Bacillus thuringiensis (Bt) is a rod-shaped, Gram-positive soil bacterium that belongs to the phylum Firmicutes and the genus Bacillus. It is a spore-forming bacterium. During sporulation, it produces a wide range of crystalline proteins that are toxic to different orders of insects. Sporulation, structure assembly, and germination are essential stages in the cell cycle of B. thuringiensis. The majority of studies on these issues have focused on the model organism Bacillus subtilis, followed by Bacillus cereus and Bacillus anthracis. The machinery for sporulation and germination extrapolated to B. thuringiensis. However, in the light of recenthe findings concerning the role of the sporulation proteins (SPoVS), the germination receptors (Gr), and the cortical enzymes in Bt, the theory strengthened that conservation in sporulation, structure assembly, and germination programs drive the survival and success of B. thuringiensis in the environment and the insect host.
- spore-forming bacteria
- Gram-positive bacillus
- Bacillus thuringiensis
- insecticidal crystal proteins
1. Introduction
2. The Soil Spore-Forming Bacterium Bacillus thuringiensis
The identity of Bacillus thuringiensis relies on a set of pore-forming proteins, known as Cry and Cyt toxins, to kill insect larvae. Therefore, Bt is considered an insect pathogen [34][35][36][37][38]. B. thuringiensis belongs to the genus Bacillus, a rod-shaped Gram-positive soil bacterium that contains genomic DNA and extrachromosomal DNA (plasmids). Interestingly, many plasmids encode the delta-endotoxins or Cry proteins, a strategy of B. thuringiensis to survive in the harsh environment of the soil’s rhizosphere and for insect and mammalian targeting [34][35][39][40]. Commitment in the life cycle of B. thuringiensis consists of a series of morphological and cytological changes that end with spore formation and crystal production. This series includes gene expression and biochemical and genetic programs [41][42].The Plasmid-Encoded
Bt
Crystalline Proteins
The insecticidal delta-endotoxins of Bacillus thuringiensis or Cry (Crystalline) proteins have been the subject of intense research during the last three or four decades [6][35][39]. These crystals comprise an array of immature protoxins with a molecular weight of 130 kDa, encoded in large plasmids [43][44][45]. To be active in the insect larvae host, protoxins are first solubilized and then processed in the C-terminal region favored by the enzymatic action, yielding a toxin with a molecular weight of 60–70 kDa [46][47]. The 3D structure of several Cry toxins was elucidated by X-ray resolution crystallography [48], including Cry1Ac [49], Cry2Aa [50], Cry3Aa [46], Cry3Ba [51], Cry4Aa [52], and Cry4Ba [53]. Moreover, based on sequence identity, it has been determined that the majority of the Cry toxins share three-domain structures with five highly conserved blocks in domain I [46][47]. Domain I is formed by a bundle of seven alpha-helices, with one central helix surrounded by six other alpha-helices [46]. The secondary structure of the alpha helices of domain I resemble bacterial pore-forming proteins, such as bacterial colicin I.3. The General Sporulation Mechanism in the Genus Bacillus
The challenge and the goal objective of sporulation in the genus Bacillus and in other spore-forming bacteria such as Clostridium is DNA protection and survival [17][54][55]. The latter process is accomplished by the Firmicutes phylum, despite pressure selection, evolution, and diversity in the set of molecular components comprising the program and that crosstalk [24][54][56][57][58][59][60] One of these systems is the Rap-Phr quorum sensing system, which regulates different bacterial processes, remarkably the commitment to sporulation in the Bacillus species [61][62][63][64][65][66][67][68]. How do Rap proteins act in sporulation? Rap proteins act as quorum sensors, forming a response regulator with a TPR (tetratricopeptide repeat) domain, a hydrophobic pocket able to bind the signaling peptide, thus inducing a conformational change and modulating regulator activities [66][69][70]. Therefore, RAP proteins act on phosphatases, an intermediary component of the sporulation phosphorelay system in Spo0F. Rap63 exhibited moderate activity during sporulation and is inhibited by the Phr63 peptide [42][71]. In Bacillus subtilis (frequently used as a model of the genus Bacillus), the starting sporulation programs is characterized by the phosphorylation of the master regulator Spo0A [24][31][72][73][74][75][76][77][78]. Across the genus Bacillus, the sporulation process is regulated by a cascade of sigma factors as follows: sigma F (σF); sigma E (σE): sigma G (σG), and sigma K (σK). Sigma factor K (σ(K) is a sigma factor conserved among the Bacillus genera, except in the genus Clostridium [24][31][42][78][79]. The sporulation program conserved among the members of the genus Bacillus comprises the following seven cytological and morphological changes [56][57][80] (Figure 1A): Stage 0 to Stage I, Axial filamentation; Stage II, Polar septum formation; Stage III, Forespore engulfment (ơF, ơE); Stage IV to Stage V, Cortex and coat assembly, and Stage VI to Stage VII, Spore maturation and mother-cell lysis. The morphological and cytological changes were impaired in spoIID, spoIIM, and spoIIP mutants [60][81][82][83][84][85][86] and in the spoIIB–spoVG double mutant. However, the deletion of the spoVS gene, controlled by ơH, permitted the spoIIB–spoVG double-mutant, to complete engulfment [75][76][87][88]. SpoVA proteins are involved in the uptake and release of nutrients from the core during the uptake of Ca2+ dipicolinic acid. The lytic enzymes SleB and CwlJ, found in bacilli, hydrolyze the spore cortex [42]. The spore is formed by an assembly process that involves a four-layer coat. The coating proteins described for Bacillus subtilis include cot, cot B, saf A, cot H, cot O, cot E, ger E, and cot E ger E [31] (Figure 1A). Assembly starts from the external outermost amorphous (crust) layer, followed by the rodlet, the honeycomb, the fibrous, and the nanodot particle layers, and finally, the undercoat/basement layer. Interestingly, under the exosporium of B. thuringiensis [89][90][91][92][93], a hexagonal honeycomb is exposed.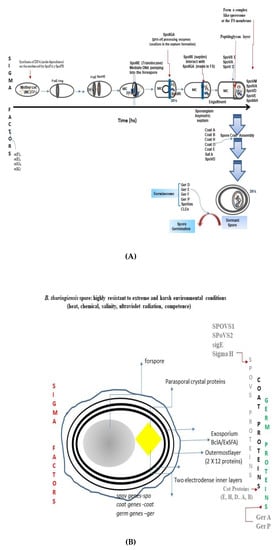
4. The General Spore Germination Program in the Genus Bacillus
In spore germination, molecular and morphogenetic changes are carried out as crosstalk among signals, germinant nutrients, and spore components in the committed endospore in order to awaken or break dormancy [118]. For an endospore, the fate and the decision to germinate encodes in the dormant spore. The program of spore germination refers to the multistep mechanism through which spores return to life, an awakening process that enables them to reenter into metabolic activity [118][119][120][121][122]. The knowledge of the components and signals in spore germination derives from studies of the model organism Bacillus subtilis [11][86][123][124]. However, there are current efforts to update and focus on other members of the genus Bacillus, especially those that constitute a problem in pathogenesis, health, agriculture, and in the food industry as well [122][125][126]. Which are the signals that initiate the process of awakening the dormant spore? What is known is that external signals (germinants, small molecules) that sense germination-specific proteins (GR)-like receptors localize in the outer and the inner coat of the spore [104][126][127][128][129][130][131]. The phenotypical characteristic of the spore-germination stage is considered a weak stage or spot in the life cycle of Bacilli species. During this stage, the spores become susceptible to physical, chemical, and environmental conditions, starting from the inner membrane coat of the spore, to render these accessible to nutrients of low molecular weight, ions, nutrients, Dodecylamine, and water, in order to flow through the core cortex [123][127][132][133][134]. The general mechanism of spore germination can be outlined as follows: (1) Germinant sensing; (2) Commitment to germinate; (3) Release of spore depot of dipicolinic acid (DPA); (4) Hydrolysis of peptidoglycan cortex spores; (5) Spore-core swelling and water uptake; (6) Cell-wall peptidoglycan remodeling, and (7) Restoration of core protein and inner-spore membrane–lipid mobility. This mechanism resembles a detailed general program for spore germination that is well characterized in B. subtilis, B. anthracis, and B. cereus. In B. thuringiensis, the differences in the molecular components that are involved in these processes are yet to be defined. One of the unsolved questions is related to the import and export of DPA and how the nutrients are sensed in the commitment spore [122]. Detailed program for spore germination in which all of the molecular components are outlined: (1) Activation. Nutrient germinant plus spores (minutes to hours). Lag phase, and (2) Commitment (a major change in IM permeability and structure). GERP proteins allow the access of nutrients into the inner membrane, low-molecular-weight, i.e., Dodecylamine, ions (H+, Na+, and K+). Channel formation by the multiple spore-specific SpoVA) (n = 7) in Bacillus subtilis (Setlow and Christie., 2020; [104][127][135][136]; (3) Release of pyridine-2, 6-dicarboxylic acid (dipicolinic acid [DPA]) chelated at 1:1 with divalent cations, predominantly Calcium (Ca2+DPA) through the IM channels; (4) Enzymatic lysozyme-mediated cleavage of the cortex, favoring permeability into small molecules in the inner coat, triggering spore germination; (5) Stage I. All of the Ca2+ DPA is released by the CLE cortex degradation, and this event leads to passage into stage II [137], and 6. Stage II cortex degradation is complete. The germ-cell wall and the core take up water and expand. This marks the initiation of germination, giving rise to growing cells and to the activation of metabolic activity [123][138].5. Implication of the Knowledge of Sporulation Structural Assembly and Germination in the Soil Bacterium B. thuringiensis
The stable and resistant nature of spores and the possibility of germinating and growing in a gut environment render them suitable for treatment in the form of probiotics and as vehicles for vaccine and drug delivery. Spore treatments have shown great promise in animal studies. However, human trials require going further. Nonetheless, spores might open the door to safe, effective, and easy-to-administer therapeutics [60][125][139]. It is pivotal to elucidate and understand the life cycle of spore-forming bacteria, especially those bacilli that threaten agriculture, the food industry, and health care [18][140][141]. t has become a promising and potential new avenue of alternatives against the biological control of insects and the application of biotechnology biomedicine. Furthermore, Bacillus thuringiensis is viewed as a biofactory for the production of proteins, but also of other products for bioremediation and for improvement as bioinsecticides. Moreover, parasporal delta endotoxins are highly specific against different orders of insects. However, Cyt proteins can exert a cytopathic effect on mammalian cells, specifically those changed by some types of cancer. This double sword of Bt marked the biotechnological success of B. thuringiensis; due to the versatility of Bt, great interest has emerged during the last two decades [3][4][35][96]. Work in this area ranges from basic research (mechanism of toxicity in insects) to applied science (the genetic engineering of economic crops with cry genes), the assembly of proteins for crystal formation (structural biology), and nanotechnology (drug-vehicle delivery or vehicles of subunit vaccines) [142][143][144]. Furthermore, B. thuringiensis can produce floating biofilms with a ring and a pellicle [145]. During sporulation, the spores remaining in the biofilm ring are of great utility for the food industry, because they confer spore resistance on washing and cleaning procedures. The spores can restart a new biofilm when food production has resumed [78].References
- Liu, S.; Moayeri, M.; Leppla, S.H. Anthrax lethal and edema toxins in anthrax pathogenesis. Trends Microbiol. 2014, 22, 317–325.
- Kokilaramani, S.; AlSalhi, M.S.; Devanesan, S.; Narenkuma, J.; Rajasekar, A.; Govarthanan, M. Bacillus megaterium-induced biocorrosion on mild steel and the effect of Artemisia pallens methanolic extract as a natural corrosion inhibitor. Arch. Microbiol. 2020, 202, 2311–2321.
- Melo, A.L.D.A.; Soccol, V.T.; Soccol, C.R. Bacillus thuringiensis: Mechanism of action, resistance, and new applications: A review. Crit. Rev. Biotechnol. 2016, 36, 317–326.
- Wang, C.; Li, W.; Kessenich, C.R.; Petrick, J.S.; Rydel, T.J.; Sturman, E.J.; Lee, T.C.; Glenn, K.C.; Edrington, T.C. Safety of the Bacillus thuringiensis-derived Cry1A.105 protein: Evidence that domain exchange preserves mode of action and safety. Regul.Toxicol. Pharmacol. 2018, 99, 50–60.
- Malovichko, Y.V.; Nizhnikov, A.A. Repertoire of the Bacillus thuringiensis virulence factors unrelated to major classes of protein toxins and its role in specificity of host-pathogen interactions. Antonets KS. Toxins 2019, 11, 347.
- Bach, E.; Rangel, C.P.; Ribeiro, I.D.A.; Passaglia, L.M.P. Pangenome analyses of Bacillus pumilus, Bacillus safensis, and Priestia megaterium exploring the plant-associated features of bacilli strains isolated from canola. Mol. Genet. Genom. 2022, 297, 1063–1079.
- Liu, L.; Li, Z.; Luo, X.; Zhang, X.; Chou, S.H.; Wang, J.; He, J. Which is stronger? A continuing battle between Cry toxins and insects. Front. Microbiol. 2021, 12, 665101.
- Stenfois Arnesen, L.P.; Fagerlund, A.; Granum, P.E. From soil to gut: Bacillus cereus and its food poisoning toxins. FEMS Microbiol. Rev. 2008, 32, 579–606.
- Logan, N.A. Bacillus and relatives in foodborne illness. J. Appl. Microbiol. 2012, 112, 417–429.
- Dalla-Vecchia, E.; Visser, M.; Stams, A.J.; Bernier-Latmani, R. Investigation of sporulation in the Desulfotomaculum genus: A genomic comparison with the genera Bacillus and Clostridium. Environ. Microbiol. Rep. 2014, 6, 756–766.
- Talukdar, P.K.; Sarker, M.R. Characterization of putative sporulation and germination genes in Clostridium perfringens Food-Poisoning Strain SM101. Microorganisms 2022, 10, 1481.
- Chakraborty, A.; Jayne ERattray, S.; Matthews, D.S.; Li, C.; Barker, B.; Jørgensen, B.B.; Hubert, C.R.J. Metabolic responses of thermophilic endospores to sudden heat-induced perturbation in marine sediment samples. Front. Microbiol. 2022, 13, 958417.
- Burke, K.E.; Lamont, J.T. Clostridium difficile infections: A worldwide disease. Gut Liver 2014, 8, 1–6.
- Sandhu, B.K.; McBride, S.H.M. Clostridioides difficile. Trends Microbiol. 2018, 26, 1049–1050.
- Shrestha, R.; Song, J.A. Terbium chloride influences Clostridium difficile spore germination. Anaerobe 2019, 58, 80–88.
- Diallo, M.; Kengen, S.W.M.; López-Contreras, A.M. Sporulation in solventogenic and acetogenic clostridia. Appl. Microbiol. Biotechnol. 2021, 105, 3533–3557.
- Setlow, P.; Johnson, E.A. Spores and their significance. In Food Microbiology, Fundamentals and Frontiers, 4th ed.; Doyle, M.P., Buchanan, R., Eds.; ASM Press: Washington, WA, USA, 2012; pp. 45–79.
- Bertuso, P.C.; Marangon, C.A.; Nitschke, M. Susceptibility of Vegetative Cells and Endospores of Bacillus cereus to Rhamnolipid Biosurfactants and Their Potential Application in Dairy. Microorganisms 2022, 10, 1860.
- Auger, S.; Ramarao, N.; Faille, C.; Fouet, A.; Aymerich, S.; Gohar, M. Biofilm formation and cell surface properties among pathogenic and nonpathogenic strains of the Bacillus cereus group. Appl. Environ. Microbiol. 2009, 75, 6616–6618.
- Vary, P.S.; Biedendieck, R.; Fuerch, T.; Meinhardt, F.; Rohde, M.; Deckwer, D.W.; Jahn, D. Bacillus megaterium--from simple soil bacterium to industrial protein production host. Appl. Microbiol. Biotechnol. 2007, 76, 957–967.
- Mallozzi, M.; Viswanathan, V.K.; Vedantam, G. Spore-forming bacilli and clostridia in human disease. Future Microbiol. 2010, 5, 1109–1123.
- Chi, Y.; Wang, D.; Jiang, M.; Chu Sh Wang, B.; Zhi, Y.; Zhou, P.; Zhang, D. Microencapsulation of Bacillus megaterium NCT-2 and its effect on remediation of secondary salinization soil. J. Microencapsul. 2020, 37, 134–143.
- Yang, M.; Zhu, G.; Korza, G.; Sun, X.; Setlow, P.; Li, J. Engineering Bacillus subtilis as a versatile and stable platform for production of nanobodies. Appl. Environ. Microbiol. 2020, 86, e02938-19.
- Galperin, M.Y.; Yutin, N.; Wolf, Y.I.; Vera-Álvarez, R.; Koonin, E.V. Conservation and evolution of the sporulation gene set in diverse members of the Firmicutes. J. Bacteriol. 2022, 204, e0007922.
- Hutchison, E.A.; Miller, D.A.; Angert, E.R. Sporulation in bacteria: Beyond the standard model. Microbiol. Spectr. 2014, 2, 1–15.
- Bate, A.R.; Bonneau, R.; Eichenberger, P. Bacillus subtilis Systems Biology: Applications of- Omics Techniques to the Study of Endospore Formation. Microbiol. Spectr. 2014, 2, 1–15.
- Kolek, J.; Diallo, M.; Vasylkivska, M.; Branska, B.; Sedlar, K.; López-Contreras, A.M.; Patakova, P. Comparison of expression of key sporulation, solventogenic and acetogenic genes in C. beijerinckii NRRL B-598 and its mutant strain overexpressing spo0A. Appl Microbiol Biotechnol. 2017, 101, 8279–8291.
- Al-Hinai, M.A.; Jones, S.W.; Papoutsakis, E.T. σK of Clostridium acetobutylicum is the first known sporulation-specific sigma factor with two developmentally separated roles, one early and one late in sporulation. J. Bacteriol. 2014, 196, 287–299.
- Al-Hinai, M.A.; Jones, S.W.; Papoutsakis, E.T. The Clostridium sporulation programs: Diversity and preservation of endospore differentiation. Microbiol. Mol. Biol. Rev. 2015, 79, 19–37.
- Zhou, Y.; Choi, Y.L.; Sun, M.; Yu, Z. Novel roles of Bacillus thuringiensis to control plant diseases. Appl. Microbiol. Biotechnol. 2008, 80, 563–572.
- Tu, Z.; Dekker, H.L.; Roseboom, W.; Swarge, B.N.; Setlow, P.; Brul, S.; Kramer, G. High resolution analysis of proteome dynamics during Bacillus subtilis sporulation. Int. J. Mol. Sci. 2021, 22, 9345.
- Wang, J.; Mei, H.; Qian, H.; Tang, Q.; Liu, X.; Yu, Z.; He, J. Expression profile and regulation of spore and parasporal crystal formation-associated genes in Bacillus thuringiensis. J. Proteome Res. 2013, 12, 5487–5501.
- Wang, J.; Mei, H.; Zheng, C.; Qian, H.; Cui, C.; Fu, Y.; Su, J.; Liu, Z.; Yu, Z.; He, J. The metabolic regulation of sporulation and parasporal crystal formation in Bacillus thuringiensis revealed by transcriptomics and proteomics. Mol. Cell. Proteomics. 2013, 12, 1363–1376.
- De Maagd, R.A.; Bravo, A.; Berry, N.; Crickmore, N.; Schnepf, H.E. Structure, diversity, and evolution of proteins toxins from spore-forming entomopathogenic bacteria. Annu. Rev. Genet. 2003, 37, 409–433.
- Bel, Y.; Ferré, J.; Hernández-Martínez, P. Bacillus thuringiensis toxins: Functional characterization and mechanism of action. Toxins 2020, 12, 785.
- Aronson, A.I.; Shai, Y. Why Bacillus thuringiensis insecticidal toxins are so effective: Unique features of their mode of action. FEMS Microbiol. Lett. 2001, 195, 1–8.
- Bravo, A.; Gill, S.; Soberón, M. Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control. Toxicon 2007, 49, 423–435.
- Bravo, A.; Likitvivatanavong, S.; Gill, S.S.; Soberón, M. Bacillus thuringiensis: A story of a successful bioinsecticide. Insect Biochem. Mol. Biol. 2011, 41, 423–431.
- Jurat-Fuentes, J.L.; Crickmore, N. Specificity determinants for Cry insecticidal proteins: Insights from their mode of action. J. Invertebr. Pathol. 2017, 142, 5–10.
- Pardo-López, L.; Soberón, M.; Bravo, A. Bacillus thuringiensis insecticidal three-domain Cry toxins: Mode of action, insect resistance and consequences for crop protection. FEMS Microbiol Rev. 2012, 37, 3–22.
- Ultee, E.; Ramijan, K.; Dame, R.T.; Briegel, A.; Claessen, D. Stress-induced adaptive morphogenesis in bacteria. Adv. Microb. Physiol. 2019, 74, 97–141.
- Fimlaid, K.A.; Shen, A. Diverse mechanisms regulate sporulation sigma factor activity in the Firmicutes. Curr. Opin. Microbiol. 2015, 24, 88–95.
- Wilcks, A.; Jayaswal, N.; Lereclus, D.; Andrup, L. Characterization of plasmid pAW63, a second self-transmissible plasmid in Bacillus thuringiensis subsp. kurstaki HD73. Microbiology 1998, 144, 1263–1270.
- Wilcks, A.; Smidt, L.; Økstad, O.A.; Kolstø, A.B.; Mahillon, J.; Andrup, L. Replication mechanism and sequence analysis of the replicon of pAW63, a conjugative plasmid from Bacillus thuringiensis. J. Bacteriol. 1999, 181, 3193–3200.
- Van der Auwera, G.; Andrup, L.; Mahillon, J. Conjugative plasmid pAW63 brings new insights into the genesis of the Bacillus anthracis virulence plasmid pXO2 and of the Bacillus thuringiensis plasmid pBT9727. BMC Genom. 2005, 6, 103–114.
- Li, J.D.; Carroll, J.; Ellar, D.J. Crystal structure of insecticidal delta-endotoxin from Bacillus thurigiensis at 2.5 Ǻ resolution. Nature 1991, 352, 815–821.
- Bravo, A. Phylogenetic relationships of Bacillus thuringiensis δ-endotoxin family proteins and their functional domains. J. Bacteriol. 1997, 179, 2793–2801.
- Grochulski, P.; Masson, L.; Borisova, S.; Pusztai-Carey, M.; Schwartz, J.L.; Brousseau, R.; Cygler, M. Bacillus thuringiensis Cry1A(a) insecticidal toxin: Crystal structure and channel formation. J. Mol. Biol. 1995, 254, 447–464.
- Derbyshire, D.J.; Ellar, D.J.; Li, J. Crystallization of the Bacillus thuringiensis toxin Cry2Ac and its complex with the receptor ligand N-Acetylgalactosamine. Acta Cryst. Sect. 2001, 57, 1938–1944.
- Morse, R.J.; Yamamoto, T.; Stroud, R.M. Structure of Cry2Aa suggests an unexpected receptor binding epitope. Structure 2001, 9, 409–417.
- Galistsky, N.; Cody, V.; Wojtczak, D.; Ghosh, J.R.; Luft, W.P.; English, L. Structure of the insecticidal bacterial δ-endotoxin Cry3Bb1 of Bacillus thuringiensis. Acta Crystallogr. Sect. D 2001, 57, 1101–1109.
- Boonserm, P.; Davis, P.; Ellar, D.J.; Li, J. Crystal structure of the mosquito-larvacidal toxin Cry4Ba and its biological implications. J. Mol. Biol. 2005, 348, 363–382.
- Boonserm, P.; Mo, M.; Angsuthanasombat, A.; Lescar, J. Structure of the functional form of the mosquito larvicidal Cry4Aa toxin from Bacillus thuringiensis at a 2.8 angstrom resolution. J. Bacteriol. 2006, 188, 3391–3401.
- Setlow, P. I will survive: DNA protection in bacterial spores. Trends Microbiol. 2007, 15, 172–180.
- Yutin, N.; Galperin, M.Y. A genomic update on clostridial phylogeny: Gram-negative spore formers and other misplaced clostridia. Environ. Microbiol. 2013, 15, 2631–2641.
- Veering, J.W.; Stewart, E.J.; Berngruber, T.W.; Taddei, F.; Kuipens, O.; Hamoen, L.W. Bet hedging and epigenetic inheritance in bacterial cell development. Proc. Natl. Acad. Sci. USA 2008, 105, 4393–4398.
- Tan, I.S.; Ramamurthi, K.S. Spore formation in Bacillus subtilis. Environ. Microbiol. Rep. 2014, 6, 212–225.
- Rigden, D.J.; Galperin, M.Y. Sequence analysis of GerM and SpoVS, uncharacterized bacterial sporulation’ proteins with widespread phylogenetic distribution. Bioinformatics 2018, 24, 1793–1797.
- Traag, B.A.; Pugliese, A.; Eisen, J.A.; Losick, R. Gene conservation among endospore-forming bacteria reveals additional sporulation genes in Bacillus subtilis. J. Bacteriol. 2013, 195, 253–260.
- Dworkin, J. Protein targeting during Bacillus subtilis sporulation. Microbiol. Spectr. 2014, 2, 1–12.
- Perego, M.; Hoch, J.A. Cell-cell communication regulates the effects of protein aspartate phosphatases on the phosphorelay controlling development in Bacillus subtilis. Proc. Natl. Acad. Sci. USA 1996, 93, 1549–1553.
- Perego, M. A peptide export-import control circuit modulating bacterial development regulates protein phosphatases of the phosphorelay. Proc. Natl. Acad. Sci. USA 1997, 94, 8612–8617.
- Perego, M. A new family of aspartyl phosphate phosphatases targeting the sporulation transcription factor Spo0A of Bacillus subtilis. Mol. Microbiol. 2001, 42, 133–143.
- Jiang, M.; Shao, W.; Perego, M.; Hoch, J.A. Multiple histidine kinases regulate entry into stationary phase and sporulation in Bacillus subtilis. Mol. Microbiol. 2000, 38, 535–542.
- Grenha, R.; Rzechorzek, N.J.; Brannigan, J.A.; de Jong, R.N.; Ab, E.; Diercks, T.; Truffault, V.; Ladds, J.C.; Fogg, M.J.; Bongiorni, C.; et al. Structural characterization of Spo0E-like protein-aspartic acid phosphatases that regulate sporulation in bacilli. J. Biol. Chem. 2006, 281, 37993–38003.
- Grenha, R.; Slamti, L.; Nicaise, M.; Refes, Y.; Lereclus, D.; Nessler, S. Structural basis for the activation mechanism of the PlcR virulence regulator by the quorum-sensing signal peptide, P.a.p.R. Proc. Natl. Acad. Sci. USA 2013, 110, 1047–1052.
- Hayashi, K.; Kensuke, T.; Kobayashi, K.; Ogasawara, N.; Ogura, M. Bacillus subtilis RghR (YvaN) represses rapG and rapH, which encode inhibitors of expression of the srfA operon. Mol. Microbiol. 2006, 59, 1714–1729.
- Díaz, A.R.; Stephenson, S.; Green, J.M.; Levdikov, V.M.; Wilkinson, A.J.; Perego, M. Functional role for a conserved aspartate in the Spo0E signature motif involved in the dephosphorylation of the Bacillus subtilis sporulation regulator Spo0A. J. Biol. Chem. 2008, 283, 2962–2972.
- Declerck, N.; Bouillaut, L.; Chaix, D.; Rugani, N.; Slamti, L.; Hoh, F.; Lereclus, D.; Arold, S.T. Structure of PlcR: Insights into virulence regulation and evolution of quorum sensing in Gram-positive bacteria. Proc. Natl. Acad. Sci. USA 2007, 104, 18490–18495.
- Slamti, L.; Perchat, S.; Huillet, E.; Lereclus, D. Quorum sensing in Bacillus thuringiensis is required for completion of a full infectious cycle in the insect. Toxins 2014, 6, 2239–2255.
- Ishikawa, S.; Core, L.; Perego, M. Biochemical characterization of aspartyl phosphate phosphatase interaction with a phosphorylated response regulator and its inhibition by a pentapeptide. J. Biol. Chem. 2002, 77, 20483–20489.
- Sonenshein, A.L. Control of sporulation initiation in Bacillus subtilis. Curr. Opin. Microbiol. 2000, 3, 561–566.
- Burbulys, D.; Trach, K.A.; Hoch, J.A. Initiation of sporulation in B. subtilis is controlled by a multicomponent phosphorelay. Cell 1991, 64, 545–552.
- Molle, V.; Fujita, M.; Jensen, S.T.; Eichenberger, P.; González-Pastor, J.E.; Liu, J.S.; Losick, R. The Spo0A regulon of Bacillus subtilis. Mol. Microbiol. 2003, 50, 1683–1701.
- Barák, I.; Youngman, P. SpoIIE mutants of Bacillus subtilis comprise two distinct phenotypic classes consistent with a dual functional role for the SpoIIE protein. J. Bacteriol. 1996, 178, 4984–4989.
- Barák, I.; Muchová, K.; Labajová, N. Asymmetric cell division during Bacillus subtilis sporulation. Future Microbiol. 2019, 14, 353–363.
- Pottathil, M.; Lazazzera, B.A. The extracellular Phr peptide-Rap phosphatase signaling circuit of Bacillus subtilis. Front. Biosci. 2003, 8, d32–d45.
- Fagerlund, A.; Dubois, T.; Okstad, O.A.; Verplaetse, E.; Gilois, N.; Bennaceur, I.; Perchat, M.; Myriam, G.; Aymerich, S.; Kolstø, A.-B.; et al. SinR controls enterotoxin expression in Bacillus thuringiensis biofilms. PLoS ONE 2014, 9, e87532.
- Dyrdahl-Young, R.; Hu, W.; DiGennaro, P. Temporal expression patterns of Pasteuria spp. sporulation genes. J. Nematol. 2019, 29, e2019–e2039.
- Boonstra, M.; de Jong, I.G.; Scholefield, G.; Murray, H.; Kuipers, O.P.; Veering, J.W. SpoOA regulates chromosome copy number during sporulation by directly binding to the origin of replication in Bacillus subtilis. Mol. Microbiol. 2013, 87, 925–938.
- Eichenberger, P.; Jensen, S.T.; Conlon, E.M.; van Ooij, C.; Silvaggi, J.; González-Pastor, J.E.; Fujita, M.; Ben-Yehuda, S.; Stragier, P.; Liu, J.S.; et al. The Sigma E regulon and the identification of additional sporulation genes in Bacillus subtilis. J. Mol. Biol. 2003, 327, 945–972.
- McKenney, P.T.; Driks, A.; Eichenberger, P. The Bacillus subtilis endospore: Assembly and functions of the multilayered coat. Nat. Rev. Microbiol. 2013, 11, 33–44.
- Higgins, D.; Dworkin, J. Recent progress in Bacillus subtilis sporulation. FEMS Microbiol. Rev. 2012, 36, 131–148.
- Pérez, A.R.; Abanes-De Mello, A.; Pogliano, K. Suppression of engulfment defects in Bacillus subtilis by elevated expression of the motility regulon. J. Bacteriol. 2006, 188, 1159–1164.
- Plomp, M.; Carroll, A.M.; Setlow, P.; Malkin, A.J. Architecture and assembly of the Bacillus subtilis spore coat. PLoS ONE 2014, 9, e108560.
- Daniel, R.A.; Drake, S.; Buchanan, C.E.; Scholle, R.; Errington, J. The Bacillus subtilis spoVD gene encodes a mother-cell-specific Penicillin-binding protein required for spore morphogenesis. J. Mol. Biol. 1994, 235, 209–220.
- Resnekov, O.; Driks, A.; Losick, R. Identification and characterization of sporulation gene spoVS from Bacillus subtilis. J. Bacteriol. 1995, 177, 5628–5635.
- Matsuno, K.; Sonenshein, A.L. Role of SpoVG in asymmetric septation in Bacillus subtilis. J. Bacteriol. 1999, 181, 3392–3401.
- Radford, D.S.; Wan, Q.; Tzokov, S.; Moir, A.; Bullough, P.A. Molecular tiling on the surface of a bacterial spore- the exosporium of the Bacillus anthracis/cereus/thuringiensis group. Mol Microbiol. 2017, 104, 539–552.
- Henriques, A.O.; Morán Jr, C.H.P. Structure, assembly, and function of the spore surface layers. Annu. Rev. Microbiol. 2007, 61, 555–588.
- Chen, X.; Gao, T.; Peng, Q.; Zhang, J.; Chai, Y.; Song, F. Novel cell wall hydrolase CwlC from Bacillus thuringiensis is essential for mother cell lysis. Appl. Environ. Microbiol. 2018, 84, e02640-17.
- Plomp, M.; Leighton, J.T.; Wheeler, K.E.; Malkin, A.J. Architecture and high-resolution structure of Bacillus thuringiensis and Bacillus cereus spore coat surfaces. Langmuir 2005, 21, 7892–7898.
- Peng, Q.; Wu, J.; Chen, X.; Qiu, L.; Zhang, J.; Tian, H.; Song, F. Disruption of two-component system LytSR affects forespore engulfment in Bacillus thuringiensis. Front. Cell. Infect. Microbiol. 2017, 7, 468.
- Cardoso, P.; Fazion, F.; Perchat, S.; Buisson, C.; Vilas-Bôas, G.; Lereclus, D. RapPhr systems from plasmids pAW63 and pHT8-1 act together to regulate sporulation in the Bacillus thuringiensis serovar kurstaki HD73 strain. Appl. Environ. Microbiol. 2020, 86, e01238–e20.
- Hou, S.H.; Zhang, R.; Lereclus, D.; Peng, Q.; Zhang, J.; Slamti, L.; Song, F. The transcription factor CpcR determines cell fate by modulating the initiation of sporulation in Bacillus thuringiensis. Appl. Environ. Microbiol. 2022, 88, e0237421.
- Liu, X.; Zhang, R.; Hou, S.; HLiu, H.; Wang, J.; Yu, Q.; Peng, O.; Song, F. Identification and functional characterization of two homologous SpoVS proteins involved in sporulation of Bacillus thuringiensis. Microbiol. Spectr. 2021, 9, e0088121.
- Dubois, T.; Lemy, C.; Perchat, S.; Lereclus, D. The signaling peptide NprX controlling sporulation and necrotrophism is imported into Bacillus thuringiensis by two oligopeptide permease systems. Mol Microbiol. 2019, 112, 219–232.
- Li, Z.; Yu, Z.; Liu, L.; Chou, S.H.-H.; Wang, J.; He, J. 6S-1 RNA contributes to sporulation and parasporal crystal formation in Bacillus thuringiensis. Front. Microbiol. 2020, 11, 1–10.
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685.
- Byrd, B.; Camilleri, E.; Korza, G.; Craft, D.L.; Green, J.; Rocha, G.M.; Mok, W.W.K.; Cairmano, M.J.; Setlow, P. Levels and characteristics of mRNAs in Spores of Firmicute Species. J. Bacteriol. 2021, 203, e0001721.
- Riley, E.P.; López-Garrido, J.; Sugie, J.; Liu, R.B.; Pogliano, K. Metabolic differentiation and intercellular nurturing underpin bacterial endospore formation. Sci. Adv. 2021, 7, eabd6385.
- Korza, G.; Camilleri, E.; Green, J.; Robinson, J.; Nagler, K.; Moeller, R.; Caimano, M.J.; Setlow, P. Analysis of mRNAs in spores of Bacillus subtilis. J. Bacteriol. 2019, 201, e00007-19.
- Swarge, B.; Abhyankar, W.; Jonker, M.; Hoefsloot, H.; Kramer, G.; Setlow, P.; Brul, S.; de Koning, L.J. Integrative analysis of proteome and transcriptome dynamics during Bacillus subtilis spore revival. mSphere 2020, 5, e00463-20.
- Setlow, P.; Christie, G. Bacterial spore mRNA: What’s up with that? Front. Microbiol. 2020, 11, 596092.
- Jeng, Y.H.; Doi, R. Messenger ribonucleic acid of dormant spores of Bacillus subtilis. J. Bacteriol. 1974, 119, 514–521.
- Segev, E.; Smith, Y.; Ben-Yehuda, S. RNA dynamics in aging bacterial spores. Cell 2012, 148, 139–149.
- Camp, A.H.; Losick, R. A novel pathway of intercellular signalling in Bacillus subtilis involves a protein with similarity to a component of type III secretion channels. Mol. Microbiol. 2008, 69, 402–417.
- Camp, A.H.; Losick, R. A feeding tube model for activation of a cell specific transcription factor during sporulation in Bacillus subtilis. Genes. Dev. 2009, 23, 1014–1024.
- Crawshaw, A.D.; Serrano, M.; Stanley, W.A.; Henriques, A.O.; Salgado, P.S. A mother cell-to-forespore channel: Current understanding and future challenges. FEMS Microbiol. Lett. 2014, 358, 129–136.
- Meisner, J.; Wang, X.; Serrano, M.; Henriques, A.O.; Moran, C.P., Jr. A channel connecting the mother cell and forespore during bacterial endospore formation. Proc. Natl. Acad. Sci. USA 2008, 105, 15100–15105.
- Singh, R.P.; Setlow, B.; Setlow, P. Levels of small molecules and enzymes in the mother cell compartment and the forespore of sporulatin Bacillus megaterium. J. Bacteriol. 1977, 130, 1130–1138.
- Ghosh, A.; Manton, J.D.; Mustafa, A.R.; Gupta, M.; Ayuso-García, A.; Rees, E.J.; Graham, C. Proteins encoded by the gerP operon are localized to the inner coat in Bacillus cereus spores and are dependent on GerPA and SafA for assembly. Appl. Environ. Microbiol. 2018, 84, e00760-18.
- De Francesco, M.; Jacobs, J.Z.; Nunes, F.; Serrano, M.; McKenney, P.T.; Chua, M.H.; Henriques, A.O.; Eichenberger, P. Physical interaction between coat morphogenetic proteins SpoVID and CotE is necessary for spore encasement in Bacillus subtilis. J. Bacteriol. 2012, 194, 4941–4950.
- Stewart, G.C. The exosporium layer of bacterial spores: A connection to the environment and the infected host. Microbiol. Mol. Biol. Rev. 2015, 79, 437–457.
- Takamatsu, H.; Watabe, K. Assembly and genetics of spore protective structures. Cell. Mol. Life Sci. 2002, 59, 434–444.
- Popham, D.L.; Bernhards, C.B. Spore peptidoglycan. Microbiol. Spectr. 2015, 3, 1–21.
- Abanes-De Mello, A.; Sun, Y.-I.; Aung, S.; Pogliano, K. A cytoskeleton-like role for the bacterial cell wall during engulfment of the Bacillus subtilis fore-spore. Genes. Dev. 2002, 16, 3253–3264.
- Paidhungat, M.; Setlow, P. Spore germination and outgrowth. In Bacillus subtilis and Its Relatives: From Genes to Cells; Onenshein, A.L., Hoch, J.A., Losick, R., Eds.; American Society for Microbiology: Washington, WA, USA, 2002; pp. 537–548.
- Paredes-Sabja, D.; Setlow, P.; Sarker, M.R. Germination of spores of Bacillales and Clostridiales species: Mechanisms and proteins involved. Trends Microbiol. 2011, 19, 85–94.
- Behravan, J.; Chirakkal, H.; Masson, A.; Moir, A. Mutations in the gerP locus of Bacillus subtilis and Bacillus cereus affect access of germinants to their targets in spores. J. Bacteriol. 2000, 182, 1987–1994.
- Moir, A.; Cooper, G. Spore germination. Microbiol. Spectr. 2015, 3, 1–19.
- Setlow, P.; Wang SHLi, Y.Q. Germination of spores of the orders Bacillales and Clostridiales. Annu. Rev. Microbiol. 2017, 71, 459–477.
- Kaieda, S.; Setlow, B.; Setlow, P.; Halle, B. Mobility of core water in Bacillus subtilis spores by 2H NMR. Biophys. J. 2013, 105, 2016–2123.
- Edwards, A.N.; McBride, S.M. Initiation of sporulation in Clostridium difficile: A twist on the classic model. FEMS Microbiol. Lett. 2014, 358, 110–118.
- Koopman, N.; Remijas, L.; Seppen, J.; Setlow, P.; Brul, S. Mechanisms and applications of bacterial sporulation and germination in the intestine. Int. J. Mol. Sci. 2022, 23, 3405.
- Christie, G.; Setlow, P. Bacillus spore germination: Knowns, unknowns and what we need to learn. Cell. Signal. 2020, 74, 109729.
- Setlow, P. Spore germination. Curr. Opin. Microbiol. 2003, 6, 550–556.
- Cortezzo, D.E.; Setlow, B.; Setlow, P. Analysis of the action of compounds that inhibit the germination of spores of Bacillus species. J. Appl. Microbiol. 2004, 96, 725–741.
- Chen, D.; Huang, S.S.; Li, Y.Q. Real-time detection of kinetic germination and heterogeneity of single Bacillus spores by laser tweezers Raman spectroscopy. Anal. Chem. 2006, 78, 6936–6941.
- Shah, I.M.; Laaberki, M.H.; Popham, D.L.; Dworkin, J. A eukaryotic Ser/Thr kinase signals bacteria to exit dormancy in response to peptidoglycan fragments. Cell 2008, 135, 486–496.
- Ramírez-Peralta, A.; Zhang, P.; Li, Y.; Setlow, P. Effects of sporulation conditions on the germination and germination protein levels of Bacillus subtilis spores. Appl. Environ. Microbiol. 2012, 78, 2689–2697.
- Setlow, P. When the sleepers wake: The germination of spores of Bacillus species. J. Appl. Microbiol. 2013, 115, 1251–1268.
- Setlow, P. Germination of spores of Bacillus species: What we know and do not know. J. Bacteriol. 2014, 196, 1297–1305.
- Knudsen, S.M.; Cermak, N.; Delgado, F.F.; Setlow, B.; Setlow, P.; Manalis, S.R. Water and small-molecule permeation of dormant Bacillus subtilis spores. J. Bacteriol. 2016, 198, 168–177.
- Yi, X.; Setlow, P. Studies of the commitment step in the germination of spores of Bacillus species. J. Bacteriol. 2010, 192, 3424–3433.
- Stewart, G.S.A.; Johnstone, K.B.; Hagelberg, F.; Ellar, D.J. Commitment of bacterial spores to germinate: A measure of the trigger reaction. Biochem. J. 1981, 198, 101–106.
- Griffiths, K.K.; Zhang, J.; Cowan, A.E.; Yu, J.; Setlow, P. Germination proteins in the inner membrane of dormant Bacillus subtilis spores colocalize in a discrete cluster. Mol. Microbiol. 2011, 81, 1061–1077.
- Xiao, Y.; Francke, C.; Abee, T.; Wells-Bennik, M.H. Clostridial spore germination versus bacilli: Genome mining and current insights. Food Microbiol. 2011, 28, 266–274.
- Setlow, P.; Christie, G. What’s new and notable in bacterial spore killing! World J. Microbiol. Biotechnol. 2021, 37, 144.
- Tehri, N.; Kumar, N.; Raghu, H.V.; Thakur, G.; Kumar-Sharma, K. Role of stereospecific nature of germinants in Bacillus megaterium spores germination. J. Biotech. 2017, 7, 259.
- Andryukov, B.G.; Karpenko, A.A.; Lyapun, I.N. Learning from nature: Bacterial spores as a target for current technologies in medicine. Sovrem. Tekhnologii Med. 2021, 12, 105–122.
- Moreno-Fierros, L.; García, N.; Gutiérrez, R.; López-Revilla, R.; Vázquez-Padrón, R.I. Intranasal, rectal and intraperitoneal immunization with protoxin Cry1Ac from Bacillus thurengiensis induces compartmentalized serum, intestinal, vaginal and pulmonary immune responses in Balb/c mice. Microbes Infect. 2000, 2, 885–890.
- Favela-Hernández, J.M.; Balderas, R.I.; Guerrero, G.G. The potential of a commercial product based on Bacillus thuringiensis Cry1A-Cry2A as a immunogen and adjuvant. Madridge J. Immunol. 2018, 2, 58–64.
- Guerrero, G.G.; Tuero, I. Adjuvant compounds: Friends in vaccine formulations against infectious diseases. Hum. Vaccines Immunother. 2021, 17, 3539–3550.
- Peng, Q.; Yu, Q.; Song, F. Expression of cry genes in Bacillus thuringiensis biotechnology. Appl. Microbiol. Biotechnol. 2019, 103, 1617–1626.
