2D sheets are less than 100 nm in a single coordinate direction. This specifies them as a thin paper or sheet-like material with a relatively expansive basal plane. Common examples are graphene (Gr), molybdenum disulfide (MoS
2), and hexagonal boron nitride (hBN)
[1]. Their unique structures induce anomalous chemical and electrochemical properties
[2][3]. The synthetic methods of vdW materials have been recently reviewed by Di Bartolomeo
[4], and therefore will not be detailed in this paper. In general, we can observe that despite the fact that many sophisticated methods have been developed for the synthesis of vdW materials, they can be fabricated in a rather easy way. One of the most frequently discussed is an exfoliation or “scotch tape” method categorized as a top-down approach. A 2D sheet may be exfoliated from its 3D crystal counterpart using this method. A bottom-up approach focuses on growing 2D sheets from small molecule precursors
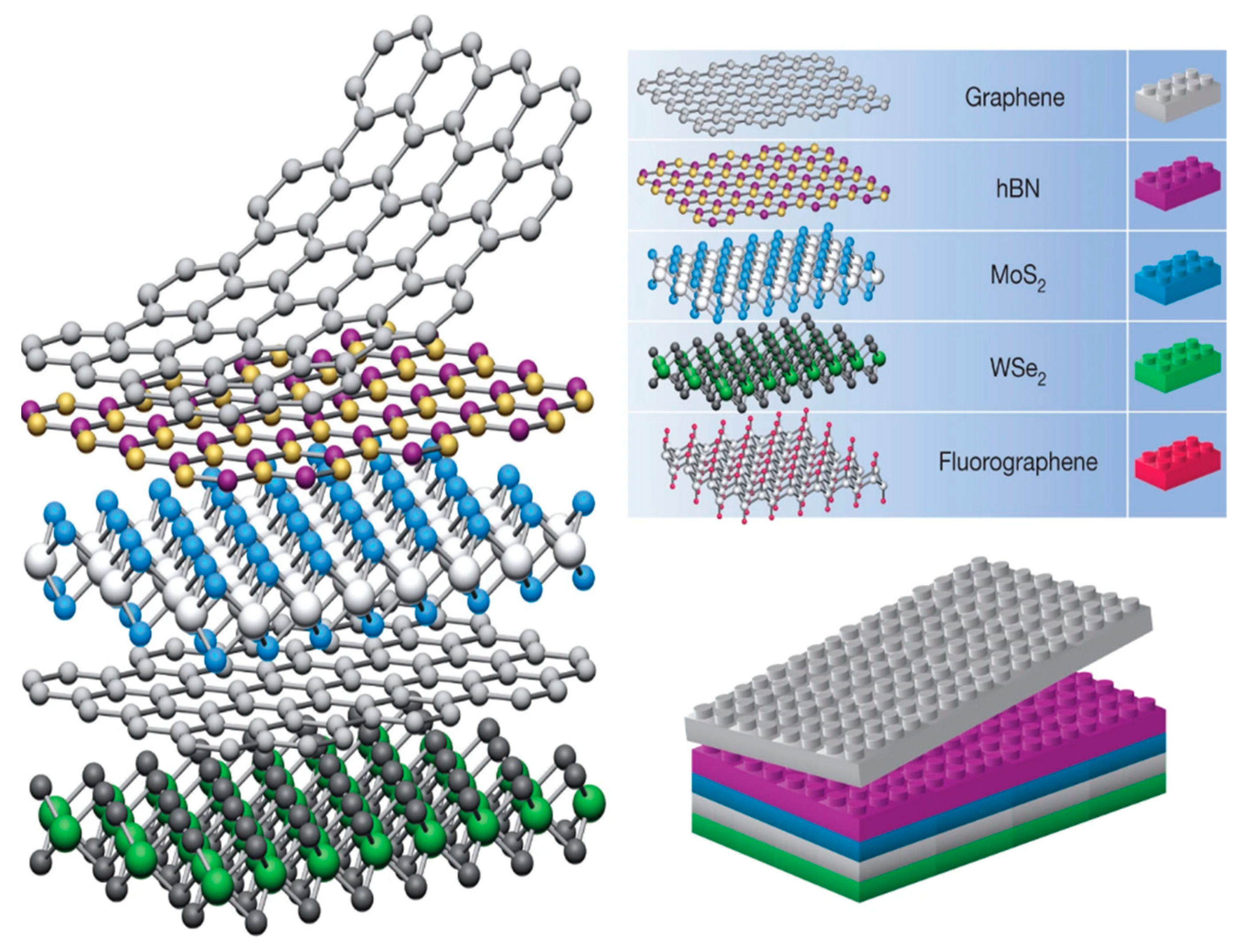
[2]. van der Waals heterostructures are stacks of 2D sheets illustrated in
Figure 1. As perfect as Lego pieces match each other, vdW building blocks fit exceptionally well. Electronic coupling at the interface unlocks unique molecular characteristics, which are presumably tunable depending on the 2D sheet materials used, and other physical properties, such as layering and inter-layer distance
[5]. Stability and contaminant issues heighten the difficulty of synthesizing heterostructures and limit the amount of available 2D building blocks. Typically, high stability of the 3D bulk crystal is required before one can begin peeling off 2D sheets
[1].
Figure 1. Structure of van der Waals heterostructures, similar to that of Legos. Reprinted (adapted) from reference
[1], copyright (2013) Springer Nature.