ThLiquis paper provides insight into the various studid crystals are generally referred to as substances that have already been carried out onblend the structure and properties of solid and liquid crystalline materials based on copper(I) complexes. Even though the study of copper(I) complexestates; they share with liquids the ability to flow but also exhibit some structural arrangement similarities with respect to theirsolids. With so many compounds synthesized, liquid crystalline property is quite few,s containing metals, also known as metallomesogens prepared with different structural components and ligands from groups such as aza macrocycles, alkyl thiolates, ethers, isocyanides, phenanthroline, Schiff bases, pyrazoles, phosphine, biquinoline, and benzoyl thiourea have, have become a major subject of study. The incorporation of metal into organic matrices enhances and induces unique magnetic, spectroscopic, and redox properties of the resulting materials. At least one liquid crystalline complex has been reported. A special section is dedicated to the discussion of the emission properties of copper(I) metallomesogens in the literature for most metals.
- copper(I)
- metallomesogens
- luminescence
- complex
- ligand
- Introduction
1. Introduction
LiquidCopper(I) crystals are generally referromplexes have not been studied to as substances that blend the structure and properties of solid and great extent as luminescent materials in the liquid states, they share with liquids, the ability to flow but alscrystalline state, but they do possess luminescence properties with great potentials [1][2][3][4][5][6][7][8][9][10]. Copper, exhibit some structural arrangement similarities with the solidswhich is somewhat abundant and affordable, is a suitable alternative to noble metal complexes [11][12]. The ratis intriguing combination of both the properties of the liquid and solid states gives liquid crystals the ability to induce certain properties that have gained useful applications in the displays of devices such as wristwatches, calculators, portable computers, and flat-screen televiso of triplet to singlet excitons is 3:1; consequently, for luminescent materials to be used in OLEDs, they should essentially be able to harvest all the excitons. Because copper(I) complexes exhibit various metal-to-ligand charge-transfer (MLCT) behaviors, they can induce spin orbital coupling of the triplet and singlet states, leading to small energy separations between the energy levels [13]. Thions as well as in sensors, smart windows, optical switches, etc.[1].
Wllows for reverse intersystem crossing (RISC), i.e., singlet harvesting, resulting in th so many compounds syntermally activated delayed fluorescence (TADF) [14]. Theresizedfore, liquid crystals containing metals, also known as metallomesogens,based on copper(I) complexes have become a major subject of study [2]. Incorporation of the metal into the organic matrix enhances and induces unique magnetic, spectroscopic, and redox properties of the resulting materials [3–9]. At least one liquid-crystallineconsiderable promise for producing effective luminescent materials for a wide range of optical or electro-optical applications due to the large diversity of possible structures, including mononuclear or polynuclear complex has been reported in the literature for most of the metals. However, issues often encountered with these complexes are those relating to the high transition temperatures usually >100 Ces, and the great potential of emission behavior. In addition, the range of coordination geometries (such as linear, pland the low thermal stability associat-trigonal, or tetrahedral) combined with the metallomesogens at elevated temperatures, which are major drawbacks that hinder the study of the physicalligands’ structural design provide a significant benefit for controlling the LC properties of these materials. Consequently, the preceding challenges make it puzzling to observe light emission (luminescence) at elevated temperatures due to the strong tendencies of the excited states t, including their enhanced thermal stability and mesophase type related to both calamitic and discotic materials.
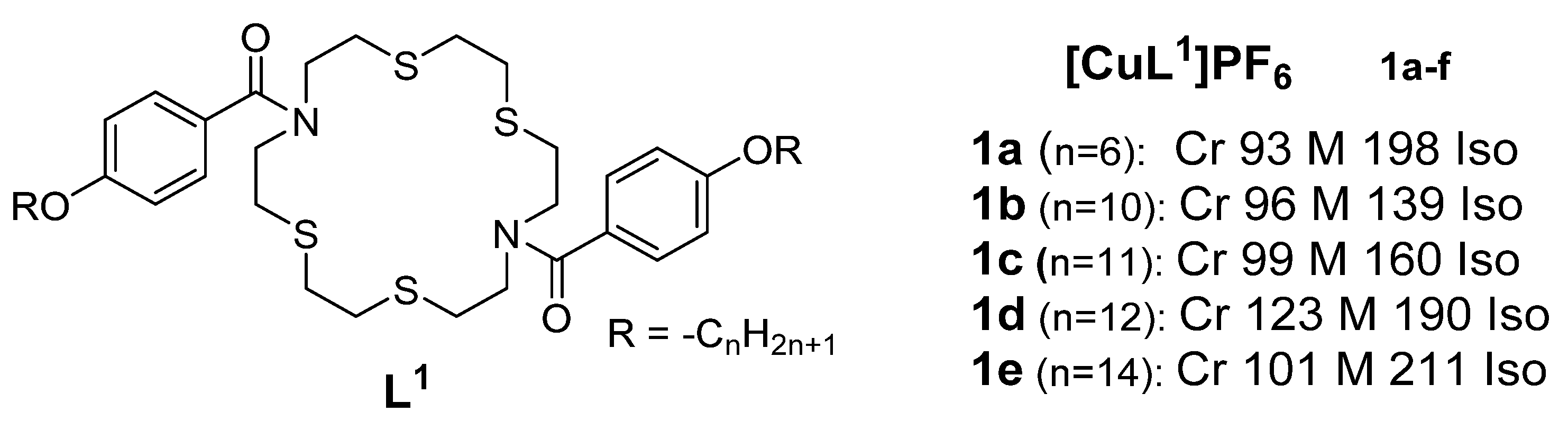
2. Copper(I) Metallomesogens with Sulfur-Containing Ligands
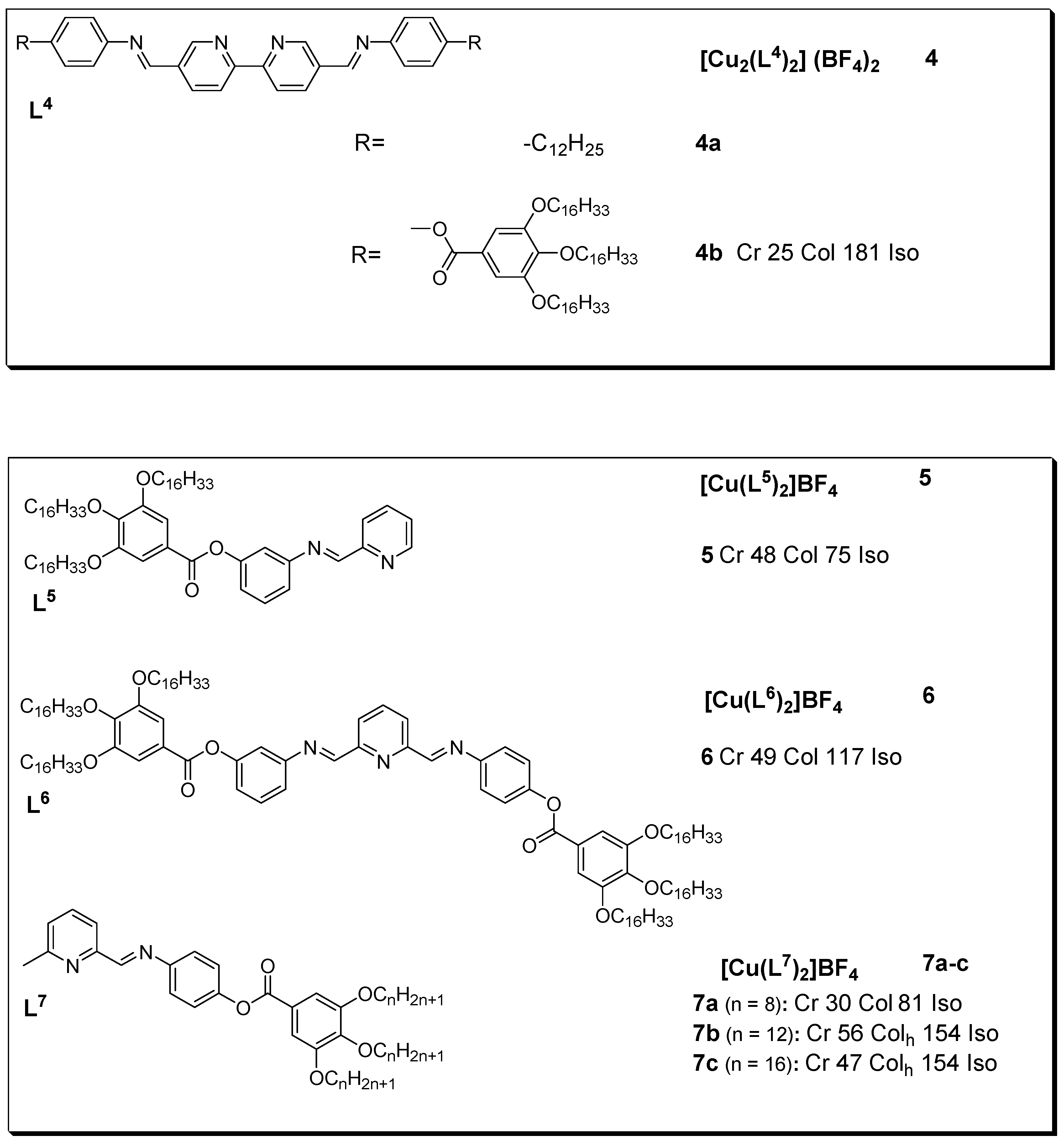
3. Copper(I) Metallomesogens with N-Donor Ligands
4. Copper(I) metallomesogens with isocyanide ligands
Isocyanides are a class of organic compounds of the type R-NC, where R is a combination of groups obtained by the removal of a hydrogen atom from an organic compound and the carbon therein is triply bonded to nitrogen whose site is also capable of coordinating with metals. The isocyanides are isomers of the nitriles [45]. These kinds of ligands have been used to design a large variety of transition metal complexes [46] and copper(I) metallomesogens are also well known for this. In general, the reaction of isocyanides with CuX (X is halide) gave a mononuclear complex but, on the contrary, the reaction of two equivalents of the isocyanide derivatives with CuX yielded the binuclear copper(I) complexes as depicted in Figure 6.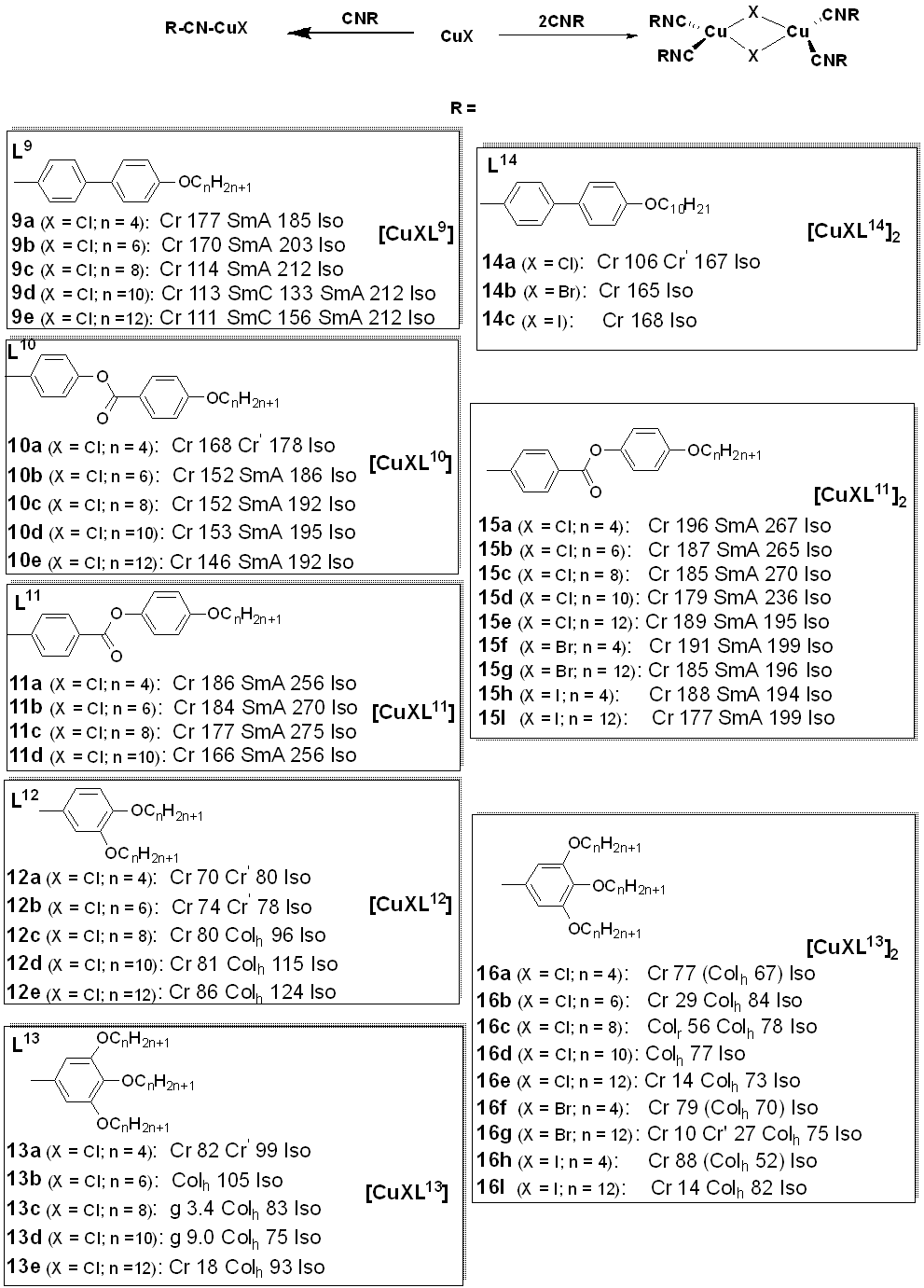
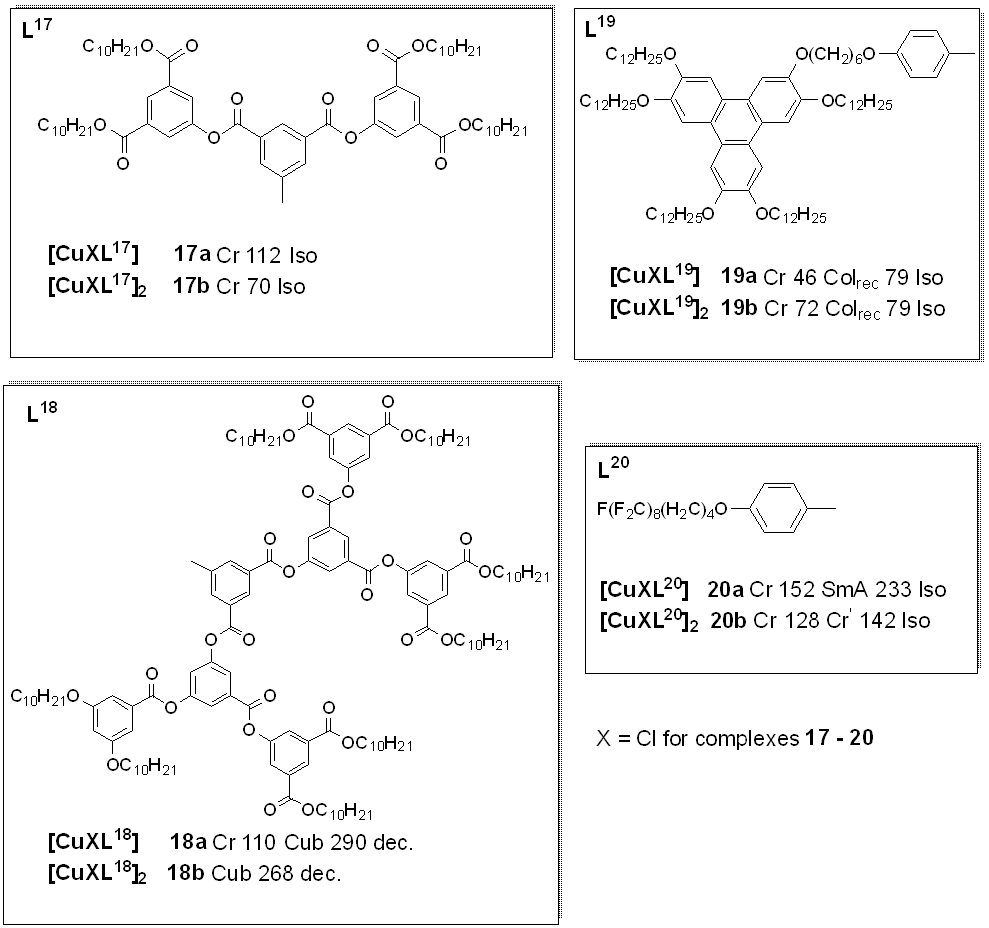
Figure 6. Mo undergo de-activation through nonradiative transitions. Theonuclear and dinuclear coppefore, luminescence studies on metallomesogens in many cases were performed on samples in t(I) isocyanide complexes. Transition temperatures are in °C [49–53].
The solfid state or those dissolved in organic solvents [10].
- Copper(I) metallomesogens with sulfur-containing ligands
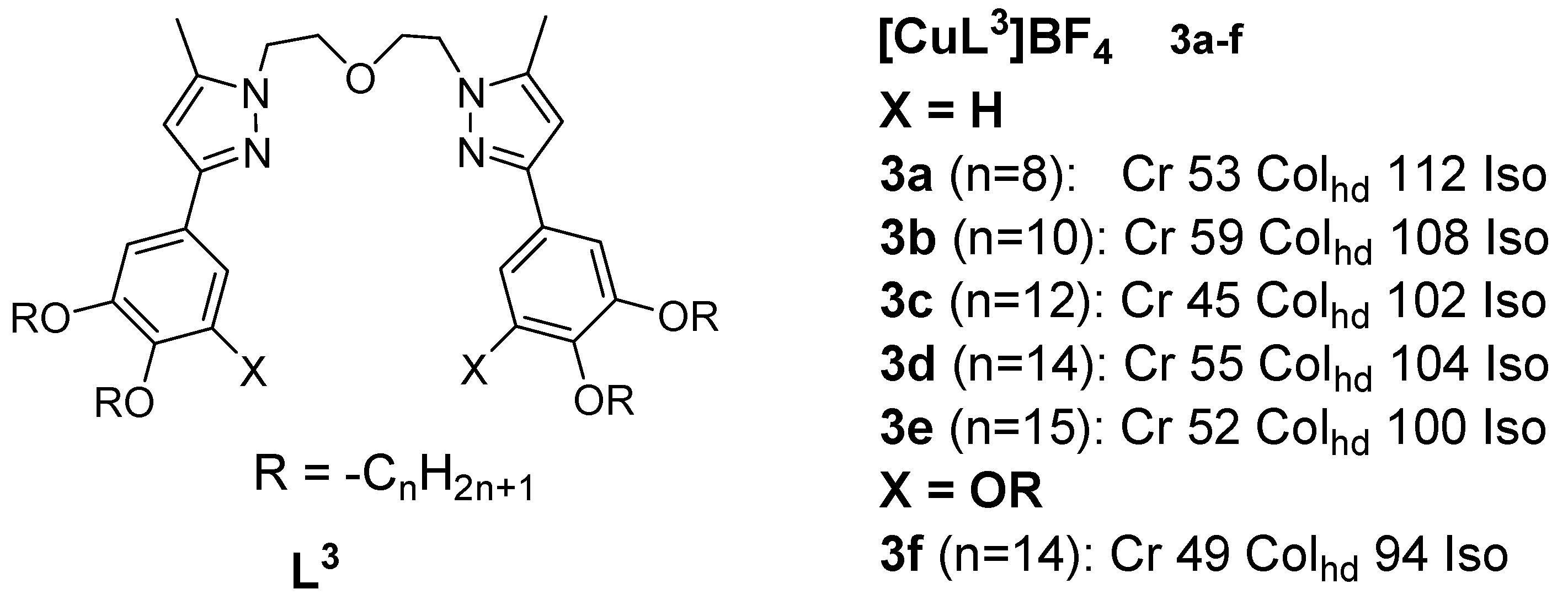
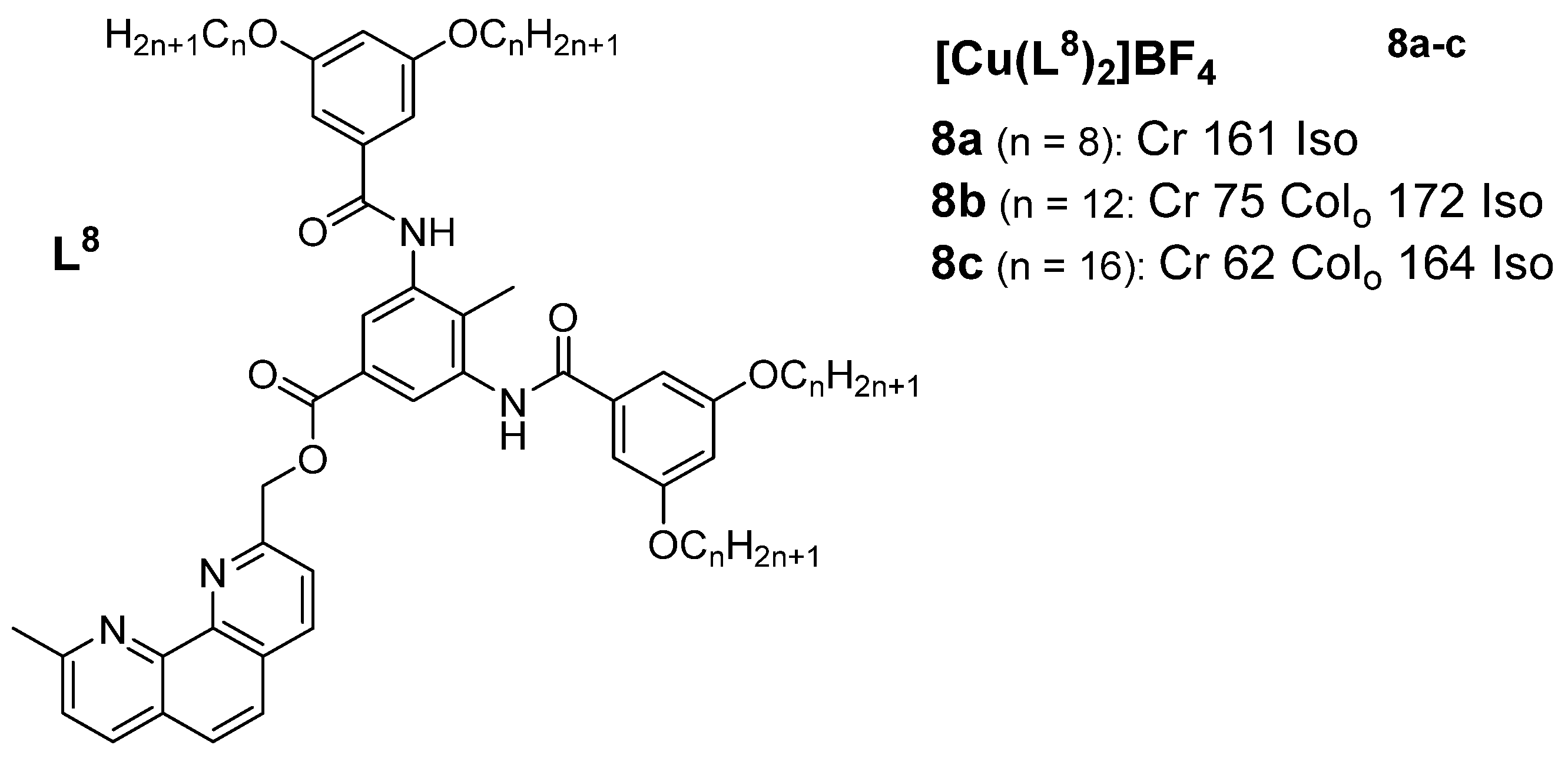
A rst set of liquid crystals baseriesd of cationic macrocyclic con copper(I) cisocyanide complexes based on non-mesogenic bis[4-(n-alkyloxy) benzamide derivatives of 1,10-diaza-4,7,13,16-tetrathiacyclooctadecanewas reported in 2001 by Benouazzane et al. [52]. Some of the isocyanide ligands (L9−11) were reported to in 1994 by Ghedinidisplay nematic and/or smectic et al.A [11]phases. The copper complexes (9a-e) studihowed had transition temperatures from the solid to liquid crystalline statSmA and SmC mesophases, while complexes r10b-e angingd from11a-d 93-123oC. XRD mweasurements for Complex found to display 1 (Figureonly 1)SmA in the series and thephases [52]. Further study revealed an X-ray diffraction pattern consistent with a disordered layered structure associated with a smectic phase of A or C type.
Figure 1: Cindicated that the range of the SmC phase increases and that of the SmA phase decreases as the length of the chain increases. All the copper(I) complexes from azamacrocycle derivative ligand. Transition temps.isocyanides reported in Figure 6 are mesogenic, except complexes 10a, are12a, in12b, (OC)13a, [11]
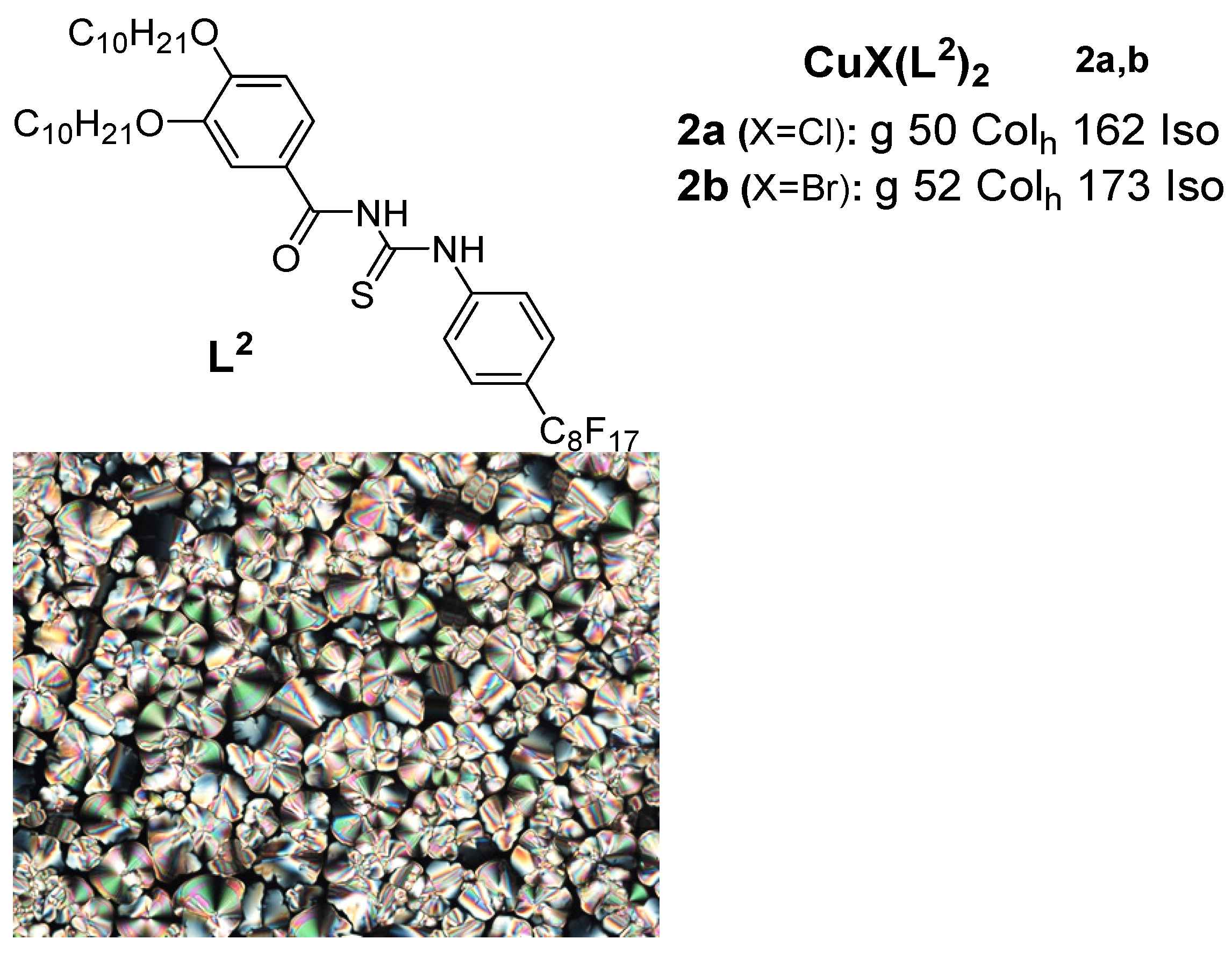
Another14a-c, cl20, 17a-b, ass ind 20b. tThe Sulphur containingisocyanide ligands groupL12 and L13, wis the benzoyl thiourea. An important class of copper(I) metalloth a single aromatic ring, did not appear to be mesogens based on ic, but upon complexation with copper(I) halid, all the complexes (except 13a with thiourea-based ligands having long chain alkoxy groups and a perfluorooctyl group was reported in 2018 by Ilis and Circu [12] (Figure 2). They found no shortest chain (n = 4)) showed liquid crystalline properbehavior.
Afty foer the ligand butsuccessful obseprved a hexagonal columnar phase for both eparation of stable linear copper(I) liquid crystals (complexes 2a9–13) with isocyand 2b ovider a high temperature above 100 C vligands reported ia a combination study of POM, DSC, and XRD, while the thermal stability studied by TGA indicated a higher stability (160oC) for th [52], in 2002, the same research group subsequently reported another se correspondingies of binuclear copper(I) complexes compared to (14–16) withat of the BTU (180oC) ligand.

Figure 2: Copmesogenic per(I) complexes with benzoyl thioureaoperties. The free isocyanide ligand. Transition temperatures are in (OC) [12].
- Copper(I) metallomesogens with N-donor ligands
Cos were reported as pper(I) metallomesogensic, with three-coordinate geometry were first reported by Linnematic and/or smectic et al.A in 2001 [13]phases. TheCopper complexes were14a–c derivedlack from bis{2-[3’-(3’’,4’’-dialkoxyphenyl)-5’-methyl-1’-pyrazolyl]ethyl} ethers and from bis{2-[3’-(3’’,4’,5’-trialkoxyphenyl)-5’-methyl-1’-pyrazolyl]ethyl} ethers. These novel mesogenic properties, whereas, copper complexes 15a-i were fobtained by complexing the ethers with [Cu(MeCN)4]BF4. Aund to be mesogenic. On the other hand, example (although the uncomplexed 3) is gocyaniven in Figude ligands (L13) are 3.
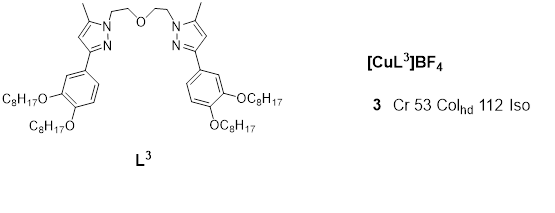
Figure 3. Cnopper(I)t complex with ether-type ligand. All temperatures are in (oC) [13]
Many liquid crystals, all their dinucoppler(I) complexes with N-donor ligands in the form of Schiff bases are known. Thesar copper isocyanide compounds slexes (16a-i) how varyingd liquid crystalline properties with modification of their structural units. Although non mesogenic, the free ligands L4, , displaying columnar mesophases [53]. These liquid crystalline binupon complexation with lear copper(I) showed mesogenic character as describcomplexes with isocyanide ligands reported by the DSC and POM experiments. The results indicated that complexBenouazzane et al. were the first examples 4 showedf high stability after several heating cycles as against those for some of the other Schiff base complexes, this attribute is explained to be due to the lack of substituents at position 6 (as seen in figure 3). In addition, the optical textures observed for complexliquid crystals with a core formed by two tetrahedral structures sharing an edge. Dendrimers based on isocyanides were first reported by Coco et al. (2008) [51]. The authors found that whereas all the free, 4 durhing slow cooling from the isotropic melt are typical ofhly branched isocyanide ligands (L17 and columnarL18) phase (with pseudo-focal-conic textures [14].

Figurend the metal complexes (17a and 4:17b) Coppwer(I) metallomesogens with Schiff base. All transition temperature not liquid crystals, complexes 18a are ind 18b (oC)showed [14].
Aa stcudy by Ziessebic mesophase.
Chico et al et. al. (2004) [15] presented mesomorphic materials based on0] reported two isocyano-triphenylene copper(I) complexes (19a and 19b), both of whith phenanthroline-based ligands. The study aimed to engineer a structural framework with additional supramolecular binding factors (hydrogen bonding) so as to stabilize the mesophase both as a free ligand and within ch displayed good thermal stability in the range of study. The free isocyanide ligand appeared not to be mesomorphic, as observed by POM. The identification of the complex. The thermotropic properties oflumnar mesophase for the lig19a and 19b was anchieved complexes of the phenanthroline derivatives were investigated via a combinaby small-angle X-ray scattering on powder samples, which was measured as a function of POM, DSC, and XRD. The ligand used ftemperature, consistent with the DSC and POM experiments. For these complexes showed distinct cubic and disordered lamell, the columnar mesophases at the related copper(I) complexes with longer chains showing mesomorphic behaviors displaying mesophases characteristic of the oblique columnar pwere stable in the temperature range of 46 to 79 °C.
Thase. An example of such compounds is described in Figure 5.
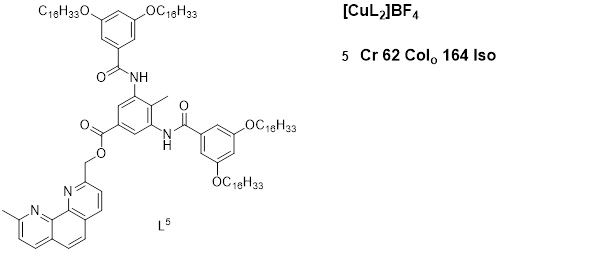
Figure 5: C effect of incorporating a fluopper(I) complex with phenanthroline-based ligand. Temperatures are in (OC) [15]
- Copper(I) metallomesogens with isocyanide ligands
The reinated chain in action of isocyanides ligand with CuX (X is halide) gives a mononuclear complex but, on the contrary, the reaction of two equivalents of the isocyaniderespect to its mesomorphic behavior when compared to its hydrocarbon derivatives with CuX yields binuclear copper(I) complexes, an example of metallomesogens from the aforementioned ligand is shown in Figures 6.

Figure 6: M was studied by Dembinski and coworkers [49]. Previously, several studies [54–56] had investigated the fluorophobic effect of single aromatic ring-cononucletar and dinuclear copper(I) Isocyanide complexes. Transition temperatures are in bracket (OC)ining organic molecules containing a perfluoroalkyl chain; as such, a [16] [17]
The firstudy set of liquid crystals based on copper(I) isocyanidewas carried out on fluorinated analogs of hydrocarbon complexes was reported in 2001 by Benouazzanein which mesomorphic behaviors had not been previously observed. et alThe [16]. Soseme of thei-perfluorinated isocyanide ligands (L20), werein reported to display nematic and/or smectic A phases. The mononuclear copper complex 6 (fcontrast to its alkyl analog, exhibited liquid crystalline propertigure 6) shows SmA and SmCs, showing a smectic A mesophases.
Chic upon et al. [17]both rheported two isocyanate-triphenylene copper(I) complexes boating and cooling. This was attributed to the fluorophobic effecth, of which display good thermal stability in the range of study. The free allowing for the mesogenicity of the isocyanide ligand appeared not to be mesomorphic as observed by POM. The identification of the columnar mesophase for the dincompound with a single benzene unit. The corresponding mononuclear copper complex 7 (figure 6) wtas achieved by small-angle X-ray scattering on powder samples which was measured as a function of ined the mesophase but with a high crystalline–mesophase transition temperature (152 °C), conswhistent with the DSC and POM experiments. Furthermore, the columnar mesophase was stable in the temperature range of 46 to 7le the dinuclear analog did not show liquid crystalline properties [49OC].
- Luminescent metallomesogens based on copper(I) complexes
5. Luminescent Metallomesogens Based on Copper(I) Complexes
A major drawback in the study of the physical properties of metallomesogens is massostly due tociated with issues relating to their high transition temperature and stability at those eelevated temperatures. It becomes increasingly difficult to study the emissions at thosesuch high temperatures due to the strong tendencies of the excited electrons to undergo deactivation via non-radiative transitions [102]. Few attempts have been made in this regard, and interesting findings have been madereported. The luminescence data of the copper(I) complexes discussed in this section are summarized in Table 1.
Table 1. Summary of luminescence data for copper(I) metallomesogens reported in [57–60].
Complex |
Phase (T/°C) |
λmaxem [λexc] (nm) |
t (µs) |
QY (%) |
|
|
21a |
Iso (80) |
650 [280] |
- |
- |
|
Cr (20) [na] |
640 [280] |
11 |
- |
|
|
Cr (20) [a] |
615 [280] |
9 |
- |
|
|
21b |
Iso (80) |
650 [280] |
- |
- |
|
Cr (20) [na] |
650 [280] |
15 |
- |
|
|
Cr (20) [a] |
610 [280] |
14 |
- |
|
|
22 |
Cr (20) [na] |
650 [280] |
7 |
- |
|
Cr (20) [a] |
640 [280] |
- |
- |
|
|
23a |
Colh (25) |
661 [290] |
28 |
42 |
|
23b |
Cr (25) |
664 [290] |
26 |
14 |
|
Colh (50) |
664 [290] |
22 |
- |
|
|
23c |
Cr (25) |
663 [290] |
21 |
24 |
|
24 |
SmA (25) |
404, 519 [280], 519 [400] |
- |
- |
|
g (-196) |
401, 487, 522 [280], 490, 522 [400] |
- |
9 |
|
|
24 [pg] |
SmA (25) |
385, 549 [280], 536 [400] |
- |
- |
|
|
g (-196) |
397, 494, 529 [280], 496, 529 [400] |
- |
1 |
|
25a |
Cr (25) |
578 [570] |
- |
2.4 |
|
Lcolg (50) |
- |
- |
0.7 |
|
|
Lcol (80) |
- |
- |
0.2 |
|
|
Lcol (100) |
- |
- |
0.06 |
|
|
25b |
Cr (25) |
578 [570] |
- |
2.2 |
|
Lcolg (50) |
- |
- |
0.5 |
|
|
Lcol (80) |
- |
- |
0.2 |
|
|
Lcol (100) |
- |
- |
0.04 |
|
|
26a |
Cr (25) |
560 [570] |
- |
1.5 |
|
Colh (50) |
- |
- |
0.4 |
|
|
Colh (80) |
- |
- |
0.1 |
|
|
Iso (100) |
- |
- |
<0.01 |
|
|
26b |
Cr (25) |
560 [570] |
- |
1.1 |
|
Colh (50) |
- |
- |
0.4 |
|
|
Colh (80) |
- |
- |
0.2 |
|
|
Iso (100) |
- |
- |
<0.01 |
t = averaged lifetime; QY = absolute quantum yield; λem = wavelength of maximum emission; λexc = excitation wavelength; na = non-aged; a = aged; Pg = ground sample; SmA = smectic A phase; g = glassy state; Cr = crystalline phase; Iso = isotropic liquid; Lcol = lamello-columnar phase; Colh = columnar hexagonal phase; Lcolg = frozen lamello-columnar phase
Kishimura et et alal. were the first to describe the emission properties of copper(I) complexes in their liquid crystalline phase [1857] in 2005. They reported a number of dendritic Cucopper(I) pyrazolate complexes 21 and 22(Figure 7), which were used to produce some thermally rewritable phosphorescent papers useful for security purposes.
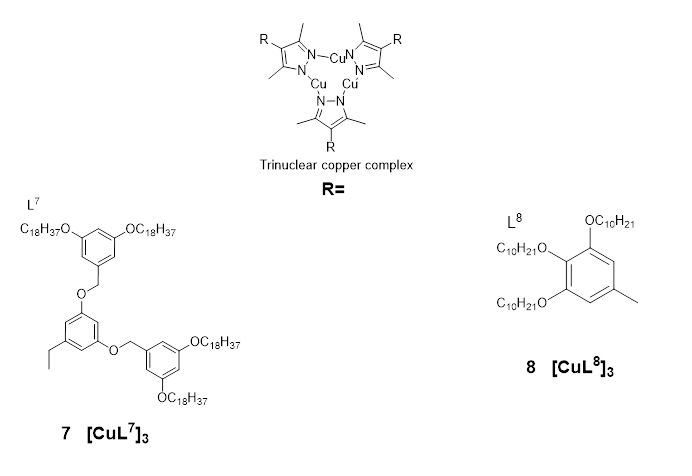
Figure 7:. Ligands and representative structure of the copper pyrazole atrinucleare complexes 7 & 8 reported in [157,58] [19]
.
The luminescence investigation of complexes from the pyrazolate ligands L21 [1857] revealed dichroism at room temperature for the solid form of complex 721a. Cooling of the hot melt (which emitted a red luminescence at λmax650 nm) naturally or by slow cooling leads to blue -shift emission at 640 or640 and 610 nm respectively. In essence, the red and yellow luminescence observed for 721a could be thermally changed from one form to the other, depending on the manner and rate of cooling, and thiswhich was also found to be the case for its liquid crystalline properties. Both the aged and non-agednalyze complex (21a), 7 both in at was analyzed ged and non-aged form, appeared to be phosphorescent, which is thought to be a result of Cu(I) to Cu(I) interactions [1857].
The photoluminescent data are summarized in Table 1.
Furthermore, XRD analysis of the aged sample of complex 721a showed diffraction patterns synonymous with a one-dimensional columnar phase, and the same, complex 7(21a) viewed iunder a polarized optical microscope showed a fan-shaped texture that is characteristic of Ddiscotic liquid crystals and on the basis of this,; accordingly, it was concluded that the aged complex 7 i(21a) was composed of long-range discotic columnar assembly. The XRD pattern of the non-aged complex after natural cooling also indicated the presence of a columnar structure. DSC measurements carried out on the aged and non-aged complex 7 (21a) revealed patterns, based upon which it was concluded that the discotic columnar assembly, which is believed to involve metallophilic interactions of the Cu(I) to Cu(I) units havingwith long alkyl chains, is formed in the aging process at about 40–50 °C. The stability of the dichroic luminescence was therefore found to be dependent on the pyrazolate ligand structure.
B Complex 21b exhibited similar phosphorescent properties as complex 21b upon rapildind and slow cooling of its hot melt; however, the red luminescence turned yellow spontaneously, even at very low temperatures. Complex 22 (Figure 7), upon thewhich has more dendritic units than the other studied complexes, did not show clear luminescence dichroism, and aging of the complex (22) by slow cooling of its hot melt resulted in only afo 10 nm red shift of the luminescence [57].
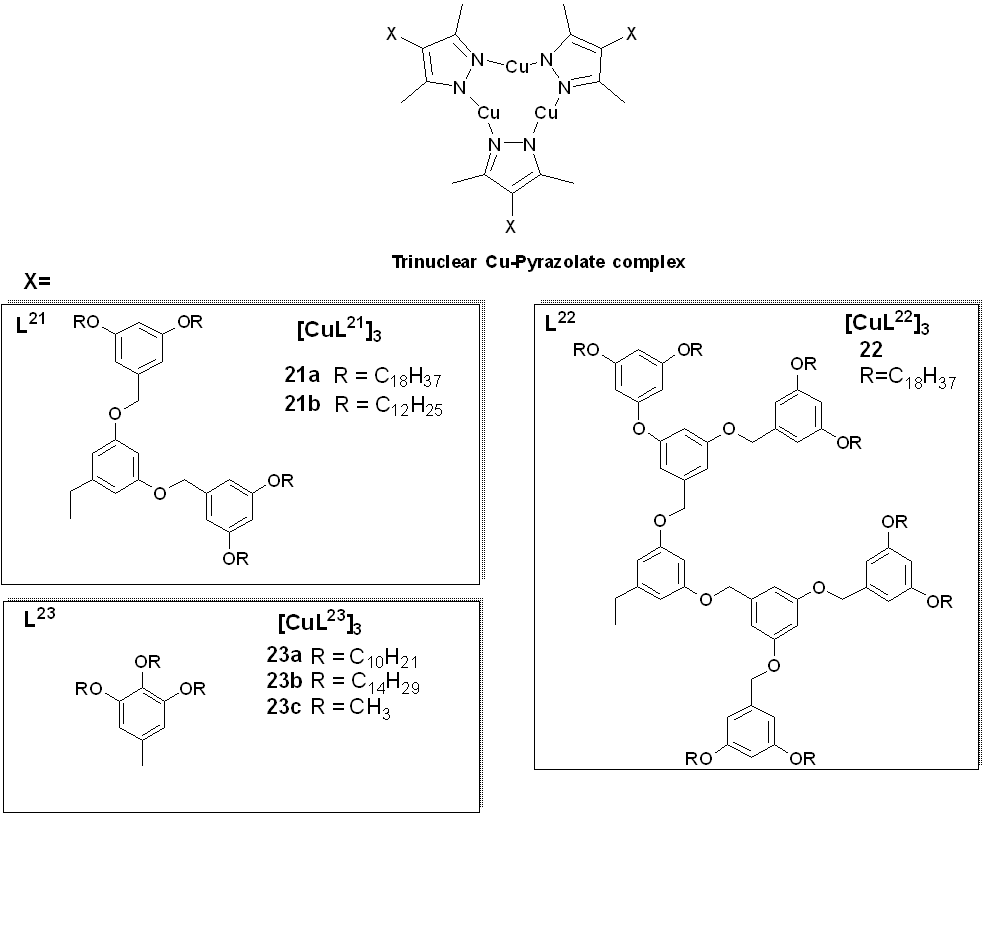
Building on the aforementioned work by Kishimura and co-workers, Gimenez et al. (2020) [1958] recently reported a series of liquid crystals achieved with cyclic trinuclear copper(I) complexes (23a and 23b) prepared using 3,5-dimethyl-4-(trialkoxyphe most important inyl) pyrazolate ligands (Figure 7). The compounds displayed well-organized hexagonal columnar mesophases, which were found to be stable at room temperature or near room temperature (Figure 8).
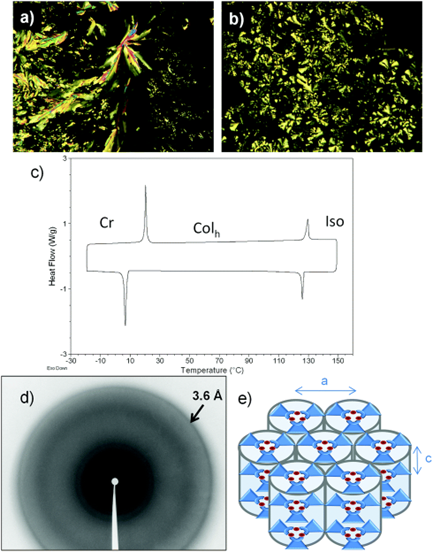
Figure 8. (a) Microphotograph of the region bethe series is given in Figure 7ween crossed polarizers observed for the texture of the Colh phase of 23a upon coomplexling of the isotropic liquid at 126 °C. 8(b), Microprepared uhotograph of the region between crossed polarizers observed for the texture of the Colh phase of 23b upon cooling of 3,5-dithe isotropic liquid at 98 °C. (c) DSC thermogram of 23a. (d) XRD patterns of thye Col-4-(th phase of 23a. The arrow ialkoxyphenyndicates the halo corresponding to the stacking distance. (e) Self-assembl)pyrazolate ligands in the hexagonal columnar mesophase. (Reproduced from ref. [58] with permission from the Royal Society of Chemistry).
C
Both complexes 8 23a and 23b wasere reported to have orange-red –red-colored emissions at room temperature, and the photoluminescence exhibited a broad band centered at around 661-–664 nm (Figure 9). A landmark reported in this study [1958] is the high Qquantum yield (QY) value of 42%, measured in the liquid crystalline state, which is the highest recorded so farto date for any copper(I) metallomesogens. The QY obtained for complex23a 7 in its liquid crystalline state was higher than thoseat obtained in the crystalline states of complexes for the rest of the series23b and 23c. Thus, the work carried out by Giminez et al. [19] have shown58] showed that low-temperature phosphorescent metallomesogens can be obtained from the more affordable and abundant copper metal.
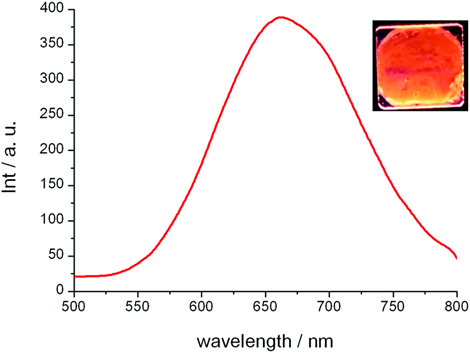
Figure 9. Photoluminescence spectrum in the columnar mesophase for 23a at 25 °C (excitation wavelength, 290 nm) and emission of the film observed under irradiation with a 254 nm handheld lamp (reproduced from ref. [58 with permission from the Royal Society of Chemistry).
Camerel et et al. al. (2016) [2059] reported a new class of copper(I) liquid crystals with a cubane core and based on phosphine ligands functionalized with pro mesogenic gallate-based moieties bearing either long alkyl chains of C8, C12, and C16 or cyanobiphenyl (CBP) fragments. The c (Figure 10). Copper(I) cubanes have already been are known for being abletheir ability to display both luminescence mechanochromism and thermochromism behavior [21–261–65].
This study is an inventive example of integrating luminescence characteristics of copper iodide clusters [Cu4I4(L24)4] with the flexible self-assembly of liquid crystals. Only the compound functionalized with a cyano biphenyl group (CBP), i.e., Complex 8 (figure24, 8) showed liquid crystalline behaviourr, displaying a Sm An SmA mesophase from room temperature to about 1000 °C.
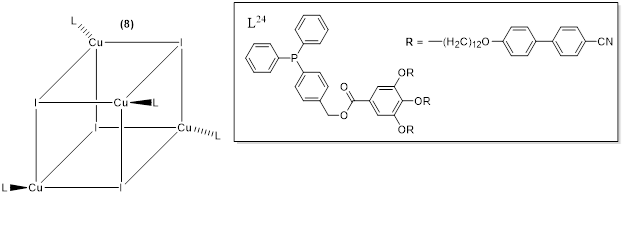
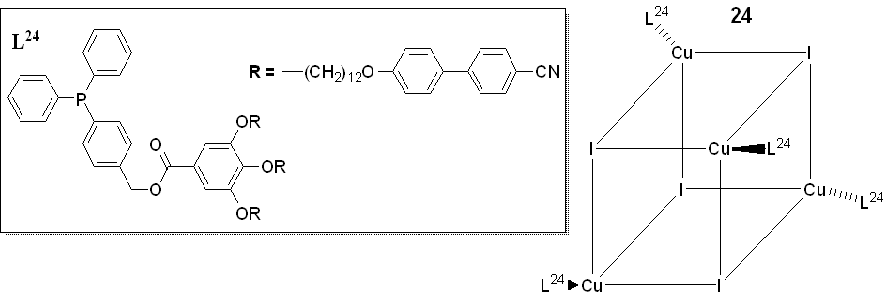
Figure 810. General structure of the functionalized [Cu4I4(L24)4] copper iodide clusters with phosphine ligands [2059].
All the complexes from the work by Camerel et
All the complexes reported in the work by Camerel et al.
al.
[59] revealed luminescence thermochromism for all the studied compounds; however, complex 24 displayed an unclassical behavior, which was attributed to the intrinsic luminescence properties of the cyanobiphenyl moiety itself, with a dual-emissive system that presented interesting emission properties. In addition to its liquid crystalline properties, compound 24 displayed luminescence mechanochromism with a modification of the emission wavelength in response to grinding (Figure 11).
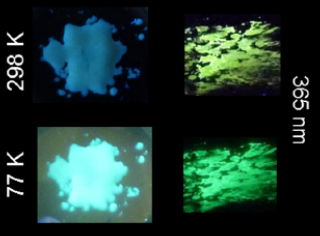
Figure 11. Photos of 24 under 365 nm (UV lamp) before and after grinding at 298 and 77 K. Reprinted (adapted) with permission from Chem. Mater. 2016, 28, 22, 8190–8200 (ref. [59]). Copyright 2016 American Chemical Society.
Cretu et al.
[20] revealed
(2018) [60] reported a new class of Cu(I) coordination complexes (25a, 25b, 26a, and 26b) with 4,4’-bisubstituted-2,2’-biquinolines as depicted in figure 12, which showed low-temperature lamello-columnar and columnar hexagonal thermotropic liquid crystalline phases. The highest deduction from the luminescence study is the presence of a medium–low-intensity band with a series of shoulders at 578 nm for 25a and 25b and at 560 nm for 26a and 26b, which is believed to be due to metal-to-ligand charge-transfer (MLCT) electronic transitions. When the solid samples were heated, they moved towards the liquid crystalline phases and retained the luminescence displayed in the solid state; however, with increased temperature, the intensity of the luminescence band decreased, and at temperatures over 120 °C, luminescence was completely quenched, although reversed with subsequent cooling of the samples. This behavior is attributed to the gain of the non-radiative kinetic constants when vibrational modes were enhanced by heating the samples [60].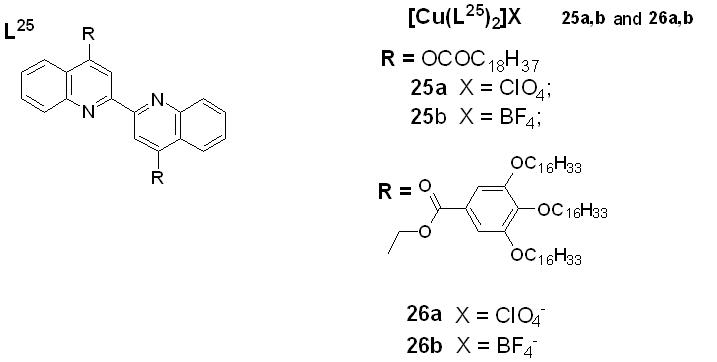
Figure 12.
luminescence t
Copper(I) complexes with bisubstituted biquinoline ligands [60].
6.Conclusions
Thermochromism for all the studied compounds however, complex 8 d revisplayed an unclassical behavior which was attributed to the intrinsic luminescence properties of the cyano biphenyl moiety itself, complex 8 displayed a dual-emissiw prove system which presented interesting emission properties. And also, in addition to its liquid crystalline properties, the compound 8 displayed luminescence mechanochromism with a modification of the emission wades an ovelength in response to grinding.
Cretu et al. (2018) [26] reported a new class of Cu(I) coordination complexes harving 4,4’-bisubstituted-2,2’-biquinolines which shows low-temperature lamella-columnar and columnar hexagonal thermotropic liquid crystalline phases. The highest deduction from the luminescence study is the presence, at 578 and 560 nm of a medium-low intensity band, with a series of shoulders, believed to be due to metal-to-ligand charge-transfer (MLCT) electronic transitions. By heating the solid samples, they move towards the liquid-crystalline phases and still retain the luminescence displayed in the solid state but by increasing the temperature, the intensity of the luminescence band decreases, and when over 120°C, luminescence is completely quenched; but returns with subsequent cooling of the samples. This behavior is attributed to the gain of the non-radiative kinetic constants when vibrational modes are enhanced by heating the samples [26]. An example of this class’s structure is given in Figure 9.
Figure 9. Cw of the different strategies imagined by chemists to control the LC properties of copper(I) complex with bisubstituted biquinoline ligand [26]
Conclusion
The aim of this, article was to make an overview of the various liquid crystalline materials based on copper(I) complexes with an emphasis on theifocusing on their luminescent properties. Examples of copper(I) metallomesogens based on isocyanide ligands are, by far, the most prevalent. By utilizing the suitable mesogenic groups, their LC characteristics maycan be simply modified. While a few other isocyanides produced hexagonal and rectangular columnar phases, as well as a cubic phase for the dendritic isocyanide supermolecules, the bulk of these complexes exhibit calamitic behavior with either SmA or SmC phases. The reported copper(I) complexes of Schiff base -base-type ligands of bipyridine imine, 2-a-, 2-iminopyridine-, and picoline -substituted imino ligands and those of alkyl thiolates all displayed varying columnar mesophases with different transition temperature ranges. Both the ether type and benzoyl thiourea ligands displayed characteristics of a hexagonal columnar mesophase for the corresponding metallomesogens, while the copper(I) complexes from phenanthroline ligands had an oblique columnar mesophase. Furthermore, interesting luminescent properties in liquid crystalline states were observed for copper(I) complexes from ligands with pyrazolate derivatives, phosphine ligands functionalized with copper iodide and s, and those of bisubstituted biquinoline ligands. The complex from the functionalized phosphine ligand displayed interesting mechanochromic luminescence propertiecharacteristics. A high quantum yield of 42% in the liquid crystalline phase of the copper(I) metallomesogens from a pyrazolate ligand was reported. This work provides a pillar to build on basis for the design and preparation of many more new multifunctional materials based on more copper(I) complexes havingwith liquid crystalline behavior withand improved properties.
References
- Handbook of Liquid Crystals. Handbook of Liquid Crystals 2014, doi:10.1002/9783527671403.
- Date, R.W.; Iglesias, E.F.; Rowe, K.E.; Elliott, J.M.; Bruce, D.W. Metallomesogens by Ligand Design. Dalton Transactions 2003, 10, 1914–1931, doi:10.1039/B212610A.
- Flora, S.J.S. Chelation Therapy. Comprehensive Inorganic Chemistry II (Second Edition): From Elements to Applications 2013, 3, 987–1013, doi:10.1016/B978-0-08-097774-4.00340-5.
- Gilli, J.M.; Thiberge, S.; Manaila-Maximean, D. New Aspect of the Voltage/Confinement Ratio Phase Diagram for a Confined Homeotropic Cholesteric. http://dx.doi.org/10.1080/15421400490478858 2010, 417, doi:10.1080/15421400490478858.
- Micutz, M.; Iliş, M.; Staicu, T.; Dumitraşcu, F.; Pasuk, I.; Molard, Y.; Roisnel, T.; Cîrcu, V. Luminescent Liquid Crystalline Materials Based on Palladium(II) Imine Derivatives Containing the 2-Phenylpyridine Core. Dalton Transactions 2013, 43, 1151–1161, doi:10.1039/C3DT52137K.
- Al-Karawi, A.J.M. From Mesogens to Metallomesogens. Synthesis, Characterisation, Liquid Crystal and Luminescent Properties. https://doi.org/10.1080/02678292.2017.1371799 2017, 44, 2285–2300, doi:10.1080/02678292.2017.1371799.
- Cuerva, C.; Cano, M.; Lodeiro, C. Advanced Functional Luminescent Metallomesogens: The Key Role of the Metal Center. Chem Rev 2021, 121, 12966–13010, doi:10.1021/ACS.CHEMREV.1C00011.
- Thaker, B.; Limbachiya, N.; Patel, K.; Patel, N. Calamitic Liquid Crystals Involving Fused Ring and Their Metallomesogens. Emerging Materials Research 2017, 6, 331–347, doi:10.1680/JEMMR.15.00060/ASSET/IMAGES/SMALL/JEMMR6-0331-F10.GIF.
- Zou, G.; Zhang, S.; Feng, S.; Li, Q.; Yang, B.; Zhao, Y.; Luo, K.; Wen, T. Bin Cyclometalated Platinum(II) Metallomesogens Based on Half-Disc-Shaped β-Diketonate Ligands with Hexacatenar: Crystal Structures, Mesophase Properties, and Semiconductor Devices. Inorg Chem 2022, 61, 11702–11714, doi:10.1021/ACS.INORGCHEM.2C01327.
- Binnemans, K. Luminescence of Metallomesogens in the Liquid Crystal State. J Mater Chem 2009, 19, 448–453, doi:10.1039/B811373D.
- Neve, F.; Ghedini, M.; Levelut, A.M.; Francescangeli, O. Ionic Metallomesogens. Lamellar Mesophases in Copper(I) Azamacrocyclic Complexes. Chemistry of Materials 1994, 6, 70–76, doi:10.1021/CM00037A016/ASSET/CM00037A016.FP.PNG_V03.
- Iliş, M.; Cîrcu, V. Discotic Liquid Crystals Based on Cu(I) Complexes with Benzoylthiourea Derivatives Containing a Perfluoroalkyl Chain. J Chem 2018, 2018, doi:10.1155/2018/7943763.
- Lin, H. da; Lai, C.K. Ionic Columnar Metallomesogens Formed by Three-Coordinated Copper(I) Complexes. Journal of the Chemical Society, Dalton Transactions 2001, 2383–2387, doi:10.1039/B103427H.
- Douce, L.; El-Ghayoury, A.; Skoulios, A.; Ziessel, R. Columnar Mesophases from Tetrahedral Copper( I ) Cores and Schiff-Base Derived Polycatenar Ligands. Chemical Communications 1999, 0, 2033–2034, doi:10.1039/A904245H.
- Ziessel, R.; Pickaert, G.; Camerel, F.; Donnio, B.; Guillon, D.; Cesario, M.; Prangé, T. Tuning Organogels and Mesophases Will Phenanthroline Ligands and Their Copper Complexes by Inter- to Intramolecular Hydrogen Bonds. J Am Chem Soc 2004, 126, 12403–12413, doi:10.1021/JA047091A/SUPPL_FILE/JA047091ASI20040708_044005.PDF.
- Benouazzane, M.; Coco, S.; Espinet, P.; Barberá, J. Supramolecular Organization in Copper(I) Isocyanide Complexes: Copper(I) Liquid Crystals from a Simple Molecular Structure. J Mater Chem 2001, 11, 1740–1744, doi:10.1039/B100548K.
- Chico, R.; de Domingo, E.; Domínguez, C.; Donnio, B.; Heinrich, B.; Termine, R.; Golemme, A.; Coco, S.; Espinet, P. High One-Dimensional Charge Mobility in Semiconducting Columnar Mesophases of Isocyano-Triphenylene Metal Complexes. Chemistry of Materials 2017, 29, 7587–7595, doi:10.1021/ACS.CHEMMATER.7B02922/ASSET/IMAGES/LARGE/CM-2017-029226_0009.JPEG.
- Kishimura, A.; Yamashita, T.; Yamaguchi, K.; Aida, T. Rewritable Phosphorescent Paper by the Control of Competing Kinetic and Thermodynamic Self-Assembling Events. Nat Mater 2005, 4, 546–549, doi:10.1038/NMAT1401.
- Giménez, R.; Crespo, O.; Diosdado, B.; Elduque, A. Liquid Crystalline Copper(i) Complexes with Bright Room Temperature Phosphorescence. J Mater Chem C Mater 2020, 8, 6552–6557, doi:10.1039/d0tc00642d.
- Huitorel, B.; Benito, Q.; Fargues, A.; Garcia, A.; Gacoin, T.; Boilot, J.P.; Perruchas, S.; Camerel, F. Mechanochromic Luminescence and Liquid Crystallinity of Molecular Copper Clusters. Chemistry of Materials 2016, 28, 8190–8200, doi:10.1021/ACS.CHEMMATER.6B03002/SUPPL_FILE/CM6B03002_SI_001.PDF.
- Benito, Q.; Baptiste, B.; Polian, A.; Delbes, L.; Martinelli, L.; Gacoin, T.; Boilot, J.P.; Perruchas, S. Pressure Control of Cuprophilic Interactions in a Luminescent Mechanochromic Copper Cluster. Inorg Chem 2015, 54, 9821–9825, doi:10.1021/ACS.INORGCHEM.5B01546/SUPPL_FILE/IC5B01546_SI_002.CIF.
- Tsuge, K.; Chishina, Y.; Hashiguchi, H.; Sasaki, Y.; Kato, M.; Ishizaka, S.; Kitamura, N. Luminescent Copper(I) Complexes with Halogenido-Bridged Dimeric Core. Coord Chem Rev 2016, 306, 636–651, doi:10.1016/J.CCR.2015.03.022.
- Volz, D.; Zink, D.M.; Bocksrocker, T.; Friedrichs, J.; Nieger, M.; Baumann, T.; Lemmer, U.; Bräse, S. Molecular Construction Kit for Tuning Solubility, Stability and Luminescence Properties: Heteroleptic MePyrPHOS-Copper Iodide-Complexes and Their Application in Organic Light-Emitting Diodes. Chemistry of Materials 2013, 25, 3414–3426, doi:10.1021/CM4010807/SUPPL_FILE/CM4010807_SI_001.PDF.
- Liu, Z.; Qiu, J.; Wei, F.; Wang, J.; Liu, X.; Helander, M.G.; Rodney, S.; Wang, Z.; Bian, Z.; Lu, Z.; et al. Simple and High Efficiency Phosphorescence Organic Light-Emitting Diodes with Codeposited Copper(I) Emitter. Chemistry of Materials 2014, 26, 2368–2373, doi:10.1021/CM5006086/SUPPL_FILE/CM5006086_SI_001.PDF.
- Cariati, E.; Lucenti, E.; Botta, C.; Giovanella, U.; Marinotto, D.; Righetto, S. Review. Coord Chem Rev 2016, Part 2, 566–614, doi:10.1016/J.CCR.2015.03.004.
26. Cretu, C.; Alumindelescu, A.A.; Candreva, A.; Crispini, A.; Szerb, E.I.; la Deda, M. Bisubstituted-Biquinoline Cu(I) Complexes: Synthesis, Mesomorphism and Photophysical Studies in Solution and Condensed Statesent properties. J Mater Chem C Mater 2018, 6, 10073–10082, doi:10.1039/C8TC02999G.
References
- Tudor, C.A.; Iliş, M.; Secu, M.; Ferbinteanu, M.; Cîrcu, V. Luminescent heteroleptic copper(I) complexes with phosphine and N-benzoyl thiourea ligands: Synthesis, structure and emission properties. Polyhedron 2022, 211, 115542.
- Favarin, L.R.V.; Rosa, P.P.; Pizzuti, L.; Machulek, A.; Caires, A.R.L.; Bezerra, L.S.; Pinto, L.M.C.; Maia, G.; Gatto, C.C.; Back, D.F.; et al. Synthesis and structural characterization of new heteroleptic copper(I) complexes based on mixed phosphine/thiocarbamoyl-pyrazoline ligands. Polyhedron 2017, 121, 185–190.
- Chan, K.C.; Cheng, S.C.; Lo, L.T.L.; Yiu, S.M.; Ko, C.C. Luminescent Charge-Neutral Copper(I) Phenanthroline Complexes with Isocyanoborate Ligand. Eur. J. Inorg. Chem. 2018, 2018, 897–903.
- Bergmann, L.; Friedrichs, J.; Mydlak, M.; Baumann, T.; Nieger, M.; Bräse, S. Outstanding luminescence from neutral copper(i) complexes with pyridyl-tetrazolate and phosphine ligands. Chem. Commun. 2013, 49, 6501–6503.
- Enikeeva, K.R.; Shamsieva, A.V.; Strelnik, A.G.; Fayzullin, R.R.; Zakharychev, D.V.; Kolesnikov, I.E.; Dayanova, I.R.; Gerasimova, T.P.; Strelnik, I.D.; Musina, E.I.; et al. Green Emissive Copper(I) Coordination Polymer Supported by the Diethylpyridylphosphine Ligand as a Luminescent Sensor for Overheating Processes. Molecules 2023, 28, 706.
- Tsuge, K.; Chishina, Y.; Hashiguchi, H.; Sasaki, Y.; Kato, M.; Ishizaka, S.; Kitamura, N. Luminescent copper(I) complexes with halogenido-bridged dimeric core. Coord. Chem. Rev. 2016, 306, 636–651.
- Sun, Y.; Lemaur, V.; Beltrán, J.I.; Cornil, J.; Huang, J.; Zhu, J.; Wang, Y.; Fröhlich, R.; Wang, H.; Jiang, L.; et al. Neutral Mononuclear Copper(I) Complexes: Synthesis, Crystal Structures, and Photophysical Properties. Inorg. Chem. 2016, 55, 5845–5852.
- Borges, A.P.; Carneiro, Z.A.; Prado, F.S.; Souza, J.R.; Silva, L.H.F.E.; Oliveira, C.G.; Deflon, V.M.; de Albuquerque, S.; Leite, N.B.; Machado, A.E.H.; et al. Cu(I) complexes with thiosemicarbazides derived from p-toluenesulfohydrazide: Structural, luminescence and biological studies. Polyhedron 2018, 155, 170–179.
- Brown, C.M.; Li, C.; Carta, V.; Li, W.; Xu, Z.; Stroppa, P.H.F.; Samuel, I.D.W.; Zysman-Colman, E.; Wolf, M.O. Influence of Sulfur Oxidation State and Substituents on Sulfur-Bridged Luminescent Copper(I) Complexes Showing Thermally Activated Delayed Fluorescence. Inorg. Chem. 2019, 58, 7156–7168.
- Bergmann, L.; Braun, C.; Nieger, M.; Bräse, S. The coordination- and photochemistry of copper(i) complexes: Variation of N^N ligands from imidazole to tetrazole. Dalton Trans. 2018, 47, 608–621.
- Li, X.; Xie, Y.; Li, Z. Diversity of Luminescent Metal Complexes in OLEDs: Beyond Traditional Precious Metals. Chem. Asian J. 2021, 16, 2817–2829.
- Dumur, F. Recent advances in organic light-emitting devices comprising copper complexes: A realistic approach for low-cost and highly emissive devices? Org. Electron. 2015, 21, 27–39.
- Yersin, H.; Czerwieniec, R.; Shafikov, M.Z.; Suleymanova, A.F. TADF Material Design: Photophysical Background and Case Studies Focusing on Cu(I) and Ag(I) Complexes in Highly Efficient OLEDs: Materials Based on Thermally Activated Delayed Fluorescence; Yersin, H., Ed.; Wiley-VCH: Hoboken, NJ, USA, 2018; pp. 1–60.
- Housecroft, C.E.; Constable, E.C. TADF: Enabling luminescent copper(i) coordination compounds for light-emitting electrochemical cells. J. Mater. Chem. C 2022, 10, 4456–4482.
- Neve, F.; Ghedini, M.; Levelut, A.-M.; Francescangeli, O. Ionic metallomesogens. Lamellar mesophases in copper(I) azamacrocyclic complexes. Chem. Mater. 1994, 6, 70–76.
- Espinet, P.; Carmen Lequerica, M.; Martín-Alvarez, J.M.Â. Synthesis, Structural Characterization and Mesogenic Behavior of Copper(I) n-Alkylthiolates. Chem. Eur. J. 1999, 5, 1982–1986.
- Dance, I.G.; Fisher, K.J.; Banda, R.M.H.; Scudder, M.L. Layered Structure of Crystalline Compounds AgSR. Inorg. Chem. 1991, 30, 183–187.
- Iliş, M.; Cîrcu, V. Discotic Liquid Crystals Based on Cu(I) Complexes with Benzoylthiourea Derivatives Containing a Perfluoroalkyl Chain. J. Chem. 2018, 2018, 7943763.
- Lin, H.-D.; Lai, C.K. Ionic columnar metallomesogens formed by three-coordinated copper(I) complexes. J. Chem. Soc. Dalton Trans. 2001, 2383–2387.
- Raczuk, E.; Dmochowska, B.; Samaszko-Fiertek, J.; Madaj, J. Different Schiff Bases—Structure, Importance and Classification. Molecules 2022, 27, 787.
- Khalaji, A.D. Structural Diversity on Copper(I) Schiff Base Complexes, Current Trends in X-Ray Crystallography. Chandrasekaran, Q., Ed.; InTech: London, UK, 2011; pp. 161–190. ISBN 978-953-307-754-3.
- Jamain, Z.; Azman, A.N.A.; Razali, N.A.; Makmud, M.Z.H. A Review on Mesophase and Physical Properties of Cyclotriphosphazene Derivatives with Schiff Base Linkage. Crystals 2022, 12, 1174.
- Rananavarem, S.B.; Pisipati, V.G.K.M. An Overview of Liquid Crystals Based on Schiff Base Compounds, Liquid Crystalline Organic Compounds and Polymers as Materials XXI Century: From Synthesis to Applications; Iwan, A., Schab-Balcerzak, E., Eds.; Transworld Research Network: Trivandrum, Kerala, 2011; ISBN 978-81-7895-496-7.
- Hoshino, N. Liquid crystal properties of metal–salicylaldimine complexes.: Chemical modifications towards lower symmetry. Coord. Chem. Rev. 1998, 174, 77–108.
- Torroba, J.; Bruce, D.W. Comprehensive Inorganic Chemistry II: From Elements to Applications, 2nd ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2013; Volume 8, pp. 837–917.
- El-Ghayoury, A.; Douce, L.; Skoulios, A.; Ziessel, R. Cation-Induced Macroscopic Ordering of Non-Mesomorphic Modules—A New Application for Metallohelicates. Angew. Chem. Int. Ed. 1998, 37, 2205–2208.
- Douce, L.; Diep, T.H.; Ziessel, R.; Skoulios, A.; Césario, M. Columnar liquid crystals from wedge-shaped tetrahedral copper(i) complexes. J. Mater. Chem. 2003, 13, 1533–1539.
- Douce, L.; El-Ghayoury, A.; Ziessel, R.; Skoulios, A. Columnar mesophases from tetrahedral copper(I) cores and Schiff-base derived polycatenar ligands. Chem. Commun. 1999, 2033–2034.
- Ziessel, R.; Pickaert, G.; Camerel, F.; Donnio, B.; Guillon, D.; Cesario, M.; Prangé, T. Tuning Organogels and Mesophases Will Phenanthroline Ligands and Their Copper Complexes by Inter- to Intramolecular Hydrogen Bonds. J. Am. Chem. Soc. 2004, 126, 12403–12413.