. Reprinted with permission from ref. [17]. Copyright © 2012 John Wiley & Sons Ltd.| Disease Type | Diseases | Ref. |
|---|---|---|
| Respiratory | Asthma, COPD, obstructive sleep apnea syndrome, pulmonary arterial hypertension, cystic fibrosis | [19] |
| Malignant | Lung, gastric, head and neck, breast, colon, prostate cancer | [15] |
| Neurodegenerative | Alzheimer’s disease, Parkinson’s diseases, multiple sclerosis | [15] |
| Metabolic | Diabetes, hyperglycemia | [12][31] |
| Bacterial infections | Upper respiratory tract infection, Mycobacterium tuberculosis, Pseudomonas, Helicobacter pylori infection | [32][22] |
| Viral infections | SARS-CoV-2 | [24][25][26] |
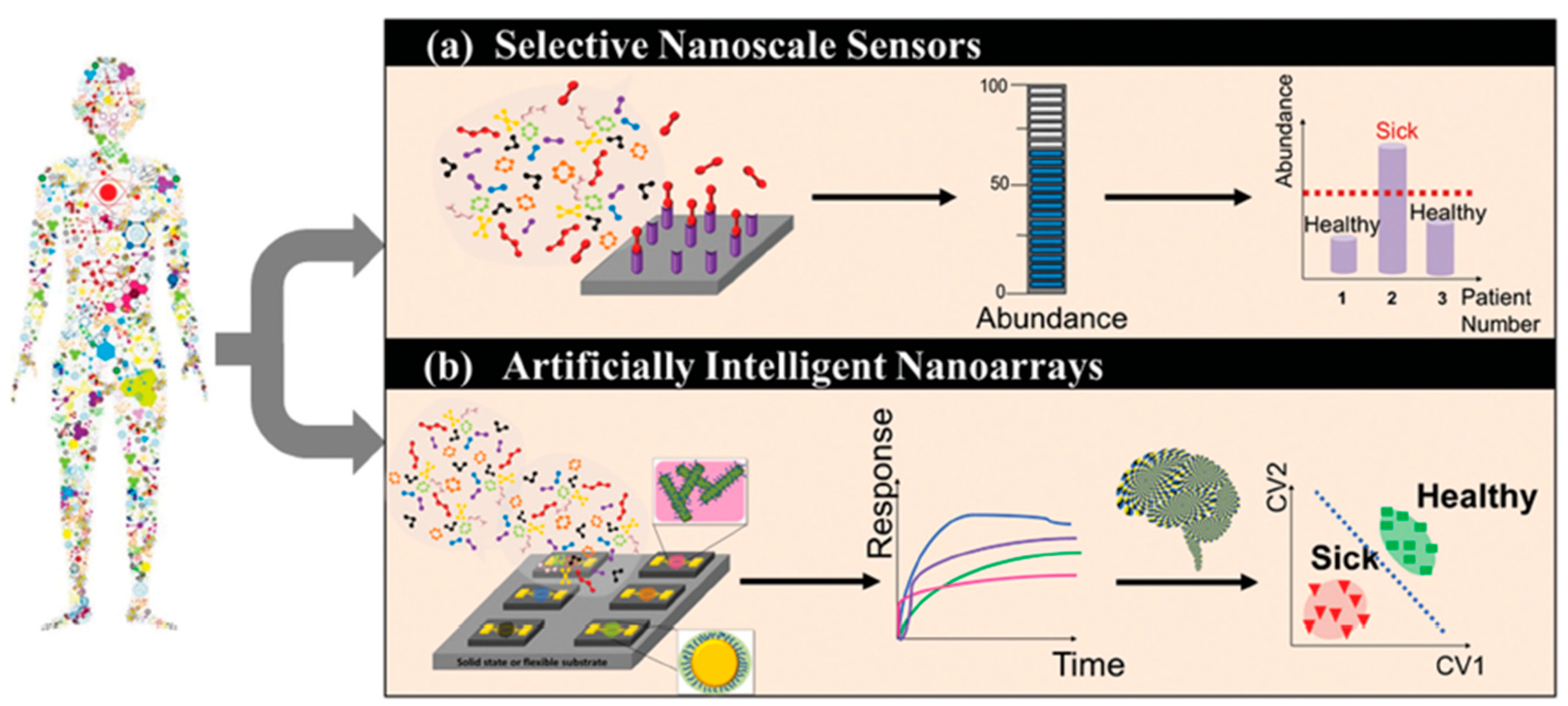 Figure 2. Schematic representation of the working principle of selective sensors and artificially intelligent cross-reactive sensor arrays. Selective sensors contain highly selective elements in order to detect a specific gas-analyte in the presence of a composite gas-mixture. Cross-reactive arrays feature sensors that are sensitive to the majority of the gases present in the gas-mixture. In any case, detecting analyte concentration above a critical value leads to the differentiation between sick and healthy subjects. The response of gas-sensing arrays can be then processed by employing artificial intelligence, machine-learning, and pattern recognition techniques. Reprinted with permission from Ref. [6] Copyright © 2015 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figure 2. Schematic representation of the working principle of selective sensors and artificially intelligent cross-reactive sensor arrays. Selective sensors contain highly selective elements in order to detect a specific gas-analyte in the presence of a composite gas-mixture. Cross-reactive arrays feature sensors that are sensitive to the majority of the gases present in the gas-mixture. In any case, detecting analyte concentration above a critical value leads to the differentiation between sick and healthy subjects. The response of gas-sensing arrays can be then processed by employing artificial intelligence, machine-learning, and pattern recognition techniques. Reprinted with permission from Ref. [6] Copyright © 2015 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim.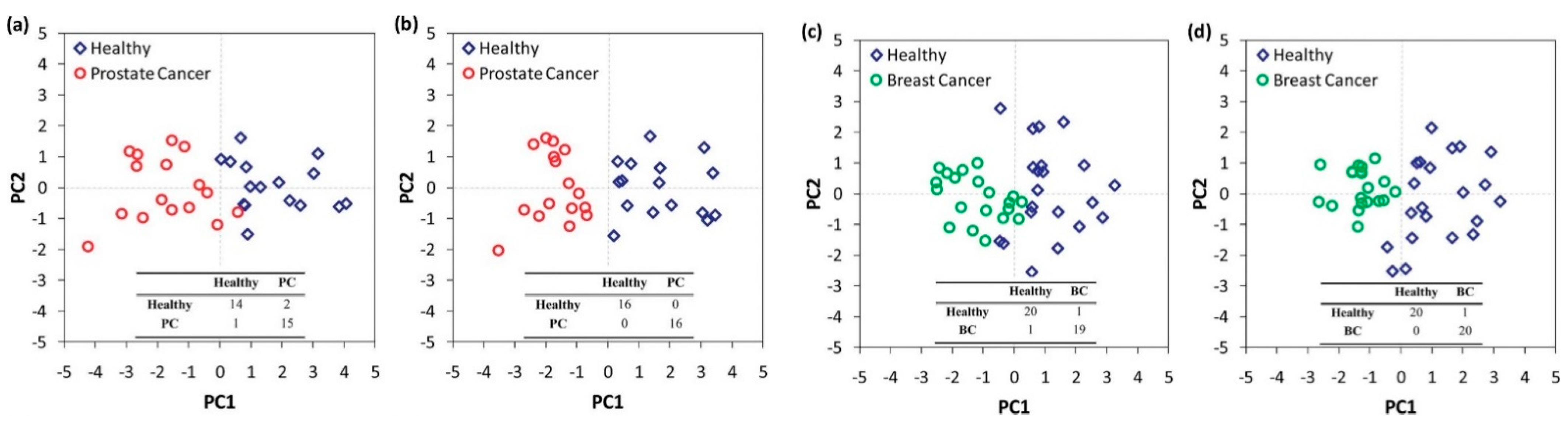 Figure 3. Statistical analysis of the response of a nanomaterial-based, cross-reactive chemiresistor for real-world samples of sick and healthy subjects. The use of PCA permits the differentiation of the groups. Notably, relative humidity compensation reduces the dispersion of different clusters thereby improving the discrimination between healthy and sick subjects. Representative 2D breath-analysis PCA plots for prostate cancer diagnosis: (a) without relative humidity compensation; (b) with relative humidity compensation. PCA plots for breast cancer diagnosis: (c) without relative humidity compensation; (d) with relative humidity compensation. Adapted with permission from Ref. [4243] Copyright © 2012, American Chemical Society.
Figure 3. Statistical analysis of the response of a nanomaterial-based, cross-reactive chemiresistor for real-world samples of sick and healthy subjects. The use of PCA permits the differentiation of the groups. Notably, relative humidity compensation reduces the dispersion of different clusters thereby improving the discrimination between healthy and sick subjects. Representative 2D breath-analysis PCA plots for prostate cancer diagnosis: (a) without relative humidity compensation; (b) with relative humidity compensation. PCA plots for breast cancer diagnosis: (c) without relative humidity compensation; (d) with relative humidity compensation. Adapted with permission from Ref. [4243] Copyright © 2012, American Chemical Society.2. Differential Diagnosis and Disease Phenotyping and Staging in Breath Analysis
As can be observed on Table 2, exhaled VOCs have been used not only for disease diagnosis in comparison to healthy subjects, but also for the discrimination between different diseases (e.g., GCa, LC, and asthma/COPD [4344]) or even for the discrimination between the different stages of a particular disease (e.g., CKD stages [4647]). One of the main prerequisites for the development of clinically applicable diagnostic tests is the effective discrimination between different diseases with similar symptoms and biochemical pathways [4748]. The uncertainty in the differentiation of patients with distinct diseases comprises one of the main drawbacks for studies distinguishing a specific disease from HC [4849]. Thus, the use of breath analysis for differential diagnosis as well as disease staging or phenotyping, using either analytical techniques or sensing devices (Table 3), attracted significant research interest over the last years. As expected, the contribution of nanomaterials is of great importance with many recent publications of nanomaterial-based sensors focusing on disease differentiation, staging or phenotyping, rather than the simple discrimination between patients and HC (Table 3). Table 2. Sensor arrays for exhaled VOCs detection as biomarkers of several diseases, in real-world or synthetic samples, using conventional materials and/or nanomaterials.| Sensing Element | Disease | Targeted VOCs | LOD | Subjects | Classifier | Results | T | Ref. | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| In Vivo Studies—Real-World Samples | ||||||||||||
| Chemiresistor—arrays | ||||||||||||
| Molecularly capped AuNPs—14 different ligands | Lung cancer | 1-Methyl-4-(1-methyl ethyl) benzene, Toluene, 3,3-Dimethyl pentane, 2,3,4-Trimethyl hexane, Dodecane, 1,1′-1-Butenylidene)bis benzene |
NA | 30 LC, 26 CC, 22 BC, 18 PC, 22 HC | PCA | Good discrimination of cancer types from HC, but not between them. No VOC overlap in abundance between HC and cancer patients. |
RT | [16] | ||||
| Colorectal cancer | 1,1′-(1-Butenylidene)bis benzene, 1,3-Dimethyl benzene, 1-Iodo nonane, (1,1-Dimethylethyl)thio acetic acid, 4-(4-Propylcyclohexyl)-4′-cyano1,1′-biphenyl-4-yl ester benzoic acid, 2-Amino-5-isopropyl-8-methyl-1-azulene carbonitrile |
|||||||||||
| Breast cancer | 3,3-Dimethyl pentane, 2-Amino-5-isopropyl-8-methyl-1-azulene carbonitrile, 5-(2-Methylpropyl)nonane, 2,3,4-Trimethyl decane, 6-Ethyl-3-octyl ester 2-trifluoromethyl benzoic acid | |||||||||||
| Prostate cancer | Toluene, 2-Amino-5-isopropyl-8-methyl-1-azulene carbonitrile, 2,2-Dimethyl decane, p-Xylene |
|||||||||||
| Molecularly capped AuNPs—7 different ligands | Prostate cancer | Toluene, 2-Amino-5-isopropyl-8-methyl-1-azulene carbonitrile, 2,2-Dimethyl decane, p-Xylene |
NA | 9 PC, 10 HC | DFA | 100% specificity, 100% sensitivity | RT | [4243] | ||||
| Breast cancer | 3,3-Dimethyl pentane, 2-Amino-5-isopropyl-8-methyl-1-azulene carbonitrile, 5-(2-Methylpropyl)nonane, 2,3,4-Trimethyl decane, 6-Ethyl-3-octyl ester 2-trifluoromethyl benzoic acid | 10 BC, 11 HC | 100% sensitivity, 95% specificity | |||||||||
| Molecularly capped AuNPs—3 different ligands | Chronic kidney disease | healthy vs. stage 2: Isoprene, Acetone, Styrene, Toluene, 2-Butatone, 2,2,6-Trimethyl octane, 2,4-Dimethyl heptane Stage 2 vs. 3: Isoprene, Acetone, 2,2,6-Trimethyl octane, 2-Butatone, 2,4-Dimethyl heptane Stage 3 vs. 4: Acetone, Ethylene Glycol, Acetoin |
1–5 ppb | 42 CKD, 20 HC | SVM | 79% accuracy early-stage CKD vs. HC 85% accuracy CKD stage 4 vs. stage 5 |
RT | [4647] | ||||
| Molecularly capped AuNPs—5 different ligands | Ovarian cancer | Styrene, Nonanal, 2-Ethylhexanol, 3-Heptanone, Decanal, Hexadecane |
ppb level | 17 OV, 26 HC | DFA | 82% accuracy | RT | [4445] | ||||
| Molecularly capped AuNPs—8 different ligands | COVID-19 | NA | NA | 49 COVID-19, 33 non-COVID symptomatic, 58 HC |
DFA | 76% accuracy COVID-19 vs. HC 95% accuracy COVID-19 vs. non-COVID symptomatic |
RT | [24] | ||||
| PAH-coated random SWCNTs network—4 different PAHs | Multiple sclerosis | Hexanal, 5-Methyl-undecane | NA | 37 MS, 18 HC | DFA | 80.4% accuracy | RT | [4950] | ||||
| Molecularly caped AuNPs/CDs-coated random SWCNTs network—20 different sensing films | Alzheimer’s and Parkinson’s disease | 24 VOCs | 1–5 ppb | 15 AD, 30 PD, 12 HC | DFA | 85% accuracy AD vs. HC 78% accuracy PD vs. HC 84% accuracy AD vs. PD |
RT | [5051] | ||||
| Molecularly caped AuNPs/PAH-coated random SWCNTs network—20 different sensing films | 17 diseases (LC, CC, HNC, OC, BLC, PC, KC, GC, CD, UC, IBS, IPD, MS, PDISM, PH, PET, CKD) | 2-Ethylhexanol, 3-Methylhexane, 5-Ethyl-3-methyloctane, Acetone, Ethanol, Ethyl acetate, Ethyl benzene, Isononane, Isoprene, Nonanal, Styrene, Toluene, Undecane |
10 ppb | 813 any of 17 diseases, 591 HC |
DFA, HCA | 86% average accuracy | RT | [5152] | ||||
| Ligand capped Au, Pt, and CuNPs—6 different sensing films | Human cutaneous leishmaniasis | 2,2,4-trimethyl pentane, 4-methyl-2-ethyl-1-pentanol, methyl vinyl ketone, nonane, 2,3,5-trimethyl hexane, hydroxy-2,4,6-trimethyl-5-(3-methyl-2 butenyl)cyclohexyl) methyl acetate, 3-ethyl-3-methyl heptane, octane, 2-methyl-6-methylene-octa-1,7-dien-3-ol | NA | 28 HCL, 32 HC | PCA, DFA | 98.2% accuracy, 96.4% sensitivity, 100% specificity | RT | [5253] | ||||
| pristine, COOH-, Hex-4T-Hex/DNA/oligomers, PTCDA/TAPC/TCTA monomers or PANI-functionalized SWCNTs | COPD | NH3, NO2, H2S, benzene, 2-propanol, acetone, ethanol, sodium hypochlorite, water | sub-ppb | 12 COPD, 9 HC | PCA | Acetone, ethanol and 2-propanol selective PANI-, TAPC- and COOH-CNTs, respectively. NO2 relevant driver of real-samples classification. Larger clinical trials needed. |
RT | [5354] | ||||
| Pristine WO3, 0.008 wt % | and 0.042 wt % Pt-WO3 macroporous NFs | Halitosis | H2S and Methyl mercaptan (in presence of Toluene and Acetone) |
sub-ppm | 4 simulated halitosis breath samples (1 ppm), 4 HC | PCA | Successful classification | 350 °C | [5455] | |||
| 7 different commercial MOS | Lung cancer | Ethyl benzene, 4-Methyl octane, Undecane, 2,3,4-trimethyl hexane |
Down to a few ppb | 37 NSCLC (81.1% I, II), 48 HC |
PCA | 75% accuracy Promising prognostic tool after LC resection surgery |
300 °C | [5556] | ||||
| 5 different commercial MOS | Lung cancer, COPD | NA | NA | 32 LC, 38 COPD, 72 HC | PCA, SVM, k-nearest neighbors | LC vs. HC: 91.3% accuracy, 84.4% sensitivity and 94.4% specificity COPD vs. HC: 90.9% accuracy, 81.6% sensitivity and 95.8% specificity |
NA | [5657] | ||||
| Field Effect Transistor (FET)—arrays | ||||||||||||
| Molecularly modified SiNWs | Gastric cancer | 2-Propenenitril, Furfural, 6-Methyl-5-heptene-2-one |
Down to a few ppb | 30 GC, 77 HC | DFA | >85% accuracy | RT | [5758] | ||||
| Molecularly modified SiNWs | Gastric cancer | 2-Propenenitril, Furfural, 6-Methyl-5-heptene-2-one |
Down to a few ppb | 149 LC, 40 GC, 56 Asthma/COPD, 129 HC |
DFA, ANN | >80% accuracy | RT | [4344] | ||||
| Lung cancer | Heptane, Decane, 2-Methyl pentane, 2-Ethyl-1-hexanol, Propanal, Pentanal, Acetone |
|||||||||||
| Asthma/COPD | Pentane | |||||||||||
| Electrochemical sensor | ||||||||||||
| Commercial NO, CO sensors, carbon electrode with linear-aldehyde selective porous poly tetrafluoroethylene membrane | Diabetes | NO, CO, Formaldehyde, Acrolein, Propanal, Crotonaldehyde, Butanal, Pentanal, Hexanal, Heptanal, Octanal, Nonanal, Decanal, Acetaldehyde | Low ppb | 15 diabetic, 14 HC | LC vs. HC, diabetic vs. HC Cross-sensitivity for aldehyde sensor: Moderate for high level of ethanol and isopropanol/Weak for H2S, NO, methanol, 3-heptanone/None for NO2, propofol, isoprene, or acetone |
RT | [5859] | |||||
| Lung cancer | 3 LC, 3 smokers, 3 HC | |||||||||||
| Optical—Colorimetric sensor arrays | ||||||||||||
| 24 chemically reactive colorants | Lung cancer | NA | Low ppm | 92 LC, 137 HC | LPM | Accuracy 81.1% LC vs. HC, 82.5–89% one histology vs. HC, 86.4% ADC vs. SCC |
RT | [5960] | ||||
| Optical sensors | ||||||||||||
| PMTFP-coated optical fiber | Vit. E deficiency | Ethane | pmol/L | 20 HC | NA | NA | RT | [6061] | ||||
| Liver diseases, Schizophrenia, Breast cancer, Rheumatoid Arthritis | Pentane | |||||||||||
| Lung cancer | Heptane, Octane, Decane, Benzene, Toluene, Styrene | |||||||||||
| Piezoelectric (SAW) sensor arrays | ||||||||||||
| GC-column/Polyisobutylene-coated SAW, non-coated SAW sensors | Lung cancer | Styrene, Decane, Isoprene, Benzene, Undecane, 1-Hexene, Hexanal, Propyl benzene, Heptanal, 1,2,4-Trimethyl benzene, Methyl cyclopentane | 500 ppb | 20 LC, 15 HC, 7 chronic bronchitis |
ANN | 80% sensitivity and specificity | RT | [6][6162] | ||||
| Piezoelectric (QCM) sensor arrays | ||||||||||||
| 7 different | metalloporphyrins | COPD | NA | NA | 5 COPD per GOLD stage (20), 5 HC | PLS-DA | Fair repeatability of measurements within HC and hypoxemic COPD patients (stage 4) Potential COPD severity assessment |
RT | [6263] | |||
| 8 different | metalloporphyrins | Asthma | NA | NA | 27 asthma, 24 HC | PCA, FNN | 87.5% accuracy | RT | [6364] | |||
| 8 different | metalloporphyrins | Lung cancer | NA | NA | 20 LC, 10 HC | PLS-DA | 85% accuracy LC vs. HC 75% accuracy ADC vs. SCC |
RT | [6465] | |||
| 8 different | metalloporphyrins | Lung cancer | NA | NA | 70 LC, 76 HC | PLS-DA | 81% sensitivity, 100% specificity | RT | [6566] | |||
| 8 different | metalloporphyrins | Tuberculosis | NA | NA | 51 TB (31/51 +HIV), 20 HC | PCA, k-nearest neighbors | 94.1% sensitivity, 90% specificity | RT | [6667] | |||
| 7 different | metalloporphyrins | Halitosis | H2S, Butyric acid, Valeric acid | 10–15 ppb | Oral malodor subjects, HC | PCA | PC1 78% of data variance | 50 °C | [6768] | |||
| 8 different anthocyanins | Asthma | NA | NA | 15 asthma, 27 HC | Factor Analysis | 75% of total variance Repeatability similar to spirometry and eNO |
RT | [6869] | ||||
| In vitro studies—Cell lines/Synthetic samples | ||||||||||||
| Chemiresistor arrays | ||||||||||||
| CNT-conductive polymer nanocomposites—5 different polymers | Lung cancer | Isopropanol, Tetrahydrofuran, Dichloromethane, Toluene, n-Heptane, Cyclohexane, Methanol, Ethanol, Water | NA | PCA | High sensitivity and selectivity for all the analytes, PC1-PC3 98% of total variance, except the two alkanes |
RT | [6970] | |||||
| Pristine rGO and rGO functionalized with 8 different amine ligands—9 elements | Cancer | Ethanol, 2-Ethylhexanol, Ethyl benzene, Nonanal | 25 ppm | NA | PCA | Successful discrimination of VOCs The LOD and the effect of humidity have to be decreased |
RT | [7071] | ||||
| Pristine Pd, ZnO and polypyrrole NWs | Breast cancer | Heptanal | 8.98 ppm | NA | PCA | 73.2% PC1 variance High sensitivity and specificity |
RT | [7172] | ||||
| Acetophenone | 798 ppb | |||||||||||
| 2-Propanol | 129.5 ppm | |||||||||||
| Isopropyl myristate | 134 ppm | |||||||||||
| Pristine In2O3 and WO3 NRs, Au, Pt, or Pd NPs-decorated In2O3 and WO3 NRs—8 elements | Diabetes | Acetone | 1.48 ppb | NA | Polar plot | Effective visual discrimination between the gases. Future PCA, DFA, HCA analysis. |
150–300 °C | [7273] | ||||
| Asthma | NO2 | 1.9 ppt | ||||||||||
| Halitosis | H2S | 2.47 ppb | ||||||||||
| WO3 NTs | Pt NPs—WO3 NTs, | Pd NPs—WO3 NTs | Asthma | NO | 50 ppb | NA | NA | NA | 350 °C | [7374] | ||
| Lung cancer | Toluene | 100 ppb | 400 °C | |||||||||
| Pristine, 0.1% wt GO- and 0.1 wt % thin layered graphite WO3 Hemi tubes | Diabetes | Acetone | 1 ppm | NA | NA | NA | 350 °C | [7475] | ||||
| Halitosis | H2S | |||||||||||
| Electrochemical sensor | ||||||||||||
| MWCNTs/Au-Ag NPs/GCE | Gastric cancer | 3-Octanone | 0.3 ppb | MGC-803 GC and GES-1 gastric mucosa cell lines | NA | Easy cell line discrimination, high sensitivity, good VOCs selectivity in presence of CO2, acetone and ethanol | RT | [7576] | ||||
| Butanone | 0.5 ppb | |||||||||||
| SiNWs-rGO | Infectious diseases | Cyclohexane, Formaldehyde in presence of Methanol, Ethanol, Acetonitrile, Acetaldehyde and humidity | 1 ppm | NA | Novel electrode platform with increased sensitivity, selectivity and repeatability | [7677] | ||||||
| Piezoelectric (SAW) sensor arrays | ||||||||||||
| SH-Calix4arene, | AuNRs, AgNCs, | Calix4arene-AuNRs, Calix4arene-AgNCs | Lung cancer | Chloroform, Toluene, Isoprene, Acetone, n-Hexane, Ethanol | 1.52–12.34 ppm for CHCl3 1.54–2.64 ppm for toluene |
NA | NA | Sensitivity ↑ for all VOCs Chloroform, toluene: 6–8 times higher sensitivity than individual responses Selectivity ↑: modified AuNRs for CHCl3, modified AgNCs for Toluene |
RT | [7778] | ||
| Pristine or AuNPs-functionalized zeolitic-imidazole-framework nanocrystals (ZIF-8, ZIF-67) | Diabetes | Acetone, Ammonia, Ethanol | acetone 1.1–3.6 ppm, ethanol 0.5–3 ppm NH3 1.6–3.2 ppm |
NA | PCA | Effective discrimination of diabetes biomarkers | RT | [7879] | ||||
| Piezoelectric (QCM) sensor arrays | ||||||||||||
| TiO2-MWCNTS and Cobalt (II) phthalocyanine-silica on Au layers | Diabetes | Acetone | 4.33 ppm | NA | NA | High sensitivity | RT | [7980] | ||||
| Asthma | NO | 5.75 ppb | ||||||||||
| Optical—Colorimetric arrays | ||||||||||||
| 36 chemically responsive dyes (porphyrin derivatives, NaFluo) | Lung cancer | p-Xylene, Styrene, Isoprene, Hexanal | 50 ppb | NA | HCA, PCA, BPNN | 100% accuracy of kind and concentration discrimination, promising for real-sample experiments | RT | [8081] | ||||
| AuNRs-modified metalloporphyrins and pH responsive dyes—36 spots | Lung cancer | Decane, Undecane, Hexanal, Heptanal, 1,2,4-Trimethylbenzene, Benzene | <1 ppm | NA | PCA, HCA | 64.2% accuracy of structurally similar VOCs, 93% photoprotection of metalloporphyrins, ↑ repeatability and long-term stability | RT | [8182] | ||||
| Sensor | Diseases/Phenotypes/Stages | Subjects | Classifier | Results | Ref. | ||
|---|---|---|---|---|---|---|---|
| Differential diagnosis | |||||||
| Cyranose 320 | NSCLC vs. COPD (GOLD stage I-III) | 10 NSCLC, 10 COPD | PCA, CDA | 85% acc. | [8283] | ||
| LC vs. non-cancer (COPD, asthma, pneumonia, pulmonary embolism, BPN) | 165 LC, 91 non-cancer | SVM | 87.3% acc. | [8384] | |||
| LC vs. COPD vs. LC/COPD vs. HC | 63 LC, 15 COPD, 79 both, 78 HC | 77.4% acc., 100% accurate LC/COPD classification | |||||
| LC vs. non-cancer (COPD, asthma, pneumonia, pulmonary embolism, bronchiectasis, BPN, TB) | 252 LC, 223 non-cancer | LRA | Sens.: 95.8% (S), 96.2% (NS) Spec.: 92.3% (S) 90.6% (NS) |
[8485] | |||
| Asthma vs. COPD | 20 asthma, 30 COPD | PCA, CDA | 96% acc. Within/between day repeatability, reproducibility of e-Noses |
[8586] | |||
| Fixed and classic asthma vs. COPD (GOLD stages II-III) | 21 fixed asthma, 40 COPD | PCA, CDA | 88% acc., 85% sens., 90% spec. | [8687] | |||
| 39 classic asthma, 40 COPD | 83% acc., 91% sens., 90% spec. | ||||||
| IPF vs. COPD | 32 IPF, 33 COPD | PCA, CDA | 80% cross-validated acc., Wider patient cohorts and inclusion of more comorbidities needed | [8788] | |||
| COPD vs. LC vs. BC | 50 COPD, 30 LC, 50 BC | PCA, CDA, CAP | Correct classification values: LC vs. COPD 96.47%, LC vs. BC 93.05%, BC vs. COPD 100%, COPD vs. LC vs. BC 91.35% |
[4849] | |||
| Bronchial vs. Laryngeal SCC (advanced) | 10 bronchial, 10 laryngeal | JMP Pro | 10% misclassification, 100% sens., 80% spec. | [8889] | |||
| AD vs. PD vs. HC | 18 AD, 16 PD, 19 HC | LDA | 76.2% sens., 45.8% spec., p < 0.0001 | [8990] | |||
| AeoNose | ILDs (COP, CTD) vs. COPD, ILDs subgroups (COP, HP, IPF, sarcoidosis, uILD, asbestosis, NSIP, RB-ILD, DIP) |
28 COP, 23 COPD | Athena program, t-test | AUC 0.77, 75% sens., 71% spec. | [9091] | ||
| 25 CTD-ILD, 23 COPD | AUC 0.85, 88% sens., 71% spec. | ||||||
| 174 ILDs, 23 COPD | Less accurate discrimination of ILDs subgroups (e.g., AUC IPF vs. CTD-ILD 0.86, COP vs. CTD-ILD 0.82) | ||||||
| Asthma vs. CF | 20 asthma (moderate-severe), 13 CF | ANN | AUC 0.90, 89% sens., 77% spec. | [9192] | |||
| HNSCC vs. LC | 52 HNSCC, 32 LC | ANN | Acc., sens., spec.: 93%, 96%, 88% (best fit), 85%, 85%, 84% (cross-validation) |
[9293] | |||
| Cancer types | 100 HNSCC, 40 bladder, 28 colon cancer | ANN | Acc., sens., spec.: 81%, 79%, 81% HNSCC vs. colon cancer, 84%, 80%, 86% HNSCC vs. bladder cancer, 84%, 88%, 79% Colon vs. bladder cancer |
[9394] | |||
| SpiroNose | LC vs. COPD vs. asthma vs. HC | 31 LC, 31 COPD, 37 asthma, 45 HC | PCA | Cross-validation values 78–88%, repeatability ↑ | [9495] | ||
| ILD subgroups: | 141 sarcoidosis, 85 IPF, 33 CTD-ILD, 25 HP, 11 IPAF, 10 NSIP | PLS-DA | Acc., sens., spec.: 77%, 75%, 84% IPF vs. HP, 94%, 98%, 85% IPF vs. CTD-ILD, 92%, 92%, 90% IPF vs. NSIP, 89%, 87%, 100% IPF vs. IPAF, 75%, 100%, 67% CTD-ILD vs. IPAF, 98%, 90%, 100% CTC-ILD vs. NSIP, 90%, 94%, 72% HP vs. sarcoidosis, 91%, 92%, 88% (training), 91%, 95%, 79% (validation) IPF vs. non-IPF |
[9596] | |||
| Chemiresistor-based | alkane sensor | LC vs. HC LC vs. COPD |
12 LC, 12 COPD, 13 HC | MANOVA | LC: 83.3% sens., 88% spec. Sensor acc no smoke-dependence |
[9697] | |
| MOS, electrochemical, hot wire, and catalytic | LC vs. COPD | 48 LC, 52 COPD | 8 different | 76.9–84.75% acc., 75–81.36% sens., 78.79–88.14 spec. Highest acc. With KPCA-XGBoost |
[9798] | ||
| LC vs. COPD | 33 LC, 28 COPD | PCA-SVM, KPCA-SVM, PCA-XGBoost, KPCA-XGBoost | 82.52–96% acc., 78.33–95% sens., 85–96.67% spec. Highest acc. With KPCA-XGBoost |
[9899] | |||
| Organically-coated AuNPs and SWCNTs based chemiresistor | LC (I/II) vs. BPN | 16 LC, 30 BPN | DFA | 87% acc., 75% sens., 93% spec. Low LC sample → careful interpretation |
[99100] | ||
| BC vs. benign | 30 HC, 15 BBT, 13 DCIS, 96 BC | DFA | Acc., sens., spec.: 88.3%, 90.6%, 83.3% BC vs. BBT/HC, 71.2–82%, 62.6–80%, 75.7–82.3% BC vs. BBT, 81.4–84.4%, 83–83.3%, 81–92% BC vs. DCIS |
[100101] | |||
| Gca vs. OLGIM groups (0-IV) | 99 Gca, 155 OLGIM 0, 136 OLGIM I-II, 34 OLGIM III-IV, 53 PUD | DFA | Acc., sens., spec.: 92%, 73%, 98% Gca vs. 0–IV, 84%, 90%, 80% Gca vs. 0, 87%, 97%, 84% Gca vs. 0–II, 90%, 93%, 80% Gca vs. III-IV, 85%, 93%, 80% Gca vs. I–IV, 87%, 87%, 87% Gca vs. PUD |
[101102] | |||
| Gca vs. benign gastric conditions | 37 Gca, 32 ulcers, 61 less severe conditions | DFA | 89% sens., 90% spec. 84% sens., 87% spec. |
[102103] | |||
| ulcer vs. less severe | |||||||
| AD vs. PD AD vs. PD vs. HC |
15 AD, 30 PD, 12 HC | DFA | AD vs. PD: 84% acc., 80% sens., 87% spec. Feasible overall discrimination, with large PD/HC overlap |
[5051] | |||
| NA-NOSE | BC, benign breast conditions, normal mammographs | 11 BC, 14 benign, 7 normal mammographs | PCA/ANOVA/Student’s t-test, SVM | 94% sens., 80% spec. for benign vs. BC and negative mammography, Similar results with both methods | [103104] | ||
| MCNPs-based chemiresistor—6 sensors-array | IBD vs. IBS | 71 IBD (35 UC, 36 CD), 26 IBS | ANN | 81/88% acc., 92/73% sens., 53/100% spec. (real/artificial) | [104105] | ||
| CD vs. UC | 75/96% acc., 75/100% sens., 47/93% spec. (real/artificial) | ||||||
| Molecularly modified SiNW FET | Gca vs. LC | 40 Gca, 149 LC, 56 asthma/COPD | DFA, ANN | 92% acc., 93% LC, and 85% Gca correct classification | [4344] | ||
| LC vs. asthma and COPD | 89% acc., 92% sens., 80% spec. | ||||||
| MOS gas sensor array | Gca vs. gastric ulcer patients | 49 Gca, 30 gastric ulcer | Back-propagation Neural network | 93% acc., 94.38% sens., 89.93% spec. Classification acc. Of malignant, benign, normal subjects: 92.54%, 93.17%, 92.49%. |
[105106] | ||
| OC vs. benign and HC | 86 OC, 51 benign, 114 HC | PCA, k-NN | Acc., sens., spec.: 85%, 6%, 84% (cross-validation/strict prediction), 87%, 89%, 86% (prediction/strict prediction), 86%, 84%, 85% (cross-validation/most probable pred.), 100%, 100%, 100% (prediction/most probable pred.) |
[106107] | |||
| AD vs. PD vs. HC | 20 AD, 20 PD, 20 HC | PCA | Effective discrimination of AD vs. PD and HC | [107108] | |||
| BIONOTE | CLD vs. NC-CLD | 65 CLD, 39 NC-CLD | PLS-DA, radar plot | Successful discrimination, 16 cirrhotic patients misclassified | [108109] | ||
| Commercial (MQ) gas sensors | CKD vs. diabetes vs. HC high creatinine, HC low creatinine | 84 CKD, 24 diabetes, 54 HC high creatinine, 54 HC low creatinine | Radar plots, PCA, SVM, PLS-regression, HCA | PCA: 96.64% of total variance expressed in PC1–3 SVM: 100% correct classification of samples |
[109110] | ||
| Disease histology/phenotyping | |||||||
| Tor Vergata e-Nose | SCC vs. ADC | 10 SCC, 10 ADC | PLS-DA | 75% correct classification | [6465] | ||
| 24 colorants | SCC vs. ADC | 22 SCC, 50 ADC | LPM | 86.4% acc. | [5960] | ||
| SCLC vs. NSCLC | 9 SCLC, 83 NSCLC | 78.1% acc. (moderate) | |||||
| UV-irradiated pristine, Au, Pt, Au/Pt, Ni, Fe-doped WO3NWs | LC vs. HC | 4 SCLC, 8 SCC, 10 ADC, 12 HC | PCA | 98.6 % acc. | [110111] | ||
| SCLC vs. NSCLC, SCC vs. ADC | DFA | Acc.: 84.5% SCLC vs. NSCLC,77.5% SCC vs. ADC | |||||
| Molecularly capped AuNPs and SWCNT based chemiresistors | LC with vs. without EGFR mutation | 19 with EGFR, 34 without EGFR | DFA | 83% acc., 79% sens., 85% spec. | [99100] | ||
| Cyranose 320, Tor Vergata, Common In-vent, Owlstone Lonestar | Clinically stable vs. unstable episodes of asthma | 22 partly controlled persistent asthma | PCA | Correct classification: 95% baseline vs. loss of control, 86% loss of control vs. recovery Owlstone Lonestar the most prominent |
[111112] | ||
| Cyranose 320 | Asthma inflammatory phenotypes | 24 EOS., 10 NEUTR., 18 PAUC. | PCA | Acc., sens., spec.: 73%, 60%, 79% EOS. vs. NEUTR., 74%, 55%, 87% EOS vs. PAUC., 89%, 94%, 80% NEUTR. vs. PAUC. | [112113] | ||
| Uncontrolled asthma-like symptoms | Training set: 65 cluster 1, 22 cluster 2, 34 cluster 3 | one-way ANOVA, Kruskal-Wallis |
Significant differences concerning chest tightness during exercise, dyspnea and gender | [113114] | |||
| HC and controlled vs. partly controlled and uncontrolled asthma | 10 HC, 9 controlled, 7 partly, 12 uncontrolled | PCA, radar plot | Good predictive ability Cross-validated AUC 0.80, 79% sens., 84% spec. |
[114115] | |||
| Organically-coated AuNPs and SWCNT-based chemiresistor | BC subtypes | 12 LuminalA, 42 LuminalB, 12 Triple Negative, 16 HER2+, 14 HER2 equivocal | DFA | Acc., sens., spec.: LuminalA vs. others 81.3–87.7%, 75–87.5%, 82.1–87.5% LuminalB vs. others78.1–86.3%, 83.3–85.3%, 74.1–87.2% HER2+ vs. others 81.3–82.4%, 81.3–91%, 80.7–81.3% Triple neg, vs. others 82.9–90.3%, 83.3–93.3%, 82.9–89.4% Luminal vs. non-Luminal 70.8–87.7%, 70.4–88.1%, 71.4–87.1% LuminalA vs. LuminalB 85.7–94%, 75–91.7%, 88.2–95.2% HER2 status/luminal 85.7–100%, 85.7–100%, 83.3–100% HER2 status/non-luminal 90.9%, 90.9%, 90.9% |
[100101] | ||
| Disease staging | |||||||
| Tor Vergata e-Nose | LC Stage I vs. II/III/IV | 40 stage I, 18 stage II, 6 III/IV | PLS-DA | Sens.: stage I 90% vs. stage II-IV 57% (+ metabolic diseases), stage I 96% vs. stage II-IV 60% (LC only) |
[6566] | ||
| 24 colorants | LC Stage I/II vs. LC stage III/IV | 41 SCLC, 42 NSCLC | LPM | 79.3 % acc. (moderate) | [5960] | ||
| 11 sensor-array (MOS, electrochemical, hot wire and catalytic) | LC Stage III vs. IV | 44 stage II, 46 stage IV | PCA-SVM, KPCA-SVM, PCA-XGBoost, KPCA-XGBoost | 70.42–82.42% acc., 45–81% sens., 79–95.5% spec. | [9899] | ||
| Organically-coated AuNPs and SWCNTs based chemiresistor | OLGIM stages | 155 OLGIM 0, 136 OLGIM I-II, 34 OLGIM III-IV, 7 Dysplasia | DFA | Acc., sens., spec.: 0-II vs. III-IV and dysplasia 61%, 83%, 60%, 0 vs. I-II 43%, 45%, 41%, 0 vs. III-IV 66%, 90%, 61%, 0 vs. I-IV 50%, 50%, 50%, I-II vs. III-IV 64%, 80%, 60% | [101102] | ||
| GCa I-II vs. III-IV | 17 GCa I-II, 18 GCa III-IV | DFA | 89% sens., 94% spec. | [102103] | |||
| Molecularly modified SiNW FET | LC staging (I-II vs. III-IV) | 34 early stage, 110 advanced stage | DFA, ANN | 81% acc., 34.5%sens.,95% spec. | [4344] | ||
| GCa staging (I-II vs. III-IV) | 86.5% correct classification, 84.6 early stage, 87.5 advanced | ||||||
| Cyranose 320 | Bronchial/Laryngeal in situ vs. advanced |
bronchial: 10 in situ, 10 advanced, laryngeal: 12 in situ, 10 advanced | JMP Pro | 21% misclassification rate, 82% sens., 75% spec. | [8889] | ||
| BIONOTE | Liver cirrhosis (A, B, C Child–Pugh) | NA | PLS-DA | Successful discrimination | [108109] | ||
2.1. The Case for Lung Diseases
Chronic and acute lung diseases such as asthma, COPD, idiopathic pulmonary fibrosis (IPF), LC, mesothelioma, and sarcoidosis have been connected with similar metabolic alternations [115116]. Especially asthma and COPD are also characterized by similar symptoms [37] with COPD being commonly underdiagnosed or diagnosed at late stages [116117]. Concerning LC, no symptoms are expressed in early stages [117118] while disease manifestation is limited to non-specific symptoms [117118] including cough, short breath, chest pain, and weight loss [37]. Disease phenotyping, on the other hand, is mandatory in some cases. Asthma subtypes such as eosinophilic, neutrophilic, mixed granulocytic, and paucigranulocytic asthma [118119] are characterized by similar symptoms while different treatment is required [119120]. Similarly, immunosuppressive, antifibrotic, or a combination of medications may be needed for fibrotic interstitial lung diseases (ILDs), depending on the respective phenotype (inflammatory, more fibrotic, or combination) [9596]. Thus, reliable phenotyping is needed for appropriate medication to be administered [33][119120][9596]. LC is subdivided into different categories with different clinical characteristics as well. Small cell LC (SCLC), with 20–25% percentage of occurrence [120121], is characterized by increased metabolic and proliferation rates compared to other cancer cells [121122], while non-small cell LC (NSCLC) accounts for 70–75% of LC cases and is subdivided into the smoking-related [37][122123] squamous cell carcinoma (SCC) [120121] and the non-squamous cell carcinomas [37] including adenocarcinomas (ADC) (minor smoking correlation) and large-cell carcinoma (LCC) [120121]. Consequently, the accurate discrimination of different lung diseases and subtypes of a lung disease, especially using breath analysis of exhaled VOCs, is of particular importance. The use of GC-MS has rendered lung disease differentiation, phenotyping and staging feasible in many cases. To be more specific, LC discrimination from patients with other lung diseases has been investigated in several studies. In an attempt to discriminate between NSCLC, COPD, and HC patients, by taking smoking habits into account, 4 VOCs were identified (in varying concentrations) for NSCLC and COPD [123124]. In another study, Wang et al. attempted to discriminate LC from COPD, asthma, pneumonia, pulmonary embolism and benign lung tumor patients; however, the selected 10 VOCs could not discriminate accurately between the two groups, implying their potential confounding role during LC-biomarkers determination [18]. Koureas et al. have also attempted to discriminate LC from other respiratory diseases, using 19 distinctive VOCs, based on the underlying disease mechanisms (targeted method); only the discrimination of LC patients from HC, using ethylbenzene, toluene, styrene, 2- and 1-propanol was achieved [4748]. However, in a more recent study of the same group, the discrimination of LC patients from patients suffering from sarcoidosis, hypersensitivity pneumonitis, interstitial lung diseases or pulmonary infections was achieved with an increased accuracy of 75.3%; the 29 VOCs were selected following a hypothesis-generating non-targeted strategy [124125]. In a different study, LC was accurately distinguished from pulmonary non-malignant diseases (PNMD; COPD, pulmonary tuberculosis, asthma) using 10 VOCs while 5 were selected as characteristic of LC in contrast to both PNMD and HC [125126]. The differentiation of LC patients from patients with benign pulmonary nodules (BPN) has also been extensively reported. Apart from a study by Wang et al. [18] which included patients with benign lung tumors, Fu et al. have investigated the respective discriminant ability of carbonyl VOCs [126][127][128][129]. Four carbonyl VOCs, captured by a silicon micro-reactor, were found to present increased concentration in LC patients when compared to BPN patients and HC [126127]. In a subsequent study, the same group achieved the differentiation of both early and III, IV stage LC patients from BPN patients, with high sensitivity (83%) and particularly increased specificity (74%) in comparison to positron emission tomography (90% and 39%, respectively) [127128], while 6 carbonyl VOCs have permitted a classification accuracy of 89% of LC vs. BPN patients [128129]. More recently, Chen et al. identified 19 VOCs able to distinguish not only LC and BPN patients (this with an accuracy of 80.9%) but also early-stage LC patients from BPN (with an accuracy of 75.6%), being remarkably promising for early LC diagnosis [117118]. LC histology and staging characterization using analytical methods is another important target of this research field. It was reported that 1-butanol, 3-hydroxy-2-butanone [9], as well as 4-hydroxyhexenal [126127], can differentiate SCC from ADC patients with the former being decreased for SCC in contrast to the other 2 VOCs. Similarly, SCLC and NSCLC can be potentially distinguished from 4-hydroxynonenal and C5H10O [126127]. Hexanal has also been found in higher concentrations for SCLC patients compared to NSCLC, potentially due to increased metabolic rates [121122]; Chen et al. have achieved NSCLC and SCLC differentiation, with an accuracy of 93.9%, using a pattern of 20 VOCs [117118]. Concerning LC staging (I, II, III, or IV), a pattern of 19 VOCs was used to distinguish between early (I, II) and advanced LC stages (III, IV) with 82.7% accuracy [117118] while Fu et al. demonstrated that exhaled 2-butanone concentration is significantly different between stages I and II–IV [126127]. Apart from LC, other lung diseases are also studied for accurate diagnosis. A series of studies have focused on asthma phenotyping using GC-MS. Brinkman et al. identified 3 VOCs significantly correlated with sputum eosinophils [111112], while Ibrahim et al. identified VOC-patterns differentiating eosinophilic from non-eosinophilic (6 VOCs) and neutrophilic from non-neutrophilic (7 VOCs) [129130]. Recently, Schleich et al. identified 4 VOCs discriminating eosinophilic from neutrophilic, eosinophilic from paucigranulocytic and neutrophilic from paucigranulocytic asthma, with accuracy similar to blood eosinophils and FeNO tests [118119]. In a more recent study, the same group used two-dimensional GC-high resolution-time-of-flight-MS, selected ion flow tube mass spectrometry (SIFT-MS), 10 VOCs, and 9 ion channels so as to achieve asthma phenotyping with an accuracy of 75% [119120]. COPD phenotyping and staging has also been attempted using analytical techniques. Fens et al. identified 8 eosinophils- and 17 neutrophils-related VOCs, with only one VOC overlapping between the two subgroups. More VOCs were related with cell counts for Global Initiative for Obstructive Lung Disease (GOLD) stage II, in comparison to GOLD stage I [130131]. In another study, 11 COPD patients with >1% and 6 with >2% eosinophil count were discriminated from non-eosinophilics (<1% and <2% eosinophil count, respectively) with accuracies of 79% and 92% [131132]. Exacerbation prediction of both asthmatic children [132][133][134][135] and adults [111112][129130] as well as COPD patients [135136] also comprises a subject of study. Following the promising applications of analytical methods that highlight the potential capabilities of breath analysis in phenotyping, staging and differential diagnosis of lung diseases, sensing devices have also been used in respective applications with remarkable results. Research interest has focused on LC discrimination from other lung diseases such as COPD and asthma. Cyranose 320 has achieved separation of NSCLC from COPD (GOLD stage I–III) with an accuracy of 85%, in an article by Dragonieri et al. [8283]. Tirzīte et al. have used this e-Nose to effectively discriminate, not only LC patients from COPD, asthma, pneumonia, pulmonary embolism, benign lung tumor patients, and HC, with 87.3% accuracy, but also between LC patients, COPD patients, LC patients suffering also from COPD and HC with 77.4% accuracy, and totally correct classification of the 79 LC/COPD patients [8384]. In a more recent study, the same group discriminated LC from patients with non-malignant lung diseases as well as bronchiectasis, tuberculosis, and HC by taking into account smoking habits. An overall sensitivity and specificity of 95.8% and 92.3% for smokers and 96.2% and 90.6% for non-smokers, respectively, was observed using Cyranose 320 [8485]. More recently, the same e-Nose was used by Rodriguez et al. for the discrimination of COPD from LC and BC, achieving an overall correct classification of 91.35% while LC correct classification in relation to COPD was equal to 96.47% [4849]. Interestingly, the contribution of the 32 sensors in the discrimination was also assessed [4849]. Tor Vergata e-Nose has been used effectively for discriminating LC patients from COPD, Interstitial lung disease, Pleurisy and Bronchitis patients, with a sensitivity of 89.3% for LC patients [136137]. In a particularly promising study, SpiroNose discriminated LC, asthma, COPD, and HC, with the respective accuracy values presented in Figure 4a (68–88%) [9495]. The applicability of breath sampling and analysis was tested as the collection of asthma breath samples at two different sites led to similar results (Figure 4b) [9495]. In the field of non-commercial and self-developed sensors, Tan et al. have attempted to develop a cross-reactive alkane-based chemiresistor combining carbon powder and tetracosane, achieving not only high affinity for alkanes and low sensitivity for polar VOCs (water, ethanol, ethanal) but also effective differentiation of 12 LC patient from 13 HC and 12 COPD patients [9697]. In a more recent study, researchers attempted to differentiate LC, COPD, and asthma patients from HC, using an array of 8 sensors of 4 different types (MOS, electrochemical, hot wire, and catalytic type). The array achieved accuracy between 76.9–84.75%, using different machine learning methods [9798]. Accuracy values were greater for LC and COPD prediction; however, the maximum accuracy value of 84.75% was attained using kernel principal component analysis—extreme gradient boosting (KPCA-XGBoost), which indicates excellent discriminatory capability for LC and COPD patients [9798]. Similarly, using an array of 11 sensors of 4 different types (namely MOS, electrochemical, hot wire, and catalytic type), Liu et al. differentiated non-smoking LC and COPD patients, with the best discriminatory accuracy (96%) being achieved using the same machine learning technique [9899]. The discrimination of LC from asthma and COPD patients was also achieved by Haick’s group with particularly high classification accuracies [4344]. Early-stage LC discrimination from BPN has been reported by Haick et al. using an array of 40 chemiresistors based on MCNPs (Au NPs) and molecularly-coated SWCNTs, achieving an accuracy of 87%. Considering that the required treatment may change in the occurrence of genetic alternations, the differentiation of patients with and without epidermal growth factor receptor (EGFR) mutation was also attempted with an accuracy of 83% [99100].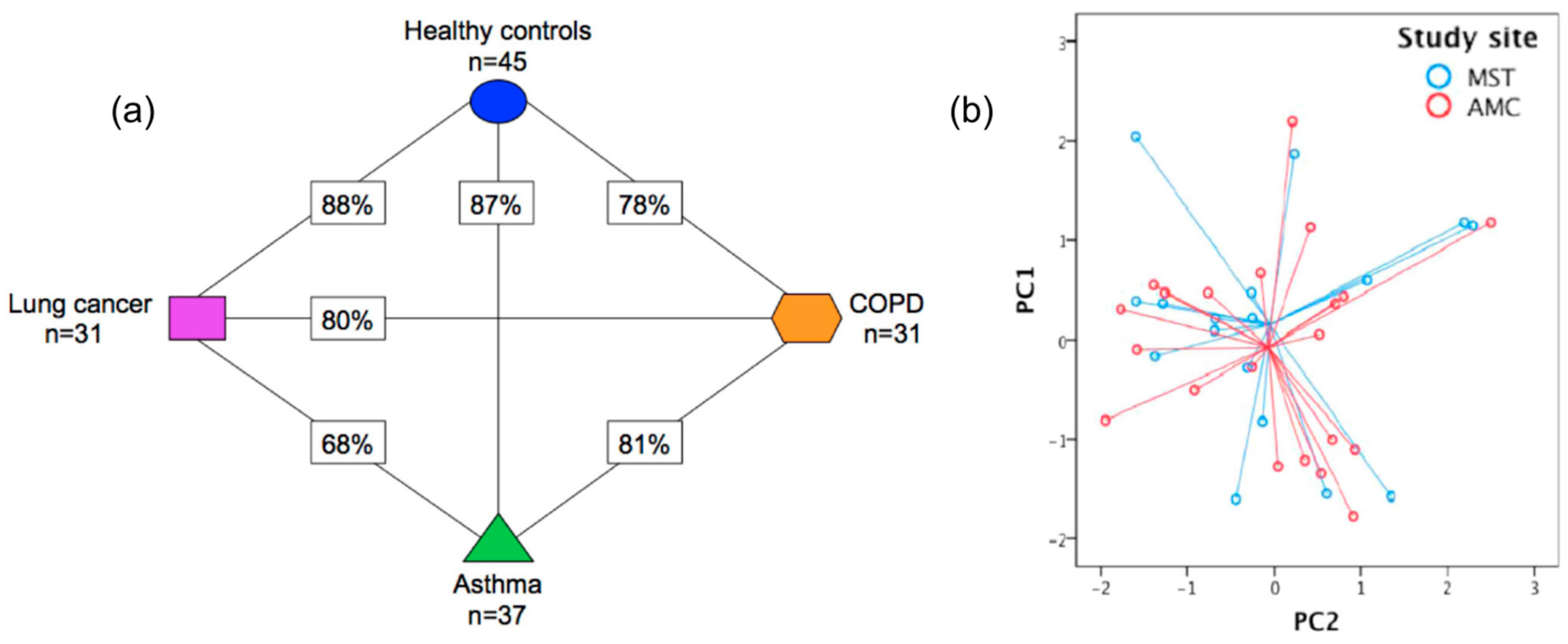 Figure 4. (a) Presentation of the cross-validation percentages of the differentiation of asthma, COPD, LC patients, and HC, using SpiroNose; (b) PCA plot of breathprints collected from asthmatic patients at the Academic Medical Center (AMC), Amsterdam and Medical Spectrum Twente (MST), Enschede, for which no significant differentiation is observed (p = 0.892). Adapted with permission from Ref. [9495]. Copyright © 2015 IOP Publishing Ltd.
Concerning LC histology and staging with sensing devices, promising studies have been reported in the literature. The discrimination of NSCLC subtypes ADC and SCC has been permitted using Tor Vergata e-Nose with an accuracy of 75%, by applying endoscopic breath sampling [6465] as well as by using a colorimetric sensor-array of 24 elements developed by Mazzone et al. ultimately achieving an accuracy of 86.4% [5960]. SCLC and NSCLC differentiation and LC staging (I/II vs. III/IV) were also examined by Mazzone et al. though with moderate accuracies [5960]. A 6-sensor-array based on UV-irradiated (394 nm) pristine or metal-doped WO3NWs (Table 3) differentiated effectively not only ADC from SCC, but also between SCLC and NSCLC with 77.5% and 84.5% accuracy values, respectively (Figure 5) [110111]. In another study aiming at the discrimination of LC patients from HC while taking into account the existence of metabolic comorbidities, Tor Vergata e-Nose exhibited far higher sensitivity for stage I LC in comparison to the rest of stages, either in the presence or absence of metabolic diseases (Table 3) [6566]. LC staging was recently attempted by Liu et al. along with COPD discrimination as mentioned above, with stage III LC being effectively discriminated from stage IV with an accuracy higher than 80%, using KPCA-XGBoost [9899]. Haick’s team has achieved LC staging with an accuracy of 81% and with low sensitivity, using a molecularly modified Si NW FET (Table 3) [4344].
Figure 4. (a) Presentation of the cross-validation percentages of the differentiation of asthma, COPD, LC patients, and HC, using SpiroNose; (b) PCA plot of breathprints collected from asthmatic patients at the Academic Medical Center (AMC), Amsterdam and Medical Spectrum Twente (MST), Enschede, for which no significant differentiation is observed (p = 0.892). Adapted with permission from Ref. [9495]. Copyright © 2015 IOP Publishing Ltd.
Concerning LC histology and staging with sensing devices, promising studies have been reported in the literature. The discrimination of NSCLC subtypes ADC and SCC has been permitted using Tor Vergata e-Nose with an accuracy of 75%, by applying endoscopic breath sampling [6465] as well as by using a colorimetric sensor-array of 24 elements developed by Mazzone et al. ultimately achieving an accuracy of 86.4% [5960]. SCLC and NSCLC differentiation and LC staging (I/II vs. III/IV) were also examined by Mazzone et al. though with moderate accuracies [5960]. A 6-sensor-array based on UV-irradiated (394 nm) pristine or metal-doped WO3NWs (Table 3) differentiated effectively not only ADC from SCC, but also between SCLC and NSCLC with 77.5% and 84.5% accuracy values, respectively (Figure 5) [110111]. In another study aiming at the discrimination of LC patients from HC while taking into account the existence of metabolic comorbidities, Tor Vergata e-Nose exhibited far higher sensitivity for stage I LC in comparison to the rest of stages, either in the presence or absence of metabolic diseases (Table 3) [6566]. LC staging was recently attempted by Liu et al. along with COPD discrimination as mentioned above, with stage III LC being effectively discriminated from stage IV with an accuracy higher than 80%, using KPCA-XGBoost [9899]. Haick’s team has achieved LC staging with an accuracy of 81% and with low sensitivity, using a molecularly modified Si NW FET (Table 3) [4344].
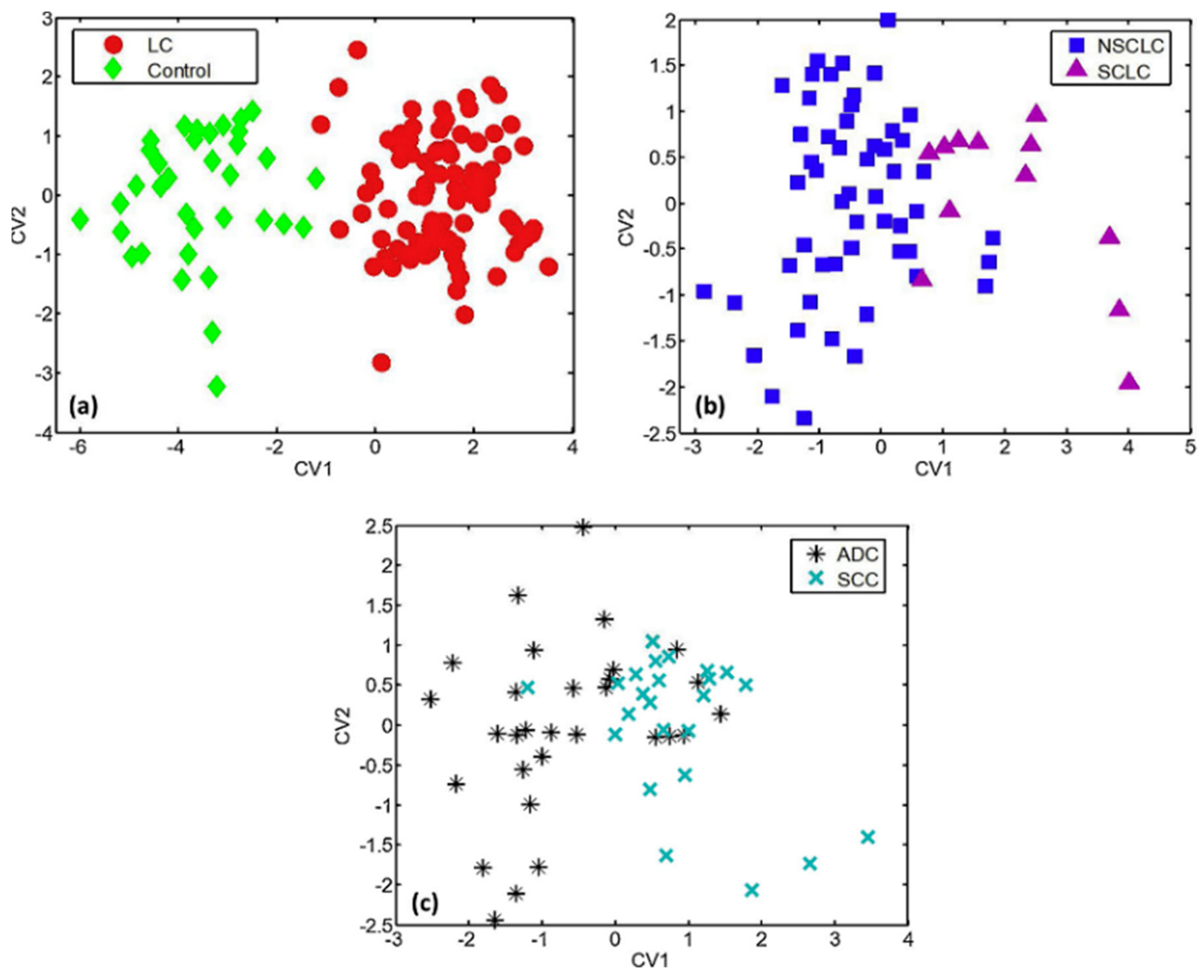 Figure 5. DFA plots representing the discrimination of (a) LC patients from HC; (b) SCLC from NSCLC patients; and (c) SCC from ADC patients, using a 6-sensor array of UV-irradiated (394 nm) pristine or metal-doped WO3NWs. The arrays achieved the detection of lung cancer but also the prediction of LC histological subtypes. Reprinted with permission from Ref. [110111]. Copyright © 2020 Published by Elsevier B.V.
As in the case of breath analysis with analytical methods, precise diagnosis of lung diseases other than LC via sensing devices is an extensive field of research. The effective discrimination of COPD and asthma has been reported in the literature by Fens at al. using Cyranose 320 and taking into consideration smoking habits, leading to high cross-validated accuracy values (Table 3) [8586][8687]. More recently, asthma and CF discrimination was also reported for pediatric population using AeoNose and with high accuracy values, excluding the confounding factors of diet, exercise, comorbidities and inhaled drugs [9192]. Concerning ILDs, Krauss et al. used the AeoNose in an attempt to differentiate between ILDs subgroups (Table 3), with moderate accuracy, as well as between ILDs cryptogenic organizing pneumonia and connective-tissue diseases-associated ILD from COPD patients with good sensitivity and specificity [9091]. COPD and IPF differentiation has been recently investigated by Dragonieri et al., with a high accuracy of 80%, verified by external validation using Cyranose 320 [8788]. In contrast to Krauss et al., Moor’s group achieved to reliably discriminate patients suffering from different ILDs by using SpiroNose as well as greater cohorts of ILD-patients (Table 3) [9596]; the group demonstrated the applicability of e-Noses in ILDs differential diagnosis and specifically in IPF discrimination from non-IPF patients with high accuracies (91%) [9596].
Disease phenotyping using sensing devices seems to be also feasible. Plaza et al. achieved differentiation between the three inflammatory phenotypes of asthma with high accuracy values (Table 3), with the participants’ phenotypes being characterized by differential leukocyte counts in induced sputum [112113] while asthma-control assessment has been also reported. Brinkman et al. used 4 different e-Noses in order to discriminate between stable and unstable periods, comparing baseline (control) vs. loss of control and loss of control vs. recovery breath samples, with the Owlstone Lonestar being the most prominent concerning the discrimination of unstable periods (Table 3) [111112]. More recently, Moreira et al. demonstrated the ability of Cyranose 320 to discriminate the uncontrolled asthma-like symptoms, using 3 different groups of asthmatic or suspicious of asthma participants divided by unsupervised hierarchical clustering [113114]. The division of participants was based on asthma, lung function, symptoms of the last month, age, and food/drink intake 2 h before breath sampling [113114]. In another recent study, the same e-Nose was used for the effective discrimination of HC and asymptomatic-controlled asthmatic children from the symptomatic partly-controlled and uncontrolled asthmatic children, after assessing the discriminatory ability of subsets of the 32 sensors of Cyranose 320 for the six different possible combinations of the 4 studied groups; increased feasibility and modest to good diagnostic accuracy values were obtained [114115]. Cyranose 320 has been also used for COPD phenotyping permitting (especially in the case of GOLD stage I) the detection of activation of inflammatory cells, indicating increased inflammatory activity in mild rather than severe COPD [130131].
Figure 5. DFA plots representing the discrimination of (a) LC patients from HC; (b) SCLC from NSCLC patients; and (c) SCC from ADC patients, using a 6-sensor array of UV-irradiated (394 nm) pristine or metal-doped WO3NWs. The arrays achieved the detection of lung cancer but also the prediction of LC histological subtypes. Reprinted with permission from Ref. [110111]. Copyright © 2020 Published by Elsevier B.V.
As in the case of breath analysis with analytical methods, precise diagnosis of lung diseases other than LC via sensing devices is an extensive field of research. The effective discrimination of COPD and asthma has been reported in the literature by Fens at al. using Cyranose 320 and taking into consideration smoking habits, leading to high cross-validated accuracy values (Table 3) [8586][8687]. More recently, asthma and CF discrimination was also reported for pediatric population using AeoNose and with high accuracy values, excluding the confounding factors of diet, exercise, comorbidities and inhaled drugs [9192]. Concerning ILDs, Krauss et al. used the AeoNose in an attempt to differentiate between ILDs subgroups (Table 3), with moderate accuracy, as well as between ILDs cryptogenic organizing pneumonia and connective-tissue diseases-associated ILD from COPD patients with good sensitivity and specificity [9091]. COPD and IPF differentiation has been recently investigated by Dragonieri et al., with a high accuracy of 80%, verified by external validation using Cyranose 320 [8788]. In contrast to Krauss et al., Moor’s group achieved to reliably discriminate patients suffering from different ILDs by using SpiroNose as well as greater cohorts of ILD-patients (Table 3) [9596]; the group demonstrated the applicability of e-Noses in ILDs differential diagnosis and specifically in IPF discrimination from non-IPF patients with high accuracies (91%) [9596].
Disease phenotyping using sensing devices seems to be also feasible. Plaza et al. achieved differentiation between the three inflammatory phenotypes of asthma with high accuracy values (Table 3), with the participants’ phenotypes being characterized by differential leukocyte counts in induced sputum [112113] while asthma-control assessment has been also reported. Brinkman et al. used 4 different e-Noses in order to discriminate between stable and unstable periods, comparing baseline (control) vs. loss of control and loss of control vs. recovery breath samples, with the Owlstone Lonestar being the most prominent concerning the discrimination of unstable periods (Table 3) [111112]. More recently, Moreira et al. demonstrated the ability of Cyranose 320 to discriminate the uncontrolled asthma-like symptoms, using 3 different groups of asthmatic or suspicious of asthma participants divided by unsupervised hierarchical clustering [113114]. The division of participants was based on asthma, lung function, symptoms of the last month, age, and food/drink intake 2 h before breath sampling [113114]. In another recent study, the same e-Nose was used for the effective discrimination of HC and asymptomatic-controlled asthmatic children from the symptomatic partly-controlled and uncontrolled asthmatic children, after assessing the discriminatory ability of subsets of the 32 sensors of Cyranose 320 for the six different possible combinations of the 4 studied groups; increased feasibility and modest to good diagnostic accuracy values were obtained [114115]. Cyranose 320 has been also used for COPD phenotyping permitting (especially in the case of GOLD stage I) the detection of activation of inflammatory cells, indicating increased inflammatory activity in mild rather than severe COPD [130131].
2.2. Cancers
Discrimination between different cancer types and cancer stages/histologies as well as between malignant and benign tumors (additionally to LC which has been mentioned earlier) using breath analysis of VOCs is also a hot research topic. Analytical techniques have been used for such applications. Phillips at al., for example, detected 5 VOCs and were able to differentiate BC patients from patients with abnormal mammograms and negative biopsies, with 93.8% sensitivity and 84.6% specificity [137138]. Haick’s group identified 21 exhaled VOCs that were significantly different between HC and patients suffering from breast benign tumors (BBT), ductal carcinoma in situ (DCIS, early-stage BC) and BC; a potentially cancer-related set of 14 VOCs that were significantly different between malignant and non-malignant patients was also identified in this study thus permitting group differentiation with 78% sensitivity and 72% accuracy [100101]. In another study by the same group, the detection of GCa and the presence/absence and risk level of precancerous lesions was attempted using GC-MS so as to identify 8 VOCs statistically different between GCa and operative link on gastric intestinal metaplasia (OLGIM) groups (e.g., GCa vs. OLGIM 0-IV, GCa vs. OLGIM 0-II, GCa vs. OLGIM 0), as well as between GCa and peptic ulcer disease (PUD) and OLGIM 0-IV and PUD (p-values < 0.017) [101102]. Those 8 VOCs, in different combinations, are considered to correspond to the breathprints of OLGIM groups [101102]. Remarkably, respective applications of sensing devices have been extensively investigated for various cancer types. Haick’s group has used NA-NOSE in order to discriminate between subjects with BC, benign breast conditions or normal mammographs, achieving increased sensitivity and specificity values for the BBT patients in comparison to the other 2 groups [103104]. In another study by the same group a chemiresistor based on organically-coated Au NPs and SWCNTs was successfully used for the differentiation of BC from BBT and HC, BBT only or DCIS only, as well as for the differentiation of different molecular BC sub-groups as presented on Table 3. Larger studies are necessary, though for significant statistical results and more information to be obtained [100101]. Very recently, differentiation between BC and LC has been achieved as well by Rodriguez et al. with a correct classification of 93.05% [4849]. Concerning GCa, the differentiation of GCa and OLGIM groups as well as between different OLGIM stages (e.g., OLGIM 0 vs. I–II, 0 vs. I–IV, I–II vs. III–IV) has been attempted by Haick’s group using the same type of chemiresistor-array and leading to high validated accuracy values in some cases (Table 3) [101102]. In another promising study, Haick et al. used a nanomaterial-based chemiresistor (Table 3) that permitted the successful discrimination of Gca from benign conditions along with Gca staging (early vs. late stages) [102103]. Gca differentiation from gastric ulcer patients has been achieved by Daniel et al. with a great classification ability, using an array of commercial MOS gas sensors and various ANN types [105106]. Remarkably, a molecularly modified Si NW FET, developed by Haick’s group, has permitted Gca staging with an accuracy of 87% as well as Gca differentiation from LC with an accuracy of 92% [4344]. More recently, discrimination of ovarian cancer (OC) from women with benign tumors and HC was achieved by Raspagliesi et al. with great classification performance, both in the case of strict and most probable prediction, using again MOS sensors [106107]. Notably in class prediction application, 4/23 early-stage OC patients were misclassified as benign/HC along with 2/14 OC patients with tumor size < 3 cm in cross validation phase, while in prediction phase only 1/9 early-stage patients were misdiagnosed [106107]. Another common cancer type, i.e., head and neck cancer (HNC), has been studied for differential diagnosis with sensing devices. As an example, Hooren et al. attempted to discriminate HNC from LC with a high accuracy of 93% analyzing patients’ exhaled breath with AeoNose, excluding cutaneous tumors and salivary glands malignancies [9293]. HNC differentiation from colon and bladder cancer with AeoNose was also reported, by the same group along with the discrimination between bladder and colon cancer, demonstrating the discriminant ability of the e-Nose for those cancer types after double cross-validation [9394]. More recently, Cyranose 320 was used for the differentiation of advanced bronchial (LC) and laryngeal (HNC) SCC, as well as for the discrimination of advanced and in situ stages of bronchial and laryngeal SCC, leading to successful classification of the groups (Table 3) [8889].2.3. Liver, Renal, and Intestinal Diseases
Liver cirrhosis, chronic hepatitis [108109], CKD [4647], and inflammatory bowel diseases (IBD) comprise common liver, kidney, and intestinal diseases, respectively. As far as CKD is concerned, it is characterized by gradual loss of kidney function within months or years, while different treatment is demanded depending on disease stage (stages I–V) [4647]. Similarly, in the case of chronic liver disease (CLD), disease staging is of great importance; following CLD diagnosis, using invasive biopsy, liver function assessment is conducted biochemically [108109]. On the other hand, early stage and precise IBD and IBS diagnosis, as well as the invasive diagnostic methods followed for IBD, comprise challenging issues [104105]. Consequently, precise diagnosis and staging of liver, kidney and intestinal diseases are particularly important and have been attempted using breath analysis with sensing devices. Pennazza et al. for instance used BIONOTE e-nose to successfully differentiate not only liver cirrhosis and CLD from non-cirrhotic CLD (chronic hepatitis) but also liver cirrhosis stages by taking into account smoking habits and potential comorbidities (e.g., diabetes, lung, and heart diseases) [108109]. Concerning renal diseases, Haick’s group achieved CKD staging using organically functionalized Au NPs-based chemiresistors and SVM [4647]. The classification of CKD stage IV in relation to stage V was permitted by 2 or 3 sensors with an accuracy of 85%, a sensitivity of 75% and specificity of 92% while only one sensor allowed for the discrimination of early and advanced stages with 76% accuracy, 75% sensitivity, and 77% specificity [4647]. The discrimination of CKD from other diseases has been also attempted. Specifically, discrimination of CKD, diabetes, and HC with high or low creatinine has been attempted with success using an array of commercial (MQ) sensors along with different classification methods, with SVM and PCA leading to good group classification [109110]. Remarkably, pre-concentration or dehumidification were not needed for clear classification to be accomplished [109110]. The effective discrimination between the intestinal diseases IBD and IBS has been also reported along with further differentiation of the IBD into ulcerative colitis (UC) and Crohn’s disease (CD), using not only artificial but also real-breath samples and a MCNPs-based chemiresistor (Table 3) [104105]. The higher accuracy values observed when using artificial samples is expected and attributed to the standard concentration of VOCs, contrary to the variable concentration of VOCs in breath [104105].2.4. Neurodegenerative Diseases
Neurodegenerative diseases are characterized by gradually augmented occurrence, as a direct consequence of the increased lifespan of human population [138139], with Alzheimer (AD) and Parkinson (PD) being the most frequent [5051]. Concerning AD, early disease detection is of great importance for preventing, decelerating and terminating the disease [138139] while diagnosis for both diseases is based on the assessment of clinical symptoms [5051]. Remarkably, the analysis of exhaled VOCs has been investigated for precise AD diagnosis as well as for differential diagnosis between AD and PD [5051][138139]. Recently, Tiele et al. attempted to discriminate mild cognitive impairment due to AD (MCI) from AD, using GC-MS, achieving 60% sensitivity and 84% specificity along with the detection of 6 potential discriminant VOCs [138139]. Haick’s group, on the other hand, achieved the discrimination of AD and PD as well as an overall discrimination of AD, PD, and HC, using an array of 20 nanomaterial-based chemiresistors and GC-MS (Table 3) [5051]. In a similar study, the ability of IMS and Cyranose 320 to differentiate AD, PD, and HC was demonstrated, achieving a high overall discriminant capability (Table 3) [8990]. IMS analysis revealed five VOCs significantly different between the groups [8990]. The discrimination of the same groups of subjects has been also attempted using different arrays of MOS sensors (TGS, MICS) with one of the combinations (8 MOS sensors) demonstrating the best discriminant ability [107108].3. Conclusions
Exhaled breath analysis, especially using selective or cross-reactive sensors, comprises a non-invasive method that holds a great promise for application in early-stage and differential diagnosis of not only respiratory but also systemic diseases. This entry presents the main categories of nanomaterials and sensors that have been used up to now in exhaled breath analysis for disease diagnosis as well as to demonstrate the applicability of breath analysis in differential diagnosis, phenotyping, and staging of several types of diseases, especially via the use of cross-reactive sensing devices. The progressive development of novel nanomaterials offers a great opportunity to develop more effective sensing elements, both for selective and cross-reactive sensors and especially for point-of-care diagnosis, treatment monitoring and population screening. However, fundamental challenges in this novel research field inhibit the application of breath analysis in clinical practice and should therefore be addressed. Concerning analytical techniques used for exhaled VOCs identification, the use of bulky, expensive, and complex analytical devices is limited in hospitals while their incorporation in portable point-of-care systems is still unattainable [139140]. In addition, the validity of breath analysis results is of major concern since the trace levels of exhaled VOCs affect the analysis accuracy [140141]. At the same time the lack of clear breath sampling protocols [140141], e.g., breath collection [139140] and breath storage, could potentially change sample composition [139140] and therefore emerge as important challenges. Sample composition can be also affected by confounding factors, i.e., age, gender, place of living, habits, and nutrition. In the case of sensors exhalation rate, a hardly controlled parameter, may also play a confusing role hence complicating the procedure [139140].References
- Chen, T.; Liu, T.; Li, T.; Zhao, H.; Chen, Q. Exhaled breath analysis in disease detection. Clin. Chim. Acta 2021, 515, 61–72.
- Gashimova, E.; Temerdashev, A.; Porkhanov, V.; Polyakov, I.; Perunov, D.; Azaryan, A.; Dmitrieva, E. Investigation of different approaches for exhaled breath and tumor tissue analyses to identify lung cancer biomarkers. Heliyon 2020, 6, e04224.
- Phillips, M.; Cataneo, R.N.; Saunders, C.; Hope, P.; Schmitt, P.; Wai, J. Volatile biomarkers in the breath of women with breast cancer. J. Breath Res. 2010, 4, 026003.
- Oakley-Girvan, I.; Davis, S.W. Breath based volatile organic compounds in the detection of breast, lung, and colorectal cancers: A systematic review. Cancer Biomarkers 2017, 21, 29–39.
- Pite, H.; Morais-Almeida, M.; Rocha, S.M. Metabolomics in asthma: Where do we stand? Curr. Opin. Pulm. Med. 2018, 24, 94–103.
- Vishinkin, R.; Haick, H. Nanoscale Sensor Technologies for Disease Detection via Volatolomics. Small 2015, 11, 6142–6164.
- Broza, Y.Y.; Vishinkin, R.; Barash, O.; Nakhleh, M.K.; Haick, H. Synergy between nanomaterials and volatile organic compounds for non-invasive medical evaluation. Chem. Soc. Rev. 2018, 47, 4781–4859.
- Tisch, U.; Haick, H. Chemical sensors for breath gas analysis: The latest developments at the Breath Analysis Summit 2013. J. Breath Res. 2014, 8, 027103.
- Song, G.; Qin, T.; Liu, H.; Xu, G.B.; Pan, Y.Y.; Xiong, F.X.; Gu, K.S.; Sun, G.P.; Chen, Z.D. Quantitative breath analysis of volatile organic compounds of lung cancer patients. Lung Cancer 2010, 67, 227–231.
- Ma, H.; Li, X.; Chen, J.; Wang, H.; Cheng, T.; Chen, K.; Xu, S. Analysis of human breath samples of lung cancer patients and healthy controls with solid-phase microextraction (SPME) and flow-modulated comprehensive two-dimensional gas chromatography (GC × GC). Anal. Methods 2014, 6, 6841–6849.
- Hakim, M.; Broza, Y.Y.; Barash, O.; Peled, N.; Phillips, M.; Amann, A.; Haick, H. Volatile organic compounds of lung cancer and possible biochemical pathways. Chem. Rev. 2012, 112, 5949–5966.
- Wilson, A.D. Advances in electronic-nose technologies for the detection of volatile biomarker metabolites in the human breath. Metabolites 2015, 5, 140–163.
- Bag, A.; Lee, N.E. Recent Advancements in Development of Wearable Gas Sensors. Adv. Mater. Technol. 2021, 6, 2000883.
- Bos, L.D.; Sterk, P.J.; Fowler, S.J. Breathomics in the setting of asthma and chronic obstructive pulmonary disease. J. Allergy Clin. Immunol. 2016, 138, 970–976.
- Broza, Y.Y.; Haick, H. Nanomaterial-based sensors for detection of disease by volatile organic compounds. Nanomedicine 2013, 8, 785–806.
- Peng, G.; Hakim, M.; Broza, Y.Y.; Billan, S.; Abdah-Bortnyak, R.; Kuten, A.; Tisch, U.; Haick, H. Detection of lung, breast, colorectal, and prostate cancers from exhaled breath using a single array of nanosensors. Br. J. Cancer 2010, 103, 542–551.
- Fens, N.; van der Schee, M.P.; Brinkman, P.; Sterk, P.J. Exhaled breath analysis by electronic nose in airways disease. Established issues and key questions. Clin. Exp. Allergy 2013, 43, 705–715.
- Wang, M.; Sheng, J.; Wu, Q.; Zou, Y.; Hu, Y.; Ying, K.; Wan, H.; Wang, P. Confounding effect of benign pulmonary diseases in selecting volatile organic compounds as markers of lung cancer. J. Breath Res. 2018, 12, 046013.
- Hashoul, D.; Haick, H. Sensors for detecting pulmonary diseases from exhaled breath. Eur. Respir. Rev. 2019, 28, 190011.
- Meyer, N.; Dallinga, J.W.; Nuss, S.J.; Moonen, E.J.C.; van Berkel, J.J.B.N.; Akdis, C.; van Schooten, F.J.; Menz, G. Defining adult asthma endotypes by clinical features and patterns of volatile organic compounds in exhaled air. Respir. Res. 2014, 15, 136.
- Corradi, M.; Poli, D.; Bouza, M. Observation of nonanoic acid and aldehydes in exhaled breath of patients with lung cancer Observation of nonanoic acid and aldehydes in exhaled breath of patients with lung cancer. J. Breath Res. 2017, 11, 26004.
- Boots, A.W.; Van Berkel, J.J.B.N.; Dallinga, J.W.; Smolinska, A.; Wouters, E.F.; Van Schooten, F.J. The versatile use of exhaled volatile organic compounds in human health and disease. J. Breath Res. 2012, 6, 027108.
- Haick, H.; Cohen-Kaminsky, S. Detecting lung infections in breathprints: Empty promise or next generation diagnosis of infections. Eur. Respir. J. 2015, 45, 21–24.
- Shan, B.; Broza, Y.Y.; Li, W.; Wang, Y.; Wu, S.; Liu, Z.; Wang, J.; Gui, S.; Wang, L.; Zhang, Z.; et al. Multiplexed Nanomaterial-Based Sensor Array for Detection of COVID-19 in Exhaled Breath. ACS Nano 2020, 14, 12125–12132.
- Giovannini, G.; Haick, H.; Garoli, D. Detecting COVID-19 from Breath: A Game Changer for a Big Challenge. ACS Sens. 2021, 6, 1408–1417.
- Grassin-Delyle, S.; Roquencourt, C.; Moine, P.; Saffroy, G.; Carn, S.; Heming, N.; Fleuriet, J.; Salvator, H.; Naline, E.; Couderc, L.J.; et al. Metabolomics of exhaled breath in critically ill COVID-19 patients: A pilot study. EBioMedicine 2021, 63, 103154.
- BreFenceTM Go COVID-19 Breath Test System Rapid Breath Test for COVID-19 Detection. Available online: https://secureservercdn.net/160.153.138.177/8g1.9f8.myftpupload.com/wp-content/uploads/2021/05/Breathonix-BreFence-Go-Breath-Test-System.pdf (accessed on 25 November 2021).
- HSA Grants Provisional Authorisation for ‘BreFence Go COVID-19 Breath Test System’ and ‘Traciex Breathalyser’ for Detection of COVID-19 Infection. Available online: https://www.hsa.gov.sg/announcements/regulatory-updates/hsa-grants-provisional-authorisation-for-brefence-go-covid-19-breath-test-system-and-traciex-breathalyser-for-detection-of-covid-19-infection (accessed on 25 November 2021).
- Provisional Authorisation for COVID-19 Tests—Breathonix Pte Ltd. Available online: https://www.hsa.gov.sg/docs/hprg-mdb/breathonix_brefence-go-covid-19-breath-test-system_provisional-authorisation-for-covid-19-tests_19052021.pdf (accessed on 25 November 2021).
- Wang, C.; Sun, B.; Guo, L.; Wang, X.; Ke, C.; Liu, S.; Zhao, W.; Luo, S.; Guo, Z.; Zhang, Y.; et al. Volatile organic metabolites identify patients with breast cancer, cyclomastopathy, and mammary gland fibroma. Sci. Rep. 2014, 4, 5383.
- Pereira, J.; Porto-Figueira, P.; Cavaco, C.; Taunk, K.; Rapole, S.; Dhakne, R.; Nagarajaram, H.; Câmara, J.S. Breath analysis as a potential and non-invasive frontier in disease diagnosis: An overview. Metabolites 2015, 5, 3–55.
- Gaida, A.; Holz, O.; Nell, C.; Schuchardt, S.; Lavae-Mokhtari, B.; Kruse, L.; Boas, U.; Langejuergen, J.; Allers, M.; Zimmermann, S.; et al. A dual center study to compare breath volatile organic compounds from smokers and non-smokers with and without COPD. J. Breath Res. 2016, 10, 026006.
- Neerincx, A.H.; Vijverberg, S.J.H.; Bos, L.D.J.; Brinkman, P.; van der Schee, M.P.; de Vries, R.; Sterk, P.J.; Maitland-van der Zee, A.H. Breathomics from exhaled volatile organic compounds in pediatric asthma. Pediatr. Pulmonol. 2017, 52, 1616–1627.
- Amal, H.; Haick, H. Point of care breath analysis systems. In Advanced Nanomaterials for Inexpensive Gas Microsensors: Synthesis, Integration and Applications, 1st ed.; Llobet, E., Ed.; Elsevier Inc.: Amsterdam, The Netherlands; Oxford, UK; Cambridge, MA, USA, 2019; pp. 315–334.
- Basanta, M.; Jarvis, R.M.; Xu, Y.; Blackburn, G.; Tal-Singer, R.; Woodcock, A.; Singh, D.; Goodacre, R.; Paul Thomas, C.L.; Fowler, S.J. Non-invasive metabolomic analysis of breath using differential mobility spectrometry in patients with chronic obstructive pulmonary disease and healthy smokers. Analyst 2010, 135, 315–320.
- Zhou, X.; Xue, Z.; Chen, X.; Huang, C.; Bai, W.; Lu, Z.; Wang, T. Nanomaterial-based gas sensors used for breath diagnosis. J. Mater. Chem. B 2020, 8, 3231–3248.
- Ratiu, I.A.; Ligor, T.; Bocos-Bintintan, V.; Mayhew, C.A.; Buszewski, B. Volatile Organic Compounds in Exhaled Breath as Fingerprints of Lung Cancer, Asthma and COPD. J. Clin. Med. 2020, 10, 32.
- Tisch, U.; Haick, H. Nanomaterials for cross-reactive sensor arrays. MRS Bull. 2010, 35, 797–803. Oliveira, Luciana Fontes de; Mallafré-Muro, Celia; Giner, Jordi; Perea, Lidia; Sibila, Oriol; Pardo, Antonio; Marco, Santiago Breath analysis using electronic nose and gas chromatography-mass spectrometry: A pilot study on bronchial infections in bronchiectasis. Clinica Chimica Acta 2021, 526, 6-13.
- Van Der Schee, M.P.; Fens, N.; Brinkman, P.; Bos, L.D.J.; Angelo, M.D.; Nijsen, T.M.E.; Raabe, R.; Knobel, H.H.; Vink, T.J.; Sterk, P.J. Effect of transportation and storage using sorbent tubes of exhaled breath samples on diagnostic accuracy of electronic nose analysis. J. Breath Res. 2013, 7, 016002. Tisch, U.; Haick, H. Nanomaterials for cross-reactive sensor arrays. MRS Bull. 2010, 35, 797–803.
- Behera, B.; Joshi, R.; Anil Vishnu, G.K.; Bhalerao, S.; Pandya, H.J. Electronic nose: A non-invasive technology for breath analysis of diabetes and lung cancer patients. J. Breath Res. 2019, 13, 24001. Van Der Schee, M.P.; Fens, N.; Brinkman, P.; Bos, L.D.J.; Angelo, M.D.; Nijsen, T.M.E.; Raabe, R.; Knobel, H.H.; Vink, T.J.; Sterk, P.J. Effect of transportation and storage using sorbent tubes of exhaled breath samples on diagnostic accuracy of electronic nose analysis. J. Breath Res. 2013, 7, 016002.
- Madianos, L.; Skotadis, E.; Patsiouras, L.; Filippidou, M.K.; Chatzandroulis, S.; Tsoukalas, D. Nanoparticle based gas-sensing array for pesticide detection. J. Environ. Chem. Eng. 2018, 6, 6641–6646. Behera, B.; Joshi, R.; Anil Vishnu, G.K.; Bhalerao, S.; Pandya, H.J. Electronic nose: A non-invasive technology for breath analysis of diabetes and lung cancer patients. J. Breath Res. 2019, 13, 24001.
- Konvalina, G.; Haick, H. Effect of humidity on nanoparticle-based chemiresistors: A comparison between synthetic and real-world samples. ACS Appl. Mater. Interfaces 2012, 4, 317–325. Madianos, L.; Skotadis, E.; Patsiouras, L.; Filippidou, M.K.; Chatzandroulis, S.; Tsoukalas, D. Nanoparticle based gas-sensing array for pesticide detection. J. Environ. Chem. Eng. 2018, 6, 6641–6646.
- Shehada, N.; Cancilla, J.C.; Torrecilla, J.S.; Pariente, E.S.; Brönstrup, G.; Christiansen, S.; Johnson, D.W.; Leja, M.; Davies, M.P.A.; Liran, O.; et al. Silicon Nanowire Sensors Enable Diagnosis of Patients via Exhaled Breath. ACS Nano 2016, 10, 7047–7057. Konvalina, G.; Haick, H. Effect of humidity on nanoparticle-based chemiresistors: A comparison between synthetic and real-world samples. ACS Appl. Mater. Interfaces 2012, 4, 317–325.
- Kahn, N.; Lavie, O.; Paz, M.; Segev, Y.; Haick, H. Dynamic Nanoparticle-Based Flexible Sensors: Diagnosis of Ovarian Carcinoma from Exhaled Breath. Nano Lett. 2015, 15, 7023–7028. Shehada, N.; Cancilla, J.C.; Torrecilla, J.S.; Pariente, E.S.; Brönstrup, G.; Christiansen, S.; Johnson, D.W.; Leja, M.; Davies, M.P.A.; Liran, O.; et al. Silicon Nanowire Sensors Enable Diagnosis of Patients via Exhaled Breath. ACS Nano 2016, 10, 7047–7057.
- Phillips, C.O.; Syed, Y.; Parthaláin, N.M.; Zwiggelaar, R.; Claypole, T.C.; Lewis, K.E. Machine learning methods on exhaled volatile organic compounds for distinguishing COPD patients from healthy controls. J. Breath Res. 2012, 6, 036003. Kahn, N.; Lavie, O.; Paz, M.; Segev, Y.; Haick, H. Dynamic Nanoparticle-Based Flexible Sensors: Diagnosis of Ovarian Carcinoma from Exhaled Breath. Nano Lett. 2015, 15, 7023–7028.
- Marom, O.; Nakhoul, F.; Tisch, U.; Shiban, A.; Abassi, Z.; Haick, H. Gold nanoparticle sensors for detecting chronic kidney disease and disease progression. Nanomedicine 2012, 7, 639–650. Phillips, C.O.; Syed, Y.; Parthaláin, N.M.; Zwiggelaar, R.; Claypole, T.C.; Lewis, K.E. Machine learning methods on exhaled volatile organic compounds for distinguishing COPD patients from healthy controls. J. Breath Res. 2012, 6, 036003.
- Koureas, M.; Kirgou, P.; Amoutzias, G.; Hadjichristodoulou, C.; Gourgoulianis, K.; Tsakalof, A. Target analysis of volatile organic compounds in exhaled breath for lung cancer discrimination from other pulmonary diseases and healthy persons. Metabolites 2020, 10, 317. Marom, O.; Nakhoul, F.; Tisch, U.; Shiban, A.; Abassi, Z.; Haick, H. Gold nanoparticle sensors for detecting chronic kidney disease and disease progression. Nanomedicine 2012, 7, 639–650.
- Rodríguez-Aguilar, M.; Díaz de León-Martínez, L.; Gorocica-Rosete, P.; Pérez-Padilla, R.; Domínguez-Reyes, C.A.; Tenorio-Torres, J.A.; Ornelas-Rebolledo, O.; Mehta, G.; Zamora-Mendoza, B.N.; Flores-Ramírez, R. Application of chemoresistive gas sensors and chemometric analysis to differentiate the fingerprints of global volatile organic compounds from diseases. Preliminary results of COPD, lung cancer and breast cancer. Clin. Chim. Acta 2021, 518, 83–92. Koureas, M.; Kirgou, P.; Amoutzias, G.; Hadjichristodoulou, C.; Gourgoulianis, K.; Tsakalof, A. Target analysis of volatile organic compounds in exhaled breath for lung cancer discrimination from other pulmonary diseases and healthy persons. Metabolites 2020, 10, 317.
- Ionescu, R.; Broza, Y.; Shaltieli, H.; Sadeh, D.; Zilberman, Y.; Feng, X.; Glass-Marmor, L.; Lejbkowicz, I.; Müllen, K.; Miller, A.; et al. Detection of multiple sclerosis from exhaled breath using bilayers of polycyclic aromatic hydrocarbons and single-wall carbon nanotubes. ACS Chem. Neurosci. 2011, 2, 687–693. Rodríguez-Aguilar, M.; Díaz de León-Martínez, L.; Gorocica-Rosete, P.; Pérez-Padilla, R.; Domínguez-Reyes, C.A.; Tenorio-Torres, J.A.; Ornelas-Rebolledo, O.; Mehta, G.; Zamora-Mendoza, B.N.; Flores-Ramírez, R. Application of chemoresistive gas sensors and chemometric analysis to differentiate the fingerprints of global volatile organic compounds from diseases. Preliminary results of COPD, lung cancer and breast cancer. Clin. Chim. Acta 2021, 518, 83–92.
- Tisch, U.; Schlesinger, I.; Ionescu, R.; Nassar, M.; Axelrod, N.; Robertman, D.; Tessler, Y.; Azar, F.; Marmur, A.; Aharon-Peretz, J.; et al. Detection of Alzheimer’s and Parkinson’s disease from exhaled breath using nanomaterial-based sensors. Nanomedicine 2013, 8, 43–56. Ionescu, R.; Broza, Y.; Shaltieli, H.; Sadeh, D.; Zilberman, Y.; Feng, X.; Glass-Marmor, L.; Lejbkowicz, I.; Müllen, K.; Miller, A.; et al. Detection of multiple sclerosis from exhaled breath using bilayers of polycyclic aromatic hydrocarbons and single-wall carbon nanotubes. ACS Chem. Neurosci. 2011, 2, 687–693.
- Nakhleh, M.K.; Amal, H.; Jeries, R.; Broza, Y.Y.; Aboud, M.; Gharra, A.; Ivgi, H.; Khatib, S.; Badarneh, S.; Har-Shai, L.; et al. Diagnosis and Classification of 17 Diseases from 1404 Subjects via Pattern Analysis of Exhaled Molecules. ACS Nano 2017, 11, 112–125. Tisch, U.; Schlesinger, I.; Ionescu, R.; Nassar, M.; Axelrod, N.; Robertman, D.; Tessler, Y.; Azar, F.; Marmur, A.; Aharon-Peretz, J.; et al. Detection of Alzheimer’s and Parkinson’s disease from exhaled breath using nanomaterial-based sensors. Nanomedicine 2013, 8, 43–56.
- Welearegay, T.G.; Diouani, M.F.; Österlund, L.; Ionescu, F.; Belgacem, K.; Smadhi, H.; Khaled, S.; Kidar, A.; Cindemir, U.; Laouini, D.; et al. Ligand-Capped Ultrapure Metal Nanoparticle Sensors for the Detection of Cutaneous Leishmaniasis Disease in Exhaled Breath. ACS Sens. 2018, 3, 2532–2540. Nakhleh, M.K.; Amal, H.; Jeries, R.; Broza, Y.Y.; Aboud, M.; Gharra, A.; Ivgi, H.; Khatib, S.; Badarneh, S.; Har-Shai, L.; et al. Diagnosis and Classification of 17 Diseases from 1404 Subjects via Pattern Analysis of Exhaled Molecules. ACS Nano 2017, 11, 112–125.
- Freddi, S.; Emelianov, A.V.; Bobrinetskiy, I.I.; Drera, G.; Pagliara, S.; Kopylova, D.S.; Chiesa, M.; Santini, G.; Mores, N.; Moscato, U.; et al. Development of a Sensing Array for Human Breath Analysis Based on SWCNT Layers Functionalized with Semiconductor Organic Molecules. Adv. Healthc. Mater. 2020, 9, 2000377. Welearegay, T.G.; Diouani, M.F.; Österlund, L.; Ionescu, F.; Belgacem, K.; Smadhi, H.; Khaled, S.; Kidar, A.; Cindemir, U.; Laouini, D.; et al. Ligand-Capped Ultrapure Metal Nanoparticle Sensors for the Detection of Cutaneous Leishmaniasis Disease in Exhaled Breath. ACS Sens. 2018, 3, 2532–2540.
- Choi, S.J.; Ku, K.H.; Kim, B.J.; Kim, I.D. Novel Templating Route Using Pt Infiltrated Block Copolymer Microparticles for Catalytic Pt Functionalized Macroporous WO3 Nanofibers and Its Application in Breath Pattern Recognition. ACS Sens. 2016, 1, 1124–1131. Freddi, S.; Emelianov, A.V.; Bobrinetskiy, I.I.; Drera, G.; Pagliara, S.; Kopylova, D.S.; Chiesa, M.; Santini, G.; Mores, N.; Moscato, U.; et al. Development of a Sensing Array for Human Breath Analysis Based on SWCNT Layers Functionalized with Semiconductor Organic Molecules. Adv. Healthc. Mater. 2020, 9, 2000377.
- Chang, J.; Lee, D.; Ban, S.; Oh, J.; Youn, M.; Kim, S.; Park, S.; Persaud, K.; Jheon, S. Chemical Analysis of volatile organic compounds in exhaled breath for lung cancer diagnosis using a sensor system. Sens. Actuators B. Chem. 2018, 255, 800–807. Choi, S.J.; Ku, K.H.; Kim, B.J.; Kim, I.D. Novel Templating Route Using Pt Infiltrated Block Copolymer Microparticles for Catalytic Pt Functionalized Macroporous WO3 Nanofibers and Its Application in Breath Pattern Recognition. ACS Sens. 2016, 1, 1124–1131.
- Binson, V.A.; Subramoniam, M.; Mathew, L. Discrimination of COPD and lung cancer from controls through breath analysis using a self-developed e-nose. J. Breath Res. 2021, 15, 46003. Chang, J.; Lee, D.; Ban, S.; Oh, J.; Youn, M.; Kim, S.; Park, S.; Persaud, K.; Jheon, S. Chemical Analysis of volatile organic compounds in exhaled breath for lung cancer diagnosis using a sensor system. Sens. Actuators B. Chem. 2018, 255, 800–807.
- Shehada, N.; Brönstrup, G.; Funka, K.; Christiansen, S.; Leja, M.; Haick, H. Ultrasensitive silicon nanowire for real-world gas sensing: Noninvasive diagnosis of cancer from breath volatolome. Nano Lett. 2015, 15, 1288–1295. Binson, V.A.; Subramoniam, M.; Mathew, L. Discrimination of COPD and lung cancer from controls through breath analysis using a self-developed e-nose. J. Breath Res. 2021, 15, 46003.
- Obermeier, J.; Trefz, P.; Wex, K.; Sabel, B.; Schubert, J.K.; Miekisch, W. Electrochemical sensor system for breath analysis of aldehydes, CO and NO. J. Breath Res. 2015, 9, 016008. Shehada, N.; Brönstrup, G.; Funka, K.; Christiansen, S.; Leja, M.; Haick, H. Ultrasensitive silicon nanowire for real-world gas sensing: Noninvasive diagnosis of cancer from breath volatolome. Nano Lett. 2015, 15, 1288–1295.
- Mazzone, P.J.; Wang, X.; Xu, Y. Exhaled Breath Analysis with a Colorimetric Sensor Array for the Identification and Characterization of Lung Cancer. J. Thorac. Oncol. 2012, 7, 137–142. Obermeier, J.; Trefz, P.; Wex, K.; Sabel, B.; Schubert, J.K.; Miekisch, W. Electrochemical sensor system for breath analysis of aldehydes, CO and NO. J. Breath Res. 2015, 9, 016008.
- Silva, L.I.B.; Freitas, A.C.; Rocha-Santos, T.A.P.; Pereira, M.E.; Duarte, A.C. Breath analysis by optical fiber sensor for the determination of exhaled organic compounds with a view to diagnostics. Talanta 2011, 83, 1586–1594. Mazzone, P.J.; Wang, X.; Xu, Y. Exhaled Breath Analysis with a Colorimetric Sensor Array for the Identification and Characterization of Lung Cancer. J. Thorac. Oncol. 2012, 7, 137–142.
- Chen, X.; Cao, M.; Li, Y.; Hu, W.; Wang, P.; Ying, K.; Pan, H. A study of an electronic nose for detection of lung cancer based on a virtual SAW gas sensors array and imaging recognition method. Meas. Sci. Technol. 2005, 16, 1535–1546. Silva, L.I.B.; Freitas, A.C.; Rocha-Santos, T.A.P.; Pereira, M.E.; Duarte, A.C. Breath analysis by optical fiber sensor for the determination of exhaled organic compounds with a view to diagnostics. Talanta 2011, 83, 1586–1594.
- Incalzi, R.A.; Pennazza, G.; Scarlata, S.; Santonico, M.; Petriaggi, M.; Chiurco, D.; Pedone, C.; D’Amico, A. Reproducibility and Respiratory Function Correlates of Exhaled Breath Fingerprint in Chronic Obstructive Pulmonary Disease. PLoS ONE 2012, 7, e45396. Chen, X.; Cao, M.; Li, Y.; Hu, W.; Wang, P.; Ying, K.; Pan, H. A study of an electronic nose for detection of lung cancer based on a virtual SAW gas sensors array and imaging recognition method. Meas. Sci. Technol. 2005, 16, 1535–1546.
- Montuschi, P.; Santonico, M.; Mondino, C. Diagnostic Performance of an Electronic Nose, Fractional Exhaled Nitric Oxide, and Lung Function Testing in Asthma. Chest 2010, 137, 790–796. Incalzi, R.A.; Pennazza, G.; Scarlata, S.; Santonico, M.; Petriaggi, M.; Chiurco, D.; Pedone, C.; D’Amico, A. Reproducibility and Respiratory Function Correlates of Exhaled Breath Fingerprint in Chronic Obstructive Pulmonary Disease. PLoS ONE 2012, 7, e45396.
- Santonico, M.; Lucantoni, G.; Pennazza, G.; Capuano, R.; Galluccio, G.; Roscioni, C.; La Delfa, G.; Consoli, D.; Martinelli, E.; Paolesse, R.; et al. In situ detection of lung cancer volatile fingerprints using bronchoscopic air-sampling. Lung Cancer 2012, 77, 46–50. Montuschi, P.; Santonico, M.; Mondino, C. Diagnostic Performance of an Electronic Nose, Fractional Exhaled Nitric Oxide, and Lung Function Testing in Asthma. Chest 2010, 137, 790–796.
- Gasparri, R.; Santonico, M.; Valentini, C.; Sedda, G.; Borri, A.; Petrella, F.; Maisonneuve, P.; Pennazza, G.; D’Amico, A.; Di Natale, C.; et al. Volatile signature for the early diagnosis of lung cancer. J. Breath Res. 2016, 10, 016007. Santonico, M.; Lucantoni, G.; Pennazza, G.; Capuano, R.; Galluccio, G.; Roscioni, C.; La Delfa, G.; Consoli, D.; Martinelli, E.; Paolesse, R.; et al. In situ detection of lung cancer volatile fingerprints using bronchoscopic air-sampling. Lung Cancer 2012, 77, 46–50.
- Zetola, N.M.; Modongo, C.; Matsiri, O.; Tamuhla, T.; Mbongwe, B.; Matlhagela, K.; Sepako, E.; Catini, A.; Sirugo, G.; Martinelli, E.; et al. Diagnosis of pulmonary tuberculosis and assessment of treatment response through analyses of volatile compound patterns in exhaled breath samples. J. Infect. 2017, 74, 367–376. Gasparri, R.; Santonico, M.; Valentini, C.; Sedda, G.; Borri, A.; Petrella, F.; Maisonneuve, P.; Pennazza, G.; D’Amico, A.; Di Natale, C.; et al. Volatile signature for the early diagnosis of lung cancer. J. Breath Res. 2016, 10, 016007.
- Pennazza, G.; Marchetti, E.; Santonico, M.; Mantini, G.; Mummolo, S.; Marzo, G.; Paolesse, R.; D’Amico, A.; Di Natale, C. Application of a quartz microbalance based gas sensor array for the study of halitosis. J. Breath Res. 2008, 2, 017009. Zetola, N.M.; Modongo, C.; Matsiri, O.; Tamuhla, T.; Mbongwe, B.; Matlhagela, K.; Sepako, E.; Catini, A.; Sirugo, G.; Martinelli, E.; et al. Diagnosis of pulmonary tuberculosis and assessment of treatment response through analyses of volatile compound patterns in exhaled breath samples. J. Infect. 2017, 74, 367–376.
- Fasola, S.; Ferrante, G.; Sabatini, A.; Santonico, M.; Zompanti, A.; Grasso, S.; Antonelli Incalzi, R.; La Grutta, S. Repeatability of exhaled breath fingerprint collected by a modern sampling system in asthmatic and healthy children. J. Breath Res. 2019, 13, 036007. Pennazza, G.; Marchetti, E.; Santonico, M.; Mantini, G.; Mummolo, S.; Marzo, G.; Paolesse, R.; D’Amico, A.; Di Natale, C. Application of a quartz microbalance based gas sensor array for the study of halitosis. J. Breath Res. 2008, 2, 017009.
- Castro, M.; Kumar, B.; Feller, J.F.; Haddi, Z.; Amari, A.; Bouchikhi, B. Chemical Novel e-nose for the discrimination of volatile organic biomarkers with an array of carbon nanotubes (CNT) conductive polymer nanocomposites (CPC) sensors. Sens. Actuators B. Chem. 2011, 159, 213–219. Fasola, S.; Ferrante, G.; Sabatini, A.; Santonico, M.; Zompanti, A.; Grasso, S.; Antonelli Incalzi, R.; La Grutta, S. Repeatability of exhaled breath fingerprint collected by a modern sampling system in asthmatic and healthy children. J. Breath Res. 2019, 13, 036007.
- Liu, B.; Huang, Y.; Kam, K.W.; Cheung, W.F.; Zhao, N.; Zheng, B. Functionalized graphene-based chemiresistive electronic nose for discrimination of disease-related volatile organic compounds. Biosens. Bioelectron. X 2019, 1, 100016. Castro, M.; Kumar, B.; Feller, J.F.; Haddi, Z.; Amari, A.; Bouchikhi, B. Chemical Novel e-nose for the discrimination of volatile organic biomarkers with an array of carbon nanotubes (CNT) conductive polymer nanocomposites (CPC) sensors. Sens. Actuators B. Chem. 2011, 159, 213–219.
- Xu, Y.; Lee, H.; Hu, Y.; Huang, J.; Kim, S.; Yun, M. Detection and identification of breast cancer volatile organic compounds biomarkers using highly-sensitive single nanowire array on a chip. J. Biomed. Nanotechnol. 2013, 9, 1164–1172. Liu, B.; Huang, Y.; Kam, K.W.; Cheung, W.F.; Zhao, N.; Zheng, B. Functionalized graphene-based chemiresistive electronic nose for discrimination of disease-related volatile organic compounds. Biosens. Bioelectron. X 2019, 1, 100016.
- Kim, G.S.; Park, Y.; Shin, J.; Song, Y.G.; Kang, C.Y. Metal oxide nanorods-based sensor array for selective detection of biomarker gases. Sensors 2021, 21, 1922. Xu, Y.; Lee, H.; Hu, Y.; Huang, J.; Kim, S.; Yun, M. Detection and identification of breast cancer volatile organic compounds biomarkers using highly-sensitive single nanowire array on a chip. J. Biomed. Nanotechnol. 2013, 9, 1164–1172.
- Koo, W.T.; Choi, S.J.; Kim, N.H.; Jang, J.S.; Kim, I.D. Catalyst-decorated hollow WO3 nanotubes using layer-by-layer self-assembly on polymeric nanofiber templates and their application in exhaled breath sensor. Sens. Actuators B Chem. 2016, 223, 301–310. Kim, G.S.; Park, Y.; Shin, J.; Song, Y.G.; Kang, C.Y. Metal oxide nanorods-based sensor array for selective detection of biomarker gases. Sensors 2021, 21, 1922.
- Choi, S.J.; Fuchs, F.; Demadrille, R.; Grévin, B.; Jang, B.H.; Lee, S.J.; Lee, J.H.; Tuller, H.L.; Kim, I.D. Fast responding exhaled-breath sensors using WO3 hemitubes functionalized by graphene-based electronic sensitizers for diagnosis of diseases. ACS Appl. Mater. Interfaces 2014, 6, 9061–9070. Koo, W.T.; Choi, S.J.; Kim, N.H.; Jang, J.S.; Kim, I.D. Catalyst-decorated hollow WO3 nanotubes using layer-by-layer self-assembly on polymeric nanofiber templates and their application in exhaled breath sensor. Sens. Actuators B Chem. 2016, 223, 301–310.
- Zhang, Y.; Gao, G.; Liu, H.; Fu, H.; Fan, J.; Wang, K.; Chen, Y.; Li, B.; Zhang, C.; Zhi, X.; et al. Identification of volatile biomarkers of gastric cancer cells and ultrasensitive electrochemical detection based on sensing interface of Au-Ag alloy coated MWCNTs. Theranostics 2014, 4, 154–162. Choi, S.J.; Fuchs, F.; Demadrille, R.; Grévin, B.; Jang, B.H.; Lee, S.J.; Lee, J.H.; Tuller, H.L.; Kim, I.D. Fast responding exhaled-breath sensors using WO3 hemitubes functionalized by graphene-based electronic sensitizers for diagnosis of diseases. ACS Appl. Mater. Interfaces 2014, 6, 9061–9070.
- Gautam, V.; Kumar, A.; Kumar, R.; Jain, V.K.; Nagpal, S. Silicon nanowires/reduced graphene oxide nanocomposite based novel sensor platform for detection of cyclohexane and formaldehyde. Mater. Sci. Semicond. Process. 2021, 123, 105571. Zhang, Y.; Gao, G.; Liu, H.; Fu, H.; Fan, J.; Wang, K.; Chen, Y.; Li, B.; Zhang, C.; Zhi, X.; et al. Identification of volatile biomarkers of gastric cancer cells and ultrasensitive electrochemical detection based on sensing interface of Au-Ag alloy coated MWCNTs. Theranostics 2014, 4, 154–162.
- Kus, F.; Altinkok, C.; Zayim, E.; Erdemir, S.; Tasaltin, C.; Gurol, I. Surface acoustic wave (SAW) sensor for volatile organic compounds (VOCs) detection with calixarene functionalized Gold nanorods (AuNRs) and silver nanocubes (AgNCs). Sens. Actuators B Chem. 2021, 330, 129402. Gautam, V.; Kumar, A.; Kumar, R.; Jain, V.K.; Nagpal, S. Silicon nanowires/reduced graphene oxide nanocomposite based novel sensor platform for detection of cyclohexane and formaldehyde. Mater. Sci. Semicond. Process. 2021, 123, 105571.
- Bahos, F.A.; Sainz-Vidal, A.; Sánchez-Pérez, C.; Saniger, J.M.; Gràcia, I.; Saniger-Alba, M.M.; Matatagui, D. ZIF nanocrystal-based surface acousticwave (SAW) electronic nose to detect diabetes in human breath. Biosensors 2019, 9, 4. Kus, F.; Altinkok, C.; Zayim, E.; Erdemir, S.; Tasaltin, C.; Gurol, I. Surface acoustic wave (SAW) sensor for volatile organic compounds (VOCs) detection with calixarene functionalized Gold nanorods (AuNRs) and silver nanocubes (AgNCs). Sens. Actuators B Chem. 2021, 330, 129402.
- Tao, W.; Lin, P.; Ai, Y.; Wang, H.; Ke, S.; Zeng, X. Multichannel quartz crystal microbalance array: Fabrication, evaluation, application in biomarker detection. Anal. Biochem. 2016, 494, 85–92. Bahos, F.A.; Sainz-Vidal, A.; Sánchez-Pérez, C.; Saniger, J.M.; Gràcia, I.; Saniger-Alba, M.M.; Matatagui, D. ZIF nanocrystal-based surface acousticwave (SAW) electronic nose to detect diabetes in human breath. Biosensors 2019, 9, 4.
- Zhao, S.; Lei, J.; Huo, D.; Hou, C.; Luo, X.; Wu, H.; Fa, H.; Yang, M. Chemical A colorimetric detector for lung cancer related volatile organic compounds based on cross-response mechanism. Sens. Actuators B. Chem. 2018, 256, 543–552. Tao, W.; Lin, P.; Ai, Y.; Wang, H.; Ke, S.; Zeng, X. Multichannel quartz crystal microbalance array: Fabrication, evaluation, application in biomarker detection. Anal. Biochem. 2016, 494, 85–92.
- Huo, D.; Xu, Y.; Hou, C.; Yang, M.; Fa, H. A novel optical chemical sensor based AuNR-MTPP and dyes for lung cancer biomarkers in exhaled breath identification. Sens. Actuators B Chem. 2014, 199, 446–456. Zhao, S.; Lei, J.; Huo, D.; Hou, C.; Luo, X.; Wu, H.; Fa, H.; Yang, M. Chemical A colorimetric detector for lung cancer related volatile organic compounds based on cross-response mechanism. Sens. Actuators B. Chem. 2018, 256, 543–552.
- Dragonieri, S.; Annema, J.T.; Schot, R.; Van Der Schee, M.P.C.; Spanevello, A.; Carratú, P.; Resta, O.; Rabe, K.F.; Sterk, P.J. An electronic nose in the discrimination of patients with non-small cell lung cancer and COPD. Lung Cancer 2009, 64, 166–170. Huo, D.; Xu, Y.; Hou, C.; Yang, M.; Fa, H. A novel optical chemical sensor based AuNR-MTPP and dyes for lung cancer biomarkers in exhaled breath identification. Sens. Actuators B Chem. 2014, 199, 446–456.
- Tirzite, M.; Bukovskis, M.; Strazda, G.; Jurka, N.; Taivans, I. Detection of lung cancer in exhaled breath with an electronic nose using support vector machine analysis. J. Breath Res. 2017, 11, 036009. Dragonieri, S.; Annema, J.T.; Schot, R.; Van Der Schee, M.P.C.; Spanevello, A.; Carratú, P.; Resta, O.; Rabe, K.F.; Sterk, P.J. An electronic nose in the discrimination of patients with non-small cell lung cancer and COPD. Lung Cancer 2009, 64, 166–170.
- Tirzïte, M.; Bukovskis, M.; Strazda, G.; Jurka, N.; Taivans, I. Detection of lung cancer with electronic nose and logistic regression analysis. J. Breath Res. 2019, 13, 016006. Tirzite, M.; Bukovskis, M.; Strazda, G.; Jurka, N.; Taivans, I. Detection of lung cancer in exhaled breath with an electronic nose using support vector machine analysis. J. Breath Res. 2017, 11, 036009.
- Fens, N.; Zwinderman, A.H.; Van Der Schee, M.P.; De Nijs, S.B.; Dijkers, E.; Roldaan, A.C.; Cheung, D.; Bel, E.H.; Sterk, P.J. Exhaled Breath Profiling Enables Discrimination of Chronic Obstructive Pulmonary Disease and Asthma. Am. J. Respir. Crit. Care Med. 2009, 180, 1076–1082. Tirzïte, M.; Bukovskis, M.; Strazda, G.; Jurka, N.; Taivans, I. Detection of lung cancer with electronic nose and logistic regression analysis. J. Breath Res. 2019, 13, 016006.
- Fens, N.; Roldaan, A.C.; van der Schee, M.P.; Boksem, R.J.; Zwinderman, A.H.; Bel, E.H.; Sterk, P.J. External validation of exhaled breath profiling using an electronic nose in the discrimination of asthma with fixed airways obstruction and chronic obstructive pulmonary disease. Clin. Exp. Allergy 2011, 41, 1371–1378. Fens, N.; Zwinderman, A.H.; Van Der Schee, M.P.; De Nijs, S.B.; Dijkers, E.; Roldaan, A.C.; Cheung, D.; Bel, E.H.; Sterk, P.J. Exhaled Breath Profiling Enables Discrimination of Chronic Obstructive Pulmonary Disease and Asthma. Am. J. Respir. Crit. Care Med. 2009, 180, 1076–1082.
- Dragonieri, S.; Scioscia, G.; Quaranta, V.N.; Carratu, P.; Venuti, M.P.; Falcone, M.; Carpagnano, G.E.; Foschino Barbaro, M.P.; Resta, O.; Lacedonia, D. Exhaled volatile organic compounds analysis by e-nose can detect idiopathic pulmonary fibrosis. J. Breath Res. 2020, 14, 047101. Fens, N.; Roldaan, A.C.; van der Schee, M.P.; Boksem, R.J.; Zwinderman, A.H.; Bel, E.H.; Sterk, P.J. External validation of exhaled breath profiling using an electronic nose in the discrimination of asthma with fixed airways obstruction and chronic obstructive pulmonary disease. Clin. Exp. Allergy 2011, 41, 1371–1378.
- Fielding, D.; Hartel, G.; Pass, D.; Davis, M.; Brown, M.; Dent, A.; Agnew, J.; Dickie, G.; Ware, R.S.; Hodge, R. Volatile organic compound breath testing detects in-situ squamous cell carcinoma of bronchial and laryngeal regions and shows distinct profiles of each tumour. J. Breath Res. 2020, 14, 046013. Dragonieri, S.; Scioscia, G.; Quaranta, V.N.; Carratu, P.; Venuti, M.P.; Falcone, M.; Carpagnano, G.E.; Foschino Barbaro, M.P.; Resta, O.; Lacedonia, D. Exhaled volatile organic compounds analysis by e-nose can detect idiopathic pulmonary fibrosis. J. Breath Res. 2020, 14, 047101.
- Bach, J.P.; Gold, M.; Mengel, D.; Hattesohl, A.; Lubbe, D.; Schmid, S.; Tackenberg, B.; Rieke, J.; Maddula, S.; Baumbach, J.I.; et al. Measuring compounds in exhaled air to detect Alzheimer’s disease and Parkinson’s disease. PLoS ONE 2015, 10, e0132227. Fielding, D.; Hartel, G.; Pass, D.; Davis, M.; Brown, M.; Dent, A.; Agnew, J.; Dickie, G.; Ware, R.S.; Hodge, R. Volatile organic compound breath testing detects in-situ squamous cell carcinoma of bronchial and laryngeal regions and shows distinct profiles of each tumour. J. Breath Res. 2020, 14, 046013.
- Krauss, E.; Haberer, J.; Maurer, O.; Barreto, G.; Drakopanagiotakis, F.; Degen, M.; Seeger, W.; Guenther, A. Exploring the ability of electronic nose technology to recognize interstitial lung diseases (ILD) by non-invasive breath screening of exhaled volatile compounds (VOC): A pilot study from the european ipf registry (eurIPFreg) and biobank. J. Clin. Med. 2019, 8, 1698. Bach, J.P.; Gold, M.; Mengel, D.; Hattesohl, A.; Lubbe, D.; Schmid, S.; Tackenberg, B.; Rieke, J.; Maddula, S.; Baumbach, J.I.; et al. Measuring compounds in exhaled air to detect Alzheimer’s disease and Parkinson’s disease. PLoS ONE 2015, 10, e0132227.
- Bannier, M.A.G.E.; Van De Kant, K.D.G.; Jöbsis, Q.; Dompeling, E. Feasibility and diagnostic accuracy of an electronic nose in children with asthma and cystic fibrosis. J. Breath Res. 2019, 13, 36009. Krauss, E.; Haberer, J.; Maurer, O.; Barreto, G.; Drakopanagiotakis, F.; Degen, M.; Seeger, W.; Guenther, A. Exploring the ability of electronic nose technology to recognize interstitial lung diseases (ILD) by non-invasive breath screening of exhaled volatile compounds (VOC): A pilot study from the european ipf registry (eurIPFreg) and biobank. J. Clin. Med. 2019, 8, 1698.
- van Hooren, M.R.A.; Leunis, N.; Brandsma, D.S.; Dingemans, A.M.C.; Kremer, B.; Kross, K.W. Differentiating head and neck carcinoma from lung carcinoma with an electronic nose: A proof of concept study. Eur. Arch. Oto-Rhino-Laryngol. 2016, 273, 3897–3903. Bannier, M.A.G.E.; Van De Kant, K.D.G.; Jöbsis, Q.; Dompeling, E. Feasibility and diagnostic accuracy of an electronic nose in children with asthma and cystic fibrosis. J. Breath Res. 2019, 13, 36009.
- van de Goor, R.M.G.E.; Leunis, N.; van Hooren, M.R.A.; Francisca, E.; Masclee, A.; Kremer, B.; Kross, K.W. Feasibility of electronic nose technology for discriminating between head and neck, bladder, and colon carcinomas. Eur. Arch. Oto-Rhino-Laryngol. 2017, 274, 1053–1060. van Hooren, M.R.A.; Leunis, N.; Brandsma, D.S.; Dingemans, A.M.C.; Kremer, B.; Kross, K.W. Differentiating head and neck carcinoma from lung carcinoma with an electronic nose: A proof of concept study. Eur. Arch. Oto-Rhino-Laryngol. 2016, 273, 3897–3903.
- De Vries, R.; Brinkman, P.; Van Der Schee, M.P.; Fens, N.; Dijkers, E.; Bootsma, S.K.; De Jongh, F.H.C.; Sterk, P.J. Integration of electronic nose technology with spirometry: Validation of a new approach for exhaled breath analysis. J. Breath Res. 2015, 9, 046001. van de Goor, R.M.G.E.; Leunis, N.; van Hooren, M.R.A.; Francisca, E.; Masclee, A.; Kremer, B.; Kross, K.W. Feasibility of electronic nose technology for discriminating between head and neck, bladder, and colon carcinomas. Eur. Arch. Oto-Rhino-Laryngol. 2017, 274, 1053–1060.
- Moor, C.C.; Oppenheimer, J.C.; Nakshbandi, G.; Aerts, J.G.J.V.; Brinkman, P.; Maitland-Van Der Zee, A.H.; Wijsenbeek, M.S. Exhaled breath analysis by use of eNose technology: A novel diagnostic tool for interstitial lung disease. Eur. Respir. J. 2021, 57, 2002042. De Vries, R.; Brinkman, P.; Van Der Schee, M.P.; Fens, N.; Dijkers, E.; Bootsma, S.K.; De Jongh, F.H.C.; Sterk, P.J. Integration of electronic nose technology with spirometry: Validation of a new approach for exhaled breath analysis. J. Breath Res. 2015, 9, 046001.
- Tan, J.L.; Yong, Z.X.; Liam, C.K. Using a chemiresistor-based alkane sensor to distinguish exhaled breaths of lung cancer patients from subjects with no lung cancer. J. Thorac. Dis. 2016, 8, 2772–2783. Moor, C.C.; Oppenheimer, J.C.; Nakshbandi, G.; Aerts, J.G.J.V.; Brinkman, P.; Maitland-Van Der Zee, A.H.; Wijsenbeek, M.S. Exhaled breath analysis by use of eNose technology: A novel diagnostic tool for interstitial lung disease. Eur. Respir. J. 2021, 57, 2002042.
- Binson, V.A.; Subramoniam, M.; Sunny, Y.; Mathew, L. Prediction of Pulmonary Diseases with Electronic Nose Using SVM and XGBoost. IEEE Sens. J. 2021, 21, 20886–20895. Tan, J.L.; Yong, Z.X.; Liam, C.K. Using a chemiresistor-based alkane sensor to distinguish exhaled breaths of lung cancer patients from subjects with no lung cancer. J. Thorac. Dis. 2016, 8, 2772–2783.
- Chen, K.; Liu, L.; Nie, B.; Lu, B.; Fu, L.; He, Z.; Li, W.; Pi, X.; Liu, H. Recognizing lung cancer and stages using a self-developed electronic nose system. Comput. Biol. Med. 2021, 131, 104294. Binson, V.A.; Subramoniam, M.; Sunny, Y.; Mathew, L. Prediction of Pulmonary Diseases with Electronic Nose Using SVM and XGBoost. IEEE Sens. J. 2021, 21, 20886–20895.
- Shlomi, D.; Abud, M.; Liran, O.; Bar, J.; Gai-Mor, N.; Ilouze, M.; Onn, A.; Ben-Nun, A.; Haick, H.; Peled, N. Detection of Lung Cancer and EGFR Mutation by Electronic Nose System. J. Thorac. Oncol. 2017, 12, 1544–1551. Chen, K.; Liu, L.; Nie, B.; Lu, B.; Fu, L.; He, Z.; Li, W.; Pi, X.; Liu, H. Recognizing lung cancer and stages using a self-developed electronic nose system. Comput. Biol. Med. 2021, 131, 104294.
- Barash, O.; Zhang, W.; Halpern, J.M.; Hua, Q.L.; Pan, Y.Y.; Kayal, H.; Khoury, K.; Liu, H.; Davies, M.P.A.; Haick, H. Differentiation between genetic mutations of breast cancer by breath volatolomics. Oncotarget 2015, 6, 44864–44876. Shlomi, D.; Abud, M.; Liran, O.; Bar, J.; Gai-Mor, N.; Ilouze, M.; Onn, A.; Ben-Nun, A.; Haick, H.; Peled, N. Detection of Lung Cancer and EGFR Mutation by Electronic Nose System. J. Thorac. Oncol. 2017, 12, 1544–1551.
- Amal, H.; Leja, M.; Funka, K.; Skapars, R.; Sivins, A.; Ancans, G.; Liepniece-karele, I.; Kikuste, I.; Lasina, I.; Haick, H. Detection of precancerous gastric lesions and gastric cancer through exhaled breath. Gut 2016, 65, 400–407. Barash, O.; Zhang, W.; Halpern, J.M.; Hua, Q.L.; Pan, Y.Y.; Kayal, H.; Khoury, K.; Liu, H.; Davies, M.P.A.; Haick, H. Differentiation between genetic mutations of breast cancer by breath volatolomics. Oncotarget 2015, 6, 44864–44876.
- Xu, Z.; Broza, Y.Y.; Ionsecu, R.; Tisch, U.; Ding, L.; Liu, H.; Song, Q.; Pan, Y.; Xiong, F.; Gu, K. A nanomaterial-based breath test for distinguishing gastric cancer from benign gastric conditions. Br. J. Cancer 2013, 108, 941–950. Amal, H.; Leja, M.; Funka, K.; Skapars, R.; Sivins, A.; Ancans, G.; Liepniece-karele, I.; Kikuste, I.; Lasina, I.; Haick, H. Detection of precancerous gastric lesions and gastric cancer through exhaled breath. Gut 2016, 65, 400–407.
- Shuster, G.; Gallimidi, Z.; Reiss, A.H.; Dovgolevsky, E.; Billan, S.; Abdah-Bortnyak, R.; Kuten, A.; Engel, A.; Shiban, A.; Tisch, U.; et al. Classification of breast cancer precursors through exhaled breath. Breast Cancer Res. Treat. 2011, 126, 791–796. Xu, Z.; Broza, Y.Y.; Ionsecu, R.; Tisch, U.; Ding, L.; Liu, H.; Song, Q.; Pan, Y.; Xiong, F.; Gu, K. A nanomaterial-based breath test for distinguishing gastric cancer from benign gastric conditions. Br. J. Cancer 2013, 108, 941–950.
- Karban, A.; Nakhleh, M.K.; Cancilla, J.C.; Vishinkin, R.; Rainis, T.; Koifman, E.; Jeries, R.; Ivgi, H.; Torrecilla, J.S.; Haick, H. Programmed Nanoparticles for Tailoring the Detection of Inflammatory Bowel Diseases and Irritable Bowel Syndrome Disease via Breathprint. Adv. Healthc. Mater. 2016, 5, 2339–2344. Shuster, G.; Gallimidi, Z.; Reiss, A.H.; Dovgolevsky, E.; Billan, S.; Abdah-Bortnyak, R.; Kuten, A.; Engel, A.; Shiban, A.; Tisch, U.; et al. Classification of breast cancer precursors through exhaled breath. Breast Cancer Res. Treat. 2011, 126, 791–796.
- Daniel, D.; Thangavel, K. Breathomics for gastric cancer classification using back-propagation neural network. J. Med. Signals Sens. 2016, 6, 172–182. Karban, A.; Nakhleh, M.K.; Cancilla, J.C.; Vishinkin, R.; Rainis, T.; Koifman, E.; Jeries, R.; Ivgi, H.; Torrecilla, J.S.; Haick, H. Programmed Nanoparticles for Tailoring the Detection of Inflammatory Bowel Diseases and Irritable Bowel Syndrome Disease via Breathprint. Adv. Healthc. Mater. 2016, 5, 2339–2344.
- Raspagliesi, F.; Bogani, G.; Benedetti, S.; Grassi, S.; Ferla, S.; Buratti, S. Detection of ovarian cancer through exhaled breath by electronic nose: A prospective study. Cancers 2020, 12, 2408. Daniel, D.; Thangavel, K. Breathomics for gastric cancer classification using back-propagation neural network. J. Med. Signals Sens. 2016, 6, 172–182.
- Lau, H.C.; Yu, J.B.; Lee, H.W.; Huh, J.S.; Lim, J.O. Investigation of exhaled breath samples from patients with Alzheimer’s disease using gas chromatography-mass spectrometry and an exhaled breath sensor system. Sensors 2017, 17, 1783. Raspagliesi, F.; Bogani, G.; Benedetti, S.; Grassi, S.; Ferla, S.; Buratti, S. Detection of ovarian cancer through exhaled breath by electronic nose: A prospective study. Cancers 2020, 12, 2408.
- Pennazza, G.; Santonico, M.; Vernile, C.; Antonelli Incalzi, R.; De Vincentis, A.; Picardi, A. Breathprinting of liver diseases. In Proceedings of the 2015 18th AISEM Annual Conference, Trento, Italy, 3–5 February 2015; pp. 1–3. Lau, H.C.; Yu, J.B.; Lee, H.W.; Huh, J.S.; Lim, J.O. Investigation of exhaled breath samples from patients with Alzheimer’s disease using gas chromatography-mass spectrometry and an exhaled breath sensor system. Sensors 2017, 17, 1783.
- Saidi, T.; Zaim, O.; Moufid, M.; El Bari, N.; Ionescu, R.; Bouchikhi, B. Exhaled breath analysis using electronic nose and gas chromatography–mass spectrometry for non-invasive diagnosis of chronic kidney disease, diabetes mellitus and healthy subjects. Sens. Actuators B Chem. 2018, 257, 178–188. Pennazza, G.; Santonico, M.; Vernile, C.; Antonelli Incalzi, R.; De Vincentis, A.; Picardi, A. Breathprinting of liver diseases. In Proceedings of the 2015 18th AISEM Annual Conference, Trento, Italy, 3–5 February 2015; pp. 1–3.
- Saidi, T.; Moufid, M.; de Jesus Beleño-Saenz, K.; Welearegay, T.G.; El Bari, N.; Lisset Jaimes-Mogollon, A.; Ionescu, R.; Bourkadi, J.E.; Benamor, J.; El Ftouh, M.; et al. Non-invasive prediction of lung cancer histological types through exhaled breath analysis by UV-irradiated electronic nose and GC/QTOF/MS. Sens. Actuators B Chem. 2020, 311, 127932. Saidi, T.; Zaim, O.; Moufid, M.; El Bari, N.; Ionescu, R.; Bouchikhi, B. Exhaled breath analysis using electronic nose and gas chromatography–mass spectrometry for non-invasive diagnosis of chronic kidney disease, diabetes mellitus and healthy subjects. Sens. Actuators B Chem. 2018, 257, 178–188.
- Brinkman, P.; van de Pol, M.A.; Gerritsen, M.G.; Bos, L.D.; Dekker, T.; Smids, B.S.; Sinha, A.; Majoor, C.J.; Sneeboer, M.M.; Knobel, H.H.; et al. Exhaled breath profiles in the monitoring of loss of control and clinical recovery in asthma. Clin. Exp. Allergy 2017, 47, 1159–1169. Saidi, T.; Moufid, M.; de Jesus Beleño-Saenz, K.; Welearegay, T.G.; El Bari, N.; Lisset Jaimes-Mogollon, A.; Ionescu, R.; Bourkadi, J.E.; Benamor, J.; El Ftouh, M.; et al. Non-invasive prediction of lung cancer histological types through exhaled breath analysis by UV-irradiated electronic nose and GC/QTOF/MS. Sens. Actuators B Chem. 2020, 311, 127932.
- Plaza, V.; Crespo, A.; Giner, J.; Merino, J.L.; Ramos-Barbón, D.; Mateus, E.F.; Torrego, A.; Cosio, B.G.; Agustí, A.; Sibila, O. Inflammatory asthma phenotype discrimination using an electronic nose breath analyzer. J. Investig. Allergol. Clin. Immunol. 2015, 25, 431–437. Brinkman, P.; van de Pol, M.A.; Gerritsen, M.G.; Bos, L.D.; Dekker, T.; Smids, B.S.; Sinha, A.; Majoor, C.J.; Sneeboer, M.M.; Knobel, H.H.; et al. Exhaled breath profiles in the monitoring of loss of control and clinical recovery in asthma. Clin. Exp. Allergy 2017, 47, 1159–1169.
- Farraia, M.; Rufo, J.C.; Paciência, I.; Castro, F.; Ana, M.; Rama, T.; Rocha, S.M.; Delgado, L.; Brinkman, P.; Moreira, A. Human volatilome analysis using eNose to assess uncontrolled asthma in a clinical setting. Allergy 2020, 75, 1630–1639. Plaza, V.; Crespo, A.; Giner, J.; Merino, J.L.; Ramos-Barbón, D.; Mateus, E.F.; Torrego, A.; Cosio, B.G.; Agustí, A.; Sibila, O. Inflammatory asthma phenotype discrimination using an electronic nose breath analyzer. J. Investig. Allergol. Clin. Immunol. 2015, 25, 431–437.
- Tenero, L.; Sandri, M.; Piazza, M.; Paiola, G.; Zaffanello, M.; Piacentini, G. Electronic nose in discrimination of children with uncontrolled asthma. J. Breath Res. 2020, 14, 046003. Farraia, M.; Rufo, J.C.; Paciência, I.; Castro, F.; Ana, M.; Rama, T.; Rocha, S.M.; Delgado, L.; Brinkman, P.; Moreira, A. Human volatilome analysis using eNose to assess uncontrolled asthma in a clinical setting. Allergy 2020, 75, 1630–1639.
- van der Vliet, A.; Janssen-Heininger, Y.M.W.; Anathy, V. Oxidative stress in chronic lung disease: From mitochondrial dysfunction to dysregulated redox signaling. Mol. Aspects Med. 2018, 63, 59–69. Tenero, L.; Sandri, M.; Piazza, M.; Paiola, G.; Zaffanello, M.; Piacentini, G. Electronic nose in discrimination of children with uncontrolled asthma. J. Breath Res. 2020, 14, 046003.
- Rodríguez-Aguilar, M.; Ramírez-García, S.; Ilizaliturri-Hernández, C.; Gómez-Gómez, A.; Van-Brussel, E.; Díaz-Barriga, F.; Medellín-Garibay, S.; Flores-Ramírez, R. Ultrafast gas chromatography coupled to electronic nose to identify volatile biomarkers in exhaled breath from chronic obstructive pulmonary disease patients: A pilot study. Biomed. Chromatogr. 2019, 33, e4684. van der Vliet, A.; Janssen-Heininger, Y.M.W.; Anathy, V. Oxidative stress in chronic lung disease: From mitochondrial dysfunction to dysregulated redox signaling. Mol. Aspects Med. 2018, 63, 59–69.
- Chen, X.; Muhammad, K.G.; Madeeha, C.; Fu, W.; Xu, L.; Hu, Y.; Liu, J.; Ying, K.; Chen, L.; Yurievna, G.O. Calculated indices of volatile organic compounds (VOCs) in exhalation for lung cancer screening and early detection. Lung Cancer 2021, 154, 197–205. Rodríguez-Aguilar, M.; Ramírez-García, S.; Ilizaliturri-Hernández, C.; Gómez-Gómez, A.; Van-Brussel, E.; Díaz-Barriga, F.; Medellín-Garibay, S.; Flores-Ramírez, R. Ultrafast gas chromatography coupled to electronic nose to identify volatile biomarkers in exhaled breath from chronic obstructive pulmonary disease patients: A pilot study. Biomed. Chromatogr. 2019, 33, e4684.
- Schleich, F.N.; Zanella, D.; Stefanuto, P.; Bessonov, K.; Smolinska, A.; Dallinga, J.W.; Henket, M.; Paulus, V.; Guissard, F.; Graff, S.; et al. Exhaled Volatile Organic Compounds Are Able to Discriminate between Neutrophilic and Eosinophilic Asthma. Am. J. Respir. Crit. Care Med. 2019, 200, 444–453. Chen, X.; Muhammad, K.G.; Madeeha, C.; Fu, W.; Xu, L.; Hu, Y.; Liu, J.; Ying, K.; Chen, L.; Yurievna, G.O. Calculated indices of volatile organic compounds (VOCs) in exhalation for lung cancer screening and early detection. Lung Cancer 2021, 154, 197–205.
- Stefanuto, P.H.; Zanella, D.; Vercammen, J.; Henket, M.; Schleich, F.; Louis, R.; Focant, J.F. Multimodal combination of GC×GC-HRTOFMS and SIFT-MS for asthma phenotyping using exhaled breath. Sci. Rep. 2020, 10, 16159. Schleich, F.N.; Zanella, D.; Stefanuto, P.; Bessonov, K.; Smolinska, A.; Dallinga, J.W.; Henket, M.; Paulus, V.; Guissard, F.; Graff, S.; et al. Exhaled Volatile Organic Compounds Are Able to Discriminate between Neutrophilic and Eosinophilic Asthma. Am. J. Respir. Crit. Care Med. 2019, 200, 444–453.
- Buszewski, B.; Ligor, T.; Jezierski, T.; Wenda-Piesik, A.; Walczak, M.; Rudnicka, J. Identification of volatile lung cancer markers by gas chromatography-mass spectrometry: Comparison with discrimination by canines. Anal. Bioanal. Chem. 2012, 404, 141–146. Stefanuto, P.H.; Zanella, D.; Vercammen, J.; Henket, M.; Schleich, F.; Louis, R.; Focant, J.F. Multimodal combination of GC×GC-HRTOFMS and SIFT-MS for asthma phenotyping using exhaled breath. Sci. Rep. 2020, 10, 16159.
- Fuchs, P.; Loeseken, C.; Schubert, J.K.; Miekisch, W. Breath gas aldehydes as biomarkers of lung cancer. Int. J. Cancer 2010, 126, 2663–2670. Buszewski, B.; Ligor, T.; Jezierski, T.; Wenda-Piesik, A.; Walczak, M.; Rudnicka, J. Identification of volatile lung cancer markers by gas chromatography-mass spectrometry: Comparison with discrimination by canines. Anal. Bioanal. Chem. 2012, 404, 141–146.
- Muñoz-Lucas, M.Á.; Jareño-Esteban, J.; Gutiérrez-Ortega, C.; López-Guijarro, P.; Collado-Yurrita, L.; Quintana-Díaz, M.; Callol-Sánchez, L. Influence of Chronic Obstructive Pulmonary Disease on Volatile Organic Compounds in Patients with Non-Small Cell Lung Cancer. Arch. Bronconeumol. 2020, 56, 801–805. Fuchs, P.; Loeseken, C.; Schubert, J.K.; Miekisch, W. Breath gas aldehydes as biomarkers of lung cancer. Int. J. Cancer 2010, 126, 2663–2670.
- Poli, D.; Carbognani, P.; Corradi, M.; Goldoni, M.; Acampa, O.; Balbi, B.; Bianchi, L.; Rusca, M.; Mutti, A. Exhaled volatile organic compounds in patients with non-small cell lung cancer: Cross sectional and nested short-term follow-up study. Respir. Res. 2005, 6, 71. Muñoz-Lucas, M.Á.; Jareño-Esteban, J.; Gutiérrez-Ortega, C.; López-Guijarro, P.; Collado-Yurrita, L.; Quintana-Díaz, M.; Callol-Sánchez, L. Influence of Chronic Obstructive Pulmonary Disease on Volatile Organic Compounds in Patients with Non-Small Cell Lung Cancer. Arch. Bronconeumol. 2020, 56, 801–805.
- Koureas, M.; Kalompatsios, D.; Amoutzias, G.D.; Hadjichristodoulou, C.; Gourgoulianis, K.; Tsakalof, A. Comparison of targeted and untargeted approaches in breath analysis for the discrimination of lung cancer from benign pulmonary diseases and healthy persons. Molecules 2021, 26, 2609. Poli, D.; Carbognani, P.; Corradi, M.; Goldoni, M.; Acampa, O.; Balbi, B.; Bianchi, L.; Rusca, M.; Mutti, A. Exhaled volatile organic compounds in patients with non-small cell lung cancer: Cross sectional and nested short-term follow-up study. Respir. Res. 2005, 6, 71.
- Zou, Y.; Zhang, X.; Chen, X.; Hu, Y.; Ying, K.; Wang, P. Optimization of volatile markers of lung cancer to exclude interferences of non-malignant disease. Cancer Biomarkers 2014, 14, 371–379. Koureas, M.; Kalompatsios, D.; Amoutzias, G.D.; Hadjichristodoulou, C.; Gourgoulianis, K.; Tsakalof, A. Comparison of targeted and untargeted approaches in breath analysis for the discrimination of lung cancer from benign pulmonary diseases and healthy persons. Molecules 2021, 26, 2609.
- Fu, X.-A.; Li, M.; Knipp, R.J.; Nantz, M.H.; Bousamra, M. Noninvasive detection of lung cancer using exhaled breath. Cancer Med. 2014, 3, 174–181. Zou, Y.; Zhang, X.; Chen, X.; Hu, Y.; Ying, K.; Wang, P. Optimization of volatile markers of lung cancer to exclude interferences of non-malignant disease. Cancer Biomarkers 2014, 14, 371–379.
- Ii, M.B.; Schumer, E.; Li, M.; Knipp, R.J.; Nantz, M.H.; Van Berkel, V.; Fu, X. Quantitative analysis of exhaled carbonyl compounds distinguishes benign from malignant pulmonary disease. J. Thorac. Cardiovasc. Surg. 2014, 148, 1074–1081. Fu, X.-A.; Li, M.; Knipp, R.J.; Nantz, M.H.; Bousamra, M. Noninvasive detection of lung cancer using exhaled breath. Cancer Med. 2014, 3, 174–181.
- Li, M.; Yang, D.; Brock, G.; Knipp, R.J.; Bousamra, M.; Nantz, M.H.; Fu, X.A. Breath carbonyl compounds as biomarkers of lung cancer. Lung Cancer 2015, 90, 92–97. Ii, M.B.; Schumer, E.; Li, M.; Knipp, R.J.; Nantz, M.H.; Van Berkel, V.; Fu, X. Quantitative analysis of exhaled carbonyl compounds distinguishes benign from malignant pulmonary disease. J. Thorac. Cardiovasc. Surg. 2014, 148, 1074–1081.
- Ibrahim, B.; Basanta, M.; Cadden, P.; Singh, D.; Douce, D.; Woodcock, A.; Fowler, S.J. Non-invasive phenotyping using exhaled volatile organic compounds in asthma. Thorax 2011, 66, 804–809. Li, M.; Yang, D.; Brock, G.; Knipp, R.J.; Bousamra, M.; Nantz, M.H.; Fu, X.A. Breath carbonyl compounds as biomarkers of lung cancer. Lung Cancer 2015, 90, 92–97.
- Fens, N.; De Nijs, S.B.; Peters, S.; Dekker, T.; Knobel, H.H.; Vink, T.J.; Willard, N.P.; Zwinderman, A.H.; Krouwels, F.H.; Lutter, R.; et al. Exhaled air molecular profiling in relation to inflammatory subtype and activity in COPD. Eur. Respir. J. 2011, 38, 1301–1309. Ibrahim, B.; Basanta, M.; Cadden, P.; Singh, D.; Douce, D.; Woodcock, A.; Fowler, S.J. Non-invasive phenotyping using exhaled volatile organic compounds in asthma. Thorax 2011, 66, 804–809.
- Basanta, M.; Ibrahim, B.; Dockry, R.; Douce, D.; Morris, M.; Singh, D.; Woodcock, A.; Fowler, S.J. Exhaled volatile organic compounds for phenotyping chronic obstructive pulmonary disease: A cross-sectional study. Respir. Res. 2012, 13, 72. Fens, N.; De Nijs, S.B.; Peters, S.; Dekker, T.; Knobel, H.H.; Vink, T.J.; Willard, N.P.; Zwinderman, A.H.; Krouwels, F.H.; Lutter, R.; et al. Exhaled air molecular profiling in relation to inflammatory subtype and activity in COPD. Eur. Respir. J. 2011, 38, 1301–1309.
- Van Vliet, D.; Smolinska, A.; Jöbsis, Q.; Rosias, P.P.R.; Muris, J.W.M.; Dallinga, J.W.; Van Schooten, F.J.; Dompeling, E. Association between exhaled inflammatory markers and asthma control in children. J. Breath Res. 2016, 10, 016014. Basanta, M.; Ibrahim, B.; Dockry, R.; Douce, D.; Morris, M.; Singh, D.; Woodcock, A.; Fowler, S.J. Exhaled volatile organic compounds for phenotyping chronic obstructive pulmonary disease: A cross-sectional study. Respir. Res. 2012, 13, 72.
- Van Vliet, D.; Smolinska, A.; Jöbsis, Q.; Rosias, P.; Muris, J.; Dallinga, J.; Dompeling, E.; Van Schooten, F.J. Can exhaled volatile organic compounds predict asthma exacerbations in children? J. Breath Res. 2017, 11, 016016. Van Vliet, D.; Smolinska, A.; Jöbsis, Q.; Rosias, P.P.R.; Muris, J.W.M.; Dallinga, J.W.; Van Schooten, F.J.; Dompeling, E. Association between exhaled inflammatory markers and asthma control in children. J. Breath Res. 2016, 10, 016014.
- Robroeks, C.M.; Van Berkel, J.J.; Jöbsis, Q.; Van Schooten, F.J.; Dallinga, J.W.; Wouters, E.F.; Dompeling, E. Exhaled volatile organic compounds predict exacerbations of childhood asthma in a 1-year prospective study. Eur. Respir. J. 2013, 42, 98–106. Van Vliet, D.; Smolinska, A.; Jöbsis, Q.; Rosias, P.; Muris, J.; Dallinga, J.; Dompeling, E.; Van Schooten, F.J. Can exhaled volatile organic compounds predict asthma exacerbations in children? J. Breath Res. 2017, 11, 016016.
- Pizzini, A.; Filipiak, W.; Wille, J.; Ager, C.; Wiesenhofer, H.; Kubinec, R.; Blaško, J.; Tschurtschenthaler, C.; Mayhew, C.A.; Weiss, G.; et al. Analysis of volatile organic compounds in the breath of patients with stable or acute exacerbation of chronic obstructive pulmonary disease. J. Breath Res. 2018, 12, 036002. Robroeks, C.M.; Van Berkel, J.J.; Jöbsis, Q.; Van Schooten, F.J.; Dallinga, J.W.; Wouters, E.F.; Dompeling, E. Exhaled volatile organic compounds predict exacerbations of childhood asthma in a 1-year prospective study. Eur. Respir. J. 2013, 42, 98–106.
- D’Amico, A.; Pennazza, G.; Santonico, M.; Martinelli, E.; Roscioni, C.; Galluccio, G.; Paolesse, R.; Di Natale, C. An investigation on electronic nose diagnosis of lung cancer. Lung Cancer 2010, 68, 170–176. Pizzini, A.; Filipiak, W.; Wille, J.; Ager, C.; Wiesenhofer, H.; Kubinec, R.; Blaško, J.; Tschurtschenthaler, C.; Mayhew, C.A.; Weiss, G.; et al. Analysis of volatile organic compounds in the breath of patients with stable or acute exacerbation of chronic obstructive pulmonary disease. J. Breath Res. 2018, 12, 036002.
- Phillips, M.; Cataneo, R.N.; Ditkoff, B.A.; Fisher, P.; Greenberg, J.; Gunawardena, R.; Kwon, C.S.; Tietje, O.; Wong, C. Prediction of breast cancer using volatile biomarkers in the breath. Breast Cancer Res. Treat. 2006, 99, 19–21. D’Amico, A.; Pennazza, G.; Santonico, M.; Martinelli, E.; Roscioni, C.; Galluccio, G.; Paolesse, R.; Di Natale, C. An investigation on electronic nose diagnosis of lung cancer. Lung Cancer 2010, 68, 170–176.
- Tiele, A.; Daulton, E.; Ifeachor, E.; Eyre, V.; Clarke, S. Breath-based non-invasive diagnosis of Alzheimer ’ s disease : A pilot study Breath-based non-invasive diagnosis of Alzheimer’ s disease: A pilot study. J. Breath Res. 2020, 14, 026003. Phillips, M.; Cataneo, R.N.; Ditkoff, B.A.; Fisher, P.; Greenberg, J.; Gunawardena, R.; Kwon, C.S.; Tietje, O.; Wong, C. Prediction of breast cancer using volatile biomarkers in the breath. Breast Cancer Res. Treat. 2006, 99, 19–21.
- Das, S.; Pal, M. Review-Non-Invasive Monitoring of Human Health by Exhaled Breath Analysis: A Comprehensive Review. J. Electrochem. Soc. 2020, 167, 037562. Tiele, A.; Daulton, E.; Ifeachor, E.; Eyre, V.; Clarke, S. Breath-based non-invasive diagnosis of Alzheimer ’ s disease : A pilot study Breath-based non-invasive diagnosis of Alzheimer’ s disease: A pilot study. J. Breath Res. 2020, 14, 026003.
- Sakumura, Y.; Koyama, Y.; Tokutake, H.; Hida, T.; Sato, K.; Itoh, T.; Akamatsu, T.; Shin, W. Diagnosis by volatile organic compounds in exhaled breath from lung cancer patients using support vector machine algorithm. Sensors 2017, 17, 287. Das, S.; Pal, M. Review-Non-Invasive Monitoring of Human Health by Exhaled Breath Analysis: A Comprehensive Review. J. Electrochem. Soc. 2020, 167, 037562.
- Sakumura, Y.; Koyama, Y.; Tokutake, H.; Hida, T.; Sato, K.; Itoh, T.; Akamatsu, T.; Shin, W. Diagnosis by volatile organic compounds in exhaled breath from lung cancer patients using support vector machine algorithm. Sensors 2017, 17, 287.
