Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Alfred Zheng and Version 1 by Natalia A. Shnayder.
Valproic acid (VPA) and its salts (sodium calcium magnesium and orotic) are psychotropic drugs that are widely used in neurology and psychiatry. The long-term use of VPA increases the risk of developing adverse drug reactions (ADRs), among which metabolic syndrome (MetS) plays a special role. MetS belongs to a cluster of metabolic conditions such as abdominal obesity, high blood pressure, high blood glucose, high serum triglycerides, and low serum high-density lipoprotein. Valproate-induced MetS (VPA-MetS) is a common ADR that needs an updated multidisciplinary approach to its prevention and diagnosis.
- valproic acid
- metabolic syndrome
- Valproate-Induced
1. Introduction
Metabolic syndrome (MetS) is the medical term for a combination of diabetes mellitus, arterial hypertension, and obesity [1,2,3,4][1][2][3][4]. It puts you at greater risk of getting cardiovascular and cerebrovascular diseases. The MetS criteria [5,6,7][5][6][7] are presented in Figure 1.

Figure 1. Metabolic syndrome criteria. Note: M—male; F—female; IDF—the International Diabetes Federation; ATPIII—the Adult Treatment Panel, III edition; HDL—high-density lipoprotein cholesterol; TG—triglycerides.
According to the Adult Treatment Panel, III edition (ATPIII) [8], the prevalence of this disorder is variable in different regions of the world, including: North America (USA)—23.7%; Europe—18.4% among male and 14.4% among female; and Asia—28.8% among male and 31.8% among female. In Russia, the prevalence of MetS is 20% or more in people of reproductive age, and in people over 60 years old it is about 47%, while in women it occurs 2.4 times more often [9,10,11][9][10][11].
Drug-induced MetS is a combination of such conditions as abdominal obesity, hypercholesterolemia, hyperglycemia, and high blood pressure which occur while taking drugs [12]. Drug-induced MetS can be associated with various drugs, including psychotropic drugs [13]. Among psychotropic drugs, antipsychotics and valproic acid (VPA) drugs are the most common cause of drug-induced MetS [1,14][1][14] (Figure 2).
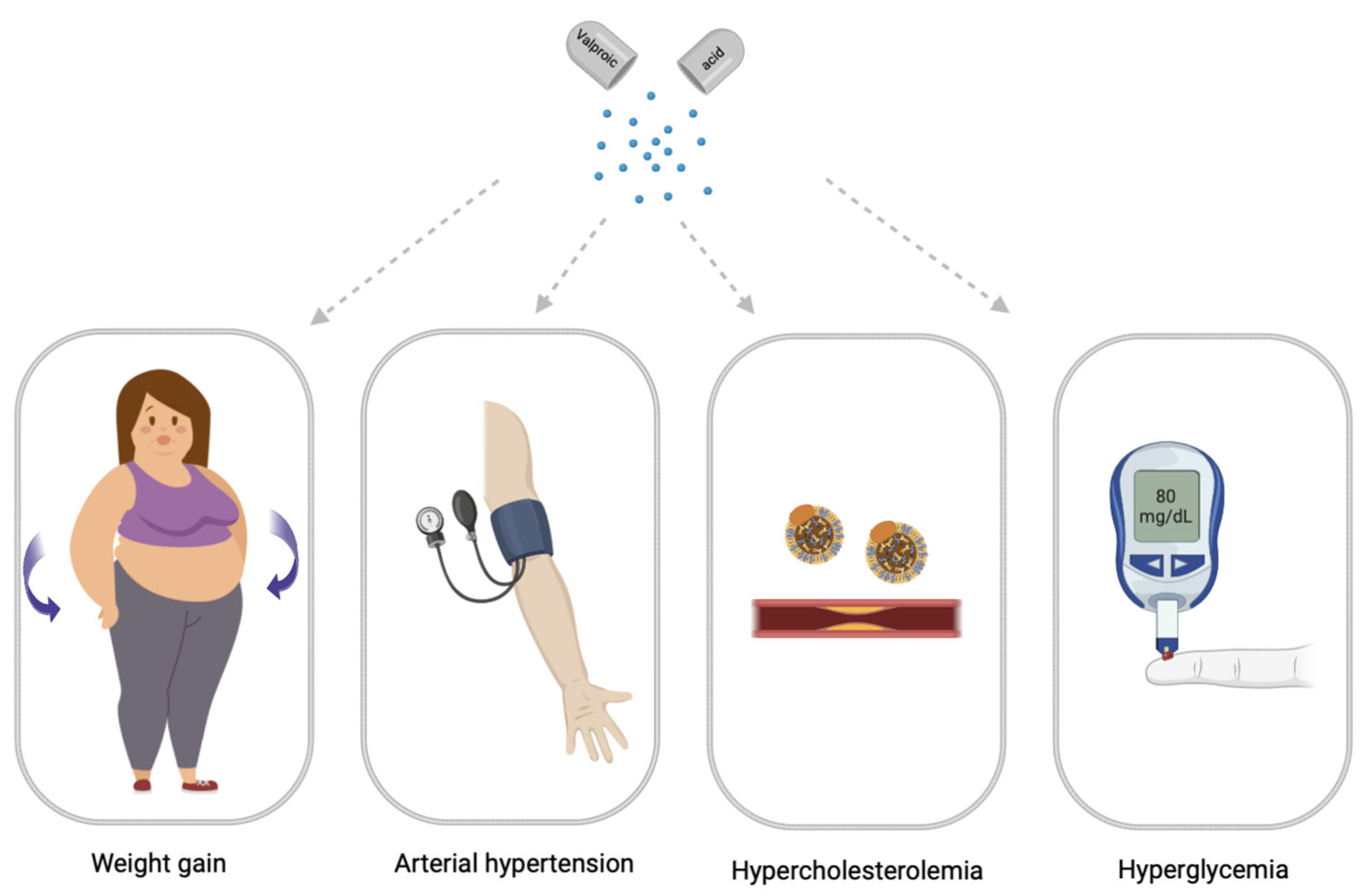
Figure 2. Main symptomes of valproate-induced metabolic syndrome (created with BioRender.com: https://www.biorender.com/ (accessed on 12 April 2023).
As is known, VPA (CH3CH2CH2)2CHCOOH 2-propylvaleric acid) is a psychotropic drug that is used in the treatment of many neurological diseases and mental disorders. The widespread use of VPA in neurology and psychiatry has led to an increased awareness of a spectrum of problematic and common adverse drug reactions (ADRs), including hepatotoxicity, nausea, vomiting, ataxia, lethargy, alopecia, thrombocytopenia, hyperammonemia, increased appetite, and weight gain. Among these ADRs, weight gain is of particular concern because the long-term use of valproates can lead to a significant increase in body weight, which is recorded in recent studies from 10% to 70% of patients and has been shown to lead to a number of metabolic disorders that can promote the development of VPA-induced MetS [15]. As a rule, the use of VPA is chronic, which increases the risk of developing VPA-induced MetS (VPA-MetS) [1,16][1][16]. The risk of developing VPA-MetS also depends on the genetic predisposition in a particular patient. This ADR may directly impair the quality of life of patients receiving valproate and be associated with a high risk of cardiovascular disease. From this position, both the prediction and prevention of the development of VPA-MetS and its early diagnosis are an urgent problem [14]. Significant weight gain is one of the most common problems with VPA in both children and adults [17]. In addition, the chronic use of valproates is associated with an increase in the level of fast plasma insulin (FPI), the development of insulin resistance, especially in a subgroup of children and women [18]. In addition to weight gain and insulin resistance, VPA-MetS is accompanied by dyslipidemia, arterial hypertension, and type 2 diabetes mellitus [15]. In recent years, there has been active research on: the pathophysiological mechanisms by which VPA and its active metabolites cause weight gain and metabolic disturbances, including increased thirst and hunger, caused by hypothalamic dysregulation due to an increased transmission of gamma-aminobutyric acid (GABA); the mechanisms of the VPA-induced modification of adipokines genes that encode the neuropeptides involved in central energy metabolism; and the VPA-induced deficiency of beta-oxidation of fatty acids [15].
Weight gain and metabolic disturbances associated with VPA intake have been investigated by various scientific groups and are of great concern to clinicians. The development of VPA-MetS reduces the quality of life of patients with neurological diseases and mental disorders, suffocates adherence to the exposure regimen, and leads to an increase in the incidence of cardiovascular and endocrine diseases.
2. Main Clinical Symptoms of Valproate-Induced Metabolic Syndrome
2.1. Valproate-Induced Weight Gain
Weight gain, also known as central obesity, is characterized by the excessive accumulation of fat in the abdomen and is associated with an increased risk of metabolic and cardiovascular diseases [19].
The features of abdominal obesity associated with VPA intake may include:
- −
-
An increase in body weight, body mass index (BMI), and waist circumference;
- −
-
An increase in the amount of fat in the abdomen, measured by the waist circumference or the ratio of waist to hips;
- −
-
Insulin resistance, which can contribute to the development of abdominal obesity.
Previous clinical studies have shown that weight gain is more common in women with epilepsy than in men. Therefore, gender can be considered one of the risk factors for VPA-induced weight gain [20]. In addition, the body fat percentage and waist-to-hip ratio are statistically significantly different between the sexes, with women having a higher body fat percentage and a lower waist-to-hip ratio. In female patients, an additional risk factor for VPA-induced weight gain is young age (weight gain is more common in post-pubertal girls taking VPA, and weight gain is more common in patients receiving VPA during puberty if epilepsy and therapy continue into adulthood) [20]. In adolescent girls, VPA-induced weight gain not only has serious psychological consequences, but can also lead to the development of important endocrinological abnormalities and reduced compliance with antiepileptic therapy. The mechanism by which VPA may cause weight gain is a matter of debate. However, various hypotheses have been put forward to explain the effect of VPA on weight gain: dysregulation of the hypothalamic system; effects on adipokines; hyperinsulinemia; and insulin resistance [21].
Experimental data have shown that VPA can cause dysregulation of the hypothalamic system [22]. This theory can be explained by an increase in gamma-aminobutyric acid (GABA) transmission in the hypothalamic axis. This is supported by studies whereby patients with epilepsy were treated with VPA, reporting weight gain, experienced an increase in appetite, and satiation with high-calorie drinks [20].
Another hypothesis is that VPA may cause weight gain by altering the expression of the adipokines genes that are expressed in the brain (pituitary gland). These genes encode the neuropeptides involved in central energy metabolism, such as resistin and starvation-induced adipose tissue factor (also known as angiopoietin-like protein 4), which have become the main targets of VPA associated with the etiology of obesity, leptin synthesis, and the development of insulin resistance [23]. Although VPA can alter gene expression in the hypothalamus in vitro [24], it is not clear whether VPA has such effects in vivo [20]. VPA may affect adipokines released by adipose tissue such as adiponectin, leptin, the soluble leptin receptor, and ghrelin [20]. VPA can increase the expression of messenger ribonucleic acid (mRNA) adiponectin-binding receptors (adipoR1) in the human hepatoma cell line HepG2 [20,25][20][25]. Since adiponectin mRNA expression is known to decrease after VPA treatment in vivo [26], the increased expression of adipoR1 mRNA in liver cells may represent a favorable response to counterbalance the suppressed secretion of adiponectin from adipocytes. Alterations in this receptor/ligand expression balance may contribute to changes in fatty acid oxidation and the development of insulin resistance in VPA-induced weight gain.
2.2. Valproate-Induced Insulin Resistance
Insulin resistance is a pathological condition in which cells do not normally respond to insulin [27]. Insulin is a hormone that facilitates the transport of glucose from the blood into the cells, thus lowering blood glucose levels. Insulin is secreted by the pancreas in response to carbohydrates consumed in the diet. In insulin resistance, high insulin levels do not have the expected effect on glucose transport and blood sugar levels [28]. Insulin resistance can be caused by a variety of factors, including obesity, a sedentary lifestyle, high-fat diet, and certain diseases such as polycystic ovary syndrome, as well as ADRs with long-term VPA [16,29][16][29].
The mechanisms by which VPA causes insulin resistance are not yet fully understood, but are thought to be mediated by several factors. VPA has been shown to increase oxidative stress, inflammation, and endoplasmic reticulum stress, all of which may contribute to the development of insulin resistance [30]. In addition, it was found that VPA increases the expression of the CEBP Alpha and SOCS-3, CEBP Alphain 3T3-L1 genes involved in the development of insulin resistance [31]. It was also found that VPA suppresses gluconeogenesis and glucagon expression, and also causes histopathological changes in the pancreas and liver [32]. The results of other studies give conflicting conclusions, including the hypothesis of an antidiabetic role of VPA in type II diabetes mellitus by modulating insulin signaling and gluconeogenesis mediated by thymus O1 protein [32]. Because VPA is a well-established drug, the detailed molecular mechanisms of the present findings may be explored for a possible new clinical application of valproate [32]. It is important to note that the relationship between VPA and insulin resistance is not yet conclusive, since there are studies in which the relationship between valproate intake and the development of insulin resistance has not been proven [33]. Further research is needed on this issue.
2.3. Valproate-Induced Arterial Hypertension
Arterial hypertension is a syndrome of increased systolic blood pressure from 140 mm Hg. Art. and above, and simultaneously or independently, diastolic blood pressure ≥90 mm Hg. Art. [34]. Arterial hypertension is one of the leading causes of death and affects the financial burden of the health care system in the world [35]. There are many studies that give conflicting results regarding the effect of VPA on the development of arterial hypertension [20].
In animal models, VPA has been shown to lower blood pressure in spontaneously hypertensive rats and prevent high-fat diet-induced hypertension [36]. VPA is known to have a broad mechanism of action, including blocking voltage-gated sodium channels, increasing GABA, and inhibiting the glutamate/N-methyl-D-aspartic acid (NMDA) receptors that mediate neuronal excitation. VPA can also influence the extracellular signal-related kinase (ERK) pathway [14]. Angiotensin leads to an increase in blood pressure by activating the ERK pathway in vascular smooth muscle cells [36,37][36][37]. On the other hand, it has been shown that under the action of angiotensin, phosphorylation and the activation of ERK occur. Since VPA can cause a decrease in ERK phosphorylation, this may be followed by a decrease in angiotensin-induced cardiac fibrosis. Therefore, it is possible that VPA also lowers blood pressure by reducing ERK phosphorylation. However, further studies of this mechanism of VPA influence on the reduction of ERK phosphorylation and blood pressure reduction are required [36,38][36][38].
Additionally, VPA acts as a direct inhibitor of histone deacetylase. Hyperacetylation of lysine residues on histones promotes the relaxation of deoxyribonucleic acid (DNA) and allows for an increase in gene transcription [1]. The long-term inhibition of histone deacetylase with VPA, regardless of the response to blood pressure, reduces hypertrophic, pro-inflammatory, and hypertensive responses by reducing reactive oxygen species and angiotensin II type 1 receptor expression in the heart, demonstrating the importance of uncontrolled histone deacetylase activity in arterial hypertension [14,39][14][39]. There are also reported cases of increased blood pressure after the initiation of valproate therapy [40].
2.4. Valproate-Induced Hypercholesterolemia
Hypercholesterolemia is a risk factor for the development of atherosclerosis [41]. Total cholesterol increases the risk of developing coronary heart disease by 2.52–3.20 times in reproductive age [42]. Several studies have also shown an increased mortality from atherosclerosis in people with epilepsy [43,44][43][44]. The effect of VPA on serum total cholesterol is still a matter of controversy. There is a significant change in lipid, lipoprotein and apolipoprotein profiles. A number of studies have demonstrated that in patients taking VPA long-term, there are no changes in the lipid profile induced by this anticonvulsant [43].
Finally, some studies show the effect of lowering serum total cholesterol levels with long-term valproate therapy [43,45][43][45]. The decrease in total cholesterol levels with the long-term use of VPA may be due to an increase in the activity of certain organs/organelles that also contribute to cholesterol metabolism. Since VPA can inhibit oxidative stress in the endoplasmic reticulum via the glycogen synthase kinase 3/β pathway, this may contribute to cholesterol metabolism and changes in the pathogenesis of atherosclerosis [43].
On the other hand, studies show that VPA can increase total and LDL cholesterol levels after 12 months of use [46]. The mean carotid intima-media thickness in patients with epilepsy treated with VPA was higher than in healthy controls. This proves that VPA can contribute to the development of atherosclerosis [47].
The mechanism of lipid profile changes under the influence of VPA and the risk of developing VPA-induced hypercholesterolemia are still unknown. A possible mechanism may be related to VPA-induced insulin resistance and hyperinsulinemia, which leads to impaired lipid transport and lipogenesis [48].
2.5. Valproate-Induced Hyperglycemia
Drug-induced hyperglycemia is a global problem, as it increases the risk of microvascular and macrovascular complications, infections, metabolic coma, and even death [49]. VPA inhibits class I (HDAC1, HDAC2, HDAC3, and HDAC8) and class II a (HDAC4, HDAC5, and HDAC7) histone deacetylase, resulting in the increased acetylation of H2, H3, and H4 histones, which alter the expression of their associated genes [14].
Recently, the use of VPA in various diseases has been investigated as a strategy for the reuse of clinically approved drugs [14]. On the other hand, there are reports that VPA reduces blood glucose and fat deposition in the adipose tissue and liver in mice and rats, and histone deacetylases classes I and IIa appear to be involved in the control of gluconeogenesis signaling and insulin production. On the other hand, VPA may reduce the microvascular complications of diabetes mellitus [50], requiring the mechanism of this drug response to be studied.
References
- Kazamel, M.; Stino, A.M.; Smith, A.G. Metabolic syndrome and peripheral neuropathy. Muscle Nerve 2021, 63, 285–293.
- Oye-Somefun, A.; Kuk, J.L.; Ardern, C.I. Associations between elevated kidney and liver biomarker ratios, metabolic syndrome and all-cause and coronary heart disease (CHD) mortality: Analysis of the U.S. National Health and Nutrition Examination Survey (NHANES). BMC Cardiovasc. Disord. 2021, 21, 352.
- Chen, W.; Pan, Y.; Jing, J.; Zhao, X.; Liu, L.; Meng, X.; Wang, Y.; Wang, Y. Recurrent Stroke in Minor Ischemic Stroke or Transient Ischemic Attack with Metabolic Syndrome and/or Diabetes Mellitus. J. Am. Heart Assoc. 2017, 6, e005446.
- Penninx, B.W.J.H.; Lange, S.M.M. Metabolic syndrome in psychiatric patients: Overview, mechanisms, and implications. Dialogues Clin. Neurosci. 2018, 20, 63–73.
- Khasanova, A.K.; Dobrodeeva, V.S.; Shnayder, N.A.; Petrova, M.M.; Pronina, E.A.; Bochanova, E.N.; Lareva, N.V.; Garganeeva, N.P.; Smirnova, D.A.; Nasyrova, R.F. Blood and Urinary Biomarkers of Antipsychotic-Induced Metabolic Syndrome. Metabolites 2022, 12, 726.
- Available online: cadiresearch.org/topic/metabolic-syndrome/metabolic-syndrome-global/ms-criteria (accessed on 21 March 2023).
- Kassi, E.; Pervanidou, P.; Kaltsas, G.; Chrousos, G. Metabolic syndrome: Definitions and controversies. BMC Med. 2011, 9, 48.
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497.
- Suplotova, L.A.; Smetanina, S.A.; Novakovskaya, N.A. Prevalence of metabolic syndrome and its components in women in different ethnic groups. Obes. Metab. 2011, 8, 48–51.
- Borisov, I.V.; Bondar, V.A.; Petrova, M.V.; Kuzovlev, A.N.; Ohlopkov, V.A.; Kanarski, M.M.; Nekrasova, J.J. Metabolic syndrome: Definition, pathogenesis and rehabilitation. Bull. All-Russ. Soc. Spec. Med. Soc. Expert. Rehabil. Rehabil. Ind. 2020, 4, 114–125.
- Chen, D.C.; Du, X.D.; Yin, G.Z.; Yang, K.B.; Nie, Y.; Wang, N.; Li, Y.L.; Xiu, M.H.; He, S.C.; Yang, F.D.; et al. Impaired glucose tolerance in first-episode drug-naïve patients with schizophrenia: Relationships with clinical phenotypes and cognitive deficits. Psychol. Med. 2016, 46, 3219–3230.
- Wofford, M.R.; King, D.S.; Harrell, T.K. Drug-induced metabolic syndrome. J. Clin. Hypertens. 2006, 8, 114–119.
- Sychev, D.A.; Ostroumova, O.D.; Pereverzev, A.P. Drug-induced diseases: Approaches to diagnosis, correction and prevention Pharmacovigilance. Pharmateka 2020, 27, 113–126.
- Shnayder, N.A.; Grechkina, V.V.; Khasanova, A.K.; Bochanova, E.N.; Dontceva, E.A.; Petrova, M.M.; Asadullin, A.R.; Shipulin, G.A.; Altynbekov, K.S.; Al-Zamil, M.; et al. Therapeutic and Toxic Effects of Valproic Acid Metabolites. Metabolites 2023, 13, 134.
- Zuo, S.; Fries, B.E.; Szafara, K.; Regal, R. Valproic Acid as a potentiator of metabolic syndrome in institutionalized residents on concomitant antipsychotics: Fat chance, or slim to none? Pharm. Ther. 2015, 40, 126–132.
- Shnaider, N.A.; Dmitrenko, D.V. Chronic valproic acid intoxication in epileptology: Diagnosis and treatment. Neurol. Neuropsychiatry Psychosom. 2016, 8, 94–99.
- Fang, J.; Chen, S.; Tong, N.; Chen, L.; An, D.; Mu, J.; Zhou, D. Metabolic syndrome among Chinese obese patients with epilepsy on sodium valproate. Seizure 2012, 21, 578–582.
- George, L.J.; Singh, P.; Aneja, S.; Singh, R.; Solanki, R.S.; Seth, A. Insulin Resistance in children on Sodium Valproate—A hospital based cross-sectional study in Indian children. Trop. Doct. 2023, 53, 91–96.
- Kim, Y.J.; Lee, Y.H.; Lee, Y.J.; Kim, K.J.; Kim, S.G. Weight Gain Predicts Metabolic Syndrome among North Korean Refugees in South Korea. Int. J. Environ. Res. Public Health 2021, 18, 8479.
- Belcastro, V.; D’Egidio, C.; Striano, P.; Verrotti, A. Metabolic and endocrine effects of valproic acid chronic treatment. Epilepsy Res. 2013, 107, 1–8.
- Romoli, M.; Mazzocchetti, P.; D’Alonzo, R.; Siliquini, S.; Rinaldi, V.E.; Verrotti, A.; Calabresi, P.; Costa, C. Valproic Acid and Epilepsy: From Molecular Mechanisms to Clinical Evidences. Curr. Neuropharmacol. 2019, 17, 926–946.
- Zhang, H.; Lu, P.; Tang, H.L.; Yan, H.J.; Jiang, W.; Shi, H.; Chen, S.Y.; Gao, M.M.; Zeng, X.D.; Long, Y.S. Valproate-Induced Epigenetic Upregulation of Hypothalamic Fto Expression Potentially Linked with Weight Gain. Cell. Mol. Neurobiol. 2021, 41, 1257–1269.
- Rehman, T.; Sachan, D.; Chitkara, A. Serum Insulin and Leptin Levels in Children with Epilepsy on Valproate-associated Obesity. J. Pediatr. Neurosci. 2017, 12, 135–137.
- Münzberg, H.; Björnholm, M.; Bates, S.H.; Myers, M.G., Jr. Leptin receptor action and mechanisms of leptin resistance. Cell Mol. Life Sci. 2005, 62, 642–652.
- Rauchenzauner, M.; Laimer, M.; Luef, G.; Kaser, S.; Engl, J.; Tatarczyk, T.; Ciardi, C.; Tschoner, A.; Lechleitner, M.; Patsch, J.; et al. Adiponectin receptor R1 is upregulated by valproic acid but not by topiramate in human hepatoma cell line, HepG2. Seizure 2008, 17, 723–726.
- Qiao, L.; Schaack, J.; Shao, J. Suppression of adiponectin gene expression by histone deacetylase inhibitor valproic acid. Endocrinology 2006, 147, 865–874.
- Brown, A.E.; Walker, M. Genetics of Insulin Resistance and the Metabolic Syndrome. Curr. Cardiol. Rep. 2016, 18, 75.
- Petersen, M.C.; Shulman, G.I. Mechanisms of Insulin Action and Insulin Resistance. Physiol. Rev. 2018, 98, 2133–2223.
- Aly, R.H.; Amr, N.H.; Saad, W.E.; Megahed, A.A. Insulin resistance in patients on valproic acid: Relation to adiponectin. Acta Neurol. Scand. 2015, 131, 169–175.
- Jian, J.; Li, L.G.; Zhao, P.J.; Zheng, R.J.; Dong, X.W.; Zhao, Y.H.; Yin, B.Q.; Cheng, H.; Li, H.L.; Li, E.Y. TCHis mitigate oxidative stress and improve abnormal behavior in a prenatal valproic acid-exposed rat model of autism. Physiol. Genom. 2022, 54, 325–336.
- Brown, R.; Imran, S.A.; Ur, E.; Wilkinson, M. Valproic acid and CEBPalpha-mediated regulation of adipokine gene expression in hypothalamic neurons and 3T3-L1 adipocytes. Neuroendocrinology 2008, 88, 25–34.
- Khan, S.; Kumar, S.; Jena, G. Valproic acid reduces insulin-resistance, fat deposition and FOXO1-mediated gluconeogenesis in type-2 diabetic rat. Biochimie 2016, 125, 42–52.
- Rauchenzauner, M.; Laimer, M.; Wiedmann, M.; Tschoner, A.; Salzmann, K.; Sturm, W.; Sandhofer, A.; Walser, G.; Luef, G.; Ebenbichler, C.F. The novel insulin resistance parameters RBP4 and GLP-1 in patients treated with valproic acid: Just a sidestep? Epilepsy Res. 2013, 104, 285–288.
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomstrӧm-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Russ. J. Cardiol. 2021, 26, 4701. (In Russian)
- Aune, D.; Huang, W.; Nie, J.; Wang, Y. Hypertension and the Risk of All-Cause and Cause-Specific Mortality: An Outcome-Wide Association Study of 67 Causes of Death in the National Health Interview Survey. Biomed Res. Int. 2021, 2021, 9376134.
- Abaseynejad, F.; Akrami, R.; Mohebbati, R.; Sehab Negah, S.; Mohammad-Zadeh, M. The Effect of Sodium Valproate on Cardiovascular Responses in Pentylenetetrazol Kindling Model of Epilepsy. Biomed. J. Sci. Tech. Res. 2022, 42, 33592–33596.
- Sousa-Lopes, A.; de Freitas, R.A.; Carneiro, F.S.; Nunes, K.P.; Allahdadi, K.J.; Webb, R.C.; Tostes, R.C.; Giachini, F.R.; Lima, V.V. Angiotensin (1-7) Inhibits Ang II-mediated ERK1/2 Activation by Stimulating MKP-1 Activation in Vascular Smooth Muscle Cells. Int. J. Mol. Cell. Med. 2020, 9, 50–61.
- Zhao, Y.; Xing, B.; Dang, Y.H.; Qu, C.L.; Zhu, F.; Yan, C.X. Microinjection of valproic acid into the ventrolateral orbital cortex enhances stress-related memory formation. PLoS ONE 2013, 8, e52698.
- Rajeshwari, T.; Raja, B.; Manivannan, J.; Silambarasan, T. Valproic acid attenuates blood pressure, vascular remodeling and modulates ET-1 expression in L-NAME induced hypertensive rats. Biomed. Prev. Nutr. 2014, 4, 195–202.
- Sivananthan, M.; Mohiuddin, S. Valproate Induced Hypertensive Urgency. Case Rep. Psychiatry 2016, 2016, 1458548.
- Zárate, A.; Manuel-Apolinar, L.; Saucedo, R.; Hernández-Valencia, M.; Basurto, L. Hypercholesterolemia as a Risk Factor for Cardiovascular Disease: Current Controversial Therapeutic Management. Arch. Med. Res. 2016, 47, 491–495.
- Peters, S.A.; Singhateh, Y.; Mackay, D.; Huxley, R.R.; Woodward, M. Total cholesterol as a risk factor for coronary heart disease and stroke in women compared with men: A systematic review and meta-analysis. Atherosclerosis 2016, 248, 123–131.
- Kusumastuti, K.; Jaeri, S. The effect of long-term valproic acid treatment in the level of total cholesterol among adult. Indian J. Pharmacol. 2020, 52, 134–137.
- Hamed, S.A. Atherosclerosis in epilepsy: Its causes and implications. Epilepsy Behav. 2014, 41, 290–296.
- Aziz, R.S.; Saeed, U.; Ali, L.; Arshad, M.; Abbas, R.; Mushtaq, S.; Asif Shahzad, A.; Shaukat, A. Effect on lipid profile due to prolong Valproic acid intake. Pak. J. Med. Health Sci. 2021, 15, 1497–1499.
- Guo, H.L.; Jing, X.; Sun, J.Y.; Hu, Y.H.; Xu, Z.J.; Ni, M.M.; Chen, F.; Lu, X.P.; Qiu, J.C.; Wang, T. Valproic Acid and the Liver Injury in Patients with Epilepsy: An Update. Curr. Pharm. Des. 2019, 25, 343–351.
- Verrotti, A.; Scardapane, A.; Franzoni, E.; Manco, R.; Chiarelli, F. Increased oxidative stress in epileptic children treated with valproic acid. Epilepsy Res. 2008, 78, 171–177.
- Verrotti, A.; la Torre, R.; Trotta, D.; Mohn, A.; Chiarelli, F. Valproate-induced insulin resistance and obesity in children. Horm. Res. 2009, 71, 125–131.
- Fathallah, N.; Slim, R.; Larif, S.; Hmouda, H.; Ben Salem, C. Drug-Induced Hyperglycaemia and Diabetes. Drug Saf. 2015, 38, 1153–1168.
- Felisbino, M.B.; Ziemann, M.; Khurana, I.; Okabe, J.; Al-Hasani, K.; Maxwell, S.; Harikrishnan, K.N.; de Oliveira, C.B.M.; Mello, M.L.S.; El-Osta, A. Valproic acid influences the expression of genes implicated with hyperglycaemia-induced complement and coagulation pathways. Sci. Rep. 2021, 11, 2163.
More
