Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Lindsay Dong and Version 1 by CHUNDO OH.
Temporomandibular joint disorders (TMDs) are conditions that affect the muscles of mastication and joints that connect the mandible to the base of the skull. Although TMJ disorders are associated with symptoms, the causes are not well proven. Chemokines play an important role in the pathogenesis of temporomandibular joint (TMJ) disease by promoting chemotaxis inflammatory cells to destroy the joint synovium, cartilage, subchondral bone, and other structures.
- chemokine
- temporomandibular joint disease
- inflammation
- β-catenin
1. Introduction
Temporomandibular joint disorders (TMDs) are conditions related to the masticatory muscles and the temporomandibular joint itself that result in pain and mandibular movement dysfunction [1]. TMDs affect patients’ quality of life by causing chronic muscle pain and limited mouth opening [2]. The prevalence of TMDs, as the second most common musculoskeletal pain, was approximately 31% in adults and 11% in children [3,4][3][4]. According to the diagnostic criteria of the Research Diagnostic Criteria for Temporomandibular Disorders (RDC/TMD) and the Diagnostic Criteria for Temporomandibular Disorders (DC/TMD), TMD can be categorized into the three groups: arthrogenous TMD (including disc disfunction and joint pathology), myogenous TMD (masticatory muscle disorders), and headache attributed to TMD [5,6,7][5][6][7]. Temporomandibular joint disorders (TMDs), including disc displacement (DD), and/or osteoarthritis (OA), often lead to joint pain and restrictions in mandibular movement [8,9][8][9]. Joint noise or crepitance during jaw movement, as well as less specific signs including ear pain and stuffiness, tinnitus, dizziness, neck pain and headache can also occur [10]. Moreover, oral disease is one of the most common public health issues worldwide, with significant socio-economic impacts [11]. Temporomandibular disorders (TMD) are prevalent and debilitating, and are reported to affect 5–12% of Americans [2], with affliction odds higher for females than males [12], and estimated annual costs of $4 billion [13]. Osseous changes are late-stage characteristics of degenerative joint disease [14,15,16][14][15][16] that are found in the TMJs of 20–30 year olds. This is over two decades earlier than in other human joints [17], and is evidence of the distinct susceptibility of the temporomandibular joint (TMJ). Why TMJ tissues prematurely fail compared to post-cranial joints is not understood. TMJ internal disc derangement (ID) and/or OA, are accompanied by synovitis characterized by chronic inflammatory changes like synovial lining hyperplasia. In these cases, the number of new capillaries and small vessels increases, followed by inflammatory cells infiltrating around these vessels [18,19][18][19]. With a frequency of 25%, TMJ OA is one of the most common end-stage TMD pathologies, and the molecular mechanisms associated with TMD remain undiscovered [20]. Chemokines are a family of small cytokines, which play a key role in the exudation of inflammatory cells from the vascular system to tissues at the inflammatory site [21]. More specifically, chemokines can stimulate the chemotaxis of neutrophils, macrophages, and T lymphocytes. In addition, inflammatory cells produce inflammatory cytokines such as IL-1β, which plays a role in OA pathology by promoting the expression of matrix-degrading enzymes and oxidative metabolites, leading to extracellular matrix degradation. Furthermore, inflammatory cytokines can stimulate the synovium and other tissues to produce more chemokines [22,23][22][23].
2. Inflammation and TMJ
TMJ OA is described as low-inflammatory arthritis, while rheumatoid arthritis (RA) is described as a high-inflammatory disease. Inflammation during TMJ OA plays a key role in the onset and progression of the disease and the pain intensity [18]. Although some inflammatory mediators are common in the local TMJ OA and systemic inflammatory diseases, local levels and compositions of these mediators may change. In fact, different cytokines, chemokines, chemokine receptors, enzymes, and bone-stimulating resorption factors that are not reported in RA are considered markers of active TMJ OA [24,25][24][25]. Increased hyperemia of capillaries and infiltration of inflammatory cells, such as T cells, monocytes, or macrophages, have been observed in the TMJ synovial lining of patients with TMJ ID and OA. Several cytokines and chemokines were detected in the synovial fluid of patients with ID or OA, which include IL-1β and TNFα [26]. These cytokines have been shown to have a role in the expression of eicosanoid acid, chemokines, and proteins in TMJ synovial cells [27].3. Chemokine and TMJ
Chemokines are pro-inflammatory peptides (8–14 kDa) that are also known as chemotactic cytokines or chemical hormones [28]. Their main biological function is to recruit leukocytes to local inflammatory sites [29]. All chemokines share major structural similarities, which include a conserved 4-cysteine motif. Some chemokines are proinflammatory cytokines, while other chemokines are thought to maintain self-regulation and control cell migration during normal tissue maintenance and development [30]. According to the arrangement of the conserved cysteine residues of mature proteins, chemokines are divided into four subfamilies: CXC(α), CC(β), C(γ) and CX3C(δ). The corresponding receptors are CXCR, CCR, CR and CX3CR [31]. Among them, α and β family members have the most extensive functions. The CXC subfamily, which has one amino acid(aa) residue separating the first two cysteine residues, includes interleukin-8 (CXCL8), growth associated oncogene GROα (CXCL1), stromal cell-derived factor 1 (SDF-1), and platelet factor 4 (PF-4 or CXCL4). Both IL-8 and GROα can send chemotactic neutrophils to inflammatory regions, while SDF-1 and PF-4 recruit lymphocytes and monocytes. The CC subfamily includes monocyte chemoattractant protein MCP-1 (CCL2), macrophage inflammatory protein MIP-1α (CCL3), MIP-1β (CCL4), exotaxin (CCL11), and RANTES (CCL5). A chemokine receptor is a transmembrane G-protein-coupled receptor that is selectively expressed on the surface of target cells. In TMJ, the inflammatory cells in synovial tissues of RA, OA and ID are increased. Additionally, some chemokines such as IL-8, GROα, RANTES, and MIP-1 have been detected in human chondrocytes and synovial cells at significantly increased levels. The expression of CXCL8 and MCP-1 have also been reported to be increased in the synovial fluid of patients with TMJ ID and/or OA [33][32]. Such chemokines can promote the infiltration of inflammatory cells in TMJ joints, which causes the release of degradation enzymes, various oxidative metabolites, and inflammatory cytokines. These can lead to joint structural damage and arthritis. Furthermore, the induction of ELR CXC (CXCL1, 2, 3, 6 and 8) may lead to the recruitment of new, small vessels in inflammatory cells and synovial tissues [30,31][30][31].3.1. CXC (α) and CX3C (δ) Subfamilies in TMJ Disease
3.1.1. IL-8
IL-8 is a member of CXC chemokines that activate leukocytes [34[33][34],35], which were formerly known as neutrophil activator protein-1 or monocyte derived neutrophil chemokine. It is a chemokine capable of inducing chemotaxis and activating neutrophils, including through direct and trans-endothelial migration, release of storage enzymes, induction of oxygen metabolites, and expression of adhesion molecules [34][33]. IL-8 can also attract T lymphocytes in vitro [36][35]. Furthermore, IL-8 is associated with many disease states, predominately angiogenic diseases such as RA. In RA, IL-8 has been shown to cause neutrophil infiltration into synovial fluid and joint inflammation. IL-8 also plays a key role in the pathogenesis of TMJ. Koch et al. reported that, compared with OA patients, the level of IL-8 in synovial fluid of RA patients increased significantly [37][36]. IL-8 is another type of IL-1β-targeting cytokine, one of the strongest chemokines of neutrophils and T lymphocytes. It can promote monocyte homing and activation in the synovium [36][35]. IL-8 can cause various pathogenic conditions, such as the release of oxidation products, apoptosis of chondrocytes, the production of MMP-13 by articular chondrocytes, and the loss of proteoglycan and subsequent cartilage degradation [38][37].3.1.2. SDF-1/CXCR4
Chemokine stromal cell-derived factor-1 (SDF-1) is a small cytokine of the CXC chemokine ligand superfamily. It is mainly expressed by bone marrow stromal cells, which include osteoblasts and endothelial cells [40][38]. C-X-C chemokine receptor-4 (CXCR4) is a specific receptor of SDF-1. The expression of SDF-1 in synovitis or OA patients increases abnormally. In synovium and articular cartilage tissues, the activation of the SDF-1 and CXCR4 signaling pathways can regulate the expression of various inflammatory factors, which include IL-1, IL-6, TNF-α, and MMPs. IL-1 is involved in joint pathology. The SDF-1 and CXCR4 signaling pathway plays a pro-inflammatory role in the experimental TMJ OA. There may be a potential relationship between the SDF-1-CXCR4 axis and extracellular signal regulated kinase (ERK) signaling pathway. In addition, there is evidence that SDF-1α activates ERK and downstream transcription factors (c-fos and c-jun) through CXCR4, which activates adaptor protein-1 on MMP-13 and leads to cartilage damage in knee arthritis. The expression of SDF-1 was high in the synovium [41,42][39][40].3.1.3. FKN (CXCL1)
Fractalkine (CX3CL1) is a chemokine of the CX3C family, and is both inflammatory and nociceptive. CX3CL1 has unique connectivity with CX3CR1, which is a single receptor in microglia. CX3CR1 has been associated with oral and facial inflammatory pain [46][41]. There is much data indicating that the activation of microglia is an effective method to treat oral and facial pain, and that glial cells play an important regulatory role in orofacial pain signaling pathway [47][42]. The pathogenesis of RA is a complex process. Proinflammatory cytokines such as Interleukin-1β (IL-1β) and tumor necrosis factor-A (TNF-α) are the central mediators of RA [48][43]. TNF-α, IL-1β, IL-6 are common cytokines. Chemokine-induced neutrophil, chemokine-1, and keratinocyte derived chemokines (KC) can trigger the release of prostaglandins and sympathetic amines, which directly act on nociceptors and cause excessive nociception [49][44]. The neurochemokine FKN has a pain-promoting effect in the spinal cord [50,51][45][46].3.2. CC(β) Subfamily in TMJ Disease
3.2.1. MCP-1 Chemokines
MCP-1 is a chemokine that is part of the CC subfamily. CC chemokines mainly act on monocytes and lymphocytes [55][47]. In IL-1β-responsive genes, monocyte chemoattractant protein (MCP)-1 mRNA was induced by IL-1β. It was observed to be highly expressed in stimulated synovial cells. The production of MCP-1 in chondrocytes and synovial cells has been shown to play an important role in joint diseases such as RA and OA through monocyte recruitment [56,57][48][49]. It has been suggested that MCP-1 is one of the markers of RA disease activity. The protein production of MCP-1 (also known as CCL2) in TMJ has been studied in vitro and in vivo.3.2.2. MIP3α-CCR6
Macrophage inflammatory protein-3α (MIP-3α), also known as CCL20, is a highly upregulated gene for IL-1β and TNF-α [60][50]. Chemokine receptor 6, also known as CCR6, is a CC chemokine receptor protein encoded in humans by the CCR6 gene. MIP-3α and CCR6 may play a role in the recruitment of monocytes and memory lymphocytes from RA peripheral blood to RA joints, which suggests that expression of the MIP-3α receptor, CCR6, may be associated with RA development [61][51]. RA synovial tissue contains many leukocytes expressing CCR6 [62[52][53],63], and both MIP-3α and CCR6 have been detected in the synovial fluid and synovium of RA patients [64][54]. The nucleotide sequence of the human MIP-3α promoter region has binding sites for Ets, AP-1, SP-1, and NFκB. This suggests that the expression of MIP-3α was regulated by several signaling molecules [65][55]. IL-1β, or TNF-α, induces MIP-3α production in human synovial fibroblast-like cells (SFCs) through ERK, p38 MAPK, JNK, and NFκB pathways. Increased levels of MIP-3α may trigger dendritic cells, T cells, and B cells to migrate into the synovial tissue and fluids of TMJ ID patients.3.2.3. RANTES-CCR1
RANTES is an 8 kDa basic polypeptide in the CC chemokine superfamily. It was originally cloned from antigen-stimulated T cell line, which is also known as CCL5. RANTES can be released by chondrocytes, synovial fibroblasts, and inflammatory cells. RANTES is an effective chemical attractant for monocytes, CD4+/CD45RO+ memory helper T lymphocytes, eosinophils, basophils, and mast cells [70,71][56][57]. It has been shown that RANTES is overexpressed in normal adult tissues and increases significantly in inflammatory sites and some tumors [72][58]. RANTES can promote the migration of monocytes, T lymphocytes, natural killer cells, eosinophils, and macrophages. RANTES can also promote the formation of osteoclasts induced by RANKL. RANTES is also highly expressed in tissues, synovial fluid, and peripheral blood of patients with RA or OA. RANTES can trigger and aggravate the inflammatory immune response by promoting the infiltration of immunocompetent cells, and activating synovial fibroblasts to produce inflammatory mediators. MMP-1, MMP-3, MMP-13, and iNOS released by synovial fibroblasts and chondrocytes promote cartilage degradation [73][59]. Migration tests confirmed that RANTES was an effective chemokine of RAW264, and was responsible for attracting macrophages to inflammatory sites. C-C chemokine receptor type 1 (CCR1) is the receptor of RANTES, and acts as an inhibitor that impairs the migration of GFP BMSCs into OA cartilage and the rescue effect of GFP BMSC injection. RANTES and CCR1 signaling plays a key and synergistic role in the recruitment of GFP BMSCs into TMJ mouse OA-degraded cartilage [74][60].3.3. Other Subfamilies in TMJ Disease
Chemerin-ChemR23
Chemerins are atypical chemokine ligand agonists that bind to many chemokine ligands. Chemerin acts as a chemoattractant for cells of the myeloid lineage, can signal through a potential adipokine of ChemR23, and can also bind CCRL2 in the absence of signaling [76][61]. Chemerin and its receptors are ubiquitous in the human body. They play a multi-functional role as chemokines, adipokines, and possible growth factors [77][62]. It was first isolated from human inflammatory fluid [78][63], which is involved in angiogenesis, osteoblast formation, and glucose homeostasis [79][64]. It is also a protein of interest in various medical fields, such as immunology, dermatology, metabolism, and development. Chemerin, a ligand protein of ChemR23, is a G-protein-coupled receptor that is expressed in macrophages (dentate cells), dendritic cells, and natural killer cells (NK) [80][65]. The interaction between Chemerin and ChemR23 is believed to be closely related to the migration of macrophages and Dendritic Cells (DC) to inflammatory sites, as well as mediating inflammatory signals to articular chondrocytes and endothelial cells (e.g., IL-1β, IL-6, IL-8 and TNF-α). It is characterized by increased secretion of MMP-1, MMP-2, MMP-3, MMP-8, and MMP-13. Wittamer et al. first reported the association between Chemerin and inflammation. They observed a high concentration of synovial fluid (SF) in patients with arthritis [81][66].3.4. TNFα Induced Chemotaxis and Chemokine Regulation in Synovial Fibroblast
TNF-α is one of the cytokines detected in the synovial fluid of patients with intercapsular lesions with disc displacement (DD) or OA [86][67]. TNF-α levels are also elevated in the synovium [87][68]. A study evaluating the role of temporomandibular joint synovial fibroblasts in chemokine release found significant increases in the expression of IL-8, GRO-α, MCP-1, and RANTES in the conditioned medium of TNF-α-treated synovial fibroblasts. The production of IL-8 and GRO-α increased soon after exposure to TNF-α (4 and 8 hrs), suggesting that neutrophil infiltration occurs in the early stages of the inflammatory response. The production of RANTES, a potent chemoattractant for CD4+/CD45RO+ memory helper T lymphocytes, increased less than other chemokines after 4 h of exposure to TNF-α. This suggests that T lymphocytes do not migrate to sites of inflammation as early as neutrophils and monocytes. These inflammatory cells produce inflammatory cytokines, such as TNF-α, matrix-degrading enzymes, and various oxidative metabolites. These enzymes and oxidative metabolism lead to extracellular matrix degradation. At the same time, inflammatory cytokines stimulate synovial fibroblasts to produce more chemokines (Figure 1).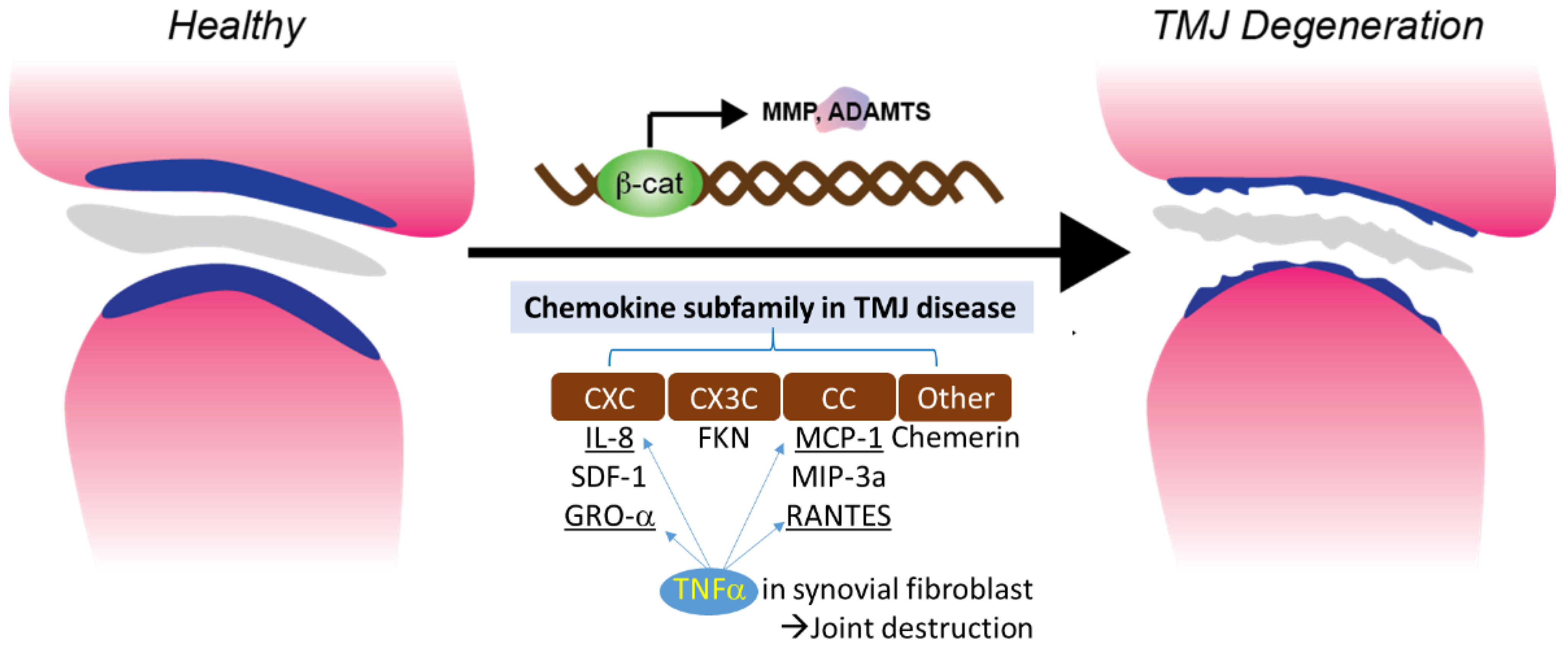
Figure 1. Chemokine subfamily associated with β-catenin and TNFα signaling in synovial fibroblast.
3.5. Chemokines and RA
Inflammatory cells in TMJ OA are thought to play a pathological role in the development and persistence of inflammation by their ability to release degrading enzymes and various oxidative metabolites. Infiltrating cells recruited from the blood in synovitis are mediated by chemokines released by activated synovial cells. IL-8 is a member of a chemokine superfamily of low molecular weight cytokines. These have a chemotactic effect on neutrophil and T cell subsets as well as activating neutrophil function, which includes calcium mobilization, threshing, and respiratory outbreaks [36,88]. The infiltration of neutrophils and T cells into synovial tissue are thought to play a pathological role in the development and persistence of RA through their ability to release degrading enzymes and various oxidative metabolites [88,89].
Chemokine subfamily associated with β-catenin and TNFα signaling in synovial fibroblast.
3.5. Chemokines and RA
Inflammatory cells in TMJ OA are thought to play a pathological role in the development and persistence of inflammation by their ability to release degrading enzymes and various oxidative metabolites. Infiltrating cells recruited from the blood in synovitis are mediated by chemokines released by activated synovial cells. IL-8 is a member of a chemokine superfamily of low molecular weight cytokines. These have a chemotactic effect on neutrophil and T cell subsets as well as activating neutrophil function, which includes calcium mobilization, threshing, and respiratory outbreaks [35][69]. The infiltration of neutrophils and T cells into synovial tissue are thought to play a pathological role in the development and persistence of RA through their ability to release degrading enzymes and various oxidative metabolites [69][70].
References
- Gil-Martinez, A.; Paris-Alemany, A.; López-de-Uralde-Villanueva, I.; La Touche, R. Management of pain in patients with temporomandibular disorder (TMD): Challenges and solutions. J. Pain Res. 2018, 11, 571–587.
- Ahmad, M.; Schiffman, E.L. Temporomandibular Joint Disorders and Orofacial Pain. Dent. Clin. 2016, 60, 105–124.
- Li, D.T.S.; Leung, Y.Y. Temporomandibular Disorders: Current Concepts and Controversies in Diagnosis and Management. Diagnostics 2021, 11, 459.
- Valesan, L.F.; Da-Cas, C.D.; Réus, J.C.; Denardin, A.C.S.; Garanhani, R.R.; Bonotto, D.; Januzzi, E.; de Souza, B.D.M. Prevalence of temporomandibular joint disorders: A systematic review and meta-analysis. Clin. Oral Investig. 2021, 25, 441–453.
- Schiffman, E.; Ohrbach, R.; Truelove, E.; Truelove, E.; Look, J.; Anderson, G.; Ceusters, W.; Smith, B. Diagnostic Criteria for Temporomandibular Disorders (DC/TMD) for Clinical and Research Applications: Recommendations of the International RDC/TMD Consortium Network and Orofacial Pain Special Interest Groupdagger. J. Oral Facial Pain Headache 2014, 28, 6–27.
- John, M.T.; Dworkin, S.F.; Mancl, L.A. Reliability of clinical temporomandibular disorder diagnoses. Pain 2005, 118, 61–69.
- Yakkaphan, P.; Smith, J.G.; Chana, P.; Renton, T.; Lambru, G. Temporomandibular disorder and headache prevalence: A systematic review and meta-analysis. Cephalalgia Rep. 2022, 5, 25158163221097352.
- Cascone, P.; Gennaro, P.; Gabriele, G.; Chisci, G.; Mitro, V.; De Caris, F.; Iannetti, G. Temporomandibular synovial chondromatosis with numerous nodules. J. Craniofac. Surg. 2014, 25, 1114–1115.
- Cascone, P.; Di Paolo, C.; Leonardi, R.; Pedullà, E. Temporomandibular disorders and orthognathic surgery. J. Craniofac. Surg. 2008, 19, 687–692.
- Yin, Y.; He, S.; Xu, J.; You, W.; Li, Q.; Long, J.; Luo, L.; Kemp, G.J.; Sweeney, J.A.; Li, F.; et al. The neuro-pathophysiology of temporomandibular disorders-related pain: A systematic review of structural and functional MRI studies. J. Headache Pain 2020, 21, 78.
- Jin, L.J.; Lamster, I.B.; Greenspan, J.S.; Pitts, N.B.; Scully, C.; Warnakulasuriya, S. Global burden of oral diseases: Emerging concepts, management and interplay with systemic health. Oral Dis. 2016, 22, 609–619.
- Slade, G.; Ohrbach, R.; Greenspan, J.D.; Fillingim, R.B.; Bair, E.; Sanders, A.E.; Dubner, R.; Diatchenko, L.; Meloto, C.B.; Smith, S.; et al. Painful temporomandibular disorder: Decade of discovery from OPPERA studies. J. Dent. Res. 2016, 95, 1084–1092.
- Stowell, A.W.; Gatchel, R.J.; Wildenstein, L. Cost-effectiveness of treatments for temporomandibular disorders: Biopsychosocial intervention versus treatment as usual. J. Am. Dent. Assoc. 2007, 138, 202–208.
- Farina, D.; Bodin, C.; Gandolfi, S.; De Gasperi, W.; Borghesi, A.; Maroldi, R. TMJ disorders and pain: Assessment by contrast-enhanced MRI. Eur. J. Radiol. 2009, 70, 25–30.
- Wiese, M.; Wenzel, A.; Hintze, H.; Petersson, A.; Knutsson, K.; Bakke, M.; List, T.; Svensson, P. Osseous changes and condyle position in TMJ tomograms: Impact of RDC/TMD clinical diagnoses on agreement between expected and actual findings. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2008, 106, e52–e63.
- Miron, D.; Raz, R.; Luder, A. Congenital toxoplasmosis in Israel: To screen or not to screen. Isr. Med. Assoc. J. IMAJ 2002, 4, 119–122.
- Iwasaki, L.R.; Gonzalez, Y.M.; Liu, Y.; Liu, H.; Markova, M.; Gallo, L.M.; Nickel, J.C. TMJ energy densities in healthy men and women. Osteoarthr. Cartil. 2017, 25, 846–849.
- Wang, X.D.; Zhang, J.N.; Gan, Y.H.; Zhou, Y.H. Current understanding of pathogenesis and treatment of TMJ osteoarthritis. J. Dent. Res. 2015, 94, 666–673.
- Liu, Q.; Yang, H.; Zhang, M.; Zhang, J.; Lu, L.; Yu, S.; Wu, Y.; Wang, M. Initiation and progression of dental-stimulated temporomandibular joints osteoarthritis. Osteoarthr. Cartil. 2021, 29, 633–642.
- Ferrillo, M.; Nucci, L.; Giudice, A.; Calafiore, D.; Marotta, N.; Minervini, G.; d’Apuzzo, F.; Ammendolia, A.; Perillo, L.; de Sire, A. Efficacy of conservative approaches on pain relief in patients with temporomandibular joint disorders: A systematic review with network meta-analysis. Cranio 2022, 1–17.
- Raman, D.; Sobolik-Delmaire, T.; Richmond, A. Chemokines in health and disease. Exp. Cell Res. 2011, 317, 575–589.
- Ogura, N.; Tobe, M.; Sakamaki, H.; Nagura, H.; Abiko, Y.; Kondoh, T. Tumor necrosis factor-α increases chemokine gene expression and production in synovial fibroblasts from human temporomandibular joint. J. Oral Pathol. Med. 2005, 34, 357–363.
- Tipton, D.A.; Christian, J.; Blumer, A. Effects of cranberry components on IL-1beta-stimulated production of IL-6, IL-8 and VEGF by human TMJ synovial fibroblasts. Arch. Oral Biol. 2016, 68, 88–96.
- Derwich, M.; Mitus-Kenig, M.; Pawlowska, E. Interdisciplinary Approach to the Temporomandibular Joint Osteoarthritis-Review of the Literature. Medicina 2020, 56, 225.
- Yang, T.; Zhang, J.; Cao, Y.; Zhang, M.; Jing, L.; Jiao, K.; Yu, S.; Chang, W.; Chen, D.; Wang, M. Wnt5a/Ror2 mediates temporomandibular joint subchondral bone remodeling. J. Dent. Res. 2015, 94, 803–812.
- Yuan, J.; Ding, W.; Wu, N.; Jiang, S.; Li, W. Protective Effect of Genistein on Condylar Cartilage through Downregulating NF-kappaB Expression in Experimentally Created Osteoarthritis Rats. BioMed Res. Int. 2019, 2019, 2629791.
- Zhang, S.; Teo, K.Y.W.; Chuah, S.J.; Lai, R.C.; Lim, S.K.; Toh, W.S. MSC exosomes alleviate temporomandibular joint osteoarthritis by attenuating inflammation and restoring matrix homeostasis. Biomaterials 2019, 200, 35–47.
- Hughes, C.E.; Nibbs, R.J.B. A guide to chemokines and their receptors. FEBS J. 2018, 285, 2944–2971.
- Gilchrist, A. Chemokines and Bone. In Bone Regulators and Osteoporosis Therapy; Handbook of Experimental Pharmacology; Stern, P.H., Ed.; Springer: Cham, Switzerland, 2020; Volume 262.
- Ridiandries, A.; Tan, J.T.M.; Bursill, C.A. The Role of Chemokines in Wound Healing. Int. J. Mol. Sci. 2018, 19, 3217.
- van der Vorst, E.P.; Doring, Y.; Weber, C. Chemokines. Arterioscler. Thromb. Vasc. Biol. 2015, 35, e52–e56.
- Legler, D.F.; Thelen, M. Chemokines: Chemistry, Biochemistry and Biological Function. Chimia 2016, 70, 856–859.
- Baggiolini, M.; Dewald, B.; Moser, B. Interleukin-8 and related chemotactic cytokines—CXC and CC chemokines. Adv. Immunol. 1994, 55, 97–179.
- Baggiolini, M.; Loetscher, P.; Moser, B. Interleukin-8 and the chemokine family. Int. J. Immunopharmacol. 1995, 17, 103–108.
- Larsen, C.G.; Anderson, A.O.; Appella, E.; Oppenheim, J.J.; Matsushima, K. The neutrophil-activating protein (NAP-1) is also chemotactic for T lymphocytes. Science 1989, 243, 1464–1466.
- Kim, H.; Lee, B.K. Anti-Inflammatory Effect of Adipose-Derived Stromal Vascular Fraction on Osteoarthritic Temporomandibular Joint Synoviocytes. Tissue Eng. Regen. Med. 2020, 17, 351–362.
- Borzi, R.M.; Mazzetti, I.; Marcu, K.B.; Facchini, A. Chemokines in cartilage degradation. Clin. Orthop. Relat. Res. 2004, 427, S53–S61.
- Villalvilla, A.; Gomez, R.; Roman-Blas, J.A.; Largo, R.; Herrero-Beaumont, G. SDF-1 signaling: A promising target in rheumatic diseases. Expert Opin. Ther. Targets 2014, 18, 1077–1087.
- Yang, J.; Li, Y.; Liu, Y.; Zhang, Q.; Zhang, Q.; Chen, J.; Yan, X.; Yuan, X. Role of the SDF-1/CXCR4 signaling pathway in cartilage and subchondral bone in temporomandibular joint osteoarthritis induced by overloaded functional orthopedics in rats. J. Orthop. Surg. Res. 2020, 15, 330.
- Lu, L.; Zhang, X.; Zhang, M.; Zhang, H.; Liao, L.; Yang, T.; Zhang, J.; Xian, L.; Chen, D.; Wang, M. RANTES and SDF-1 Are Keys in Cell-based Therapy of TMJ Osteoarthritis. J. Dent. Res. 2015, 94, 1601–1609.
- Nanki, T.; Imai, T.; Kawai, S. Fractalkine/CX3CL1 in rheumatoid arthritis. Mod. Rheumatol. 2017, 27, 392–397.
- Fan, W.; Zhu, X.; He, Y.; Zhu, M.; Wu, Z.; Huang, F.; He, H. The role of satellite glial cells in orofacial pain. J. Neurosci. Res. 2019, 97, 393–401.
- Brennan, F.M.; McInnes, I.B. Evidence that cytokines play a role in rheumatoid arthritis. J. Clin. Investig. 2008, 118, 3537–3545.
- Conte, F.D.P.; Barja-Fidalgo, C.; Verri, W.A., Jr.; Cunha, F.Q.; Rae, G.A.; Penido, C.; Henriques, M.D.G.M. Endothelins modulate inflammatory reaction in zymosan-induced arthritis: Participation of LTB4, TNF-α, and CXCL-1. J. Leukoc. Biol. 2008, 84, 652–660.
- Clark, A.K.; Yip, P.K.; Grist, J.; Gentry, C.; Staniland, A.A.; Marchand, F.; Dehvari, M.; Wotherspoon, G.; Winter, J.; Ullah, J.; et al. Inhibition of spinal microglial cathepsin S for the reversal of neuropathic pain. Proc. Natl. Acad. Sci. USA 2007, 104, 10655–10660.
- Clark, A.K.; Yip, P.K.; Malcangio, M. The liberation of fractalkine in the dorsal horn requires microglial cathepsin S. J. Neurosci. 2009, 29, 6945–6954.
- Ellingsen, T.; Hornung, N.; Møller, B.K.; Poulsen, J.H.; Stengaard-Pedersen, K. Differential effect of methotrexate on the increased CCR2 density on circulating CD4 T lymphocytes and monocytes in active chronic rheumatoid arthritis, with a down regulation only on monocytes in responders. Ann. Rheum. Dis. 2007, 66, 151–157.
- Yao, T.C.; Kuo, M.L.; See, L.C.; Ou, L.S.; Lee, W.I.; Chan, C.K.; Huang, J.L. RANTES and monocyte chemoattractant protein 1 as sensitive markers of disease activity in patients with juvenile rheumatoid arthritis: A six-year longitudinal study. Arthritis Rheum. Off. J. Am. Coll. Rheumatol. 2006, 54, 2585–2593.
- Pierer, M.; Rethage, J.; Seibl, R.; Lauener, R.; Brentano, F.; Wagner, U.; Hantzschel, H.; Michel, B.A.; Gay, R.E.; Gay, S.; et al. Chemokine secretion of rheumatoid arthritis synovial fibroblasts stimulated by Toll-like receptor 2 ligands. J. Immunol. 2004, 172, 1256–1265.
- Rossi, D.L.; Vicari, A.P.; Franz-Bacon, K.; McClanahan, T.K.; Zlotnik, A. Identification through bioinformatics of two new macrophage proinflammatory human chemokines: MIP-3alpha and MIP-3beta. J. Immunol. 1997, 158, 1033–1036.
- Schutyser, E.; Struyf, S.; Van Damme, J. The CC chemokine CCL20 and its receptor CCR6. Cytokine Growth Factor Rev. 2003, 14, 409–426.
- Cavanagh, L.L.; Boyce, A.; Smith, L.; Padmanabha, J.; Filgueira, L.; Pietschmann, P.; Thomas, R. Rheumatoid arthritis synovium contains plasmacytoid dendritic cells. Arthritis Res. Ther. 2005, 7, R230–R240.
- Tran, C.N.; Lundy, S.K.; Fox, D.A. Synovial biology and T cells in rheumatoid arthritis. Pathophysiology 2005, 12, 183–189.
- Matsui, T.; Akahoshi, T.; Namai, R.; Hashimoto, A.; Kurihara, Y.; Rana, M.; Nishimura, A.; Endo, H.; Kitasato, H.; Kawai, S.; et al. Selective recruitment of CCR6-expressing cells by increased production of MIP-3 α in rheumatoid arthritis. Clin. Exp. Immunol. 2001, 125, 155–161.
- Kwon, J.H.; Keates, S.; Simeonidis, S.; Grall, F.; Libermann, T.A.; Keates, A.C. ESE-1, an enterocyte-specific Ets transcription factor, regulates MIP-3alpha gene expression in Caco-2 human colonic epithelial cells. J. Biol. Chem. 2003, 278, 875–884.
- Schall, T.J.; Bacon, K.; Toy, K.J.; Goeddel, D.V. Selective attraction of monocytes and T lymphocytes of the memory phenotype by cytokine RANTES. Nature 1990, 347, 669–671.
- Rot, A.; Krieger, M.; Brunner, T.; Bischoff, S.C.; Schall, T.J.; Dahinden, C.A. RANTES and macrophage inflammatory protein 1 α induce the migration and activation of normal human eosinophil granulocytes. J. Exp. Med. 1992, 176, 1489–1495.
- von Luettichau, I.; Nelson, P.J.; Pattison, J.M.; Van De Rijn, M.; Huie, P.; Warnke, R.; Wiedermann, C.J.; Stahl, R.A.; Sibley, R.K.; Krensky, A.M. RANTES chemokine expression in diseased and normal human tissues. Cytokine 1996, 8, 89–98.
- Agere, S.A.; Akhtar, N.; Watson, J.M.; Ahmed, S. RANTES/CCL5 Induces Collagen Degradation by Activating MMP-1 and MMP-13 Expression in Human Rheumatoid Arthritis Synovial Fibroblasts. Front. Immunol. 2017, 8, 1341.
- Feng, S.Y.; Lei, J.; Chen, H.M.; Wang, Y.X.; Yap, A.U.J.; Fu, K.Y. Increased chemokine RANTES in synovial fluid and its role in early-stage degenerative temporomandibular joint disease. J. Oral Rehabil. 2020, 47, 1150–1160.
- Bondue, B.; Wittamer, V.; Parmentier, M. Chemerin and its receptors in leukocyte trafficking, inflammation and metabolism. Cytokine Growth Factor Rev. 2011, 22, 331–338.
- Meder, W.; Wendland, M.; Busmann, A.; Kutzleb, C.; Spodsberg, N.; John, H.; Richter, R.; Schleuder, D.; Meyer, M.; Forssmann, W.G. Characterization of human circulating TIG2 as a ligand for the orphan receptor ChemR23. FEBS Lett. 2003, 555, 495–499.
- Jin, C.H.; Yi, K.W.; Ha, Y.R.; Shin, J.H.; Park, H.T.; Kim, T.; Hur, J.Y. Chemerin Expression in the Peritoneal Fluid, Serum, and Ovarian Endometrioma of Women with Endometriosis. Am. J. Reprod. Immunol. 2015, 74, 379–386.
- Ernst, M.C.; Issa, M.; Goralski, K.B.; Sinal, C.J. Chemerin exacerbates glucose intolerance in mouse models of obesity and diabetes. Endocrinology 2010, 151, 1998–2007.
- Kaneko, K.; Miyabe, Y.; Takayasu, A.; Fukuda, S.; Miyabe, C.; Ebisawa, M.; Yokoyama, W.; Watanabe, K.; Imai, T.; Muramoto, K.; et al. Chemerin activates fibroblast-like synoviocytes in patients with rheumatoid arthritis. Arthritis Res. Ther. 2011, 13, R158.
- Wittamer, V.; Franssen, J.D.; Vulcano, M.; Mirjolet, J.F.; Le Poul, E.; Migeotte, I.; Brézillon, S.; Tyldesley, R.; Blanpain, C.; Detheux, M.; et al. Specific recruitment of antigen-presenting cells by chemerin, a novel processed ligand from human inflammatory fluids. J. Exp. Med. 2003, 198, 977–985.
- Takahashi, T.; Kondoh, T.; Fukuda, M.; Yamazaki, Y.; Toyosaki, T.; Suzuki, R. Proinflammatory cytokines detectable in synovial fluids from patients with temporomandibular disorders. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 1998, 85, 135–141.
- Emshoff, R.; Puffer, P.; Rudisch, A.; Gaßner, R. Temporomandibular joint pain: Relationship to internal derangement type, osteoarthrosis, and synovial fluid mediator level of tumor necrosis factor-α. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2000, 90, 442–449.
- Baggiolini, M.; Walz, A.; Kunkel, S.L. Neutrophil-activating peptide-1/interleukin 8, a novel cytokine that activates neutrophils. J. Clin. Investig. 1989, 84, 1045–1049.
- Harris, E.D., Jr. Rheumatoid arthritis. Pathophysiology and implications for therapy. N. Engl. J. Med. 1990, 322, 1277–1289.
More
