1000/1000
Hot
Most Recent

Lipid peroxidation refers to the process in which lipids are oxidized to generate lipid peroxides as a primary product. Cellular lipid peroxidation can occur through different reactions, but they can be categorized into enzyme and non-enzyme dependent reactions. The primary substrates in lipid peroxidation reactions are polyunsaturated lipids since carbon-carbon double bonds are susceptible to reactive oxygen species, such as the hydroxyl radical (HO•), which is a key radical that participates in peroxidation reactions.
Oxidative stress and inflammation are linked to cancer development [1][2]. Mutations in the DNA, phosphorylation of kinases, or inactivation of phosphatases can alter the cell growth, cellular control of the division, cell death, cell fate, and cell motility, which are altered in angiogenesis, inflammation, and fuel cancer progression [3][4][5][6]. A progressive increase of reactive oxygen species (ROS) marks the transition steps from a healthy tissue towards an invasive carcinoma [7]. This trend is owed to cancer cells' metabolic aberrations to adapt strategies to escape from cell death. It occurs in the presence of compensatory upregulation of the genes coding antioxidant enzymes, preventing ROS induced cell death [2][5]. Therefore, a blockage of the antioxidant cellular defenses or pro-oxidant therapies' stimulation is suggested as potential strategies to fight against cancer [8][9]. In general, lipids peroxides ability to participate in anti-inflammatory and/or pro-inflammatory signaling cascades is defined by the length of the fatty acyl chain, the number of unsaturations, and the place where the oxidation account [10]. During lipid peroxidation, oxygen molecules are added to the unsaturated fatty acyl chain of non-polar lipids, increasing their water solubility and diffusion towards the membrane surface. Cyclooxygenases or lipoxygenases accessibility for their substrates is boosted, the generation of lipid metabolites linked to inflammation is prompted, and the interaction of specific proteins and receptors recognizing lipid oxidation products is promoted [11][12][13][14][15]. All these actions are part of the lipid-dependent inflammatory cascades. Moreover, lipid peroxides can be the substrate for certain enzymes such as phospholipases, which prevent the accumulation of lipid peroxides into the membrane, which might functionally damage membrane components such as proteins by induction of covalent modifications, which might compromise membrane permeability [16][17]. The release of fatty acids and oxidized fatty acids from phospholipids by the action of phospholipases is essential in inflammation [18]. In addition, polyunsaturated fatty acids (PUFA) and their related phospholipids are very well-known signaling molecules with pro-inflammatory and anti-inflammatory functions, but also sensitive substrates for peroxidation [19][20][21][22][23][24][25][26][27][28]. PUFA can promote cell life or cell death through complex signaling cascades related to the fatty acid structure and their oxidation products [29][30]. In cancer, the connection of some of these pathways with inflammation is being unveiled by identifying a group of lipid oxidation products, known as lipid pro-resolving mediators, which can resolve the inflammation [31][32][33]. Their discovery opens the opportunity to identify new potential drugs in cancer therapy [32]. Some lipid oxidation products have also gained attention since they are suggested as biomarkers for cancer development and recurrence [34][35]. In general, lipids' ability to participate in anti-inflammatory or pro-inflammatory signaling cascades depends on the lipid's nature and its degree of oxidation.
In the non-enzymatic reactions, the Fenton and Haber–Weiss reactions producing HO• are dependent on transition metals (i.e., iron [36][37]) for the initiation of the radical chain reactions required for lipid peroxidation. In addition to this radical formation, some authors have suggested that for the initiation of lipid peroxidation reactions, the formation of a complex between iron and the lipids is required [38]. In general, it is accepted that the initiation reaction starts when a hydrogen atom is abstracted from lipid, forming an alkyl radical [39]. HO• is preferred over other radicals to performed this abstraction [38][39][40][41][42][43]. Once the alkyl radical is formed, the chain-carrying a carbon radical reacts with oxygen, leading to an alkyl peroxyl radical formation. This radical can abstract hydrogen from an organic substrate, which can be another lipid, to form a hydroperoxide plus an organic radical or be added to alkenes, such as those present in the fatty acyl chains of PUFA present in phospholipids, which provide isolated double bonds [44]. This last reaction leads to the formation of a lipid hydroperoxide with a conjugated double bond. By reaction with metals, lipid radical reactions leading to lipid peroxidation can be reinitiated as part of the propagating radical reactions [45][46]. This process occurs when the hydroperoxides react with an oxidized metal forming an alkoxyl radical. In case the reaction involves a reduced metal, e.g., Fe2+, an alkyl peroxyl radical is generated, which also contributes to the propagation of the reaction. PUFA are lipid molecules priming the Fenton's reaction, as previously indicated. Arachidonic acid (AA) and the phospholipids containing this fatty acid are essential molecules since they are precursors of pro- and anti-inflammatory mediators, sometimes enriched at cellular locations identified as signaling platforms, such as the plasma membrane lipid rafts [47][48]. Lipid oxidation at this location during the inflammation process is relevant since lipid rafts are platforms required for cell activation in the immune system [49].
HO• can also be generated by ionizing radiation [38], which is generally used and applied in patients to treat cancer. As previously indicated, HO• is a very reactive radical, leading to the generation of lipid hydroperoxides and oxidizing other biomolecules, including DNA. Radiation exposition leads to peroxides generation in membranes enriched with PUFA. Indeed, this fact should not be discarded as a relevant factor for cancer therapy's success in these patients [50][51][52][53]. Noteworthy, more efforts are required to shed light on the role that ionizing radiation generating lipid peroxides have in cancer cell death vs. other targets. These investigations could help in the characterization of pharmacological drugs prompting the cancer cell sensitivity to lipid hydroperoxides generated by ionizing radiation (i.e., glutathione peroxidase 4 (GPX4) inhibitors) and, therefore, better define or reduce the patient's exposure to ionizing radiation that can also damage non-tumoral tissues.
The enzyme-dependent reactions are executed by peroxidases, which have been elegantly classified by Vlasova [54]. Based on this classification, lipids can be oxidized by proteins that possess a true peroxidase activity, myeloperoxidase, eosinophil peroxidase, lactoperoxidases, or by proteins that do not have a peroxidase activity but acquire a pseudo-peroxidase activity under certain conditions, i.e., cytochrome c [55][56][57][58][59] upon binding to cardiolipin or other hemeproteins in defined conditions [60][61][62][63]. Lipoxygenases (LOX), cyclooxygenases (COX), and some cytochrome P450s present a pseudo-peroxidase activity that generates intermediary lipid peroxides required in their catalytic cycles, which might be a substrate for other activities, i.e.: cyclization reactions in the case of COX. The coordinated iron or iron associated with the heme group is key in peroxidases' catalytic center. Compound I, compound II, and sometimes compound III are generally typical and associated with different iron valences [64]. The function of real peroxidases might depend upon the existence of binding pockets where substrates can settle and interact with the enzyme catalytic center and upon electrons donated by organic molecules, which might be protein amino acids acting as electron donors [54]. In heme-dependent pseudo-peroxidases, substrates accessibility to the enzyme coordinating sphere depends upon the enzyme catalytic center flexibility to swift from a metal hexa- to penta-coordination, a feature that can be influenced by the redox state or the interaction with a ligand, i.e., cytochrome c upon cardiolipin binding to the protein [55].
The main enzymes using AA to generate lipid hydroperoxides and derived metabolites as signaling molecules in mammals and in cancer are cyclooxygenases (COX), lipoxygenases (LOX), and P450 families [29][30][47][65]. COX-1 is constitutively expressed in many tissues and cell types, whereas COX-2 is an inducible cyclooxygenase isoform which activation has been reported in tumoral tissues [66][67][68][69][70][71]. Some studies have also pointed out that other peroxidases (like myeloperoxidase and eosinophil peroxidase) are released from infiltrating neutrophils and eosinophils in the tumor microenvironment [72][73][74][75] or from infiltrated macrophages can also generate lipid hydroperoxide [76]. Although myeloperoxidases are potential sources of lipid peroxides and some myeloperoxidases polymorphisms have been correlated with a higher risk of suffering pulmonary, ovarian, and gastric cancer [77][78][79], there is no correlation between their activity and the lipid peroxides derived from it with disease development. In contrast, anti-inflammatory drugs have been linked to a decreased risk of cancer development and decreased tumor growth rate [28]. Notably, in this context, overexpression of enzymes generating eicosanoids in breast, lung, and pancreas cancer has been reported [28]. In particular, prostaglandins (PGs) can stimulate mitogenesis by directly affecting fibroblasts, osteoblasts, and mammary cells. The production of the proinflammatory PG named prostaglandin E2 (PGE2) (Figure 1A) through COX-2 activity can be found in mutagenesis, angiogenesis, and cell migration processes associated with cancer (Table 1). An activation mechanism for COX-2 using human colorectal HT-29, and the human prostate carcinoma DU145 cell lines has been proposed [80]. A correlation between the production of PGE2 with the resistance of cancer cells to apoptosis has been found through activation of the P2Y2/Src/p38 signaling pathway, which leads to AA release from the membranes by overexpression of some PLA2 isoforms, and the overexpression of COX-2 with the subsequent PGE2 production [80].
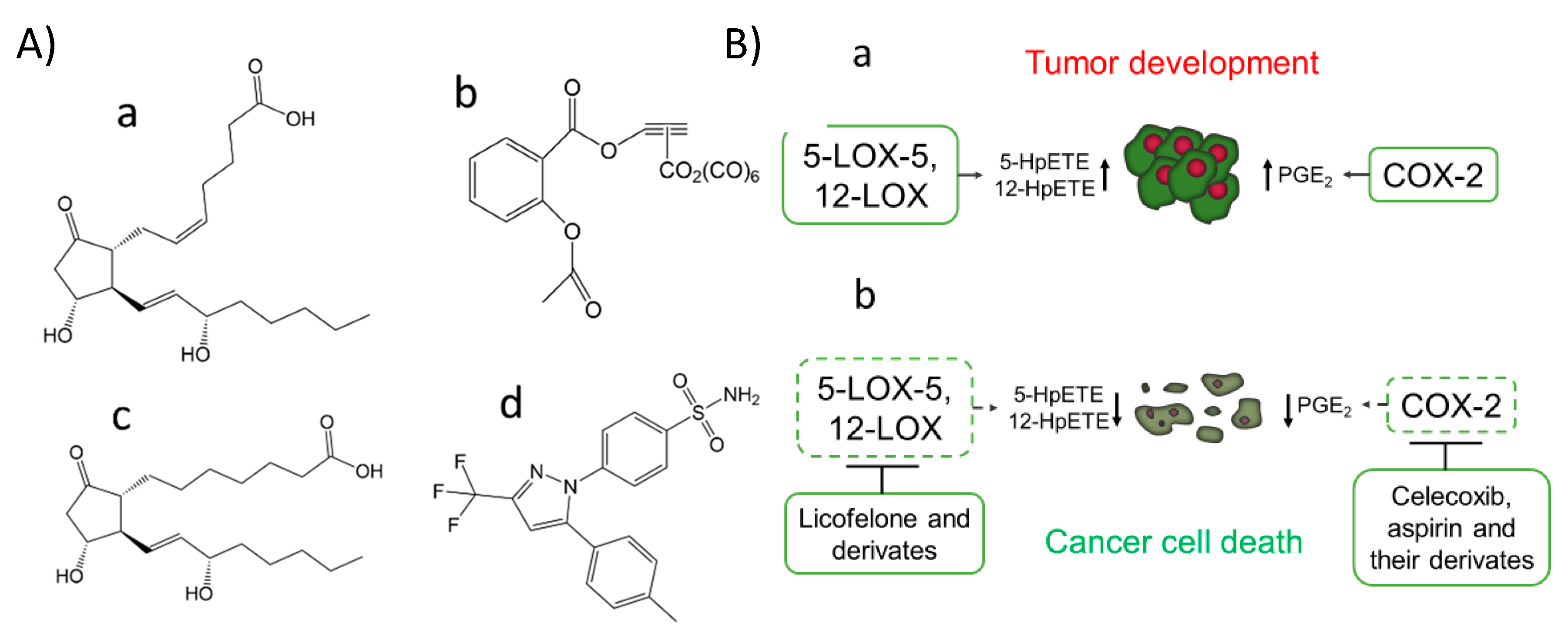
Figure 1. COX-2 inhibitors, PGE1, and PGE2 chemical structure (A). The prostaglandin E2 (PGE2) (pro-cancer) (a), the aspirin derivate named [2-acetoxy-(2-propynyl)benzoate]hexacarbonyldicobalt (Co-ASS) (b), the prostaglandin E1 (PGE1) (anticancer) (c) and the COX-2 selective inhibitor named celecoxib, (p-(5-p-Tolyl-3-(trifluoromethyl) pyrazol-1-yl)benzenesulfonamide) (d). Expression of COX-2, 5-LOX, and 12-LOX in cancer and the effect of inhibitors against these targets (B). Activation of 5-LOX-5, 12-LOX, and COX-2 has been reported in the development and progress of tumors from different tissues associated with the production of specific lipid peroxides and metabolites, such as PGE2 (a). Some inhibitors of these LOX isoforms and COX-2 have emerged as potential therapeutical agents for cancer treatment, modulating the production of previously commented metabolites and by induction of cancer cell death (b).
To better define the role of lipid peroxidation in mammals signaling, the type of lipid hydroperoxide generated should be finely characterized. In the metal-mediated lipid peroxidation based on the Fenton and Haber–Weiss reactions, random lipid peroxides are generated and differentiated from those formed by specialized enzymes that produce specific lipid signatures that can be used as fingerprints of enzymatic activities [58][81][82]. Many efforts are being made to find specific inhibitors that can provide a specific modulation of lipid hydroperoxide production that could act as mediators in signaling cascades [58][83]. For example, aspirin, which has beneficial effects in some cancer types, has been proposed to play that role [82][84][85]. Its implication in cancer has been associated with COX inhibition via acetylation of the active site, where AA binds. This accounts for COX-1 isozyme inhibition, while in COX-2, aspirin binding produces a structural rearrangement shifting the cyclooxygenase towards the lipoxygenase activity [86][87]. Therefore, AA oxygenation and cyclization to form a 15R-Prostaglandin endoperoxide is promoted, which favors the production of Prostaglandin D2 (PGD2) (with has a suggested function in inflammation resolution) instead of PGE2 [88]. Recent studies have also shown the potential therapeutically effect of aspirin organometallic derivatives as anticancer agents targeting COX-2 [89], such as the 4-[5-(4-Chlorophenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]-benzenesulfonamide (SC-236) and [2-acetoxy-(2-propynyl)benzoate]hexacarbonyldicobalt (Co-ASS) (Figure 1A), which open a promising field in the search for inhibitors derived from aspirin [90][91][92].
Table 1. Correlation between COX-2 level and other biomarkers found in tumoral tissues
|
Tissue Location and Type of Cancer |
Correlation with Other Biomarkers |
|
Colon cancer [66], primary tumors and metastatic lymph nodes resections for colorectal adenocarcinoma [93], stage II and III colorectal cancer patients [94] |
High levels of COX-2 correlates with high levels of MMP-2 and VEGF expression and shorter survival time [93][94]. |
|
Cervical cancer [67] |
Multivariate analysis of COX-2 levels in tumor/stromal compartments. The proportion of CD3+, CD4+, and CD25+ cells was lower in tumors with high tumor/stroma ratios, but in these tumors, mast cells were increased [67]. |
|
No correlation between COX-2 expression and EGFR, and HER-2/neu status [96]. |
|
|
Elevated COX-2 expression associated with a large tumor size, a high histological grade, a negative hormone receptor status, a high proliferation rate, high p53 expression, and the presence of HER-2 oncogene amplification along with axillary node metastases and a ductal type of histology [98]. COX-2 inhibition may potentially prevent the development of ER-positive and ER-negative breast cancers [98]. Expression of PGE2 and IL-8 [101]. COX-2 over-expression induces an oncogenic microRNA (miR655) in human breast cancer cells by activation of EP4 [102]. |
|
|
COX-2 expression stabilizes survivin, an inhibitor of apoptosis (IAP) [103]. CacyBP expression was significantly negatively associated with the COX expression [104]. |
|
|
Correlation between HER-2, EGFR, and COX-2 expression in patients of non-small cell lung cancer at different degrees [69] |
|
|
Laryngeal cancer [71] |
Cox-2 overexpression was significantly associated with radioresistant tumors [71]. |
|
Papillary thyroid cancer [106] |
The expression of COX-2 is increased with age in papillary thyroid cancer [106]. Immunohistochemically, expression of COX-2 and VEGF-C correlated strongly, and both were induced by the tumor promoter phorbol 12-myristate 13-acetate [107]. |
|
No correlation between COX-2 expression with estrogen (ER) or progesterone receptor (PR), p53, and neu [110]. Correlation between COX-2 (59%) and aromatase (65%) expression but not estrogen and progesterone receptor [111]. |
|
|
Invasive gallbladder cancer [112] |
COX-2, c-Met, β-catenin, c-erbB2 and EGFR were over-expressed in 80%, 74%, 71%, 62%, and 11% of invasive gallbladder cancers, respectively [112]. |
|
Prostate cancer Metastatic primary prostate carcinoma compared to non-metastatic cancers [113][114][115][116] |
COX-2 and Ki-67 antigen co-expression in 42.9% and 67% of the prostate cancer patients [113]. Patients with PSA > 7 ng/mL and high COX-2 expression had the highest probability of recurrence [114]. The expressions of COX-2 and E-cadherin are very firmly and inversely correlated as prognostic indicators. [115]. High expression of COX-2, TGF-beta, and Ki67 in metastatic primary prostate carcinoma was associated with death from prostate carcinoma [116]. |
|
A positive correlation between COX-2 and K-ras expression with the depth of invasion and lymph node metastasis in gastric cancer [117]. Epithelial MMP-2 expression in gastric cancer is associated with aggressive forms, COX-2 expression, and poor survival [118]. |
|
|
Cervical cancer [119] |
DNA hypermethylation of the COX-2 gene may be a potential prognostic marker in the early stages of cervical cancer [119]. |
|
Anaplastic pancreatic cancer [122] |
Tumor COX-2 expression portends a poor prognosis for patients with resected adenocarcinoma of the pancreas, particularly in tumors > or = 3 cm [121]. Expression of L1CAM, COX-2, and EGFR in the majority of undifferentiated pancreatic carcinomas [122]. |
Lipid hydroperoxides generated via enzyme or non-enzyme-dependent reactions can be further oxidized to form highly reactive species and lipid autoxidation products. Acrolein, malonaldehyde, and 4-hydroxynonenal can covalently modify proteins leading to functional and structural changes in proteins [123][124][125]. Lipid autoxidation products mainly react with primary amines and lysines, histidine, and cysteine residues from proteins to induce covalent crosslinking and prompt protein aggregation [125]. The amino acid residues mentioned above are also the primary targets for several protein post-transductional modifications, such as acylation, acetylation, phosphorylation, methylation, glycation, and S-nitrosylation, among other modifications [126][127]. Therefore, it can be presumed that the reaction of essential amino acid residues with lipid autoxidation products will also induce changes in the signaling pathways in which these proteins are involved. The generation of lipid autoxidation products has been reported in cancer development, angiogenesis, and invasiveness [128][129][130][131][132]. Some autoxidation products, such as 4-hydroxynonenal, have been implicated in DNA modifications that generate cancer-linked mutations [1].
Antioxidants play a central role to counteract lipid peroxidation. In the non-enzyme-dependent reactions, the radical chain reactions can terminate when antioxidants react with the alkyl peroxyl or the alkoxyl radicals. Tocopherol is the main membrane antioxidant in charge of reacting with these radicals and one of the primary membrane antioxidants against reactions generating lipid peroxides, in general [133]. Consequently, a lipid hydroperoxide or the alcohol and the radical antioxidant are products of the reaction between the lipid radical with the antioxidant. In the particular case of the tocopheroxyl radical, it can be reduced back to tocopherol by its reaction with other antioxidants, such as ascorbate and ubiquinol [134], or by enzymes in charge of reducing the antioxidant radical [135][136]. Indirectly or directly, the enzymatic activities that reduce the radicals derived from antioxidants are essential to keep optimal alpha-tocopherol levels in the membrane [135][137][138]. Other types of enzymes, such as glutathione peroxidases, can reduce lipid hydroperoxides to alcohols at the expense of glutathione (GSH). GPX4 is a pharmacological target in cancer [139][140][141], and its inhibition has been found to induce cancer cell death by the accumulation of lipid hydroperoxides [140].