Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Camila Xu and Version 1 by Rafael Antonio Nascimento Ramos.
Species within the genus Ixodiphagus (Hymenoptera: Encyrtidae) are natural parasitoid wasps of ticks (Acari: Ixodida), which were first described more than a century ago, in Haemaphysalis leporispalustris from Texas, United States (USA).
- Ixodiphagus hookeri
- biological control
- ixodid
- argasid
1. Introduction
Species within the genus Ixodiphagus (Hymenoptera: Encyrtidae) are natural parasitoid wasps of ticks (Acari: Ixodida) [1], which were first described more than a century ago, in Haemaphysalis leporispalustris from Texas, United States (USA) [2]. The etymology of the genus name Ixodiphagus (from Greek ixod = tick and phage = eater) alludes to its parasitoid behavior. After its first description, other species of “tick eaters” within this genus were formally described worldwide [3,4,5,6][3][4][5][6].
Currently, at least ten species of these parasitoids are considered valid, namely Ixodiphagus texanus Howard, 1907; Ixodiphagus hookeri Howard, 1908; Ixodiphagus mysorensis Mani, 1941; Ixodiphagus hirtus Nikolskava, 1950; Ixodiphagus theilerae Fielder, 1953; Ixodiphagus biroi Erdos, 1956; Ixodiphagus sagarensis Geevarghese, 1977; Ixodiphagus taiaroaensis Heath and Cane, 2010; Ixodiphagus sureshani Hayat and Islam, 2011; and Ixodiphagus aethes Hayat and Veenakumari, 2015. These insects are small, generally measuring less than 1 cm in length, blackish in color, and exhibiting the typical appearance of members of the superfamily Chalcidoidea, and display similar biological and ecological features [7].
Despite being known for over a century, many knowledge gaps remain about the biology of these parasitoid wasps, with most information limited to I. hookeri [5,8][5][8]. The life cycle of these wasps starts when gravid females lay eggs inside the tick’s body. After an incubation period, the larvae hatch and feed on the internal content of the tick [7]. Approximately 30–57 days after oviposition, new adult male and female wasps emerge from the dead tick, mating and continuing their life cycle [9]. Based on this life cycle, the use of Ixodiphagus spp. as an agent for biological control of ticks has inspired the interest of the scientific community [10]. In addition, populations of I. hookeri may have different developmental times, parasitism rates, and host preferences according to the geographical area of occurrence [10], which may explain the failure, or the limited efficacy, of these wasps in the control of ticks in field studies [11,12][11][12].
2. Biology of Ixodiphagus spp. and Geographic Distribution
Information on the biology of Ixodiphagus species is insufficient and mainly limited to experimental studies [10]. The entire life cycle ranges from 28 to 70 days, and starts when female wasps lay eggs into ticks through the penetration of their ovipositor into the tick’s body (Figure 1). After hatching, larvae (Figure 2) develop inside the tick. While no information is available about the pupal stage, adult wasps emerge from their tick hosts through a hole at the posterior end, with mating occurring soon after the emergence [9]. There have been no studies assessing the number of Ixodiphagus eggs released by females in natural conditions. However, based on experimental studies, it is estimated that during the entire life span, I. hookeri and I. texanus lay about 120 and 200 eggs, respectively [23,24][13][14].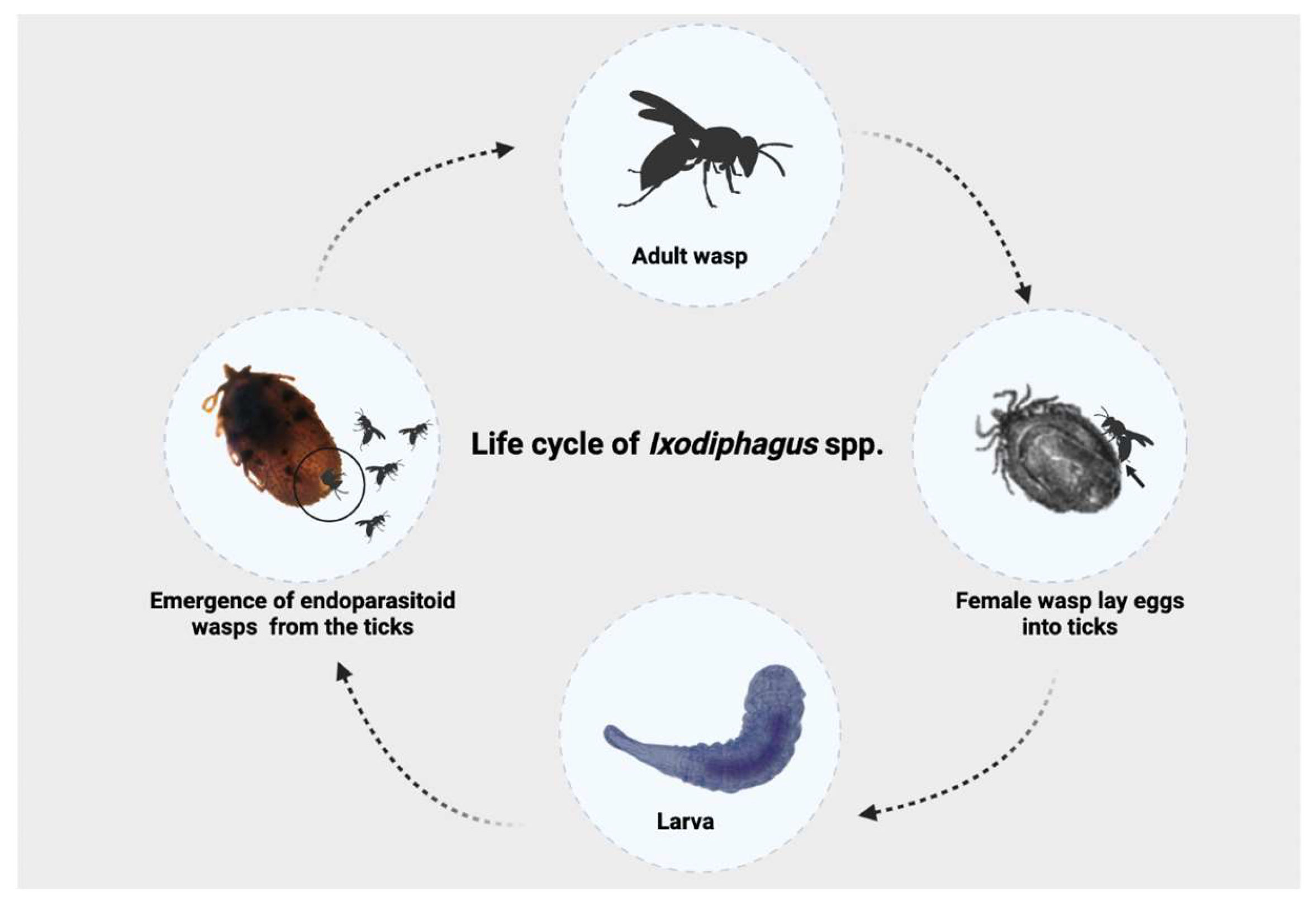
Figure 1.
Life cycle of
Ixodiphagus
spp.
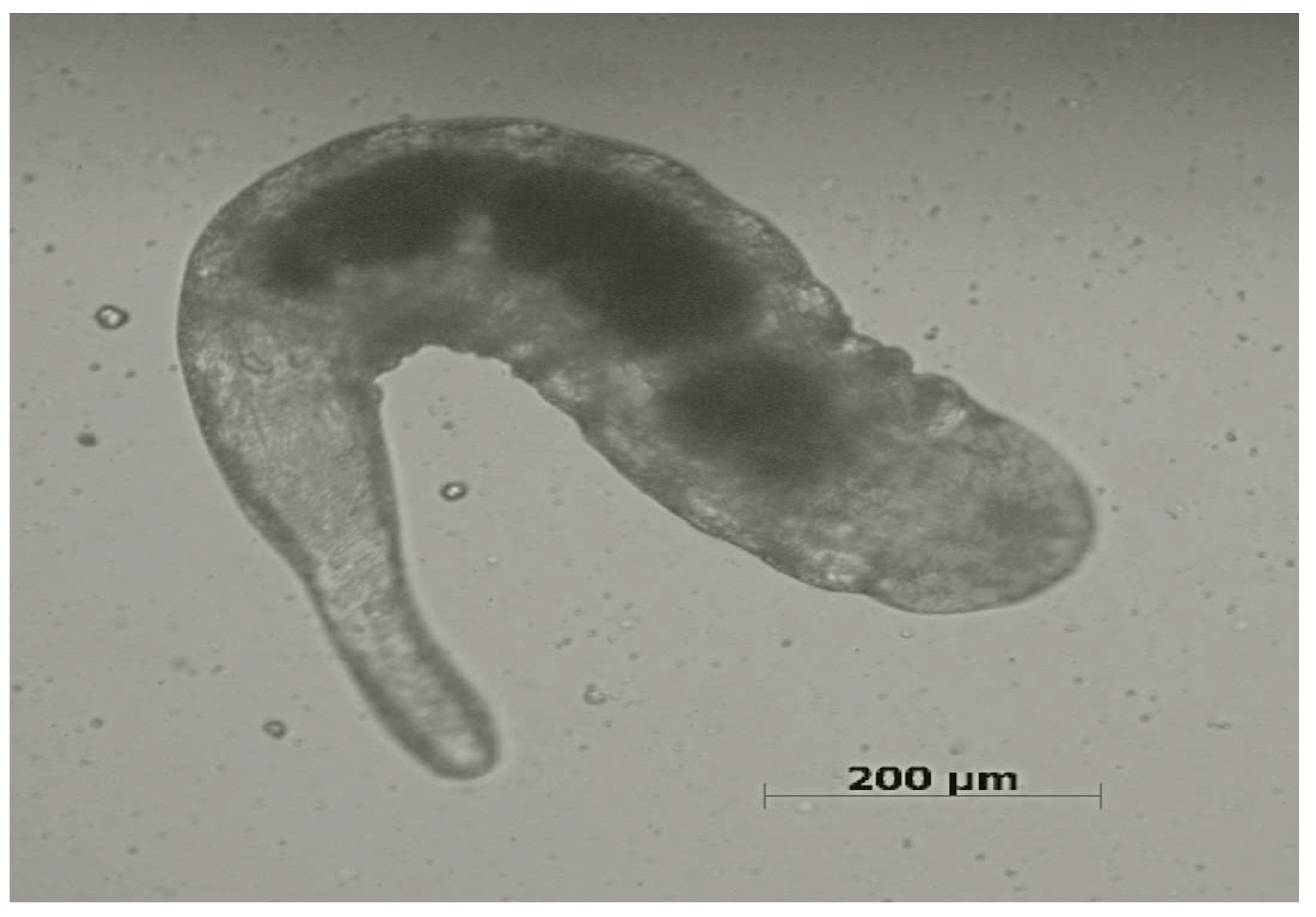
Figure 2.
Ixodiphagus
sp. larva in a
Rhipicephalus sanguineus
s.l. tick (Scale bar = 200 μm).
Table 1.
Distribution of
Ixodiphagus
spp. parasitizing different tick species in the world.
| Parasitoid | Tick | Tick Life Stage | Country | Reference |
|---|---|---|---|---|
| I. texanus | H. leporispalustris | Nymph | United States | [2] |
| I. hookeri | R. sanguineus | Nymph | United States | [41][31] |
| I. hookeri | R. sanguineus, D. marginatus | Nymph | United States | [9] |
| I. hookeri | I. ricinus | Nymph | France | [42][32] |
| I. hookeri | H. concinna, D. reticulatus, D. venustus, R. sanguineus |
NA | France | [43][33] |
| I. hookeri | R. sanguineus | Nymph | Brazil | [44][34] |
| I. hookeri | R. sanguineus | NA | India | [45][35] |
| I. hookeri | D. nitens | NA | United States | [46][36] |
| I. hookeri | D. variabilis | NA | United States | [11] |
| I. hookeri | H. aegyptium | NA | South Africa | [47][37] |
| I. hookeri | R. sanguineus | Nymph | Nigeria | [48][38] |
| I. hookeri | I. cookei | Nymph | United States | [49][39] |
| I. hookeri | R. sanguineus | NA | United States | [50][40] |
| I. texanus | H. leporispalustris | Nymph | United States | [51][41] |
| I. hookeri | R. sanguineus | Nymph | United States | [52][42] |
| I. mysorensis | Ornithodorus sp. | NA | India | [40][30] |
| I. texanus | I. persulcatus | Nymph | Russia | [53][43] |
| I. hookeri | I. ricinus | Nymph | Czech Republic/Slovakia (Czechoslovakia) | [54][44] |
| I. hookeri | R. sanguineus | Nymph | Kenya | [55][45] |
| I. hookeri | R. sanguineus | Nymph | Africa | [56][46] |
| Ixodiphagus sp. | H. bancrofti, H. bremneri, I. holocyclus, I. tasmani | NA | Australia | [57][47] |
| I. hookeri | R. sanguineus | NA | Indonesia | [58][48] |
| I. hookeri | R. sanguineus | Nymph | Malaysia | [59][49] |
| I. texanus | H. leporispalustris | Larva, Nymph | Canada | [60][50] |
| I. hookeri | I. dammini | Nymph | United States | [21][51] |
| I. hookeri | H. punctata | Nymph | Spain | [61][52] |
| I. hookeri | A. variegatum | Nymph | Kenya | [62][53] |
| I. hookeri | I. ricinus | NA | France | [63][54] |
| I. texanus | I. dammini | Nymph | United States | [64][55] |
| I. hookeri | R. sanguineus | Nymph | Mexico | [65][56] |
| I. hookeri | I. scapularis | Nymph | United States | [66][57] |
| I. hookeri | I. scapularis | Nymph | United States | [25][15] |
| I. hookeri | A. variegatum | Nymph | Kenya | [67][58] |
| I. hookeri | I. scapularis | Nymph | United States | [29][19] |
| I. hookeri | R. sanguineus | Nymph | Venezuela | [68][59] |
| I. hookeri | A. variegatum | Nymph | Kenya | [37][27] |
| I.hookeri | H. concinna | Nymph | Slovakia | [26][16] |
| I. taiaroaensis | I. uriae, I. eudyptidis | Larva, Nymph | New Zealand | [69][60] |
| I. hookeri | I. ricinus | Nymph | Germany | [10] |
| I. hookeri | I. ricinus | Nymph | Netherlands | [32][22] |
| I. hookeri, I. texanus | R. sanguineus, Amblyomma sp. | Nymph | Brazil | [70][61] |
| I. hookeri | I. ricinus | Nymph | France | [32][22] |
| I. hookeri, I. texanus | R. sanguineus | Nymph | Panama | [71][62] |
| I. hookeri | I. ricinus | Nymph, Adult | Italy | [3] |
| I. hookeri | I. ricinus | Nymph | Slovakia | [22][63] |
| Ixodiphagus sp. | R. sanguineus | Nymph, Adult | Brazil | [4] |
| I. hookeri | I. ricinus | Nymph | Finland | [5] |
| I. hookeri | R. sanguineus | Nymph | United States | [72][64] |
| I. hookeri | R. microplus, I. persulcatus, D. silvarum, H. concinna |
Adult | Côte d’Ivoire, Senegal, Russia | [39][29] |
| I. hookeri | I. ricinus | Larva, Nymph | Netherlands | [8] |
| I. hookeri | I. ricinus, H. concinna | Nymph | Slovakia | [1] |
| I. hookeri | I. ricinus | Nymph | France | [35][25] |
| I. hookeri | I. ricinus | Nymph | United Kingdom | [73][65] |
| I. hookeri | A. nodosum | Nymph, Adult | Brazil | [74][66] |
| I. hookeri | I. ricinus | Nymph | Hungary | [6] |
NA: Not available.
References
- Buczek, A.; Buczek, W.; Bartosik, K.; Kulisz, J.; Stanko, M. Ixodiphagus hookeri wasps (Hymenoptera: Encyrtidae) in two sympatric tick species Ixodes ricinus and Haemaphysalis concinna (Ixodida: Ixodidae) in the Slovak Karst (Slovakia): Ecological and biological considerations. Sci. Rep. 2021, 11, 11310.
- Howard, L.O. A chalcidoid parasite of a tick. Ent. News. 1907, 18, 375–378.
- Ramos, R.A.; Campbell, B.E.; Whittle, A.; Lia, R.P.; Montarsi, F.; Parisi, A.; Dantas-Torres, F.; Wall, R.; Otranto, D. Occurrence of Ixodiphagus hookeri (Hymenoptera: Encyrtidae) in Ixodes ricinus (Acari: Ixodidae) in southern Italy. Ticks Tick Borne Dis. 2015, 6, 234–236.
- Santos, M.A.B.; Macedo, L.O.; Souza, I.B.; Ramos, C.A.N.; Alves, L.C.; Ramos, R.A.N.; Carvalho, G.A. Larvae of Ixodiphagus wasps (Hymenoptera: Encyrtidae) in Rhipicephalus sanguineus sensu lato ticks (Acari: Ixodidae) from Brazil. Ticks Tick Borne Dis. 2017, 8, 564–566.
- Sormunen, J.J.; Sippola, E.; Kaunisto, K.M.; Vesterinen, E.J.; Sääksjärvi, I.E. First evidence of Ixodiphagus hookeri (Hymenoptera: Encyrtidae) parasitization in Finnish castor bean ticks (Ixodes ricinus). Exp. Appl. Acarol. 2019, 79, 395–404.
- Tóth, A.G.; Farkas, R.; Gyurkovszky, M.; Krikó, E.; Solymosi, N. First detection of Ixodiphagus hookeri (Hymenoptera: Encyrtidae) in Ixodes ricinus ticks (Acari: Ixodidae) from multiple locations of Hungary. Sci. Rep. 2023, 13, 1624.
- Hu, R.; Hyland, K.E.; Oliver, J.H. A review on the use of Ixodiphagus wasps (Hymenoptera: Encyrtidae) as natural enemies for the control of ticks (Acari: Ixodidae). Syst. Appl. Acarol. 1988, 3, 19–28.
- Krawczyk, A.I.; Bakker, J.W.; Koenraadt, C.J.M.; Fonville, M.; Takumi, K.; Sprong, H.; Demir, S. Tripartite Interactions among Ixodiphagus hookeri, Ixodes ricinus and deer: Differential interference with transmission cycles of tick-borne pathogens. Pathogens 2020, 9, 339.
- Wood, H.P. Notes on the life history of the tick parasite Hunterellus hookeri Howard. J. Econ. Entomol. 1911, 4, 425–431.
- Collatz, J.; Selzer, P.; Fuhrmann, A.; Oehme, R.M.; Mackenstedt, U.; Kahl, O.; Steidle, J.L.M. A hidden beneficial: Biology of the tick-wasp Ixodiphagus hookeri in Germany. J. Appl. Entomol. 2011, 135, 351–358.
- Cooley, R.A. A parasite of ticks. Mont. State Bd. Ent. Bien. Rept. 1927, 6, 15–17.
- Smith, C.N.; Cole, M.M. Studies of parasites of the American dog tick. J. Econ. Entomol. 1943, 36, 569–572.
- Knipling, E.F.; Steelman, C.D. Feasibility of controlling Ixodes scapularis ticks (Acari: Ixodidae), the vector of Lyme disease, by parasitoid augmentation. J. Med. Entomol. 2000, 37, 645–652.
- Bowman, J.L.; Logan, T.M.; Hair, J.A. Host suitability of Ixodiphagus texanus Howard on five species of hard ticks. J. Agric. Entomol. 1986, 3, 1–9.
- Hu, R.; Hyland, K.E. Prevalence and seasonal activity of the wasp parasitoid, Ixodiphagus hookeri (Hymenoptera: Encyrtidae) in its tick host, Ixodes scapularis (Acari: Ixodidae). Syst. Appl. Acarol. 1997, 2, 95–100.
- Slovák, M. Finding of the endoparasitoid Ixodiphagus hookeri (Hymenoptera, Encyrtidae) in Haemaphysalis concinna ticks in Slovakia. Biologia 2003, 58, 890.
- Demas, F.A.; Hassanali, A.; Mwangi, E.N.; Kunjeku, E.C.; Mabveni, A.R. Cattle and Amblyomma variegatum odors used in host habitat and host finding by the tick parasitoid, Ixodiphagus hookeri. J. Chem. Ecol. 2000, 26, 1079–1093.
- Korenberg, E.I. Seasonal population dynamics of Ixodes ticks and tick-borne encephalitis virus. Exp. Appl. Acarol. 2000, 24, 665–681.
- Stafford III, K.C.; Denicola, A.J.; Kilpatrick, H.J. Reduced abundance of Ixodes scapularis (Acari: Ixodidae) and the tick parasitoid Ixodiphagus hookeri (Hymenoptera: Encyrtidae) with reduction of white-tailed deer. J. Med. Entomol. 2003, 40, 642–652.
- Dantas-Torres, F.; Otranto, D. Seasonal dynamics of Ixodes ricinus on ground level and higher vegetation in a preserved wooded area in southern Europe. Vet. Parasitol. 2013, 192, 253–258.
- Alfeev, N.I.; Klimas, Y.V. On the possibility of developing ichneumon flies, Hunterellus hookeri in climatic conditions of the USSR. Soviet. Vet. 1938, 15, 55.
- Tijsse-Klasen, E.; Braks, M.; Scholte, E.J.; Sprong, H. Parasites of vectors Ixodiphagus hookeri and its Wolbachia symbionts in ticks in the Netherlands. Parasit. Vectors. 2011, 4, 228.
- Plantard, O.; Bouju-Albert, A.; Malard, M.A.; Hermouet, A.; Capron, G.; Verheyden, H. Detection of Wolbachia in the tick Ixodes ricinus is due to the presence of the hymenoptera endoparasitoid Ixodiphagus hookeri. PLoS ONE 2012, 7, e30692.
- Zchori-Fein, E.; Gottlieb, Y.; Kelly, S.E.; Brown, J.K.; Wilson, J.M.; Karr, T.L.; Hunter, M.S. A newly discovered bacterium associated with parthenogenesis and a change in host selection behavior in parasitoid wasps. Proc. Natl. Acad. Sci. USA 2001, 98, 12555–12560.
- Lejal, E.; Chiquet, J.; Aubert, J.; Robin, S.; Estrada-Peña, A.; Rue, O.; Midoux, C.; Mariadassou, M.; Bailly, X.; Cougoul, A.; et al. Temporal patterns in Ixodes ricinus microbial communities: An insight into tick-borne microbe interactions. Microbiome 2021, 9, 153.
- Lyon, S.M.; Van Driesche, R.; Edman, J.D. Ecology of Hunterellus hookeri (Hymenoptera: Encyrtidae) and evaluation of its impact on Ixodes scapularis (Acari: Ixodidae) on Nanamasset Island in Massachusetts. Environ. Entomol. 1998, 27, 463–468.
- Takasu, K.; Nakamura, S. Life history of the tick parasitoid Ixodiphagus hookeri (Hymenoptera: Encyrtidae) in Kenya. Biol. Control. 2008, 46, 114–121.
- Hayat, M.; Veenakumari, K. Description of four new species of brachypterous Encyrtidae (Hymenoptera: Chalcidoidea) from India. Zootaxa 2015, 3990, 259–271.
- Gaye, M.; Amanzougaghene, N.; Laidoudi, Y.; Niang, E.H.A.; Sekeyová, Z.; Laroche, M.; Bérenger, J.M.; Raoult, D.; Kazimírová, M.; Fenollar, F.; et al. Hymenopteran parasitoids of hard ticks in western Africa and the Russian Far East. Microorganisms 2020, 8, 1992.
- Mani, M.S. Studies on Indian parasitic Hymenoptera. Indian J. Entomol. 1941, 3, 29.
- Howard, L.O. Another chalcidoid parasite of a tick. Can. Entomol. 1908, 40, 239–241.
- Buysson, R.D. Un Hymenoptere parasite des Ixodes. Arch. Parasitol. 1912, 15, 246–247.
- Brumpt, M.E. Utilisation des insectes auxiliaires—Entomophages dans la lutte contre les insectes pathogenes. Presse Med. 1913, 36, 359–361.
- Costa, L. The Chalcid, Hunterellus hookeri, Howard, a parasite of the tick Rhipicephalus sanguineus, Latreille, observed in Rio de Janeiro. Rev. Vet. Zoot. 1915, 5, 201–203.
- Timberlake, P.H. Hunterrelus hookeri Howard in India. (Note). Haw. Ent. Soc. Proc. 1922, 5, 10.
- Wolcott, G.N. Insectae Portoricensis. J. Dept. Agr. Porto Rico 1923, 7, 60.
- Cooley, R.A. The Montana tick parasite expedition to Africa. Science 1930, 71, 419–420.
- Philip, C.B. Occurrence of a colony of the tick parasite Hunterellus hookeri in West Africa. Public Health Rep. 1931, 46, 2168–2172.
- Cooley, R.A. A search for tick parasites in South Africa. Onderstepoort J. Vet. Sci. Anim. Ind. 1934, 3, 23–42.
- Bishopp, F.C. Record of hymenopterous parasites of ticks in the United States. Proc. Entomol. Soc. Wash. 1934, 36, 87–88.
- Larson, C.A. The tick parasite Ixodiphagus texanus in nymphs and larvae of Haemaphysalis leporispalustris in Minnesota. J. Parasitol. 1937, 23, 496–498.
- Tate, H.D. The biology of the tropical cattle tick and other species of ticks in Puerto Rico, with notes on the effects on ticks of arsenical dips. J. Agric. Sci. 1941, 25, 1–24.
- Pervomaisky, G.S. On the infestation of Ixodes persulcatus by Hunterellus hookeri How. (Hymenoptera) Zool. Zhurnal. 1943, 22, 211–213.
- Boucek, Z.; Verny, V. A parasite of ticks, the chalcid Hunterellus hookeri in Czechoslovakia. Zool. List. 1954, 3, 109–111.
- Philip, C.B. A new locality record in Africa for the tick parasite, Hunterellus hookeri Howard. J. Parasitol. 1954, 40, 234–235.
- Steyn, J.J. A second locality record in Uganda for the tick parasite, Hunterellus hookeri Howard, and a few discussions on other tick parasites. East Afr. Med. J. 1955, 32, 357–360.
- Doube, B.M.; Heath, A.C.G. Observations on the biology and seasonal abundance of an encyrtid wasp , a parasite of ticks in Queensland . J. Med. Entomol. 1975, 12, 443–447.
- Munaf, H.B. The first record of Hunterellus hookeri parasitizing Rhipicephalus sanguineus in Indonesia. Southeast Asian J. Trop. Med. Public Health 1976, 7, 492.
- Cheong, W.H.; Rajamanikam, C.; Mahadevan, S. A case of Hunterellus hookeri parasitization of ticks in Pentaling Jaya, Peninsula Malaysia. Southeast Asian J. Trop. Med. Public Health 1978, 9, 456–458.
- Davis, A.J.; Campbell, V. Ixodiphagus texanus Howard (Hymenoptera: Encyrtidae) a parasite of the rabbit tick in Nova Scotia. Can. J. Zool. 1979, 57, 1164–1166.
- Mather, T.N.; Piesman, J.; Spielman, A. Absence of spirocates (Borrelia burgdorferi) and piroplasms (Babesia microti) in deer ticks Ixodes dammini parasitized by chalcid wasps (Hunterellus hookeri). Med. Vet. Entomol. 1987, 1, 3–8.
- Estradapena, A.; Sanchezacedo, C. Presence in Spain of Hunterellus hookeri Howard, 1907, a parasite of Haemaphysalis punctata (Acarina: Ixodidae). Rev. Iber. Parasitol. 1989, 49, 183–184.
- Mwangi, E.N.; Newson, R.M.; Kaaya, G.R. A hymenopteran parasitoid of the Bont tick Amblyomma variegatum Fabricius (Acarina: Ixodidae) in Kenya. Discov. Innov. 1993, 5, 331–335.
- Doby, J.M.; Laere, G. Hunterellus hookeri Howard, 1907 (Hymenoptera: Chalcididae), a parasite of the tick Ixodes ricinus in western and central France. Parasite 1993, 2, 265–270.
- Hu, R.; Hyland, K.E.; Mather, T.N. Occurrence and distribution in Rhode Island of Hunterellus hookeri (Hymenoptera: Encyrtidae), a wasp parasitoid of Ixodes dammini. J. Med. Entomol. 1993, 30, 277–280.
- Soto, M.M. Hunterellus hookeri, a parasitoid of Rhipicephalus sanguineus, a tick of public health interest in Mexico. Rev. Soc. Mex. Hist. Nat. 1995, 45, 157–161.
- Stafford III, K.C.; Denicola, A.J.; Magnarelli, L.A. Presence of Ixodiphagus hookeri (Hymenoptera: Encyrtidae) in two Connecticut populations of Ixodes scapularis (Acari: Ixodidae). J. Med. Entomol. 1996, 33, 183–188.
- Mwangi, E.N.; Hassan, S.M.; Kaaya, G.P.; Essuman, S. The impact of Ixodiphagus hookeri, a tick parasitoid, on Amblyomma variegatum (Acari: Ixodidae) in a field trial in Kenya. Exp. Appl. Acarol. 1997, 21, 117–126.
- Coronado, A. Ixodiphagus hookeri Howard, 1907 (Hymenoptera: Encyrtidae) in the brown dog tick Rhipicephalus sanguineus Latreille, 1806 (Acari: Ixodidae) in Venezuela. Entomotropica 2006, 21, 61–64.
- Heath, A.C.G.; Cane, R.P. A new species of Ixodiphagus (Hymenoptera: Chalcidoidea: Encyrtidae) parasitizing seabird ticks in New Zealand. N. Z. J. Zool. 2010, 37, 147–155.
- Lopes, A.J.; Nascimento, J.R., Jr.; Silva, C.G.; Prado, A.P.; Labruna, M.B.; Costa, L.M., Jr. Parasitism by Ixodiphagus wasps (Hymenoptera: Encyrtidae) in Rhipicephalus sanguineus and Amblyomma ticks (Acari: Ixodidae) in three regions of Brazil. J. Econ. Entomol. 2012, 105, 1979–1981.
- Miranda, R.; Kadoch, N.; Bermúdez, S.E. Notas preliminares sobre enemigos naturales de Rhipicephalus sanguineus sl (Latreille 1806) (Ixodida: Ixodidae) en condiciones urbanas y rurales de Panamá. Rev. Ibér. Aracnol. 2013, 23, 141–143.
- Bohacsova, M.; Oleg, M.; Kazimirova, M.; Raoult, D.; Sekeyova, Z. Arsenophonus nasoniae and Rickettsiae infection of Ixodes ricinus due to parasitic wasp Ixodiphagus hookeri. PLoS ONE 2016, 11, e0149950.
- Vasquez, A.; Goolsby, J.A.; Vacek, A.T.; Racelis, A.; Kariyat, R.R. Incidence of the Brown dog tick, Rhipicephalus sanguineus and its parasitoid, Ixodiphagus hookeri on dogs in South Texas. Subtrop. Agricult. Environ. 2019, 70, 6–10.
- Luu, L.; Palomar, A.M.; Farrington, G.; Schilling, A.K.; Premchand-Branker, S.; McGarry, J.; Makepeace, B.L.; Meredith, A.; Bell-Sakyi, L. Bacterial pathogens and symbionts harboured by Ixodes ricinus ticks parasitising red squirrels in the United Kingdom. Pathogens 2021, 10, 458.
- Vecchi, M.O.M.; Ramos, R.A.N.; Bitencourth, K.; Alves, M.A.S. First record of the parasitoid wasp Ixodiphagus hookeri (Hymenoptera: Encyrtidae) infesting the tick Amblyomma nodosum (Acari: Ixodidae). Parasitol. Res. 2023, 122, 1255–1259.
More
