The glycocalyx generally covers almost all cellular surfaces, where it participates in mediating cell-surface interactions with the extracellular matrix as well as with intracellular signaling molecules. The endothelial glycocalyx that covers the luminal surface mediates the interactions of endothelial cells with materials flowing in the circulating blood, including blood cells. Cardiovascular diseases (CVD) remain a major cause of morbidity and mortality around the world. The cardiovascular risk factors start by causing endothelial cell dysfunction associated with destruction or irregular maintenance of the glycocalyx, which may culminate into a full-blown cardiovascular disease. The endothelial glycocalyx plays a crucial role in shielding the cell from excessive exposure and absorption of excessive salt, which can potentially cause damage to the endothelial cells and underlying tissues of the blood vessels.
- endothelial-glycocalyx
- proteoglycans
- syndecans
- hyaluronan
- glypicans
- sodium chloride-salt
- CVD
1. Background
2. Major Components of the Glycocalyx in CVD Pathophysiology
The components of the glycocalyx involved in CVD pathophysiology include membrane-bound heparan sulfate proteoglycans (such as syndecans and glypicans), CD44, and a membrane-associated polysaccharide called hyaluronic acid (or hyaluronan) (Figure 1).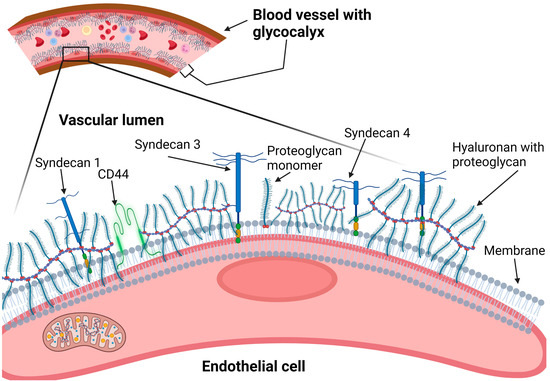
2.1. Heparan Sulfate Proteoglycans (Syndecans and Glypicans)
2.1.1. Syndecans
2.1. Heparan Sulfate Proteoglycans (Syndecans and Glypicans)
2.1.1. Syndecans
2.1.2. Glypicans
2.2. Hyaluronan
Hyaluronan is a long polysaccharide of alternating glucuronic acid (GlcA) and N-acetylglucosamine (GlcNAc) sugar residues, which are un-sulfated but result in a highly negatively charged molecule [41][57]. It is pericellularly synthesized by a family of three pericellular enzymes known as hyaluronan synthases: from HAS-1 to -3 [42][43][58,59]. It is then degraded by a family of six hyaluronidase (HYALs) members, of which HYAL-1 and HYAL-2 are the most potent enzymes [44][45][60,61]. HYAL-2 is believed to have a role in promoting pulmonary vascular remolding and pulmonary hypertension [45][61]. The deficiency of HYAL-3 increases collagen deposition, promoting post-myocardial infarction fibrosis [45][61]. Hyaluronan is most known as an extracellular matrix molecule, but, due to its close association with various cell surface structures including its cognate receptor CD44, integrins, and proteoglycans, it contributes to the surface gel-like glycocalyx of the synthesizing cell [46][62]. The hyaluronan receptor-CD44 can also be cleaved from the cell surface by proteases such as ADAM-10, ADAM-17, and MMP-14 [47][48][63,64]. A delicate balance is maintained normally between hyaluronan bio-synthesis and degradation depending on the activity of HASs and HYL enzymes, which varies in different physiological and pathological conditions [46][62]. The hyaluronan synthesis and degradation processes are indeed crucial during cardiovascular development or angiogenesis [49][50][65,66]. Native hyaluronan is believed to inhibit angiogenesis, whereas oligosaccharides formed following hyaluronan degradation seem to promote angiogenesis by increasing endothelial cell proliferation and migration [51][52][67,68]. The interactions of hyaluronan with reactive oxygen or nitrogen species (ROS/RNS) and their consequences on tissue homeostasis are well summarized by Berdiaki et al., whereby the innate high molecular weight hyaluronan is anti-angiogenic, anti-inflammatory, and anti-oncogenic [53][69]. However, following degradation by the ROS/RNS, the low molecular weight oligosaccharides produced become pro-angiogenic, pro-inflammatory, and oncogenic [53][69]. Changes in hyaluronan synthesis and degradation have mainly been associated with cancer and inflammatory conditions [54][55][56][57][70,71,72,73].3. Glycocalyx and Salt Interactions
Since the endothelial glycocalyx covers the luminal surface of the cells, where it forms a gel-like structure, it is believed to protect the endothelial cells from direct exposure to excessive NaCl salt (above 160 mEq/L) dissolved in plasma [58][86]. The normal sodium levels in plasma are kept within a narrow range of 135–145 mEq/L by a combination of ‘thirst’/water intake and hormonal (aldosterone-anti-diuretic/vasopressin) systems [59][60][87,88]. The GAG chains (including heparan sulfate, chondroitin sulfate, and hyaluronan) are major components of the glycocalyx and are highly negatively charged, making them attractive to the positively charged sodium ions flowing in circulation [61][89], Figure 2. Thus, the glycocalyx is able to play a positive role in the sodium buffering by transiently binding sodium on the luminal side of the blood vessels [62][90]. The glycocalyx, therefore, is a major player in buffering the intravascular sodium and stores a great amount sodium creating a hypertonic environment [63][64][91,92].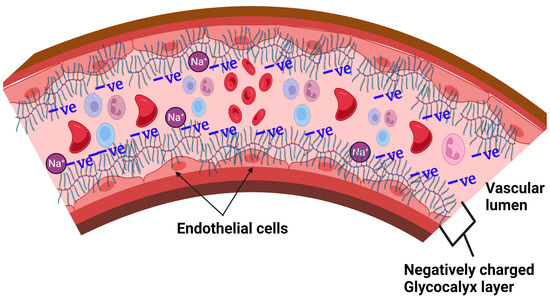
3.1. Proposed Glycocalyx-Salt Interaction Mechanisms Contributing to Hypertension and Cardiovascular Disease
3.1. Proposed Glycocalyx-Salt Interaction Mechanisms Contributing to Hypertension and Cardiovascular Disease
3.2. Possible Strategies for Reducing the Damaging Effects of NaCl-Salt Overload on Vascular Endothelium
3.2. Possible Strategies for Reducing the Damaging Effects of NaCl-Salt Overload on Vascular Endothelium
High dietary salt intake remains a big challenge as many people in various populations around the world are still unable to stop consuming high amounts of salt due to the diverse sources of dietary salt available in common foods [126,127,128]. It is therefore understandable that efforts are being made to find alternative ways of overcoming the deleterious effects of high salt overload on human health, other than simply advising people to reduce salt intake. For example, a recent clinical trial conducted among black women aged between 20 and 60 years in the USA provided evidence showing that ‘hot yoga’ can reduce the harmful effects of salt overload on endothelial function [129]. The exact mechanism is unknown. Similarly, regular aerobic exercise has also been reported to reduce endothelin-1-mediated vasoconstriction and endothelial dysfunction in postmenopausal women, as well as in obese or overweight adults [130,131]. Replacement or substitution of NaCl salt with other forms of salt with similar ‘saltness’ taste, such as potassium chloride (KCl) and monosodium glutamate (MSG), have also been piloted by the Department of Food Science at Cornell University (New York) and been found to have relatively high acceptability among the study subjects, although with a caveat of not disclosing the specific names of the salt substitute [132].
High dietary salt intake remains a big challenge as many people in various populations around the world are still unable to stop consuming high amounts of salt due to the diverse sources of dietary salt available in common foods [80][81][82]. It is therefore understandable that efforts are being made to find alternative ways of overcoming the deleterious effects of high salt overload on human health, other than simply advising people to reduce salt intake. For example, a recent clinical trial conducted among black women aged between 20 and 60 years in the USA provided evidence showing that ‘hot yoga’ can reduce the harmful effects of salt overload on endothelial function [83]. The exact mechanism is unknown. Similarly, regular aerobic exercise has also been reported to reduce endothelin-1-mediated vasoconstriction and endothelial dysfunction in postmenopausal women, as well as in obese or overweight adults [84][85]. Replacement or substitution of NaCl salt with other forms of salt with similar ‘saltness’ taste, such as potassium chloride (KCl) and monosodium glutamate (MSG), have also been piloted by the Department of Food Science at Cornell University (New York) and been found to have relatively high acceptability among the study subjects, although with a caveat of not disclosing the specific names of the salt substitute [86].
