Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Conner Chen and Version 1 by Robert T. Streeper.
Lipids are hydrophobic or amphiphilic small molecules, including fatty acids and their derivatives, in particular esters, sterols, steroids and phospholipids.
- insulin resistance
- type 2 diabetes
- natural product
1. Introduction
Insulin resistance (IR) is clinically defined as the inability of a known quantity of exogenous or endogenous insulin to increase glucose uptake and its utilization in an individual as much as it does in a normal population [1]. IR is associated with conditions beyond prediabetes and type 2 diabetes (T2D), including cardiovascular and metabolic pathologies, collectively referred to as a metabolic syndrome, nonalcoholic fatty liver disease, as well as atherosclerosis, hypertension, polycystic ovarian syndrome and infectious diseases with sepsis sequelae [2,3,4,5,6,7,8][2][3][4][5][6][7][8]. The cause of IR is poorly understood, but the major contributors are thought to comprise systemic chronic inflammation [9] associated with abnormal lipid levels, oxidative and endoplasmic reticulum stress, insulin receptor mutations and mitochondrial dysfunctions [10].
2. Lipids
Lipids are hydrophobic or amphiphilic small molecules, including fatty acids and their derivatives, in particular esters, sterols, steroids and phospholipids. Azelaic acid (AZA) [28][11] and the best characterized azelate, diethyl azelate DEA [29][12], (Figure 1A) are both MAIMs. Yet, as can be expected from the large differences in respective water–octanol partitioning coefficients, the respective interactions of AZA and DEA with plasma membrane are quite different [30][13]. DEA is a membrane fluidizer that interacts with the plasma membrane and membrane proteins, while AZA, due to its lower lipophilicity, has limited membrane interactions [12,30][13][14].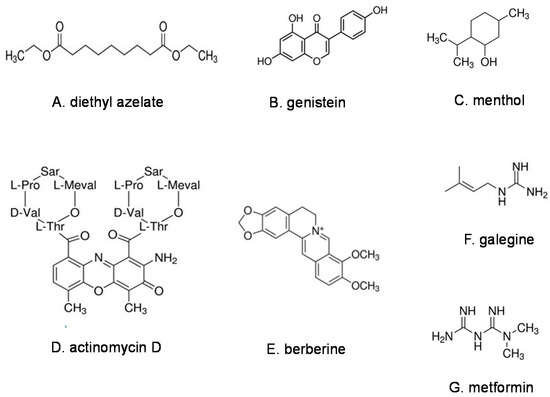
Figure 1. Representative natural products in the families of lipids (A), phenols (B), terpenes (C), antibiotics (D) and alkaloids (E–G).
References
- Lebovitz, H.E. Insulin resistance: Definition and consequences. Exp. Clin. Endocrinol. Diabetes 2001, 109 (Suppl. S2), S135–S148.
- Kouzi, S.A.; Yang, S.; Nuzum, D.S.; Dirks-Naylor, A.J. Natural supplements for improving insulin sensitivity and glucose uptake in skeletal muscle. Front. Biosci. 2015, 7, 94–106.
- Bugianesi, E.; Moscatiello, S.; Ciaravella, M.F.; Marchesini, G. Insulin resistance in nonalcoholic fatty liver disease. Curr. Pharm. Des. 2010, 16, 1941–1951.
- Affinati, A.H.; Wallia, A.; Gianchandani, R.Y. Severe hyperglycemia and insulin resistance in patients with SARS-CoV-2 infection: A report of two cases. Clin. Diabetes Endocrinol. 2021, 7, 8.
- Acquah, S.; Boampong, J.N.; Eghan Jnr, B.A.; Eriksson, M. Evidence of insulin resistance in adult uncomplicated malaria: Result of a two-year prospective study. Malar. Res. Treat. 2014, 2014, 136148.
- Van Cromphaut, S.J.; Vanhorebeek, I.; Van den Berghe, G. Glucose metabolism and insulin resistance in sepsis. Curr. Pharm. Des. 2008, 14, 1887–1899.
- Niang, F.; Sarfo, F.S.; Frimpong, M.; Guenin-Mace, L.; Wansbrough-Jones, M.; Stinear, T.; Phillips, R.O.; Demangel, C. Metabolomic profiles delineate mycolactone signature in Buruli ulcer disease. Sci. Rep. 2015, 5, 17693.
- Phillips, R.O.; Sarfo, F.S.; Landier, J.; Oldenburg, R.; Frimpong, M.; Wansbrough-Jones, M.; Abass, K.; Thompson, W.; Forson, M.; Fontanet, A.; et al. Combined inflammatory and metabolic defects reflected by reduced serum protein levels in patients with Buruli ulcer disease. PLoS Negl. Trop. Dis. 2014, 8, e2786.
- Hansson, E.; Skioldebrand, E. Coupled cell networks are target cells of inflammation, which can spread between different body organs and develop into systemic chronic inflammation. J. Inflamm. 2015, 12, 44.
- Yaribeygi, H.; Farrokhi, F.R.; Butler, A.E.; Sahebkar, A. Insulin resistance: Review of the underlying molecular mechanisms. J. Cell Physiol. 2019, 234, 8152–8161.
- Azelaic acid. Available online: https://pubchem.ncbi.nlm.nih.gov/#query=azelaic%20acid (accessed on 5 June 2023).
- Diethyl azelate. Available online: https://pubchem.ncbi.nlm.nih.gov/#query=diethyl%20azelate (accessed on 5 June 2023).
- Izbicka, E.; Streeper, R. Azelaic Acid Esters as Pluripotent Immunomodulatory Molecules: Nutritional Supplements or Drugs? Nutraceuticals 2021, 1, 42–53.
- Izbicka, E.; Streeper, R.T.; Louden, C. Adaptive Membrane Fluidity Modulation: A Feedback Regulated Homeostatic System and Target for Pharmacological Intervention. In Vivo 2021, 35, 3073–3095.
- Izbicka, E.; Streeper, R.T. Adaptive Membrane Fluidity Modulation: A Feedback Regulated Homeostatic System Hiding in Plain Sight. In Vivo 2021, 35, 2991–3000.
- Matsubara, T.; Tanaka, N.; Krausz, K.W.; Manna, S.K.; Kang, D.W.; Anderson, E.R.; Luecke, H.; Patterson, A.D.; Shah, Y.M.; Gonzalez, F.J. Metabolomics identifies an inflammatory cascade involved in dioxin- and diet-induced steatohepatitis. Cell Metab. 2012, 16, 634–644.
- Wu, C.; Hwang, S.H.; Jia, Y.; Choi, J.; Kim, Y.J.; Choi, D.; Pathiraja, D.; Choi, I.G.; Koo, S.H.; Lee, S.J. Olfactory receptor 544 reduces adiposity by steering fuel preference toward fats. J. Clin. Investig. 2017, 127, 4118–4123.
- Jung, H.W.; Tschaplinski, T.J.; Wang, L.; Glazebrook, J.; Greenberg, J.T. Priming in systemic plant immunity. Science 2009, 324, 89–91.
- Mastrofrancesco, A.; Ottaviani, M.; Aspite, N.; Cardinali, G.; Izzo, E.; Graupe, K.; Zouboulis, C.C.; Camera, E.; Picardo, M. Azelaic acid modulates the inflammatory response in normal human keratinocytes through PPARgamma activation. Exp. Dermatol. 2010, 19, 813–820.
- Muthulakshmi, S.; Saravanan, R. Protective effects of azelaic acid against high-fat diet-induced oxidative stress in liver, kidney and heart of C57BL/6J mice. Mol. Cell Biochem. 2013, 377, 23–33.
- Streeper, R.T.; Louden, C.; Izbicka, E. Oral Azelaic Acid Ester Decreases Markers of Insulin Resistance in Overweight Human Male Subjects. In Vivo 2020, 34, 1173–1186.
- Sebacic acid. Available online: https://pubchem.ncbi.nlm.nih.gov/#query=sebacic%20acid (accessed on 5 June 2023).
- Iaconelli, A.; Gastaldelli, A.; Chiellini, C.; Gniuli, D.; Favuzzi, A.; Binnert, C.; Mace, K.; Mingrone, G. Effect of oral sebacic Acid on postprandial glycemia, insulinemia, and glucose rate of appearance in type 2 diabetes. Diabetes Care 2010, 33, 2327–2332.
- Montgomery, M.K.; Turner, N. Mitochondrial dysfunction and insulin resistance: An update. Endocr. Connect. 2015, 4, R1–R15.
- Russell, A.P.; Foletta, V.C.; Snow, R.J.; Wadley, G.D. Skeletal muscle mitochondria: A major player in exercise, health and disease. Biochim. Biophys. Acta 2014, 1840, 1276–1284.
- Kang, N.; Bahk, Y.Y.; Lee, N.; Jae, Y.; Cho, Y.H.; Ku, C.R.; Byun, Y.; Lee, E.J.; Kim, M.S.; Koo, J. Olfactory receptor Olfr544 responding to azelaic acid regulates glucagon secretion in alpha-cells of mouse pancreatic islets. Biochem. Biophys. Res. Commun. 2015, 460, 616–621.
- Thach, T.T.; Wu, C.; Hwang, K.Y.; Lee, S.J. Azelaic Acid Induces Mitochondrial Biogenesis in Skeletal Muscle by Activation of Olfactory Receptor 544. Front. Physiol. 2020, 11, 329.
- Thach, T.T.; Hong, Y.J.; Lee, S.; Lee, S.J. Molecular determinants of the olfactory receptor Olfr544 activation by azelaic acid. Biochem. Biophys. Res. Commun. 2017, 485, 241–248.
- Muthulakshmi, S.; Chakrabarti, A.K.; Mukherjee, S. Gene expression profile of high-fat diet-fed C57BL/6J mice: In search of potential role of azelaic acid. J. Physiol. Biochem. 2015, 71, 29–42.
- Wu, C.; Jeong, M.Y.; Kim, J.Y.; Lee, G.; Kim, J.S.; Cheong, Y.E.; Kang, H.; Cho, C.H.; Kim, J.; Park, M.K.; et al. Activation of ectopic olfactory receptor 544 induces GLP-1 secretion and regulates gut inflammation. Gut Microbes 2021, 13, 1987782.
- Nachawi, N.; Rao, P.P.; Makin, V. The role of GLP-1 receptor agonists in managing type 2 diabetes. Cleve. Clin. J. Med. 2022, 89, 457–464.
- Lim, G.E.; Huang, G.J.; Flora, N.; LeRoith, D.; Rhodes, C.J.; Brubaker, P.L. Insulin regulates glucagon-like peptide-1 secretion from the enteroendocrine L cell. Endocrinology 2009, 150, 580–591.
- Phosphatidylcholine. Available online: https://pubchem.ncbi.nlm.nih.gov/#query=phosphatidylcholine&sort=cid&page=1 (accessed on 5 June 2023).
- Phosphatidylethanolamine. Available online: https://pubchem.ncbi.nlm.nih.gov/#query=%22Phosphatidylethanolamine%20%5BWHO-DD%5D%22 (accessed on 5 June 2023).
- Zhao, J.; Zhu, Y.; Hyun, N.; Zeng, D.; Uppal, K.; Tran, V.T.; Yu, T.; Jones, D.; He, J.; Lee, E.T.; et al. Novel metabolic markers for the risk of diabetes development in American Indians. Diabetes Care 2015, 38, 220–227.
- Newsom, S.A.; Brozinick, J.T.; Kiseljak-Vassiliades, K.; Strauss, A.N.; Bacon, S.D.; Kerege, A.A.; Bui, H.H.; Sanders, P.; Siddall, P.; Wei, T.; et al. Skeletal muscle phosphatidylcholine and phosphatidylethanolamine are related to insulin sensitivity and respond to acute exercise in humans. J. Appl. Physiol. 2016, 120, 1355–1363.
- Van der Veen, J.N.; Kennelly, J.P.; Wan, S.; Vance, J.E.; Vance, D.E.; Jacobs, R.L. The critical role of phosphatidylcholine and phosphatidylethanolamine metabolism in health and disease. Biochim. Biophys. Acta Biomembr. 2017, 1859, 1558–1572.
- Chang, W.; Hatch, G.; Wang, Y.; Yu, F.; Wang, M. The relationship between phospholipids and insulin resistance: From clinical to experimental evidence. J. Cell. Mol. Med. 2019, 23, 702–710.
- Alpha linolenic acid. Available online: https://pubchem.ncbi.nlm.nih.gov/#query=%22alpha%20Linolenic%20Acid%22 (accessed on 5 June 2023).
- Linoleic acid. Available online: https://pubchem.ncbi.nlm.nih.gov/#query=linoleic%20acid (accessed on 5 June 2023).
- Calder, P.C. Polyunsaturated fatty acids and inflammatory processes: New twists in an old tale. Biochimie 2009, 91, 791–795.
- Lepretti, M.; Martucciello, S.; Burgos Aceves, M.A.; Putti, R.; Lionetti, L. Omega-3 Fatty Acids and Insulin Resistance: Focus on the Regulation of Mitochondria and Endoplasmic Reticulum Stress. Nutrients 2018, 10, 350.
- Heskey, C.E.; Jaceldo-Siegl, K.; Sabate, J.; Fraser, G.; Rajaram, S. Adipose tissue alpha-linolenic acid is inversely associated with insulin resistance in adults. Am. J. Clin. Nutr. 2016, 103, 1105–1110.
- Taouis, M.; Dagou, C.; Ster, C.; Durand, G.; Pinault, M.; Delarue, J. N-3 polyunsaturated fatty acids prevent the defect of insulin receptor signaling in muscle. Am. J. Physiol. Endocrinol. Metab. 2002, 282, E664–E671.
- Chung, S.; Brown, J.M.; Provo, J.N.; Hopkins, R.; McIntosh, M.K. Conjugated linoleic acid promotes human adipocyte insulin resistance through NFkappaB-dependent cytokine production. J. Biol. Chem. 2005, 280, 38445–38456.
- Cholesterol. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/5997 (accessed on 5 June 2023).
- Yeagle, P.L. Cholesterol and the cell membrane. Biochim. Biophys. Acta 1985, 822, 267–287.
- Owen, D.M.; Williamson, D.J.; Magenau, A.; Gaus, K. Sub-resolution lipid domains exist in the plasma membrane and regulate protein diffusion and distribution. Nat. Commun. 2012, 3, 1256.
- Pihlajamaki, J.; Gylling, H.; Miettinen, T.A.; Laakso, M. Insulin resistance is associated with increased cholesterol synthesis and decreased cholesterol absorption in normoglycemic men. J. Lipid Res. 2004, 45, 507–512.
- Hoenig, M.R.; Sellke, F.W. Insulin resistance is associated with increased cholesterol synthesis, decreased cholesterol absorption and enhanced lipid response to statin therapy. Atherosclerosis 2010, 211, 260–265.
- Gylling, H.; Hallikainen, M.; Pihlajamaki, J.; Simonen, P.; Kuusisto, J.; Laakso, M.; Miettinen, T.A. Insulin sensitivity regulates cholesterol metabolism to a greater extent than obesity: Lessons from the METSIM Study. J. Lipid Res. 2010, 51, 2422–2427.
- Haeusler, R.A.; Astiarraga, B.; Camastra, S.; Accili, D.; Ferrannini, E. Human insulin resistance is associated with increased plasma levels of 12alpha-hydroxylated bile acids. Diabetes 2013, 62, 4184–4191.
- Staels, B.; Kuipers, F. Bile acid sequestrants and the treatment of type 2 diabetes mellitus. Drugs 2007, 67, 1383–1392.
- Tomkin, G.H.; Owens, D. Obesity diabetes and the role of bile acids in metabolism. J. Transl. Int. Med. 2016, 4, 73–80.
More
