Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by SANJAY KUMAR SINGH PATEL and Version 2 by Conner Chen.
Pancreatic cancer is challenging, with a poor progression and limited treatment options. Its tumorigenesis or metastasis involves, pathways, including phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT), RAS, janus kinase (JAK)/signal transducer, and activator of transcription (STAT), NF-κB, Hippo/yes-kinase-associated protein (hippo/YAP), and Wingless/int1 (WNT). These pathways are associated with numerous cellular processes linked to pancreatic cancer, such as apoptosis, angiogenesis, differentiation, immunological regulations, metabolism, migration, and cell proliferation.
- molecular mechanism
- cancer
1. Introduction
Pancreatic cancer is a fatal disease that affects the pancreas, a large gland in the abdomen. American Cancer Society suggested that pancreatic cancer is the third highly common type of cancer in humans. Initial recognition is critical so that patients receive the extremely efficient treatment possible and have the greatest chance of survival [1][2][3][1,2,3]. Primarily, many patients are detected at an advanced stage. In the United States, this year approximately 53,090 people will be diagnosed with pancreatic cancer, and more than 41,170 people died from the disease [4][5][4,5]. Therefore, we must continue identifying new and improved ways to diagnose the disease earlier to save lives. Pancreatic cancer is highly destructive and has a poor prognosis with only a 9% of survival rate within five-years. Unluckily, the symptoms of pancreatic cancer are often vague and easy to overlook, so many people do not know that they have it until it is too late [6][7][6,7]. Its primary symptoms include jaundice, loss of appetite/weight, and right abdomen pain [4][8][4,8]. The pancreas is also challenging to reach and diagnose, so traditional imaging methods often cannot detect cancer early enough. However, early diagnosis and treatment are critical to improving survival. Today’s early detection methods are better than ever, but there are still significant limitations to available diagnostic tools. Methods of detecting pancreatic tumors include x-rays, ultrasounds, MRI.s, and CT scans [4]. These tests can effectively identify the tumor’s location and the extent of the disease. It is difficult to diagnose early, but specific blood tests can detect it if certain risk factors exist. Specific biomarkers can also be applied to detect cancer cells [9][10][9,10]. Pancreatic cancer is diagnosed with endoscopy, a minimally invasive procedure that uses a tube with a tiny camera attached to examine the inner lining of the digestive tract. During the process, removing a tissue sample from the pancreas to test for cancer cells can be validated [11][12][11,12]. Further, staging determines how far your cancer has spread and whether it has spread beyond the pancreas and risk assessment to effective diagnostic management [13].
The use of drugs destroys cancerous cells while sparing the surrounding healthy cells, allowing them to be combined with other cancer treatments to maximize their effectiveness [14][15][14,15]. Existing treatment possibilities for patients with advanced pancreatic cancer include chemotherapy, immunotherapy, targeted therapies, and surgery [4][16][17][4,16,17]. Chemotherapy involves using powerful drugs that kill cancer cells by attacking their DNA and disrupting their growth and division [18]. Gemcitabine is the first drug approved to treat pancreatic cancer [19]. It stops the development of new cancer cells and accelerates the immune system to kill them. The current standard treatment of gemcitabine is an intravenous injection twice a day for several days. After that, the treatment is repeated every three weeks until the patient no longer responds to the drug or until their condition worsens [19][20][19,20]. Unfortunately, few patients with advanced-stage cancer will respond to these treatments. New targeted therapies continue to be consideration-based in genomics, genetics, and molecular therapies, along with integrative concepts for improved diagnostics and treatments of pancreatic cancer [21][22][23][21,22,23].
Commonly known pancreatic tumors are pancreatic ductal adenocarcinoma (PDAC), accounting of nearly 90% of pancreatic-based cancers [24][25][24,25]. In some cases, patients may also develop secondary tumors in the liver or lungs due to the disease spreading throughout the body [26]. In general, treatment options for pancreatic cancer are usually limited and ineffective. Many patients with this disease do not respond to traditional treatments and are only given a few months to live [4][27][4,27]. However, recent developments in cancer treatment have led to many new therapies that may extend the lives of patients diagnosed with pancreatic cancer [28][29][28,29]. Furthermore, global perspectives have been significantly altered in the current pandemic toward therapeutic strategies for sustainable approaches to maintain good health and the economy [30][31][30,31].
2. Pancreatic Cancer Molecular Manifestation and Pathways Regulation
Pancreatic cancer is challenging, with a poor progression and limited treatment options [32][38]. Its tumorigenesis or metastasis involves, pathways, including phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT), RAS, janus kinase (JAK)/signal transducer, and activator of transcription (STAT), NF-κB, Hippo/yes-kinase-associated protein (hippo/YAP), and Wingless/int1 (WNT). These pathways are associated with numerous cellular processes linked to pancreatic cancer, such as apoptosis, angiogenesis, differentiation, immunological regulations, metabolism, migration, and cell proliferation. In addition, histone modification is a vital feature in pancreatic cancer for epithelial-to-mesenchyme transition [4][33][4,39]. The regulation and consideration of these pathways for pancreatic cancer can be helpful in developing novel targets and therapeutics. A key feature of pancreatic cancer includes the immunosuppressive tumor microenvironment [34][35][40,41]. Several molecular and cellular factors have been identified as critical players in the induction and maintenance of immunosuppression within the pancreatic stroma. The pancreatic stroma is composed of an extracellular matrix, immune cells, and fibroblasts that surround the tumor cells, forming a barrier that impedes cancer drug effect and immune cell infiltration [36][42] (Figure 1). Macrophages, which are a critical type of innate immune cells, play a significant role in immunomodulation in pancreatic cancer via a secreting range of cytokines [37][43]. Cytokines play a crucial role in tumor growth and immune cell evasion by promoting cancer cell invasion, proliferation, and immunosuppression [38][39][44,45]. Inflammatory cytokines such as IL-1β, IL-6, IL-8, and IL-10, have been demonstrated to activate tumor-associated macrophages (TAMs). The cytokines and chemokines attract immune cells, i.e., regulatory T cells (Treg cells), TAMs, and neutrophils, which impede CD8+ cytotoxic T cells function. Regulatory T cells (Tregs) have been associated with advancing pancreatic cancer by curbing cytotoxic T cells [40][46]. Pancreatic cancer cells coordinate immune evasion by synchronizing the secretion of cytokines in a highly coordinated way through TP53-dependent or KRAS-dependent pathways [41][47]. Tregs are vital in preserving immunologic self-tolerance and regulating suppression in pancreatic tumor growth.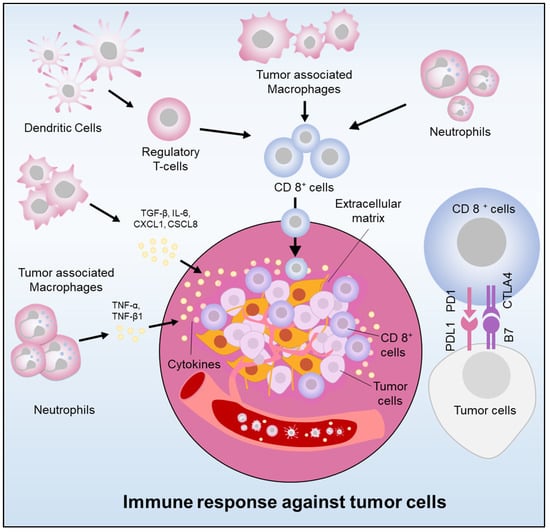
Figure 1. Immunoregulation in pancreatic cancer (Cancer cells elude the immune system by releasing cytokines and chemokines, recruiting immunosuppressive cells, and expressing PDL-1. Immune cell infiltration also stimulates tumor growth).

Figure 2.
Molecular pathways of pancreatic cancer.
