Multiple sclerosis (MS) is a disabling immune-mediated demyelinating neurodegenerative disease with an estimated prevalence of 1 in 1000 in populations of European descent. It primarily affects females (F:M = 2–3:1) mainly between the ages of 15 and 55 years.
- multiple sclerosis
- pediatric-onset multiple sclerosis
- nutrition
- diet
- gut microbiota
- gut-brain axis
- blood-brain barrier
- vitamin D
1. The Gut-Brain Axis in Multiple Sclerosis
1.1. The Intestinal Barrier
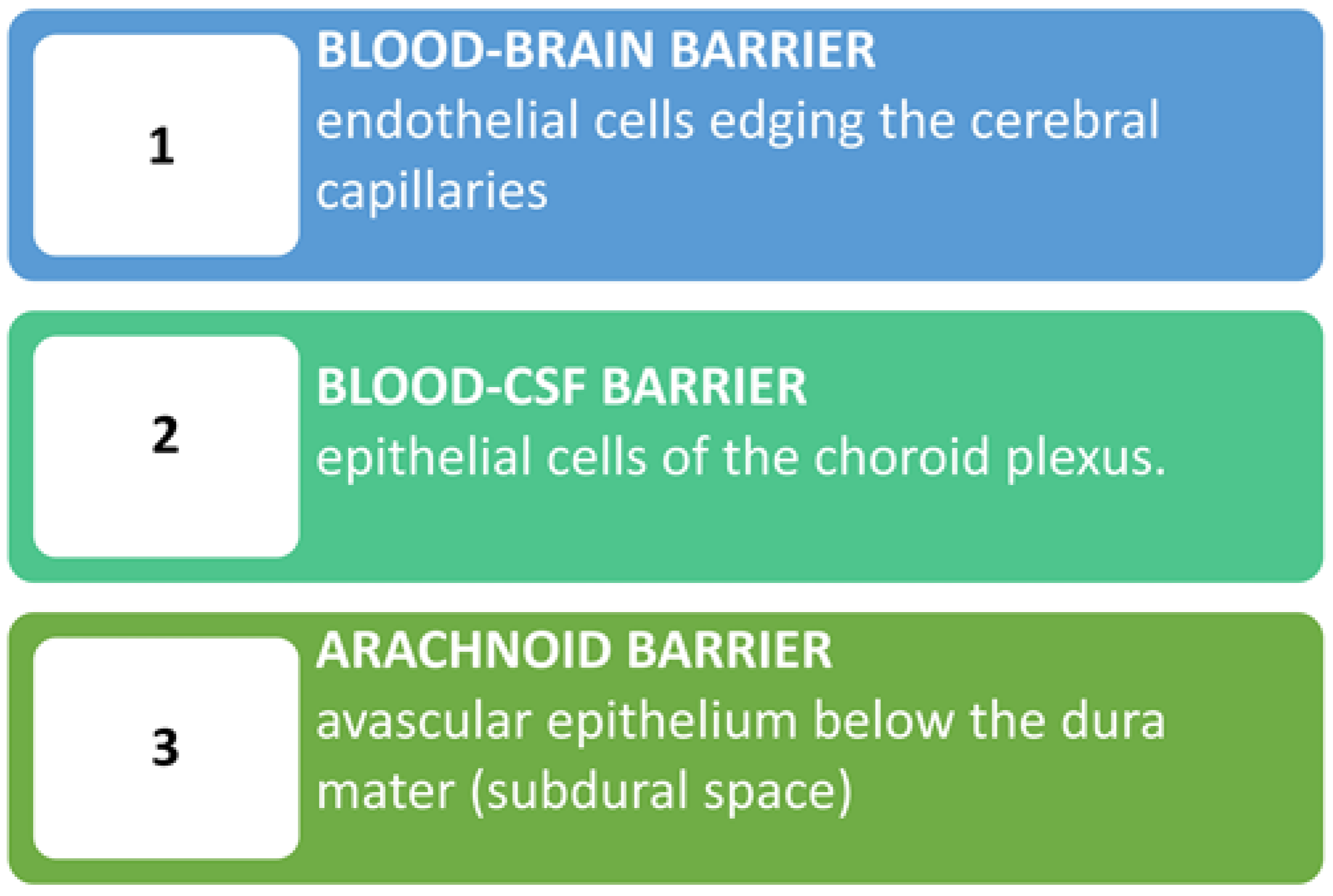
1.2. The Blood-Brain Barrier
2. Diets and Dietary Supplementations
2.1. Dietary Influence on MS
2.1. Dietary Influence on Multiple Sclerosis
| Diet Name | Main Characteristics |
|---|---|
| Allergen free/milk free | Hypoallergenic diet based on the unproven hypothesis of the association between MS and external allergens [76][65]. The milk protein butyrophilin has been implicated through antigenic mimicry with myelin oligodendrocyte glycoprotein in EAE [77][66] as well as in MS patients [78][67]. Some studies with questionnaires suggest an inverse relationship between total dairy intake and MS disability severity [79,80][68][69] with an inverse relationship between whole grain intake and MS-related disability [80][69]. |
| Gluten free | Among studies, only one clinical trial gave meaningful results, but there are methodological limitations [81,82,83][70][71][72]. All in all, the current level of evidence is inadequate to state whether gluten plays a role in MS [82][71]. |
| Mega Ascorbic | High in vitamin C diet. No well-defined link between MS and vitamin C [84][73]. |
| Multi Vitaminic | Multi vitaminic supplementation (e.g., A and D): quite convincing data show that higher vitamin intake/serum levels correlate with lower risk of MS development but not convincing on the contrary [85][74]. Possible detrimental effects of overdosing require vitamin-level monitoring [86,87][75][76]. |
| Hebener’s | Self-reported disease stability/amelioration in one study with fish oil and antioxidant drugs supplementation + Ω-6 restriction [88][77]. |
| Kousmine | High in polyunsaturated fats/low in animal fats diet to counteract a possibly increased membrane permeability [89,90,91][78][79][80]. |
| Swank (low saturated fats) | Low-saturated fats (<20 g fat/day or <20% total calories): reported lower death rates and better outcome in the more adherent patients and those with lower disability at entry [92,93][81][82]. |
| Mediterranean diets (MD) | Common features include emphasis on vegetables, fruits, beans, nuts, seeds, breads, unrefined grains, and olive oil; inclusion of fish and wine; minimal intake of full-fat dairy products and possibly lean meats [94,95][83][84]. Conflicting results on whether lean and unprocessed red meat is detrimental [96,97,98][85][86][87]. It is considered beneficial for its antioxidant properties. Negatively associated with neurological and fatigue symptoms. Adherence should be monitored through validated tests [e.g., Predimed for adults [94][83] and KidMed for children [99][88] |
| Mediterranean/DASH | It derives from the Mediterranean Dietary Approaches to Stop Hypertension (DASH) [100,101][89][90]. |
| MIND | The Mediterranean/Intervention for Neurodegenerative Delay (MIND) is a combination of MD and DASH [100,101,102][89][90][91]. |
| Paleolithic1 | Consists of high-quality foods full of nutrients and fiber and with less artificial sugar and salt compared to present-day diets [103][92]. Nutrients included in this diet are essential to myelin growth and repair. Typically, it does not permit consumption of dairy or grain products. |
| Modified Paleolithic (MD-PI intervention) |
This diet is rich in α-lipoic acid and polyphenols. It has commonalities with MD including avoidance of high-fat meats/ultra-processed foods with added sugar, sodium, and hydrogenated fats [104,105,106][93][94][95]. |
| Wahls™ Paleo diet | Differences from a traditional Paleo diet: exclusion of eggs; limited animal and fish protein. It allows legumes (e.g., soy milk), two servings of gluten-free grains (e.g., rice) per week; it specifies nine cups of fruits and vegetables (F/V)/day with 1/3 each from dark-green leafy vegetables, sulfur-rich vegetables, and deeply colored F/V; seaweed, algae and nutritional yeast are encouraged [107,108][96][97]. |
| Wahls/Elim Paleo | This is a paleo version modified by adding a restriction of lectins to reduce intestinal permeability and CNS inflammation [107,108][96][97]. |
| Overcoming MS (OMS) | Minimized saturated fats and plant-based, whole-food diet plus seafood [79,[68109]][98]. |
| Ketogenic diet (KD) | Eliminating all/almost all carbohydrates and increasing the intake of proteins. KD combined with a modified MD have been suggested to improve neuroinflammation in MS [110,111][99][100]. |
| Energy restriction (ER) | Chronic ER/Intermittent energy restriction (IER) determines a switch from glucose to fatty acids and ketones as the major fuel source for cells [112,[113,101114,115]][102][103][104]. Mice fed a “fasting mimicking” diet (very low-calorie diet lasting for 3 days every 7 days) exhibited delayed onset, reduced incidence, and decreased severity of EAE. Histological findings show reduced immune cell infiltration and demyelination in the spinal cord [116][105]. |
| McDougall Diet | A low-fat (10–15% of calories from fat), starch-based, vegan diet with no oils permitted. For 7 days, produced significant favorable changes in commonly tested biomarkers used to predict future risks for cardiovascular disease and metabolic diseases [75,117][64][106]. It appeared safe and effective in preventing clinical attacks/new MRI lesions. Drawback = long-term adherence [118][107]. |
2.2. Dietary Supplementations
MS starting in childhood (Pediatric-Onset MS, POMS) is estimated to account for between 2–5% and 5–10% of the MS population worldwide. Although youth with POMS have a lower risk of disability within the first 10 years of diagnosis than those with adult-onset MS, the disease may negatively affect their school and emotional spheres. Moreover, they reach disability milestones earlier than adults, even though they tend to take a longer time to advance to the secondary progressive phase. Overall, quite common cognitive impairment requiring specific management, decrease in QOL, and an increase in economic burden in POMS have been shown to have profound impacts not only on patients but also on their families.
Childhood obesity has been identified as a potential risk factor for increased morbidity not only due to hepatic-cardiac-metabolic comorbidities but also from MS and clinically isolated syndrome (CIS) in adolescents, particularly in girls. The underlying mechanism may involve vitamin D deficiency, as obesity is associated with lower vitamin D levels. Of note, sedentary indoor lifestyles and reduced sunlight exposure, contribute to decreased vitamin D synthesis and increased hypovitaminosis D in children.
Unbalanced diets with increased fats, especially saturated fat content, are associated with a higher risk of unfavorable disease progression. A healthy diet characterized by the consumption of fruit, yogurt and legumes during childhood, appears associated with a lower probability of developing subsequent MS in adulthood. Specific dietary strategies may therefore aid children with POMS in slowing disease progression and improving their quality of life. Overall, these data are particularly worrying if one considers that a large proportion of adolescents with POMS have been found to have a non-self-perceived elevated BMI. To improve their disease progression, they should therefore receive more accurate counseling to improve their diet and physical activity as well.
The gut microbiota (GM) has been implicated also in POMS. Differences in microbial composition and metabolic pathways have been observed in children with POMS compared to healthy controls, and have been found to predict the likelihood of recurrence.
3. Perspectives
What is around the gastrointestinal/nutritional corner of MS ? A) Next generation (NG) engineered probiotics, obtained by modifying original probiotics through gene editing modalities, have hitherto been used in inflammatory bowel disease, and in a number of bacterial infections, tumors, and metabolic diseases, mainly in MS akin EAE murine models and/or in vitro. Promising preliminary results showing they are effective, with fewer side effects than traditional treatments or wild-type strains, suggest that they will probably be proposed soon for central nervous system (CNS) diseases as well, including much probably MS. Of note, the design of NG probiotics should specifically be directed towards the production of metabolites (e.g., SCFAs) and neurotransmitters (e.g., serotonin, GABA) which are known to affect the neurobiology of CNS inflammatory diseases. B) Fecal microbiota transplantation (FMT) represents a further interesting approach to modulate GM. FMT studies in animal models and in humans with MS are still scarce and preliminary. Some data available from a cohort of RRMS patients show that FMT is safe and was well-tolerated and may have also improved their gut dysbiosis and elevated small intestinal permeability. Moreover, single case reports and a case series in addition to confirming the safety of the treatment, also showed specific clinical improvements in MS-related neurological symptoms.
4. Conclusions
In conclusion, data show that (1) no universal best diet exists, (2) healthy/balanced diets are necessary to safeguard the adequate intake of all essential nutrients, (3) diets with high intakes of fruits, vegetables, whole grains, and lean proteins that limit processed foods, sugar, and saturated fat appear beneficial for their antioxidant and anti-inflammatory properties and their ability to shape a gut microbiota that respects the gut and brain barriers, (4) obesity may trigger MS onset and/or its less favorable course, especially in pediatric-onset MS. Vitamin D and polyunsaturated fatty acids are the most studied supplements for reducing MS-associated inflammation.
More in detail, several dietary/nutritional factors play an important role both in adult and pediatric MS development and progression. Several gut-oriented nutritional interventions aiming to improve the dysregulation of the so-called Gut Brain Axis through a proper diet appear to intervene beneficially mainly against the inflammatory pathomechanisms associated with MS. The efficacy of any dietary intervention in MS, however, remains difficult to prove due to spontaneous remissions (and relapses) with temporary clinical improvement occurring by chance alone. Pending more solid evidence on specific diets, experts suggest that individuals with MS should be taught to follow a “healthy” regime and possibly enter into nutrition education programs, which, however, are largely lacking as in most other neurological diseases at present.
Because of the high prevalence of overweight/obesity, and the evidence that obesity can worsen MS prognosis, education on weight management is still an unmet need. Pediatric interventions may be hampered by the lack of self-perceived BMI elevation at this age. Because there is, at present, no robust evidence, future research is also needed to identify appropriate study designs and intervention strategies targeting physical activity participation. New solid longitudinal and experimental designs are necessary not only to better elucidate the role of diet and other modifiable lifestyle factors in this population, but also to explore other modalities of support. These should include a closer monitoring of nutritional status of patients with moderate-advanced MS in order to prevent their tendency to be overweight secondary to the decrease in basal energy expenditure and loss of muscle mass.
Further detailed and up-to-date information is available inside the article by Mandato C, Colucci A, Lanzillo R, Staiano A, Scarpato E, Schiavo L, Operto FF, Serra MR, Di Monaco C, Napoli JS, Massa G, and Vajro P. Multiple Sclerosis—Related Dietary and Nutritional Issues: An Updated Scoping Review with a Focus on Pediatrics. Children. 2023; 10(6):1022. https://doi.org/10.3390/children10061022
References
- Bierhansl, L.; Hartung, H.-P.; Aktas, O.; Ruck, T.; Roden, M.; Meuth, S.G. Thinking Outside the Box: Non-Canonical Targets in Multiple Sclerosis. Nat. Rev. Drug. Discov. 2022, 21, 578–600.
- Parodi, B.; Kerlero de Rosbo, N. The Gut-Brain Axis in Multiple Sclerosis. Is Its Dysfunction a Pathological Trigger or a Consequence of the Disease? Front. Immunol. 2021, 12, 718220.
- Timmermans, S.; Bogie, J.F.; Vanmierlo, T.; Lütjohann, D.; Stinissen, P.; Hellings, N.; Hendriks, J.J. High Fat Diet Exacerbates Neuroinflammation in an Animal Model of Multiple Sclerosis by Activation of the Renin Angiotensin System. J. Neuroimmune Pharmacol. 2014, 9, 209–217.
- Yoon, H.; Kleven, A.; Paulsen, A.; Kleppe, L.; Wu, J.; Ying, Z.; Gomez-Pinilla, F.; Scarisbrick, I.A. Interplay between Exercise and Dietary Fat Modulates Myelinogenesis in the Central Nervous System. Biochim. Biophys. Acta BBA-Mol. Basis Dis. 2016, 1862, 545–555.
- Guerrero-García, J.d.J.; Carrera-Quintanar, L.; López-Roa, R.I.; Márquez-Aguirre, A.L.; Rojas-Mayorquín, A.E.; Ortuño-Sahagún, D. Multiple Sclerosis and Obesity: Possible Roles of Adipokines. Mediators Inflamm. 2016, 2016, 4036232.
- Nyirenda, M.H.; Fadda, G.; Healy, L.M.; Mexhitaj, I.; Poliquin-Lasnier, L.; Hanwell, H.; Saveriano, A.W.; Rozenberg, A.; Li, R.; Moore, C.S. Pro-Inflammatory Adiponectin in Pediatric-Onset Multiple Sclerosis. Mult. Scler. J. 2021, 27, 1948–1959.
- Paolella, G.; Mandato, C.; Pierri, L.; Poeta, M.; Di Stasi, M.; Vajro, P. Gut-Liver Axis and Probiotics: Their Role in Non-Alcoholic Fatty Liver Disease. World J. Gastroenterol. WJG 2014, 20, 15518.
- Mandato, C.; Bovi, A.P.D.; Vajro, P. The Gut-Liver Axis as a Target of Liver Disease Management. Hepatobiliary Surg. Nutr. 2021, 10, 100.
- Spadoni, I.; Zagato, E.; Bertocchi, A.; Paolinelli, R.; Hot, E.; Di Sabatino, A.; Caprioli, F.; Bottiglieri, L.; Oldani, A.; Viale, G. A Gut-Vascular Barrier Controls the Systemic Dissemination of Bacteria. Science 2015, 350, 830–834.
- Di Tommaso, N.; Santopaolo, F.; Gasbarrini, A.; Ponziani, F.R. The Gut–Vascular Barrier as a New Protagonist in Intestinal and Extraintestinal Diseases. Int. J. Mol. Sci. 2023, 24, 1470.
- Camara-Lemarroy, C.R.; Metz, L.M.; Yong, V.W. Focus on the Gut-Brain Axis: Multiple Sclerosis, the Intestinal Barrier and the Microbiome. World J. Gastroenterol. 2018, 24, 4217.
- Boziki, M.K.; Kesidou, E.; Theotokis, P.; Mentis, A.-F.A.; Karafoulidou, E.; Melnikov, M.; Sviridova, A.; Rogovski, V.; Boyko, A.; Grigoriadis, N. Microbiome in Multiple Sclerosis: Where Are We, What We Know and Do Not Know. Brain Sci. 2020, 10, 234.
- Panera, N.; Mandato, C.; Crudele, A.; Bertrando, S.; Vajro, P.; Alisi, A. Genetics, Epigenetics and Transgenerational Transmission of Obesity in Children. Front. Endocrinol. 2022, 13, 1006008.
- Madison, A.; Kiecolt-Glaser, J.K. Stress, Depression, Diet, and the Gut Microbiota: Human–Bacteria Interactions at the Core of Psychoneuroimmunology and Nutrition. Curr. Opin. Behav. Sci. 2019, 28, 105–110.
- Altowaijri, G.; Fryman, A.; Yadav, V. Dietary Interventions and Multiple Sclerosis. Curr. Neurol. Neurosci. Rep. 2017, 17, 28.
- Freedman, S.N.; Shahi, S.K.; Mangalam, A.K. The “Gut Feeling”: Breaking down the Role of Gut Microbiome in Multiple Sclerosis. Neurotherapeutics 2018, 15, 109–125.
- Ullah, H.; Tovchiga, O.; Daglia, M.; Khan, H. Modulating Gut Microbiota: An Emerging Approach in the Prevention and Treatment of Multiple Sclerosis. Curr. Neuropharmacol. 2021, 19, 1966.
- Sorboni, S.G.; Moghaddam, H.S.; Jafarzadeh-Esfehani, R.; Soleimanpour, S. A Comprehensive Review on the Role of the Gut Microbiome in Human Neurological Disorders. Clin. Microbiol. Rev. 2022, 35, e00338-20.
- Wang, X.; Liang, Z.; Wang, S.; Ma, D.; Zhu, M.; Feng, J. Role of Gut Microbiota in Multiple Sclerosis and Potential Therapeutic Implications. Curr. Neuropharmacol. 2022, 20, 1413–1426.
- Preiningerova, J.L.; Jiraskova Zakostelska, Z.; Srinivasan, A.; Ticha, V.; Kovarova, I.; Kleinova, P.; Tlaskalova-Hogenova, H.; Kubala Havrdova, E. Multiple Sclerosis and Microbiome. Biomolecules 2022, 12, 433.
- Engelenburg, H.J.; Lucassen, P.J.; Sarafian, J.T.; Parker, W.; Laman, J.D. Multiple Sclerosis and the Microbiota: Progress in Understanding the Contribution of the Gut Microbiome to Disease. Evol. Med. Public Health 2022, 10, 277–294.
- Yu, H.; Bai, S.; Hao, Y.; Guan, Y. Fatty Acids Role in Multiple Sclerosis as “Metabokines”. J. Neuroinflammation 2022, 19, 157.
- Nouri, M.; Bredberg, A.; Weström, B.; Lavasani, S. Intestinal Barrier Dysfunction Develops at the Onset of Experimental Autoimmune Encephalomyelitis, and Can Be Induced by Adoptive Transfer of Auto-Reactive T Cells. PLoS ONE 2014, 9, e106335.
- Dargahi, N.; Matsoukas, J.; Apostolopoulos, V. Streptococcus Thermophilus ST285 Alters Pro-Inflammatory to Anti-Inflammatory Cytokine Secretion against Multiple Sclerosis Peptide in Mice. Brain Sci. 2020, 10, 126.
- Cani, P.D.; de Vos, W.M. Next-Generation Beneficial Microbes: The Case of Akkermansia Muciniphila. Front. Microbiol. 2017, 8, 1765.
- Xu, R.; Zhang, Y.; Chen, S.; Zeng, Y.; Fu, X.; Chen, T.; Luo, S.; Zhang, X. The Role of the Probiotic Akkermansia Muciniphila in Brain Functions: Insights Underpinning Therapeutic Potential. Crit. Rev. Microbiol. 2023, 49, 151–176.
- Shahi, S.K.; Freedman, S.N.; Mangalam, A.K. Gut Microbiome in Multiple Sclerosis: The Players Involved and the Roles They Play. Gut Microbes 2017, 8, 607–615.
- Farrokhi, V.; Nemati, R.; Nichols, F.C.; Yao, X.; Anstadt, E.; Fujiwara, M.; Grady, J.; Wakefield, D.; Castro, W.; Donaldson, J. Bacterial Lipodipeptide, Lipid 654, Is a Microbiome-associated Biomarker for Multiple Sclerosis. Clin. Transl. Immunol. 2013, 2, e8.
- Ling, Z.; Cheng, Y.; Yan, X.; Shao, L.; Liu, X.; Zhou, D.; Zhang, L.; Yu, K.; Zhao, L. Alterations of the Fecal Microbiota in Chinese Patients with Multiple Sclerosis. Front. Immunol. 2020, 11, 590783.
- Dias de Sousa, M.A.; Desidério, C.S.; da Silva Catarino, J.; Trevisan, R.O.; Alves da Silva, D.A.; Rocha, V.F.R.; Bovi, W.G.; Timoteo, R.P.; Bonatti, R.C.F.; da Silva, A.E. Role of Cytokines, Chemokines and IFN-Γ+ IL-17+ Double-Positive CD4+ T Cells in Patients with Multiple Sclerosis. Biomedicines 2022, 10, 2062.
- Duarte-Silva, E.; Meuth, S.G.; Peixoto, C.A. Microbial Metabolites in Multiple Sclerosis: Implications for Pathogenesis and Treatment. Front. Neurosci. 2022, 16, 885031.
- Levi, I.; Gurevich, M.; Perlman, G.; Magalashvili, D.; Menascu, S.; Bar, N.; Godneva, A.; Zahavi, L.; Chermon, D.; Kosower, N. Potential Role of Indolelactate and Butyrate in Multiple Sclerosis Revealed by Integrated Microbiome-Metabolome Analysis. Cell Rep. Med. 2021, 2, 100246.
- Silva, Y.P.; Bernardi, A.; Frozza, R.L. The Role of Short-Chain Fatty Acids from Gut Microbiota in Gut-Brain Communication. Front. Endocrinol. 2020, 11, 25.
- Takewaki, D.; Suda, W.; Sato, W.; Takayasu, L.; Kumar, N.; Kimura, K.; Kaga, N.; Mizuno, T.; Miyake, S.; Hattori, M. Alterations of the Gut Ecological and Functional Microenvironment in Different Stages of Multiple Sclerosis. Proc. Natl. Acad. Sci. USA 2020, 117, 22402–22412.
- Fyfe, I. Microbiome Varies between Multiple Sclerosis Subtypes. Nat. Rev. Neurol. 2020, 16, 243.
- Reynders, T.; Devolder, L.; Valles-Colomer, M.; Van Remoortel, A.; Joossens, M.; De Keyser, J.; Nagels, G.; D’hooghe, M.; Raes, J. Gut Microbiome Variation Is Associated to Multiple Sclerosis Phenotypic Subtypes. Ann. Clin. Transl. Neurol. 2020, 7, 406–419.
- Cekanaviciute, E.; Yoo, B.B.; Runia, T.F.; Debelius, J.W.; Singh, S.; Nelson, C.A.; Kanner, R.; Bencosme, Y.; Lee, Y.K.; Hauser, S.L. Gut Bacteria from Multiple Sclerosis Patients Modulate Human T Cells and Exacerbate Symptoms in Mouse Models. Proc. Natl. Acad. Sci. USA 2017, 114, 10713–10718.
- Berer, K.; Gerdes, L.A.; Cekanaviciute, E.; Jia, X.; Xiao, L.; Xia, Z.; Liu, C.; Klotz, L.; Stauffer, U.; Baranzini, S.E. Gut Microbiota from Multiple Sclerosis Patients Enables Spontaneous Autoimmune Encephalomyelitis in Mice. Proc. Natl. Acad. Sci. USA 2017, 114, 10719–10724.
- Luu, M.; Pautz, S.; Kohl, V.; Singh, R.; Romero, R.; Lucas, S.; Hofmann, J.; Raifer, H.; Vachharajani, N.; Carrascosa, L.C. The Short-Chain Fatty Acid Pentanoate Suppresses Autoimmunity by Modulating the Metabolic-Epigenetic Crosstalk in Lymphocytes. Nat. Commun. 2019, 10, 760.
- Cantoni, C.; Lin, Q.; Dorsett, Y.; Ghezzi, L.; Liu, Z.; Pan, Y.; Chen, K.; Han, Y.; Li, Z.; Xiao, H. Alterations of Host-Gut Microbiome Interactions in Multiple Sclerosis. EBioMedicine 2022, 76, 103798.
- Kadry, H.; Noorani, B.; Cucullo, L. A Blood–Brain Barrier Overview on Structure, Function, Impairment, and Biomarkers of Integrity. Fluids Barriers CNS 2020, 17, 69.
- Davanzo, G.G.; Castro, G.; de Brito Monteiro, L.; Castelucci, B.G.; Jaccomo, V.H.; da Silva, F.C.; Marques, A.M.; Francelin, C.; de Campos, B.B.; de Aguiar, C.F. Obesity Increases Blood-Brain Barrier Permeability and Aggravates the Mouse Model of Multiple Sclerosis. Mult. Scler. Relat. Disord. 2023, 72, 104605.
- Ji, Z.; Wu, S.; Xu, Y.; Qi, J.; Su, X.; Shen, L. Obesity Promotes EAE through IL-6 and CCL-2-Mediated T Cells Infiltration. Front. Immunol. 2019, 10, 1881.
- Babaloo, Z.; Aliparasti, M.R.; Babaiea, F.; Almasi, S.; Baradaran, B.; Farhoudi, M. The Role of Th17 Cells in Patients with Relapsing-Remitting Multiple Sclerosis: Interleukin-17A and Interleukin-17F Serum Levels. Immunol. Lett. 2015, 164, 76–80.
- Awasthi, A.; Kuchroo, V.K. Th17 Cells: From Precursors to Players in Inflammation and Infection. Int. Immunol. 2009, 21, 489–498.
- Li, X.; You, X.; Wang, C.; Li, X.; Sheng, Y.; Zhuang, P.; Zhang, Y. Bidirectional Brain-gut-microbiota Axis in Increased Intestinal Permeability Induced by Central Nervous System Injury. CNS Neurosci. Ther. 2020, 26, 783–790.
- Carabotti, M.; Scirocco, A.; Maselli, M.A.; Severi, C. The Gut-Brain Axis: Interactions between Enteric Microbiota, Central and Enteric Nervous Systems. Ann. Gastroenterol. Q. Publ. Hell. Soc. Gastroenterol. 2015, 28, 203.
- Abdollahpour, I.; Jakimovski, D.; Shivappa, N.; Hébert, J.R.; Vahid, F.; Nedjat, S.; Mansournia, M.A.; Weinstock-Guttman, B. Dietary Inflammatory Index and Risk of Multiple Sclerosis: Findings from a Large Population-Based Incident Case–Control Study. Clin. Nutr. 2020, 39, 3402–3407.
- Alhaj, O.A.; Trabelsi, K.; Younes, A.M.; Shivappa, N.; Bragazzi, N.L.; Hebert, J.R.; Jahrami, H.A. Diet-Related Inflammation Increases the Odds of Multiple Sclerosis: Results from a Large Population-Based Prevalent Case-Control Study in Jordan. Front. Nutr. 2023, 10, 554.
- Hoare, S.; Lithander, F.; Van Der Mei, I.; Ponsonby, A.-L.; Lucas, R.; Group, A.I. Higher Intake of Omega-3 Polyunsaturated Fatty Acids Is Associated with a Decreased Risk of a First Clinical Diagnosis of Central Nervous System Demyelination: Results from the Ausimmune Study. Mult. Scler. J. 2016, 22, 884–892.
- Matveeva, O.; Bogie, J.F.; Hendriks, J.J.; Linker, R.A.; Haghikia, A.; Kleinewietfeld, M. Western Lifestyle and Immunopathology of Multiple Sclerosis. Ann. N. Y. Acad. Sci. 2018, 1417, 71–86.
- Leo, E.E.M.; Campos, M.R.S. Effect of Ultra-Processed Diet on Gut Microbiota and Thus Its Role in Neurodegenerative Diseases. Nutrition 2020, 71, 110609.
- Mannino, A.; Daly, A.; Dunlop, E.; Probst, Y.; Ponsonby, A.-L.; van der Mei, I.A.F.; Chapman, C.; Coulthard, A.; Dear, K.; Dwyer, T.; et al. Higher Consumption of Ultra-Processed Foods and Increased Likelihood of Central Nervous System Demyelination in a Case-Control Study of Australian Adults. Eur. J. Clin. Nutr. 2023, 77, 611–614.
- Martín-Hersog, F.A.; Muñoz-Jurado, A.; Escribano, B.M.; Luque, E.; Galván, A.; LaTorre, M.; Giraldo, A.I.; Caballero-Villarraso, J.; Agüera, E.; Santamaría, A. Sodium Chloride-Induced Changes in Oxidative Stress, Inflammation, and Dysbiosis in Experimental Multiple Sclerosis. Nutr. Neurosci. 2022, 1–13.
- Kleinewietfeld, M.; Manzel, A.; Titze, J.; Kvakan, H.; Yosef, N.; Linker, R.A.; Muller, D.N.; Hafler, D.A. Sodium Chloride Drives Autoimmune Disease by the Induction of Pathogenic TH17 Cells. Nature 2013, 496, 518–522.
- Farez, M.F.; Fiol, M.P.; Gaitán, M.I.; Quintana, F.J.; Correale, J. Sodium Intake Is Associated with Increased Disease Activity in Multiple Sclerosis. J. Neurol. Neurosurg. Psychiatry 2015, 86, 26–31.
- Huang, J.; Kockum, I.; Stridh, P. Trends in the Environmental Risks Associated with Earlier Onset in Multiple Sclerosis. Mult. Scler. Relat. Disord. 2022, 68, 104250.
- Riccio, P.; Rossano, R. Undigested Food and Gut Microbiota May Cooperate in the Pathogenesis of Neuroinflammatory Diseases: A Matter of Barriers and a Proposal on the Origin of Organ Specificity. Nutrients 2019, 11, 2714.
- Black, L.J.; Zhao, Y.; Peng, Y.C.; Sherriff, J.L.; Lucas, R.M.; Van Der Mei, I.; Pereira, G.; The Ausimmune Investigator Group. Higher Fish Consumption and Lower Risk of Central Nervous System Demyelination. Eur. J. Clin. Nutr. 2020, 74, 818–824.
- Bayat, P.; Farshchi, M.; Yousefian, M.; Mahmoudi, M.; Yazdian-Robati, R. Flavonoids, the Compounds with Anti-Inflammatory and Immunomodulatory Properties, as Promising Tools in Multiple Sclerosis (MS) Therapy: A Systematic Review of Preclinical Evidence. Int. Immunopharmacol. 2021, 95, 107562.
- Hashimoto, M.; Yamamoto, S.; Iwasa, K.; Yamashina, K.; Ishikawa, M.; Maruyama, K.; Bosetti, F.; Yoshikawa, K. The Flavonoid Baicalein Attenuates Cuprizone-Induced Demyelination via Suppression of Neuroinflammation. Brain Res. Bull. 2017, 135, 47–52.
- Evans, E.; Levasseur, V.; Cross, A.H.; Piccio, L. An Overview of the Current State of Evidence for the Role of Specific Diets in Multiple Sclerosis. Mult. Scler. Relat. Disord. 2019, 36, 101393.
- Marck, C.H.; Probst, Y.; Chen, J.; Taylor, B.; van der Mei, I. Dietary Patterns and Associations with Health Outcomes in Australian People with Multiple Sclerosis. Eur. J. Clin. Nutr. 2021, 75, 1506–1514.
- Yadav, V.; Marracci, G.; Kim, E.; Spain, R.; Cameron, M.; Overs, S.; Riddehough, A.; Li, D.K.; McDougall, J.; Lovera, J. Low-Fat, Plant-Based Diet in Multiple Sclerosis: A Randomized Controlled Trial. Mult. Scler. Relat. Disord. 2016, 9, 80–90.
- Ashtari, F.; Jamshidi, F.; Shoormasti, R.S.; Pourpak, Z.; Akbari, M. Cow’s Milk Allergy in Multiple Sclerosis Patients. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2013, 18, S62.
- Stefferl, A.; Schubart, A.; Storch, M.; Amini, A.; Mather, I.; Lassmann, H.; Linington, C. Butyrophilin, a Milk Protein, Modulates the Encephalitogenic T Cell Response to Myelin Oligodendrocyte Glycoprotein in Experimental Autoimmune Encephalomyelitis. J. Immunol. 2000, 165, 2859–2865.
- Guggenmos, J.; Schubart, A.S.; Ogg, S.; Andersson, M.; Olsson, T.; Mather, I.H.; Linington, C. Antibody Cross-Reactivity between Myelin Oligodendrocyte Glycoprotein and the Milk Protein Butyrophilin in Multiple Sclerosis. J. Immunol. 2004, 172, 661–668.
- Hadgkiss, E.J.; Jelinek, G.A.; Weiland, T.J.; Pereira, N.G.; Marck, C.H.; van der Meer, D.M. The Association of Diet with Quality of Life, Disability, and Relapse Rate in an International Sample of People with Multiple Sclerosis. Nutr. Neurosci. 2015, 18, 125–136.
- Fitzgerald, K.C.; Tyry, T.; Salter, A.; Cofield, S.S.; Cutter, G.; Fox, R.; Marrie, R.A. Diet Quality Is Associated with Disability and Symptom Severity in Multiple Sclerosis. Neurology 2018, 90, e1–e11.
- Passali, M.; Josefsen, K.; Frederiksen, J.L.; Antvorskov, J.C. Current Evidence on the Efficacy of Gluten-Free Diets in Multiple Sclerosis, Psoriasis, Type 1 Diabetes and Autoimmune Thyroid Diseases. Nutrients 2020, 12, 2316.
- Thomsen, H.L.; Jessen, E.B.; Passali, M.; Frederiksen, J.L. The Role of Gluten in Multiple Sclerosis: A Systematic Review. Mult. Scler. Relat. Disord. 2019, 27, 156–163.
- Rodrigo, L.; Hernández-Lahoz, C.; Fuentes, D.; Mauri, G.; Alvarez, N.; Vega, J.; González, S. Randomised Clinical Trial Comparing the Efficacy of a Gluten-Free Diet versus a Regular Diet in a Series of Relapsing-Remitting Multiple Sclerosis Patients. Int. J. Neurol. Neurother. 2014, 1, 1–6.
- Peng, H.; Wu, X.; Wen, Y.; Lin, J. Plasma Circulating Vitamin C Levels and Risk of Multiple Sclerosis: A Two-Sample Mendelian Randomization Analysis. Mult. Scler. Relat. Disord. 2021, 56, 103267.
- Bowling, A. Vitamins, Minerals & Herbs in MS AN INTRODUCTION. Available online: https://www.nationalmssociety.org/Programs-and-Services/Resources/Vitamins,-Minerals,-and-Herbs-in-MS-An-Introductio (accessed on 6 May 2023).
- Zorzella-Pezavento, S.F.G.; Mimura, L.A.N.; Denadai, M.B.; De Souza, W.D.F.; de Campos Fraga-Silva, T.F.; Sartori, A. Is There a Window of Opportunity for the Therapeutic Use of Vitamin D in Multiple Sclerosis? Neural Regen. Res. 2022, 17, 1945.
- Vandebergh, M.; Dubois, B.; Goris, A. Effects of Vitamin D and Body Mass Index on Disease Risk and Relapse Hazard in Multiple Sclerosis: A Mendelian Randomization Study. Neurol.-Neuroimmunol. Neuroinflammation 2022, 9, e1165.
- Hebener, O.; Ackermann, H.; Kappel, U.; Kramer, J. Multiple Sclerosis and Additional Balanced Diet. Results of a Retrospective Study. Schweiz. Z. Für Ganzheitsmed./Swiss J. Integr. Med. 2005, 7, 104–108.
- Kousmine, C. Sauvez Votre Corps; Robert Laffont: Paris, France, 1987; ISBN 13: 9782221053843.
- Newman, P.E. Could Diet Be One of the Causal Factors of Alzheimer’s Disease? Med. Hypotheses 1992, 39, 123–126.
- Farinotti, M.; Vacchi, L.; Simi, S.; Di Pietrantonj, C.; Brait, L.; Filippini, G. Dietary Interventions for Multiple Sclerosis. Cochrane Database Syst. Rev. 2012, 12, CD004192.
- Swank, R.L.; Dugan, B.B. Effect of Low Saturated Fat Diet in Early and Late Cases of Multiple Sclerosis. Lancet 1990, 336, 37–39.
- Swank, R.L.; Goodwin, J. Review of MS Patient Survival on a Swank Low Saturated Fat Diet 1 1 (For an Additional Perspective, See Editorial Opinions). Nutrition 2003, 19, 161.
- Appel, L.J.; Van Horn, L. Did the PREDIMED Trial Test a Mediterranean Diet? N. Engl. J. Med. 2013, 368, 1353–1354.
- Öztürk, Y.E.; Helvaci, E.M.; Sökülmez Kaya, P.; Terzi, M. Is Mediterranean Diet Associated with Multiple Sclerosis Related Symptoms and Fatigue Severity? Nutr. Neurosci. 2023, 26, 228–234.
- O’Connor, L.E.; Biberstine, S.L.; Paddon-Jones, D.; Schwichtenberg, A.J.; Campbell, W.W. Adopting a Mediterranean-Style Eating Pattern with Different Amounts of Lean Unprocessed Red Meat Does Not Influence Short-Term Subjective Personal Well-Being in Adults with Overweight or Obesity. J. Nutr. 2018, 148, 1917–1923.
- Black, L.J.; Baker, K.; Ponsonby, A.-L.; Van Der Mei, I.; Lucas, R.M.; Pereira, G. A Higher Mediterranean Diet Score, Including Unprocessed Red Meat, Is Associated with Reduced Risk of Central Nervous System Demyelination in a Case-Control Study of Australian Adults. J. Nutr. 2019, 149, 1385–1392.
- Sand, I.K.; Benn, E.K.; Fabian, M.; Fitzgerald, K.C.; Digga, E.; Deshpande, R.; Miller, A.; Gallo, S.; Arab, L. Randomized-Controlled Trial of a Modified Mediterranean Dietary Program for Multiple Sclerosis: A Pilot Study. Mult. Scler. Relat. Disord. 2019, 36, 101403.
- Serra-Majem, L.; Ribas, L.; Ngo, J.; Ortega, R.M.; García, A.; Pérez-Rodrigo, C.; Aranceta, J. Food, Youth and the Mediterranean Diet in Spain. Development of KIDMED, Mediterranean Diet Quality Index in Children and Adolescents. Public Health Nutr. 2004, 7, 931–935.
- Noormohammadi, M.; Ghorbani, Z.; Naser Moghadasi, A.; Saeedirad, Z.; Shahemi, S.; Ghanaatgar, M.; Rezaeimanesh, N.; Hekmatdoost, A.; Ghaemi, A.; Razeghi Jahromi, S. MIND Diet Adherence Might Be Associated with a Reduced Odds of Multiple Sclerosis: Results from a Case–Control Study. Neurol. Ther. 2022, 11, 397–412.
- Van den Brink, A.C.; Brouwer-Brolsma, E.M.; Berendsen, A.A.; van de Rest, O. The Mediterranean, Dietary Approaches to Stop Hypertension (DASH), and Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) Diets Are Associated with Less Cognitive Decline and a Lower Risk of Alzheimer’s Disease—A Review. Adv. Nutr. 2019, 10, 1040–1065.
- Sand, I.K.; Fitzgerald, K.C.; Gu, Y.; Brandstadter, R.; Riley, C.S.; Buyukturkoglu, K.; Leavitt, V.M.; Krieger, S.; Miller, A.; Lublin, F. Dietary Factors and MRI Metrics in Early Multiple Sclerosis. Mult. Scler. Relat. Disord. 2021, 53, 103031.
- Challa, H.J.; Bandlamudi, M.; Uppaluri, K.R. Paleolithic Diet. In StatPearls ; StatPearls Publishing: Orlando, FL, USA, 2021.
- Learmonth, Y.; Dlugonski, D.; Pilutti, L.A.; Sandroff, B.M.; Klaren, R.; Motl, R.W. Psychometric Properties of the Fatigue Severity Scale and the Modified Fatigue Impact Scale. J. Neurol. Sci. 2013, 331, 102–107.
- Irish, A.K.; Erickson, C.M.; Wahls, T.L.; Snetselaar, L.G.; Darling, W.G. Randomized Control Trial Evaluation of a Modified Paleolithic Dietary Intervention in the Treatment of Relapsing-Remitting Multiple Sclerosis: A Pilot Study. Degener. Neurol. Neuromuscul. Dis. 2017, 7, 1–18.
- Bisht, B.; Darling, W.G.; Shivapour, E.T.; Lutgendorf, S.K.; Snetselaar, L.G.; Chenard, C.A.; Wahls, T.L. Multimodal Intervention Improves Fatigue and Quality of Life in Subjects with Progressive Multiple Sclerosis: A Pilot Study. Degener. Neurol. Neuromuscul. Dis. 2015, 5, 19–35.
- Wahls, T.L.; Chenard, C.A.; Snetselaar, L.G. Review of Two Popular Eating Plans within the Multiple Sclerosis Community: Low Saturated Fat and Modified Paleolithic. Nutrients 2019, 11, 352.
- Chenard, C.A.; Rubenstein, L.M.; Snetselaar, L.G.; Wahls, T.L. Nutrient Composition Comparison between a Modified Paleolithic Diet for Multiple Sclerosis and the Recommended Healthy US-Style Eating Pattern. Nutrients 2019, 11, 537.
- Jelinek, G. Overcoming Multiple Sclerosis: An Evidence-Based Guide to Recovery; Allen & Unwin: Crows Nest, Australia, 2010; ISBN 10: 1742371795/13: 9781742371795.
- Brenton, J.N.; Banwell, B.; Bergqvist, A.C.; Lehner-Gulotta, D.; Gampper, L.; Leytham, E.; Coleman, R.; Goldman, M.D. Pilot Study of a Ketogenic Diet in Relapsing-Remitting MS. Neurol.-Neuroimmunol. Neuroinflammation 2019, 6, e565.
- Di Majo, D.; Cacciabaudo, F.; Accardi, G.; Gambino, G.; Giglia, G.; Ferraro, G.; Candore, G.; Sardo, P. Ketogenic and Modified Mediterranean Diet as a Tool to Counteract Neuroinflammation in Multiple Sclerosis: Nutritional Suggestions. Nutrients 2022, 14, 2384.
- Fitzgerald, K. Diet and Disease Modification in Multiple Sclerosis: A Nutritional Epidemiology Perspective; BMJ Publishing Group Ltd.: London, UK, 2018; Volume 89, ISBN 0022-3050.
- Cignarella, F.; Cantoni, C.; Ghezzi, L.; Salter, A.; Dorsett, Y.; Chen, L.; Phillips, D.; Weinstock, G.M.; Fontana, L.; Cross, A.H. Intermittent Fasting Confers Protection in CNS Autoimmunity by Altering the Gut Microbiota. Cell Metab. 2018, 27, 1222–1235.e6.
- Ghezzi, L. Energy Restriction in People with Multiple Sclerosis: Is Time More Important than Calories? EBioMedicine 2022, 82, 104183.
- Fitzgerald, K.C.; Bhargava, P.; Smith, M.D.; Vizthum, D.; Henry-Barron, B.; Kornberg, M.D.; Cassard, S.D.; Kapogiannis, D.; Sullivan, P.; Baer, D.J. Intermittent Calorie Restriction Alters T Cell Subsets and Metabolic Markers in People with Multiple Sclerosis. EBioMedicine 2022, 82, 104124.
- Choi, I.Y.; Piccio, L.; Childress, P.; Bollman, B.; Ghosh, A.; Brandhorst, S.; Suarez, J.; Michalsen, A.; Cross, A.H.; Morgan, T.E. A Diet Mimicking Fasting Promotes Regeneration and Reduces Autoimmunity and Multiple Sclerosis Symptoms. Cell Rep. 2016, 15, 2136–2146.
- McDougall, J.; Thomas, L.E.; McDougall, C.; Moloney, G.; Saul, B.; Finnell, J.S.; Richardson, K.; Petersen, K.M. Effects of 7 Days on an Ad Libitum Low-Fat Vegan Diet: The McDougall Program Cohort. Nutr. J. 2014, 13, 99.
- Dean, C.; Parks, S.; Titcomb, T.J.; Arthofer, A.; Meirick, P.; Grogan, N.; Ehlinger, M.A.; Bisht, B.; Fox, S.S.; Daack-Hirsch, S. Facilitators of and Barriers to Adherence to Dietary Interventions Perceived by Women with Multiple Sclerosis and Their Support Persons. Int. J. MS Care 2022, 24, 235–241.
- Snetselaar, L.G.; Cheek, J.J.; Fox, S.S.; Healy, H.S.; Schweizer, M.L.; Bao, W.; Kamholz, J.; Titcomb, T.J. Titcomb Efficacy of Diet on Fatigue and Quality of Life in Multiple Sclerosis. Neurology 2023, 100, e357.
- Spain, R.I.; Piccio, L.; Langer-Gould, A.M. Langer-Gould The Role of Diet in Multiple Sclerosis. Neurology 2023, 100, 167.
- Galus, W.; Walawska-Hrycek, A.; Rzepka, M.; Krzystanek, E. Vitamin D Supplementation Practices among Multiple Sclerosis Patients and Professionals. J. Clin. Med. 2022, 11, 7278.
- Cantarel, B.L.; Waubant, E.; Chehoud, C.; Kuczynski, J.; DeSantis, T.Z.; Warrington, J.; Venkatesan, A.; Fraser, C.M.; Mowry, E.M. Gut Microbiota in Multiple Sclerosis: Possible Influence of Immunomodulators. J. Investig. Med. 2015, 63, 729–734.
- Rumah, K.R.; Vartanian, T.K.; Fischetti, V.A. Oral Multiple Sclerosis Drugs Inhibit the in Vitro Growth of Epsilon Toxin Producing Gut Bacterium, Clostridium Perfringens. Front. Cell Infect. Microbiol. 2017, 7, 11.
- Sadeghi Bahmani, D.; Kesselring, J.; Papadimitriou, M.; Bansi, J.; Pühse, U.; Gerber, M.; Shaygannejad, V.; Holsboer-Trachsler, E.; Brand, S. In Patients with Multiple Sclerosis, Both Objective and Subjective Sleep, Depression, Fatigue, and Paresthesia Improved after 3 Weeks of Regular Exercise. Front. Psychiatry 2019, 10, 265.
- Sadeghi Bahmani, D.; Gonzenbach, R.; Motl, R.W.; Bansi, J.; Rothen, O.; Niedermoser, D.; Gerber, M.; Brand, S. Better Objective Sleep Was Associated with Better Subjective Sleep and Physical Activity; Results from an Exploratory Study under Naturalistic Conditions among Persons with Multiple Sclerosis. Int. J. Environ. Res. Public. Health 2020, 17, 3522.
- Clauss, M.; Gérard, P.; Mosca, A.; Leclerc, M. Interplay between Exercise and Gut Microbiome in the Context of Human Health and Performance. Front. Nutr. 2021, 8, 637010.
- Simpson, S.; Furlong, M.; Giebel, C. Exploring the Enablers and Barriers to Social Prescribing for People Living with Long-Term Neurological Conditions: A Focus Group Investigation. BMC Health Serv. Res. 2021, 21, 1230.
- Keykhaei, F.; Norouzy, S.; Froughipour, M.; Nematy, M.; Saeidi, M.; Jarahi, L.; Amiri, F.; Malek Ahmadi, M.; Norouzy, A. Adherence to Healthy Dietary Pattern Is Associated with Lower Risk of Multiple Sclerosis. J. Cent. Nerv. Syst. Dis. 2022, 14, 11795735221092516.
- Teng, M.; Bensmail, D.; Hanachi, M.; Haddad, R.; Hugeron, C.; Lansaman, T.; Levy, J. Nutritional Status in Patients with Advanced-stage Multiple Sclerosis. Eur. J. Neurol. 2022, 29, 1730–1740.
- Robles, L.R.; De La Maza, B.P.; García, J.T.; Vieitez, J.J.G.; Gómez, M.J.F.; Mellado, I.B.; Pomar, M.D.B. Nutritional Profile of Multiple Sclerosis. Nutr. Hosp. Organo Soc. Esp. Nutr. Parenter. Enter. 2019, 36, 340–349.
- Silveira, S.L.; Jeng, B.; Cutter, G.; Motl, R.W. Diet Quality Assessment in Wheelchair Users with Multiple Sclerosis. Nutrients 2021, 13, 4352.
- Luca, M.; Ortega-Castro, N.; Patti, F. Paediatric Multiple Sclerosis: A Scoping Review of Patients’ and Parents’ Perspectives. Children 2021, 9, 11.
- Ghai, S.; Kasilingam, E.; Lanzillo, R.; Malenica, M.; van Pesch, V.; Burke, N.C.; Carotenuto, A.; Maguire, R. Needs and Experiences of Children and Adolescents with Pediatric Multiple Sclerosis and Their Caregivers: A Systematic Review. Children 2021, 8, 445.
- Langer-Gould, A.; Brara, S.M.; Beaber, B.E.; Koebnick, C. Childhood Obesity and Risk of Pediatric Multiple Sclerosis and Clinically Isolated Syndrome. Neurology 2013, 80, 548–552.
- Milles, P.; De Filippo, G.; Maurey, H.; Tully, T.; Deiva, K. Obesity in Pediatric-Onset Multiple Sclerosis: A French Cohort Study. Neurol.-Neuroimmunol. Neuroinflammation 2021, 8, e1044.
- Pakpoor, J.; Pakpoor, J. Childhood Obesity and Risk of Pediatric Multiple Sclerosis and Clinically Isolated Syndrome. Neurology 2013, 81, 1366.
- Shahi, S.K.; Ghimire, S.; Lehman, P.; Mangalam, A.K. Obesity Induced Gut Dysbiosis Contributes to Disease Severity in an Animal Model of Multiple Sclerosis. Front. Immunol. 2022, 13, 966417.
- Russell, R.D.; Langer-Gould, A.; Gonzales, E.G.; Smith, J.B.; Brennan, V.; Pereira, G.; Lucas, R.M.; Begley, A.; Black, L.J. Obesity, Dieting, and Multiple Sclerosis. Mult. Scler. Relat. Disord. 2020, 39, 101889.
- Bruce, J.M.; Cozart, J.S.; Shook, R.P.; Ruppen, S.; Siengsukon, C.; Simon, S.; Befort, C.; Lynch, S.; Mahmoud, R.; Drees, B. Modifying Diet and Exercise in MS (MoDEMS): Study Design and Protocol for a Telehealth Weight Loss Intervention for Adults with Obesity & Multiple Sclerosis. Contemp. Clin. Trials 2021, 107, 106495.
- Cercato, C.; Fonseca, F.A. Cardiovascular Risk and Obesity. Diabetol. Metab. Syndr. 2019, 11, 74.
- Marrie, R.A.; Rudick, R.; Horwitz, R.; Cutter, G.; Tyry, T.; Campagnolo, D.; Vollmer, T. Vascular Comorbidity Is Associated with More Rapid Disability Progression in Multiple Sclerosis. Neurology 2010, 74, 1041–1047.
- Schepici, G.; Silvestro, S.; Bramanti, P.; Mazzon, E. The Gut Microbiota in Multiple Sclerosis: An Overview of Clinical Trials. Cell Transplant. 2019, 28, 1507–1527.
- Sarkar, A.; Lehto, S.M.; Harty, S.; Dinan, T.G.; Cryan, J.F.; Burnet, P.W. Psychobiotics and the Manipulation of Bacteria–Gut–Brain Signals. Trends Neurosci. 2016, 39, 763–781.
- Oroojzadeh, P.; Bostanabad, S.Y.; Lotfi, H. Psychobiotics: The Influence of Gut Microbiota on the Gut-Brain Axis in Neurological Disorders. J. Mol. Neurosci. 2022, 72, 1952–1964.
- Kumar, N.; Sahoo, N.K.; Mehan, S. The Importance of Gut-Brain Axis and Use of Probiotics as a Treatment Strategy for Multiple Sclerosis. Mult. Scler. Relat. Disord. 2023, 71, 104547.
- Hashemi, B.; Abdollahi, M.; Abbaspour-Aghdam, S.; Hazrati, A.; Malekpour, K.; Kafil, H.S.; Yousefi, M.; Roshangar, L.; Ahmadi, M. The Effect of Probiotics on Immune Responses and Their Therapeutic Application: A New Treatment Option for Multiple Sclerosis. Biomed. Pharmacother. 2023, 159, 114195.
- Dziedzic, A.; Saluk, J. Probiotics and Commensal Gut Microbiota as the Effective Alternative Therapy for Multiple Sclerosis Patients Treatment. Int. J. Mol. Sci. 2022, 23, 14478.
- Stephens, S.; Shams, S.; Lee, J.; Grover, S.A.; Longoni, G.; Berenbaum, T.; Finlayson, M.; Motl, R.W.; Yeh, E.A. Benefits of Physical Activity for Depression and Fatigue in Multiple Sclerosis: A Longitudinal Analysis. J. Pediatr. 2019, 209, 226–232.e2.
- Parks, N.E.; Jackson-Tarlton, C.S.; Vacchi, L.; Merdad, R.; Johnston, B.C. Dietary Interventions for Multiple Sclerosis-related Outcomes. Cochrane Database Syst. Rev. 2020, 2020, CD004192.
- Amatya, B.; Khan, F. Do Dietary Interventions Improve Health Outcomes in People with Multiple Sclerosis? A Cochrane Review Summary with Commentary. NeuroRehabilitation 2022, 50, 161–163.
- Tredinnick, A.R.; Probst, Y.C. Evaluating the Effects of Dietary Interventions on Disease Progression and Symptoms of Adults with Multiple Sclerosis: An Umbrella Review. Adv. Nutr. 2020, 11, 1603–1615.
- Galus, W.; Chmiela, T.; Walawska-Hrycek, A.; Krzystanek, E. Radiological Benefits of Vitamin D Status and Supplementation in Patients with MS—A Two-Year Prospective Observational Cohort Study. Nutrients 2023, 15, 1465.
