The investigation aimed to study the in vitro and in silico antioxidant properties of Melissa officinalis subsp. officinalis essential oil (MOEO). The chemical composition of MOEO was determined using GC–MS analysis. Among 36 compounds identified in MOEO, the main were beta-cubebene (27.66%), beta-caryophyllene (27.41%), alpha-cadinene (4.72%), caryophyllene oxide (4.09%), and alpha-cadinol (4.07%), respectively. In vitro antioxidant properties of MOEO have been studied in 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) and 2,2-diphenyl-1-picrylhydrazyl (DPPH) free-radical scavenging, and inhibition of β-carotene bleaching assays. The half-maximal inhibitory concentration (IC50) for the radical scavenging abilities of ABTS and DPPH were 1.225 ± 0.011 μg/mL and 14.015 ± 0.027 μg/mL, respectively, demonstrating good antioxidant activity. Moreover, MOEO exhibited a strong inhibitory effect (94.031 ± 0.082%) in the β-carotene bleaching assay by neutralizing hydroperoxides, responsible for the oxidation of highly unsaturated β-carotene. Furthermore, molecular docking showed that the MOEO components could exert an in vitro antioxidant activity through xanthine oxidoreductase inhibition. The most active structures are minor MOEO components (approximately 6%), among which the highest affinity for the target protein belongs to carvacrol.
1. Introduction
Lipid oxidation represents a significant concern for the food industry because it can occur throughout processing, storage, and distribution, directly affecting food stability, safety, and quality
[1]. Furthermore, it can increase oxidative rancidity, loss of essential fatty acids, generation of off odors and off flavors, and toxic compounds, crucial for the foodstuff shelf life
[1][2][3][1,2,3]. Consequently, to extend the shelf-life of foodstuffs without any adverse effect on their sensory or nutritional qualities, antioxidants have become an indispensable group of food additives for the food industry, mainly the synthetic ones
[3]. They have been reported to act through single or combined mechanisms; particularly, by neutralizing radicals (as radical scavengers), as singlet oxygen quenchers; through synergism with other antioxidants; through complexing of pro-oxidants that catalyze the generation of radicals; and finally, as inhibition of pro-oxidant enzymes that generate radicals (i.e., lipoxygenase, xanthine oxidase, and NADPH oxidase)
[4][5][6][4,5,6]. However, due to potential health risks (i.e., carcinogenic and teratogenic effects)
[7][8][7,8], food consumers have an increasing demand for the development of natural antioxidants, which are generally supposed to be safer
[9][10][9,10].
The essential oils (EOs) exert various biological activities, prominently antioxidant, antibacterial, and antifungal activities
[11][12][11,12]. Those properties are mainly associated with EOs’ chemical composition, which is determined by pedoclimatic conditions and plant genotype
[13][14][15][13,14,15]. Numerous EOs have been confirmed as natural antioxidants
[11][16][17][18][11,16,17,18] and are recommended as possible replacements of synthetic antioxidants in the food industry. Moreover, the natural extracts’ biological activities can have applicability to the pharmaceutical industry, by inhibiting lipid peroxidative damage associated with pathological disorders, such as aging processes, coronary atherosclerosis, Alzheimer’s disease, and carcinogenesis
[19][20][19,20]. The antioxidant activity of natural extracts may be due to a combination of multiple factors that commonly result in the reduction in cellular oxidative stress. In addition to the ability of some substances to act as molecules capable of reducing free radicals with destructive oxidative potential, they can act as inducers of enzymes with antioxidant effects, inducers of endogenous antioxidant compound biosynthesis, or inhibitors of enzymes whose metabolic action generates reactive oxygen species (ROS) as a byproduct
[21]. Enzymes that can produce ROS are usually involved in metabolic oxidative degradation reactions of endogenous/exogenous compounds. Cytochromes, lipoxygenases, or xanthine oxidoreductase fall into this category. The increased activity of such enzymes can generate a high level of oxidative stress which is usually associated with pathological conditions. Oxidative stress caused by xanthine oxidoreductase hyperreactivity is associated with gout
[22], whereas the oxidizing activity of lipoxygenase plays a significant role in oxidative-stress-triggered apoptosis
[23]. Other enzymes that produce reactive oxygen species are involved in the regeneration of coenzymes (NADH, NADPH) that regulate the mitochondrial electron transport. Disruption of the physiological activity of such enzymes can cause a mitochondrial imbalance with increasing ROS levels which can have serious implications for cell proliferation, viability, or programmed cell death
[24].
Melissa officinalis L. (lemon balm), a member of the
Lamiaceae family, is a perennial subshrub endemic to Europe and Central Asia and extensively cultivated in Romania, Spain, Bulgaria, and Turkey
[25]. All three subspecies of
M. officinalis, subsp.
officinalis, subsp.
Inodona, and subsp.
Altissima, have commercial value, but only subsp.
officinalis has been extensively cultivated for its characteristic lemon-scented oil
[25][26][25,26].
M. officinalis leaves contain 0.05–0.15% EO in fresh material and 0.1–0.45% EO in dried material, respectively
[27]. Due to its digestive and antispasmodic properties, the leaves of
M. officinalis are utilized in traditional medicine to treat moderate abdominal disorders and biliary dyskinesia
[28]. The
M. officinalis essential oil mainly contains terpenic aldehydes (citral, geranial, neral, and citronellal) and terpenic alcohols (geraniol, linalool, and octen-3-ol-l)
[29]. Moreover, EOs and extracts of lemon balm possess antibacterial, antiparasitic, and antiviral activity
[18][29][30][31][18,29,30,31]. Moreover, lemon balm oil and extracts demonstrate good potential for antioxidant activity
[18][26][32][33][18,26,32,33] that recommend them for being used in lipid-containing foods.
2. Results and Discussion
2.1. MOEO Chemical Composition
A pale-yellow color oil with a lemon-like odor was isolated by steam distillation from
M. officinalis leaves with a 0.41% yield. The determined yield revealed that the plant sample from western Romania is rich in essential oil. Moreover, the results match the scientific literature’s values that report yields ranged between 0.01 and 0.45% (dry material)
[34][44]. Higher yields have been recorded for
M. officinalis from Brazil (0.97%)
[35][45], Iran (1%)
[36][46], and Spain (0.8%)
[37][47]. According to Kittler et al.
[34][44], the lemon balm EO content is strongly related to the biotic and abiotic conditions, different harvesting years, and genetic makeup of the genotypes.
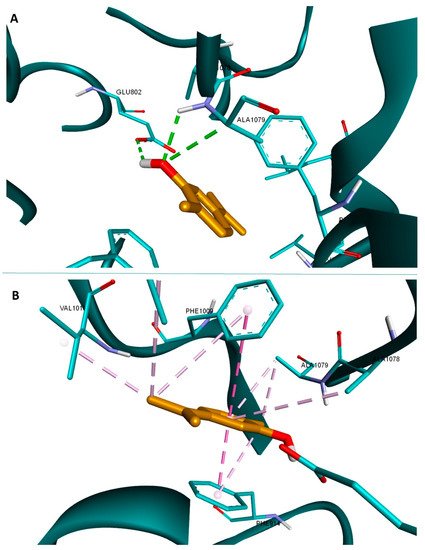
The GC–MS analysis identified 36 components, representing 98.79% of the total contents of the MOEO (
Table 12). The main constituents are beta-cubebene (27.66%), beta-caryophyllene (27.41%), alpha-cadinene (4.72%), caryophyllene oxide (4.09%), and alpha-cadinol (4.07%). A high content of sesquiterpenoids, such as beta-cubebene (15.41%), beta-caryophyllene (14.24%), alpha-cadinol (7.19%), has also been reported in a MOEO from Turkey
[38][48]. According to the scientific data, beta-cubebene is a chemical compound commonly found in lower amounts in subsp.
officinalis [15][28][15,28]. Only for the subsp.
altissima was there previously recorded a higher amount of beta-cubebene (39%)
[39][49]. However, caryophyllene, the second major compound of the analyzed oil, has been recorded in large amounts in subsp.
officinalis from Sardinia (20–39%)
[40][50] and Germany (1.17–18.64%)
[34][44]. Another peculiarity of the analyzed oil is the low content of alpha-citral (2.06%), beta-citral (1.15%), and citronellal (0.27%), compared with other subsp.
officinalis. These oxygenated monoterpenes are present in large amounts in subsp.
officinalis EOs
[15][18][28][15,18,28] and are responsible for their lemon-like aroma
[39][49]. This phytochemical polymorphism is significantly determined by genetic factors
[34][41][44,51] and also influenced by ontogenetic
[42][52] and environmental variations
[43][53].
Table 1. Chemical composition of M. officinalis subsp. officinalis essential oil analyzed by GC–MS.2. Chemical composition of M. officinalis subsp. officinalis essential oil analyzed by GC–MS.
| No |
Compounds |
RI 1 |
% |
| 1. |
Hydroperoxide, 1-ethylbutyl |
925 |
0.11 |
| 2. |
Hydroperoxide, 1-methylpentyl |
934 |
0.08 |
| 3. |
p-Cymene |
1005 |
0.07 |
| 4. |
beta-trans-Ocimene |
1017 |
0.09 |
| 5. |
beta-cis-Ocimene |
1029 |
0.51 |
| 6. |
gama-Terpinene |
1042 |
0.09 |
| 7. |
Nonanal |
1092 |
0.17 |
| 8. |
(R)-(+)-Citronellal |
1145 |
0.27 |
| 9. |
Decanal |
1206 |
0.11 |
| −6.4 |
10. |
Octyl acetate |
1211 |
0.08 |
| Decanal |
−5.3 |
−5.5 |
−4.9 |
−6.5 |
11. |
beta-Citral |
1241 |
1.15 |
| Octyl acetate |
−5.1 |
−5.9 |
12. |
(S)-(−)-Citronellic acid, methyl ester |
1264 |
0.66 |
| −5.5 |
−6.5 |
| beta-Citral (Neral) |
−4.4 |
−7.4 |
−6.8 |
−6.3 |
13. |
alpha-Citral |
| (S)-(−)-Citronellic acid, methyl ester | 1275 |
−2.8 |
−5.72.06 |
| −5.3 |
−6.5 |
14. |
Carvacrol |
1309 |
0.18 |
| 15. |
| alpha-Citral (Geranial) |
−3.8 |
−7.6 |
−7.3 |
−6.4 |
Methyl geranate |
| Carvacrol |
−3.9 | 1333 |
0.19 |
| −5.7 |
−5.6 |
−7.2 |
16. |
p-Menthane-3,8-diol |
1352 |
2.14 |
| Methyl geranate |
−0.4 |
−7.7 |
−7.2 |
−7 |
17. |
alpha-Copaene |
| p-Menthane-3,8-diol |
−5.8 | 1394 |
2.78 |
| −6.3 |
−6.1 |
−6.7 |
18. |
beta-Bourbonene |
1402 |
1.16 |
| alpha-Copaene |
−4 |
−5.9 |
−5.9 |
−6.1 |
19. |
beta-Elemene |
1408 |
2.73 |
| beta-Bourbonene |
−5.3 |
−6.5 |
−6.2 |
−6.2 |
20. |
beta-Caryophyllene |
1442 |
27.41 |
| beta-Elemene |
−3.5 |
−6.7 |
−6.4 |
−6.4 |
21. |
alpha-Cubebene |
| beta-Caryophyllene | 1450 |
0.41 |
| −4.9 |
−6.4 |
−6.1 |
−5.2 |
22. |
alpha-Caryophyllene |
1476 |
3.37 |
| alpha-Cubebene |
−5.9 |
−6.1 |
−6 |
−5.2 |
23. |
Alloaromadendrene |
1481 |
0.87 |
| alpha-Caryophyllene |
−4.4 |
−6.4 |
−5.8 |
−5.1 |
24. |
beta-Cubebene |
1504 |
27.66 |
| 25. |
(Z,E)-alpha-Farnesene |
1512 |
1.37 |
| 26. |
alpha-Muurolene |
1520 |
0.96 |
| 27. |
alpha-Farnesene |
1526 |
0.71 |
| 28. |
gamma-Cadinene |
1534 |
1.36 |
| 29. |
alpha-Cadinene |
1540 |
4.72 |
| 30. |
Germacrene D-4-ol |
1596 |
1.96 |
| 31. |
Caryophyllene oxide |
1601 |
4.09 |
| 32. |
alpha-Cadinol |
1669 |
4.07 |
| 33. |
Isoaromadendrene epoxide |
1819 |
0.98 |
| 34. |
Platambin |
1849 |
2.13 |
| 35. |
Murolan-3,9(11)-diene-10-peroxy |
1884 |
1.18 |
| 36. |
Aromadendrene oxide |
1891 |
0.92 |
| |
|
Total: |
98.79 |