Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Catherine Yang and Version 1 by Federica Re.
Bone regenerative medicine is a clinical approach combining live osteoblast progenitors, such as mesenchymal stromal cells (MSCs), with a biocompatible scaffold that can integrate into host bone tissue and restore its structural integrity.
- clinical trials
- mesenchymal stromal cells
- scaffolds
- hydrogels
1. Overview of Published Clinical Trials with MSCs and Scaffolds (2018–2023)
A total of nine clinical trials were included in the analysis: six were present in the literature and three were reported on Clinicaltrials.gov (Table 1 and Table 2, Figure 1). Background information from each trial was summarized including clinical phase, condition, controls used, and follow-up. Studies were conducted in Spain [35][1], Mexico [36][2], Norway [37][3], the Czech Republic [38][4], Italy [39[5][6][7],40,41], Western Australia (NCT01742260), and Spain and Portugal [42][8]. One trial was reported in three publications [39,40,41][5][6][7]. Two clinical trials were at Phase I [35][1] (NCT01742260), two were at Phase I/II [37[3][5],39], one was at Phase IIa [38][4], and phase was not reported in one clinical trial [36,42][2][8]. The studies were prospective, open, non-randomized [35[1][3][5][6][7],37,39,40,41], randomized [36[2][8],42], interventional (NCT01742260), and in particular, one trial was multicentric [39,40,41][5][6][7]. The most common indications concerned the treatment of lumbar intervertebral degenerative disc disease (DDD) [35][1], the alveolar ridge [37,42][3][8], large skeletal defects [38][4], and fracture of long bones [29,30,31,32,33,34,35,36,37,38,39,40][1][2][3][4][5][6][9][10][11][12][13][14]. Clinical evaluation showed that the patients achieved lumbar fusion in up to five years using TCP and autologous MSCs [35][1]. An increase in bone mineralization in association with a decrease in inflammation were obtained thanks to the combination of MSCs from dental pulp and a collagen sponge scaffold in periodontal disease at the 6-month follow-up [36][2]. In particular, Hernandez et al. evaluated 10 controls for which only collagen scaffold without DPMSCs had been placed, observing a less impressive clinical outcome with respect to the cell-added scaffold group [36][2]. Successful ridge augmentation without adverse events in maxillofacial bone defects was pursued using BCP and autologous MSCs [37][3]. No significant differences were obtained using cancellous allografts compared to the combination of TCP and MSCs in promoting the healing of bone defects, whereas significant differences were documented following the implantation of TCP only and cancellous allografts in femoral bone defects [38][4]. Clinical and radiological evaluation confirmed complete bone consolidation in long bone non-unions at 12 months using biphasic calcium phosphate bioceramic granules and autologous MSCs [39,40,41][5][6][7]. In addition, some clinical trials reported no findings, results, or publications in ClinicalTrials.gov although the study’s expected completion dates were 2017 for NCT01742260, 2018 for NCT03682315, and 2022 for NCT03797963. Hydrogels are the new generation of scaffolds for bone reconstruction and DEXGEL Bone, a hydrogel used for alveolar ridge preservation, was shown to stimulate natural bone regeneration without side effects [42][8]. DEXGEL Bone is derived from the association of Bonelike by Biosckin® (BL®), a glass-reinforced hydroxyapatite synthetic bone substitute, with a dextrin-based hydrogel named DEXGEL. In particular, Machado et al. compared the synthetic bone substitute BL® (control) to its hydrogel-reinforced version, DEXGEL Bone (test), in the preservation of alveolar ridge dimensions following tooth extraction, demonstrating bone quantity and quality and primary stability of the implant [42][8]. Finally, considering all studies, the follow-up periods ranged between 1 and 60 months.
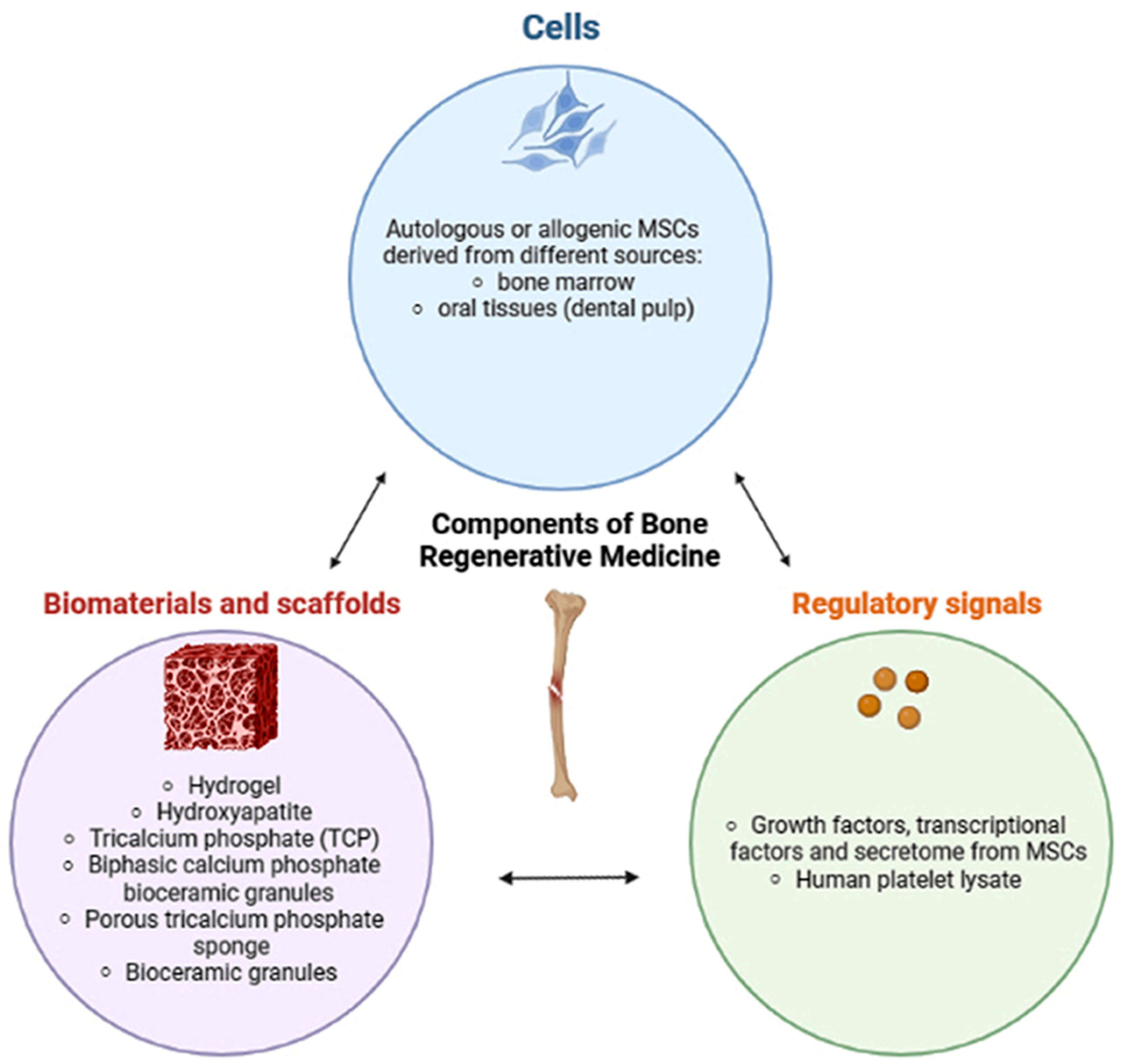
Figure 1. Bioengineering strategy beyond bone regeneration in clinical practice. The nature and structure of the scaffolds have great importance to support cell growth. The MSCs represent a source of growth factors, cytokines, and extracellular vesicles to their surrounding cells, which may favor bone regeneration and osteogenesis [10][15].
Table 1.
Summary of clinical trials using MSCs and scaffolds for bone regeneration from 2018 to 2023.
| Cells | Scaffolds | Condition | Number of Patients (Age) | Number of Cells Seeded (Scaffolds Dimensions) | Follow Up | Control | Evaluation Methods and Outcomes | References |
|---|
Table 2.
Summary of clinical trials using scaffolds without cells for bone regeneration from 2018 to 2023.
| Cells | Scaffolds | Condition | Number of Patients (Age) | Follow Up | Control | Evaluation Methods and Outcomes | References | Registration ID and Country | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Autologous bone marrow-derived MSCs | ß-tricalcium phosphate (TCP) | Lumbar degenerative disc disease (DDD) at L4-L5 or L5-S1 | 11 (18–65) | 1.5 × 10−6 cells/kg from the patient (20 mL of TCP) | 1, 3, 6, 12, 60 months | ||||||||||
| No cells | Anorganic bovine bone (BioOss Xenograft) | Bilateral Maxillary Sinus Floor Augmentation | 8 (>18 years) | 6 months | Active Comparator (contralateral: biphasic phycogenic biomaterial and autogenous cortical bone) | _ | CBCT scans before the sinus floor elevation and 6 months later before implant placement to calculate vertical bone height change from the crestal bone to the floor of the maxillary sinus. Radiography and clinical evaluation revealed that 80% of patients achieved lumbar fusion in up to five years. Both the visual analog scale (VAS) and the Oswestry disability index (ODI) improved after surgery. The Short-Form Health Survey (SF-36) evaluated the physical and mental status that showed a significant improvement in the first year after surgery. There were no adverse effects related to cell implantation. |
Histomorphometric quantification of new mineralized tissue, non-mineralized tissue and remaing graft particles in a bone biopsy collected 6 months after the grafting procedure. NO outcomes. |
No publication, no results posted. Actual study completion date 2018Blanco et al., 2019 [35][1] |
||||||
| NCT03682315 | Responsable: Pablo Galindo-Moreno, Universidad de Granada, Spain | Phase not applicable | MSCs obtained from the dental pulp of two male patients ages 7 and 8 and a 10-year-old patient (hDPSCs) |
Scaffold of lyophilized collagen-polyvinylpyrrolidone sponge (Fibroquel; Aspid, Mexico City, Mexico) |
Deep infra bony defect ≥ 4 mm deep caused by periodontal disease | 22 (55–64) | 5 × 106 hDPSCs (0.5 cm2) | 6 months | 11 scaffolds without hDPSCs | Increase in the bone mineral density of the alveolar bone; increased salivary superoxide-dismutase and decreased levels of salivary IL1β | Beatriz Hernández-Monjaraz et al., 2020 | ||||
| No cells | Anorganic bovine bone (BioOss Xenograft) + autogenous cortical bone | Bilateral Maxillary Sinus Floor Augmentation | 10 (>18 years) | 6–12–18 months | Active Comparator (contralateral: Porcine bone mineral (Symbios Xenograft) + autogenous cortical bone) |
CBCT scans after the sinus floor elevation and 6–12–18 months later before implant placement to calculate vertical bone height change. Histomorphometric quantification of new mineralized tissue, non-mineralized tissue and remaing graft particles in a bone biopsy collected 6–12–18 months after the grafting procedure. NO outcomes. |
No publication, no results posted. Actual study completion date 2022 | [36][2] | |||||||
| NCT03797963 | Responsable: Pablo Galindo-Moreno, Universidad de Granada, Spain | Phas not applicable | Autologous bone marrow-derived MSCs | Biphasic calcium phosphate granules (BCP) | Maxillofacial bone defects | 11 (52–79) | |||||||||
| No cells but BL® was mixed with autologous blood previously extracted from | 20 × 10 | 6 | cells | (1 cm3) |
1, 2, 4,12 months | _ |
the alveolar defect and applied with a spatula. | DEXGEL Bone: Bonelike by Biosckin® (BLAll patients had successful ridge augmentation and an adequate amount of bone for dental implant installation without adverse events. The alveolar ridge increased both in width and volume. | ®), a glass-reinforced hydroxyapatite synthetic bone substitute, in association to dextrin-based hydrogel, DEXGEL |
Alveolar ridge preservation | 12 (above 18 years) | 6 months | BL® granules (250–500 μm) were administered to 6 randomized participants whereas the other 6 received DEXGEL Bone. | Both treatments showed good osseointegration. DEXGEL Bone exhibited increased granule resorption accompanied by a tendency for more new bone ingrowth compared to the BL® group. DEXGEL was rapidly resorbed and accelerated BL® resorption as well, freeing up space that favored new bone ingrowth, without compromising mechanical support. The healing of defects was free of any local or systemic complications. |
Machado et al., 2023 [42][8]Gjerde et al., 2018 [37][3] |
| EUDAMED: CIV-PT-18–01–02,705. | RNEC: 30122. | Portugal | Phase not reported | Autologous bone marrow-derived MSCs | ß -tricalcium phosphate (TCP) | Femoral bone defect | 37 (44–75) | 15 ± 4.5 × 106 cells (dimensions not reported) | 6 weeks, 3, 6, 12 months | Group A: 19 patients with ß -TCP and autologous MSC, group B: 19 patients with ß -TCP alone, group C: 19 patients with cancellous allografts only | The combination between TCP and MSCs appears safe and promotes the healing of bone defects. No significant differences were observed between groups A and B. Significant differences were observed between group B and C. Adverse events emerged from the demanding and extensive character of revision hip replacement without a causal relationship to the suspension of autologous MSCs. | Pavel Sponer et al., 2018 [38][4] | |||
| Autologous bone marrow-derived MSCs | Biphasic calcium phosphate bioceramic granules (BCP) | Long bone non-unions (fractures of the femur, tibia, and humerus) | 28 (3 months), 27 (6 months), 25 (12 months) (18–65). E. Gómez-Barrena et al., 2020 [25][16] 26 (18–65). E. Gómez-Barrena et al., 2020 [26][17] 28 (18–65). E. Gómez-Barrena et al., 2020 [27][18] |
20 × 106 cells (5–10 cc of bioceramic granules) | 3, 6, 12 months For E. Gómez-Barrena et al., 2020 [27][18] subgroup analysis of gender, tobacco use, time since the original fracture |
_ | The ATMP combined with the bioceramic was surgically delivered to the non-unions, and 26/28 treated patients were found radiologically healed at one year (3 out of 4 cortices with bone bridging). E. Gómez-Barrena et al., 2020 [25][16] The REBORNE bone healing score, defined to perform an evaluation of long bone non-union consolidation in radiograph and computed tomography (CT), proved valid to assess consolidation against CT measurements with a concordance correlation of 79% and an accuracy based on ROC curves of 83%. E. Gómez-Barrena et al., 2020 [26][17] The clinical and radiological evaluation confirmed bone consolidation at 3 months (25%), 6 months (67.8%), and 12 months (92.8%), with lower consolidation scores in smokers. Femur, humerus, and tibia showed consolidation at one year. E. Gómez-Barrena et al., 2020 [27][18] |
E. Gómez-Barrena et al., 2020 [39][5] E. Gómez-Barrena et al., 2020 [40][6] E. Gómez-Barrena et al., 2020 [41][7] |
|||||||
| Bone marrow-derived MSCs from donor | Medical grade bioceramic granules of beta-tricalcium phosphate by ChronOS (Synthes GmbH, Oberdorf) placed between specially moulded plastic scaffolds (PLA such as 70:30 polyia (L-lactide-co-D,L-lactide) and insert the sandwich into the skull. | Cranial defect <80 mm diameter | 10 (18–80) | Not reported | 12 months | _ | Quantitative bone density of the tissue-engineered construct and adjacent bone from CT scan at 12 months. Assessment of cosmesis by photography. NO outcomes |
No publication, no results posted estimated study completion date 2017 |
2. MSC Tissue Sources, Characterization, and Manipulation
Bone marrow cells represented the cells most commonly used, particularly autologous cells [35,37,38,39,40,41][1][3][4][5][6][7]. Cells from donors were also used in [36][2] and NCT01742260. The term ‘stem’ was much more commonly used than ‘stromal’. MSCs from bone marrow were expanded in vitro using GMP, according to common standard operating procedures (SOP), in a specific medium enriched with PL without animal products at different concentrations: 5% PL [35][1], and 8% PL [39,40,41][5][6][7]. Cells were used at two or three passages [35,38,39,40,41][1][4][5][6][7] and one passage [37][3]. Mononuclear cell isolation after density-gradient centrifugation was performed by Blanco et al. [35][1], while details of viability analysis by flow cytometry for the positivity of CD90, CD73, and CD105 markers and the negativity of CD14 and CD45 markers were reported by Gjerde et al. [37][3] and in the ORTHO-1 study [39,40,41][5][6][7]. Other tested markers were reported: MHCI, MHCII, CD16, CD45, CD34, CD19, CD3, CD14, and CD80 [38][4]. Additional analyses, such as bacteriological tests and cell attachment using the fluorescent dye DAPI on BCP, were also performed [39][5]. Gómez-Barrena et al. used crystal violet and a live/dead assay to indicate that MSCs were attached and alive [39,40,41][5][6][7]. Moreover, sterility, endotoxins, and mycoplasma were tested [39,40,41][5][6][7]. Additional quality controls were performed according to the requests of each country-specific national competent authority [39,40,41][5][6][7]. In addition, certificates of analysis included with the investigational medicinal product (IMP) were obtained by each national competent authority (NCA) of countries participating in the REBORNE consortium [39,40,41][5][6][7]. Cells were obtained from 40–100 mL [35][1], 15–20 mL [37][3], and 10–12 mL [42][8] of bone marrow aspirate while 50,000 white blood cells per cm2 of bone marrow aspirate in a culture chamber were used by researchers in the REBORNE consortium (ORTHO-1 study) and distributed to other units [39,40,41][5][6][7]. hDPSCs from young donors were only used in the research of Hernández-Monjaraz et al. [36][2]. The nature of the growth factors used in cell culture for cell expansion was not detailed, but it was declared that the experiments were conducted under the strict criteria of GMP, using animal-origin-free reagents [36][2].
3. Scaffolds for Bone Regeneration
The majority of scaffolds were composed of calcium phosphate ceramic, such as β-tricalcium phosphate (TCP) [35[1][4],38], biphasic calcium phosphate bioceramic granules [37,39[3][5][6][7],40,41], anorganic bovine bone (NCT03682315, NCT03797963), and hydrogel in association with hydroxyapatite [42][8]. A total of 1.5 × 10−6 cells/kg from the patient were mixed with 20 mL of TCP support [35][1], 20 × 106 cells/cm3 were cultivated in BCP [37][3], 15 ± 4.5 × 106 cells were applied onto an absorbable porous β-tricalcium phosphate sponge [38][4], and 20 × 106 cells per mL were suspended in 10 mL solution with bioceramic granules to obtain the ORTHO-1 MSC tissue-engineered product [39,40,41][5][6][7]. Processing of bone biopsies, after scaffolds were seeded with implanted cells, was performed for histological staining using hematoxylin/eosin and Masson trichrome [39][5]. In addition, immunohistology was performed to identify macrophages with human CD68 primary antibody by Gómez-Barrena et al. [39,40,41][5][6][7]. Only in the research of Hernández-Monjaraz et al., 5 × 106 DPMSCs dripped suspended in PBS were seeded onto a scaffold of lyophilized polyvinylpyrrolidone sponge® (clg-PVP) in 0.5 cm2 fragments, while the control group only received PBS without DPMSCs [36][2]. Finally, in both groups, collagen membranes (Biomed extend®, ZimVie, CA, USA) were placed on the flap. Moreover, in the clinical trial of Herrmann (NCT01742260), the researchers created a skull-like scaffold composed of medical-grade bioceramic granules of beta-tricalcium phosphate by ChronOS (Synthes GmbH, Oberdorf) and cells (concentration not reported) were placed between the specially molded plastic scaffolds (PLA such as 70:30 poly(L-lactide-co-D,L-lactide) [36][2].
Not all of the studies used scaffolds in association with cells. In particular, the study by Machado et al. demonstrated the ability of the hydrogel to stimulate newly formed bone and biological compatibility with the host tissues [42][8]. The authors used DEXGEL, an in situ gelling hydrogel with oxidized dextrin as the base, as a moldable carrier of BL® granules in the management of alveolar bone regeneration. BL® is a synthetic bone graft designed to mimic the inorganic composition of bone [42][8]. Even if no cells were used in association with DEXGEL Bone, BL® (control) was mixed with autologous blood previously extracted from the alveolar defect and applied with a spatula [42][8]. Moreover, two other studies tested scaffolds without cells for sinus floor augmentation but no results were reported. The first study used xenograft bovine hydroxyapatite (BioOss) (NCT03682315) with contralateral active control of the biphasic phycogenic biomaterial and autogenous cortical bone. The second study added BioOss to the autogenous cortical bone (NCT03797963) with contralateral active control of the porcine bone mineral (Symbios Xenograft) mixed with autogenous cortical bone.
References
- Blanco, J.F.; Villarón, E.M.; Pescador, D.; Da Casa, C.; Gómez, V.; Redondo, A.M.; López-Villar, O.; López-Parra, M.; Muntión, S.; Sánchez-Guijo, F. Autologous mesenchymal stromal cells embedded in tricalcium phosphate for posterolateral spinal fusion: Results of a prospective phase I/II clinical trial with long-term follow-up. Stem Cell Res. Ther. 2019, 10, 63.
- Hernández-Monjaraz, B.; Santiago-Osorio, E.; Ledesma-Martínez, E.; Alcauter-Zavala, A.; Mendoza-Núñez, V.M. Retrieval of a periodontally compromised tooth by allogeneic grafting of mesenchymal stem cells from dental pulp: A case report. J. Int. Med. Res. 2018, 46, 2983–2993.
- Gjerde, C.; Mustafa, K.; Hellem, S.; Rojewski, M.; Gjengedal, H.; Yassin, M.A.; Feng, X.; Skaale, S.; Berge, T.; Rosen, A.; et al. Cell therapy induced regeneration of severely atrophied mandibular bone in aclinical trial. Stem Cell Res. Ther. 2018, 9, 1–15.
- Šponer, P.; Kučera, T.; Brtková, J.; Urban, K.; Kočí, Z.; Měřička, P.; Bezrouk, A.; Konrádová, Š.; Filipová, A.; Filip, S. Comparative Study on the Application of Mesenchymal Stromal Cells Combined with Tricalcium Phosphate Scaffold into Femoral Bone Defects. Cell Transplant. 2018, 27, 1459–1468.
- Gómez-Barrena, E.; Rosset, P.; Gebhard, F.; Hernigou, P.; Baldini, N.; Rouard, H.; Sensebé, L.; Gonzalo-Daganzo, R.M.; Giordano, R.; Padilla-Eguiluz, N.; et al. Feasibility and safety of treating non-unions in tibia, femur and humerus with autologous, expanded, bone marrow-derived mesenchymal stromal cells associated with biphasic calcium phosphate biomaterials in a multicentric, non-comparative trial. Biomaterials 2019, 196, 100–108.
- Gómez-Barrena, E.; Padilla-Eguiluz, N.G.; García-Rey, E.; Hernández-Esteban, P.; Cordero-Ampuero, J.; Rubio-Suárez, J.C. Validation of a long bone fracture non-union healing score after treatment with mesenchymal stromal cells combined to biomaterials. Injury 2020, 51, S55–S62.
- Gómez-Barrena, E.; Padilla-Eguiluz, N.; Rosset, P.; Gebhard, F.; Hernigou, P.; Baldini, N.; Rouard, H.; Sensebé, L.; Gonzalo-Daganzo, R.-M.; Giordano, R.; et al. Early efficacy evaluation of mesenchymal stromal cells (MSC) combined to biomaterials to treat long bone non-unions. Injury 2020, 51 (Suppl. S1), S63–S73.
- Machado, A.; Pereira, I.; Costa, F.; Brandão, A.; Pereira, J.E.; Maurício, A.C.; Santos, J.D.; Amaro, I.; Falacho, R.; Coelho, R.; et al. Randomized clinical study of injectable dextrin-based hydrogel as a carrier of a synthetic bone substitute. Clin. Oral Investig. 2023, 27, 979–994.
- Taboni, S.; Ferrari, M.; Gualtieri, T.; Chan, H.; Townson, J.; Mattavelli, D.; Eu, D.; Dey, K.; Mathews, S.; Re, F.; et al. Bioengineered scaffolding for mandibular reconstruction: A preclinical, xenograft animal study. Cytotherapy 2021, 23, S140–S141.
- Wang, X.; Thomsen, P. Mesenchymal stem cell–derived small extracellular vesicles and bone regeneration. Basic Clin. Pharmacol. Toxicol. 2020, 128, 18–36.
- Kim, K.-T.; Kim, K.G.; Choi, U.Y.; Lim, S.H.; Kim, Y.J.; Sohn, S.; Sheen, S.H.; Heo, C.Y.; Han, I. Safety and Tolerability of Stromal Vascular Fraction Combined with β-Tricalcium Phosphate in Posterior Lumbar Interbody Fusion: Phase I Clinical Trial. Cells 2020, 9, 2250.
- Spanò, R.; Muraglia, A.; Todeschi, M.R.; Nardini, M.; Strada, P.; Cancedda, R.; Mastrogiacomo, M. Platelet-rich plasma-based bioactive membrane as a new advanced wound care tool. J. Tissue Eng. Regen. Med. 2017, 12, e82–e96.
- Rauch, C. Alternatives to the use of fetal bovine serum: Human platelet lysates as a serum substitute in cell culture media. Altex 2011, 28, 305–316.
- Re, F.; Sartore, L.; Moulisova, V.; Cantini, M.; Almici, C.; Bianchetti, A.; Chinello, C.; Dey, K.; Agnelli, S.; Manferdini, C.; et al. 3D gelatin-chitosan hybrid hydrogels combined with human platelet lysate highly support human mesenchymal stem cell proliferation and osteogenic differentiation. J. Tissue Eng. 2019, 10, 2041731419845852.
- Gamie, Z.; Tran, G.T.; Vyzas, G.; Korres, N.; Heliotis, M.; Mantalaris, A.; Tsiridis, E. Stem cells combined with bone graft substitutes in skeletal tissue engineering. Expert Opin. Biol. Ther. 2012, 12, 713–729.
- Renesme, L.; Pierro, M.; Cobey, K.D.; Mital, R.; Nangle, K.; Shorr, R.; Lalu, M.M.; Thébaud, B. Definition and Characteristics of Mesenchymal Stromal Cells in Preclinical and Clinical Studies: A Scoping Review. Stem Cells Transl. Med. 2022, 11, 44–54.
- Shi, X.; Mao, J.; Liu, Y. Pulp stem cells derived from human permanent and deciduous teeth: Biological characteristics and therapeutic applications. Stem Cells Transl. Med. 2020, 9, 445–464.
- Re, F.; Gabusi, E.; Manferdini, C.; Russo, D.; Lisignoli, G. Bone Regeneration Improves with Mesenchymal Stem Cell Derived Extracellular Vesicles (EVs) Combined with Scaffolds: A Systematic Review. Biology 2021, 10, 579.
More
